PHARMACOGNOSTICAL EVALUATION, PHYTOCHEMICAL SCREENING OF HOUTTUYNIA CORDATA THUNB LEAVES AND INVESTIGATION OF ITS ANTI-TUSSIVE EFFECT ON GUINEA PIGS
HTML Full TextPHARMACOGNOSTICAL EVALUATION, PHYTOCHEMICAL SCREENING OF HOUTTUYNIA CORDATA THUNB LEAVES AND INVESTIGATION OF ITS ANTI-TUSSIVE EFFECT ON GUINEA PIGS
Ireenia Warjri and Rajat Das *
Department of Pharmacognosy, Himalayan Pharmacy Institute, Majhitar, Rangpo, East Sikkim, India.
ABSTRACT: Background: Many plants are known by people to be used traditionally as medicinal plants among those known some are investigated for their medicinal activities whereas some have not been evaluated. Houttuynia cordata is a plant most commonly found in Southeast Asian countries, it is known tradionally in many countries to be used for various diseases like diarrhea, cough and many others depending on the country find outs. Objective: To study the Pharmacognostical, phytochemical characteristics of Houttuynia cordata and to investigate its anti-tussive activity on guinea pigs. Methods: The plant Houttuynia cordata was collected, dried and powdered. The powdered plant was carried out for extraction using various solvents based on their polarity; the extracts obtained are carried out for phytochemical screening and pharmacological investigation for its medicinal activity. Results: Sub-fraction extraction of the plant was carried by various solvents from the most polar to the least using Soxhlet apparatus; Petroleum ether 10.07%, Benzene 2.27%, Chloroform 1.06%, Acetone 3.34%, Ethanol 6.27%, Aqueous extract 26.21%. Phytochemical screening of H. cordata showed the presence of Alkaloids, carbohydrates, flavonoids, glycosides, Saponins and organic acids; the phytochemical screening results determine the extract that can be used to investigate the pharmacological activity. The pharmacological activity of the plant extracts showed that the Ethanol extract of the plant showed comparatively better results than the aqueous extract when compared to the baseline and standard drug Ambroxol on guinea pigs. Conclusion: The plant Houttuynia cordata has the potential anti-tussive activity when studied on chemically cough-induced guinea pigs; the plant can be studied more in detail for the activity and other activities that have not been explored.
Keywords: Houttuynia cordata, Flavanoids, Anti-tussive, Animal activity, Medicinal plant, Traditional use
INTRODUCTION: Cough is a powerful physiological mechanism that causes the central airways to be cleared of foreign material and excess secretions; it results from the stimulation of sensory nerves in the airways 1.
Physiologically, the cough has three phases: inspiratory, compressive, and expiratory 2. There are two types of cough 3, 4. Acute cough, chronic cough.
The development of automated cough counting monitoring devices has been the goal of cough assessment for some time. They will likely provide useful data for the development and regulatory approval of anti-tussive drugs in the future 5. Anti-tussive drugs are amongst the most widely used medications worldwide. They are used to relieve cough associated with the common cold and to treat persistent cough arising from a wide range of respiratory conditions such as chronic obstructive pulmonary disease, lung cancer and pulmonary fibrosis 6. Types of antitussives 7.
Centrally acting anti-tussives, Peripherally acting anti-tussives. A variety of inhaled tussive stimuli such as capsaicin, citric acid and distilled water can be used to assess the sensitivity of the cough reflex. 8. Capsaicin is the most widely used tussive agent. The plant Houttuynia cordata Thunb, is a plant found in most countries in South East Asia. It is a plant eaten by most people as a salad and is also known to be used traditionally in these Southeast 9. Asian countries as a medicinal plant. It is commonly known in English as Chameleon Plant, Fish Mint, Fishwort, Chinese lizard tail and heart leaf and in khasi it is called Jamyrdoh. Houttuynia cordata is distributed widely in Bhutan, China, Indonesia, Japan, Korea, Myanmar, Thailand, Nepal, Taiwan, Vietnam, the Northwest Himalayan region, and North-eastern parts of India and its uses differ in each country 10.
MATERIALS AND METHODS:
Sample Collection: The leaves of Houttuynia cordata (Jamyrdoh) as sample were collected from different parts of Ri-Bhoi District, Meghalaya, India. The collected leaves were dried under shade after proper washing. For sample preparation, dried leaves were pulverized into coarse powder and stored in a tight moisture-free container.
FIG. 1: COLLECTION OF SAMPLE
FIG. 2: DRYING OF SAMPLE
FIG. 3: POWDERED SAMPLE
Identification and Authentication of Sample: The plant parts were collected during January’2022 for preparing the herbarium sheet; the plant was identified and authenticated by the Botanical Survey of India, Shillong bearing no- BSI/ERC/Tech/2021-22/390.
Macroscopic Examination: The macroscopical observation was carried out as per the standard methods to determine the shape, size, taste, colour, and odour 11.
Microscopic Evaluation:
Powder Microscopy of Leaves: The powder analysis of the drugs was performed by subjecting the powdered drug to different reagents like Fast green, Ruthenium red and Safranin and observed under the microscope at 10X for the presence of the constituents like fibres, epidermis, pollen grains, oil glands and calcium oxalate crystals.
Transverse Section of a Leaf: A fine section of the leaf of H. cordata was cut with a razor blade and mounted in a glass slide with a drop of glycerine, covered with a cover-slip, place the slide on the stage of Digital Microscope and observed using 10X and 45X lens, each distinguishes character was noted down 12.
Determination of Ash Value:
Total Ash: Ash value is generally the residue remaining after incineration. It represents the inorganic salts naturally occurring in the drug and those added for adulteration. The main objective is to remove all the traces of organic matter which may otherwise interfere in an analytical determination. Take empty silica crucible, transfer to muffle furnace, Place the empty SC inside MF, set temp, keep for 15 min to dry properly. After 15 min bring temp setting knob to zero, remove the SC from MF chamber now use a desiccator to cool at R.T. Weigh empty SC and tare the weighing balance. Add sample powder & weigh about 2gm. Spread the powder in SC and transfer it to MF & set temp to 450-600°C (Allow it for burning to white ash or nearly white) for 30 mins. Bring the temp knob to zero & remove SC. Allow to cool in desiccator remove from desiccator & weigh it 13.
% Total ash = X-Y / N × 100
Where, X= the weight of crucible dish + ash obtained, Y= the weight of the empty dish, N= weight of crude drug taken.
Determination of Acid Insoluble Ash: Prepare 40% HCl take an empty silica crucible, transfer to muffle furnace, Place the empty SC inside MF, set temp, keep for 15 min to dry properly. After 15 min bring temp setting knob to zero, remove the SC from MF chamber now use a desiccator to cool at R.T. Weigh empty SC and tare the weighing balance. Add sample powder & weigh about 2gm. Spread the powder in SC and transfer it to MF & set temp to 450-600°C (Allow it for burning to white ash or nearly white) for 30 min. Bring the temp knob to zero & remove SC Measure 25ml of 40% HCl & pour in SC containing ash. Place the SC in hot plate to boil the ash content in the HCl for 5 min remove the SC from hot plate using a tong and prepare a filter system using an ash less filter paper (Whatman filter paper). Filter the ash solution using ash less filter paper while it is still warm we can observe few particles are trapped on the filter paper. These particles will be counted as acid insoluble ash please bring hot water & wash the SC, then transfer the washing to filter paper for filtration to ensure complete transfer of ash. Add additional hot water to the filter paper washing, to ensure that no residue of HCl is left with the filter paper. Next, place a SC & lid in hot air oven at 110°C for 30 min to dry cool it in desiccators. After cooling, take weight of the dried blank crucible and note the weight. Fold the filter paper carefully to avoid the loss of the filtrate & place it to the blank SC, cover it with lid and place the SC inside the MF at 550°C for 90 min. Take out the SC from MF after cooling. (Check the ash color). Take the weight of SC containing ‘acid insoluble ash’.
% Acid insoluble ash = X-Y / N × 100
Where, X= the weight of crucible dish + ash obtained, Y= the weight of the empty dish, N= weight of crude drug taken.
Determination of Water Soluble Ash: Take empty silica crucible, transfer to muffle furnace, Place the empty SC inside MF, set temp, keep for 15 min to dry properly. After 15 min bring temp setting knob to zero, remove the SC from MF chamber now use a desiccator to cool at R.T. Weigh empty SC and tare the weighing balance. Add sample powder & weigh about 2gm. Spread the powder in SC, transfer it to MF & set temp to 450-600°C (Allow it to burn to white ash or nearly white) for 30 min. Bring the temp knob to zero & remove SC Measure 25ml of water & pour in SC containing ash. Place the SC in hot plate to boil the ash content in water for 5 min; remove the SC from hot plate using a tong and prepare a filter system using ashless filter paper (Whatman filter paper). Filter the ash solution using ashless filter paper while it is still warm we can observe few particles are trapped on the filter paper. These particles will be counted as water-soluble ash please bring hot water & wash the SC, then transfer the washing to filter paper for filtration to ensure complete ash transfer. Next, place a SC & lid in hot air oven at 110°C for 30 min to dry cool it in desiccators. After cooling, take weight of the dried blank crucible and note the weight. Fold the filter paper carefully to avoid the loss of the filtrate & place it to the blank SC, cover it with lid and place the SC inside the MF at 550°C for 90 min. Take out the SC from MF after cooling. (Check the ash colour). Take the weight of SC containing ‘water-soluble ash’ 14.
% Water soluble ash = X-Y / N × 100
Where, X= the weight of crucible dish + ash obtained, Y= the weight of the empty dish, N= weight of crude drug taken.
Determination of Loss on Drying: The air dried crude drug (1 g) placed on a porcelain dish was subjected to a temperature of 105°C for 30 min. The loss in weight is recorded as the moisture lost due to drying 15.
% Loss on drying = X-Y / N × 100
X = The weight of porcelain dish + crude drug obtained, Y= the weight of the empty porcelain dish, N= weight of crude drug taken.
Fluorescence Analysis: The organic molecules usually absorb light over a specific range of wave length, get excited to a high energy level, and many radiate while returning to the original state. Such a phenomenon of re-emission of absorbed light that occurs only when the substance is receiving the exciting rays is known as ‘Fluorescence’. The powdered drug was examined under U.V and ordinary light with different reagents. About 10gm of the powdered drug was taken in a Petri dish and treated with reagents viz., 50% sulphuric acid, 50% potassium hydroxide, 1N Hydrochloric acid, AgNO3, 1N Sodium Hydroxide. These were observed under different wavelengths i.e., visible rays and UV rays (254 nm and 365 nm).
Extractive Values: The extracts obtained by exhausting crude drugs are indicative of approximate measures of their chemical constituents. Taking into consideration the diversity in chemical nature and properties of contents of drugs, various solvents are used for the determination of extractives 16.
Determination of Water Soluble Extractives: Macerated 5gm of the air-dried coarsely leaves powder with 100ml of chloroform water in a closed flask for 24 hrs. Shaking frequently during the first 6 hours and allowed to stand for 18 hrs. Thereafter, it was filtered rapidly, taking precautions against loss of the solvent. Evaporated 25ml of the filtrate to dryness in a tarred bottom flat bottom shallow dish dried at 105°C and weighed. The percentage of water-soluble extractive value was calculated with reference to the air-dried drug.
Determination of Alcohol Soluble Extractives: Macerated 5gm of the air-dried coarsely leaves powder with 100ml of 95% ethanol in a closed flask for 24 hrs. Shaking frequently during the first 6 hours and allowed to stand for 18 hrs. After that, it was filtered rapidly, taking precautions against solvent loss. Evaporated 25ml of the filtrate to dryness in a tarred bottom flat bottom shallow dish dried at 105°C and weighed. The percentage of water-soluble extractive value was calculated concerning the air-dried drug.
Sub-fractions Extraction: 70 grams of powder was weighed and taken for successive extraction using various solvents depending on their polarity, Petroleum ether, benzene, chloroform, acetone, ethanol, and chloroform water. The extracted solvent was filtered with filter paper and evaporated using a rotary evaporator. The extract then dries in a water bath until it becomes viscous and dries in open at room temperature.
FIG. 4: SOXHLET EXTRACTION
FIG. 5: EVAPORATION OF SOLVENT USING ROTARY EVAPORATOR
Phytochemical Screening: The Ethanol, Acetone and Chloroform water leaves extract was analyzed with various tests to find out the phytoconstituents such as alkaloids, carbohydrates, flavonoid, tannins, phenols, glycosides, saponins, protein, and amino acid and organic acids.
Test for Alkaloids: Aqueous HCl (5ml, 1%v/v) was used to dissolve the extract and filter; the filtrates were divided into 2ml of aliquots subsequently the following test was carried out:
Hager’s Test: The extract was treated with Hager’s reagent (saturated picric acid solution). The formation of a yellow-colored precipitate indicates the presence of alkaloids.
Mayer’s Test: The extract was treated with Mayer’s reagent (potassium mercuric iodide solution). The formation of a cream-colored precipitate indicates the presence of alkaloids.
Dragendroff’s Test: The extract was treated with dragendroff’s reagent (potassium bismuth iodide solution). The formation of an orange-brown precipitate indicates the presence of alkaloids.
Wagner’s Test: The extract was treated with Wagner’s reagent (iodine potassium solution). The formation of a reddish-brown precipitate indicates the presence of alkaloids.
Test for Carbohydrates:
Molisch Test: Extract was treated with Molisch reagent (α-naphthol in 95% ethanol), and few drops of sulphuric acid were added through the side of the test tube. The appearance of the violet ring at the junction indicates the presence of carbohydrates.
Benedict’s Test: The extract was treated with benedict’s reagent (copper sulfate + sodium citrate + sodium carbonate in water), and heated for 10 minutes. Red-colored precipitate indicates the presence of sugars.
Fehling’s Test (Test for Reducing Sugars): Extract was dissolved in 1ml of distilled water and filter. 1ml of Fehling’s solution A and B were added to the filtrate and heated in a water bath for a few minutes. The formation of a brick-red precipitate indicates the presence of reducing sugar.
Barfoed’s Test: The extract was dissolved in an equal amount of Barfoed’s reagent it was heated in a water bath for few minutes and the cooled. The formation of red precipitate indicates the presence of monosaccharides.
Test for Flavonoids:
Zinc Test: Heat solution with Zinc dust and hydrochloric acid, pink to red indicates the presence of flavonoids.
Shinoda Test: Extract was treated with few magnesium turnings and concentrated hydrochloric acid dropwise. The formation of pink scarlet, crimson red, or occasionally green to blue indicates flavonoids.
Lead acetate Test: Few ml of extract solution was treated with 1 ml of 10% lead acetate solution in water. Yellow color precipitate indicates the presence of phenols.
Ferric Chloride Test: Few ml of extract was treated with ferric chloride solution in water. Green to black color indicates the presence of flavonoids.
Test for Glycosides:
Bontrager's Test: Powdered drug was mixed with ether, which is filtered, and to the filtrate, caustic soda and aqueous ammonia were added. Red, pink, or violet color produced indicates the presence of anthraquinone glycoside.
Keller-killiani Test: Crude extract was mixed with 2ml of glacial acetic acid containing 1- 2 drops of 2 % FeCl3 solution. To the mixture, add 2ml of concentrated sulphuric acid. A brown ring at the interphase indicates the presence of cardiac glycosides.
Legal Test: Extracts were treated with sodium nitroprusside in pyridine and sodium hydroxide. The formation of pink to blood-red color indicates the presence of cardiac glycosides.
Test for Phenols and Tannins:
- A few ml of extract solution was allowed to react with 1 ml of 10% ferric chloride solution. Green indicates the presence of tannins.
- A few ml of extract solution was allowed to react with 1ml of vanillin solution purplish red colour indicated the presence of tannins.
- A few ml of extract solution was allowed to react with 1ml of diluted iodine solution.
Test for Proteins:
Xanthoprotein Test: Extract solution was treated with 1ml of conc. In sulphuric acid, a white precipitate will be seen then boil it precipitate turns to yellow, add ammonium hydroxide precipitate turns orange; this indicates the presence of of proteins.
Test for Protein Containing Sulphur: Extract solution is treated with 40% Sodium hydroxide adding 10% lead acetate to it, boiled; black or brownish colour indicates the presence of proteins.
Biuret Test: Extract solution is allowed to react with 2% copper sulphate and 1 ml ethanol with the addition of excess of potassium hydroxide pellets, pink colour in ethanolic layer indicates the presence of ethanolic layer.
Test for Amino Acids:
Ninhydrin Test: Extract was treated with ninhydrin reagent and ammonia and heated. The Violet color indicates the presence of proteins.
Test for Cysteine: Extract solution is treated with 40% Sodium hydroxide adding 10% lead acetate to it, boiled; black precipitate indicates the presence of amino acids.
Test for Saponin:
Froth Test: Crude extract was mixed with 5ml distilled water and it was shaken vigorously. The formation of stable foam indicates the presence of saponin.
Test for Organic Acids: Extract solution was allowed to react with calcium chloride and then the following reactions were observed;
- Presence of white precipitate indicates the presence of oxalic acids.
- If on shaking gives white precipitate it indicates the presence of tartaric acids.
- The solution was boiled and cooled if it gives white precipitate it indicates the presence of citric acid.
- Formation on precipitate on addition of absolute alcohol indicates the presence of malic acid 17.
Pharmacological Investigation (Anti-tussive Activity): Guinea pigs weighing (500 - 700 g) will be used for the experiment. They will be housed and given free access to food, drinking water, and standard rodent diet at a temperature of 24 (0.5˚C) for a week 18. IAEC has approved the animal activity with reference no. HPI/2021/60/IAEC/PP-0183.
Experimental Protocol:
Cough-Induced Model: The animals will be randomly grouped in five groups, each with 5 animals (n=5).
Group I: Termed as negative control, will be treated with vehicle (Saline water)
Group II: Termed as positive control will be treated with Ambroxol Hydrochloride (250 mg/kg).
Group III: Termed as low dose of H. cordata extract (100mg/kg) treated group
Group IV: Termed as intermediate dose H. cordata (250mg/kg) treated group.
Group V: Termed as high dose H. cordata (400mg/kg) treated group.
Evaluation of Activity: The animals are placed in a transparent plastic box; the guinea pig is exposed to the aerosol of 15% citric acid or 2.8% of NH4OH 19 in water for 10 min using a nebulizer. Each animal is tested first to obtain the control response. The number of tussive responses is registered.
30 minutes later, the test substance is applied orally, and 30 minutes later, the guinea pig is subjected to aerosol again. The number of coughs during the 5 min is recorded. The standard drug used in this activity is Ambroxol HCl 20 which will be administered to the guinea pigs orally using a gavage.
The above protocol was performed 10 min after exposing animals to aerosols of the following solutions for a period of 5 min 21.
- Normal Saline solution.
- Ambroxol hydrochloride 250 mg/kg.
- Houttuynia cordata extract 100mg/kg.
- Houttuynia cordata extract 250mg/kg.
- Houttuynia cordata extract 400mg/kg.
FIG. 6: ADMINISTRATION OF DRUGS
RESULTS:
Macroscopic Examination: The leaves of the Houttuynia cordata are green, 4–8cm in length, 3–4cm in width, simple, and broad with a long petiole, ovate–cordate lamina with shortly acuminate apex bearing entire margin and are pubescent due to presence of trichomes. The leaves are slightly pungent in taste with a characteristic aromatic odour.
Microscopic Examination:
Powder Microscopy of Leaves: The powder microscopy evaluation of the sample was carried out using reagents like fast green, Ruthenium red and safranin to detect the presence of different constituents. After observing the powder under the microscope, different constituents like Raphides of calcium oxalate crystals, vascular bundles, slender-shaped fibres, trichomes, lignified vessel, vein islet, oil glands, starch grains, cork cells, epidermal cells, glandular trichomes, phloem elements and acicular crystals.
FIG. 7: POWDER MICROSCOPY OF H. CORDATA
FIG. 8: MAGNIFIED FIGURES OF MICROSCOPIC CHARACTERS
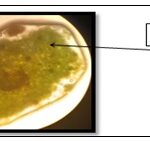
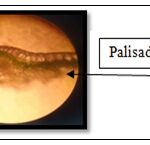
Transverse Section of a Leaf: A fine section of the leaf of H. cordata was mounted onto the microscope and was observed at 10X to identify the different microscopic characters of the leaf.
FIG. 9: TRANSVERSE SECTION OF HOUTTUYNIA CORDATA LEAF
Ash Values: The values are as follows
TABLE 1: ASH VALUES OF THE LEAVES OF HOUTTUYNIA CORDATA THUNB
| Sl. no. | Type of Ash | Percentage (W/W) |
| 1 | Total Ash | 14.5% |
| 2 | Acid Insoluble Ash | 4.5% |
| 3 | Water Soluble Ash | 17.8% |
FIG. 10: ASH VALUE
Loss on Drying: The values are as follows.
TABLE 2: LOSS ON DRYING OF HOUTTUYNIA CORDATA THUNB
| Initial weight of powdered drug and Petridish | Final weight after drying at 105°C for 30 min | Percentage of loss on Drying |
| 32.59g | 32.51g | 0.24% |
Fluorescence Analysis: The powdered drug was examined under U.V and ordinary light with different reagents, the results are found as below.
TABLE 3: FLORESCENCE ANALYSIS OF HOUTTUYNIA CORDATA
| Sl. no. | Powder + Chemical | Fluorescence under daylight | Fluorescence at ʎmax254nm | Fluorescence at ʎmax365nm |
| 1 | Powder as such | Brown | NF | NF |
| 2 | Powder + 1N NaOH in methanol | Dark green | NF | Off white |
| 3 | Powder + 1N NaOH in water | Maroon | NF | Light green |
| 4 | Powder + 1N HCL in methanol | Dark red | NF | Off White |
| 5 | Powder + 1N HCL in water | Yellowish brown | NF | Pale green |
| 6 | Powder + 1N HNO3 in methanol | Brown | NF | NF |
| 7 | Powder + 1N HNO3 in water | Orange | Golden red | Yellowish green |
| 8 | Powder + Iodine (5%) | Orange red | NF | NF |
| 9 | Powder + KOH (50%) | Maroon | Green | Spring green |
| 10 | Powder + NH3 (25%) | Dark maroon | NF | Greenish yellow |
*NF indicating No fluorescence.
FIG. 11: FLUORESCENCE ANALYSIS
Extractive Values: The extractive values of Houttuynia cordata.
TABLE 4: EXTRACTIVE VALUES OF HOUTTUYNIA CORDATA
| Initial weight of powdered drug | Solvent | Percentage |
| 5 g | Water | 36% |
| 5 g | Alcohol | 20.8% |
Yield of Sub-fractions: Successive extraction was carried out Successive Soxhlation using various solvents depending on their polarity and maceration with chloroform water.
TABLE 5: PERCENTAGE YIELD OF VARIOUS SOLVENT EXTRACTS OF HOUTTUYNIA CORDATA THUNB
| Initial amount of powdered drug | Solvent | Percentage yield |
| 70g | Petroleum ether | 10.07% |
| 70g | Benzene | 2.27% |
| 70g | Chloroform | 1.06% |
| 68.8g | Acetone | 3.34% |
| 66g | Ethanol | 6.97% |
| 70g | Aqueous | 26.21% |
Phytochemical Screening: Qualitative phytochemical screening of the extract of the leaves extract of Houttuynia cordata is shown in table.
TABLE 6: PHYTOCHEMICAL SCREENING OF THE EXTRACTS OF HOUTTUYNIA CORDATA THUNB
| Tests | Ethanolic Extract | Acetone Extract | Chloroform water extract |
| Alkaloids | ++ | - | - |
| Carbohydrates | + | ++ | +++ |
| Flavonoids | +++ | +++ | ++ |
| Glycosides | + | ++ | - |
| Tannins | - | - | ++ |
| Amino acids | - | - | +++ |
| Proteins | - | - | - |
| Saponins | + | + | + |
| Organic acids | + | - | +++ |
FIG. 12: PHYTOCHEMICAL SCREENING
Pharmacological Activity:
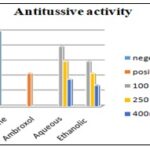
FIG. 13: ANTI-TUSSIVE ACTIVITY ON GUINEA PIGS
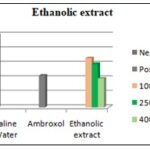
Ethanolic Extract: As shown in the figure, the number of coughs in animals that received 100 mg/kg, 250mg/kg and 400mg/kg dose of Ethanolic extract of Houttuynia cordata was seen to be decreasing gradually without much difference between the doses; this was found to be significantly different from that of the baseline value. It can also be seen that the Ethanolic extract show comparative difference when compared with the standard drug Ambroxol (250 mg/kg). According to the figure it was seen that the high dose showed similar results compared to that of Ambroxol.
FIG 14: COUGH NUMBER OBTAINED IN THE PRESENCE OF LOW DOSE (100 MG/KG), INTERMEDIATE DOSE (250 MG/KG) AND HIGH DOSE (400 MG/KG) OF ETHANOLIC EXTRACTS FROM HOUTTUYNIA CORDATA COMPARED TO THOSE OBTAINED IN THE PRESENCE OF SALINE (BASELINE) AND AMBROXOL
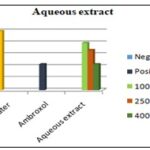
Aqueous Extract: All concentrations of aqueous extract caused a reduction in the number of coughs compared to the baseline value. As seen in Ethanolic extract, so also in aqueous extract of Houttuynia cordata (100 mg/kg, 250 mg/kg and 400 mg/kg), drug given showed decreased in cough number. It can also be seen that the aqueous extract at low doses did not differ much, but at high dose, we can see comparative results when compared with the standard drug Ambroxol (250 mg/kg).
FIG. 15: COUGH NUMBER OBTAINED IN THE PRESENCE OF LOW DOSE (100 MG/KG), INTERMEDIATE DOSE (250 MG/KG) AND HIGH DOSE (400 MG/KG) OF AQUEOUS EXTRACTS FROM HOUTTUYNIA CORDATA COMPARED TO THOSE OBTAINED IN THE PRESENCE OF SALINE (BASELINE) AND AMBROXOL
The present study demonstrated a relatively potent anti-tussive effect for both extracts obtained from Houttuynia cordata. The anti-tussive effects obtained from Ethanolic extract showed significant effect on guinea pigs compared to aqueous plant extract.
FIG. 16: COUGH NUMBER OBTAINED IN THE PRESENCE OF LOW DOSE (100 MG/KG), INTERMEDIATE DOSE (250 MG/KG) AND HIGH DOSE (400 MG/KG) OF AQUEOUS AND ETHANOLIC EXTRACTS FROM HOUTTUYNIA CORDATA COMPARED TO THOSE OBTAINED IN THE PRESENCE OF SALINE (BASELINE) AND AMBROXOL
However, the effect of the Ethanolic extract was significantly greater than that of the same concentration of the aqueous extract. In addition, the anti-tussive effects of both concentrations of extracts in different animals were highly reproducible. As seen in the figure below, we can see the difference between different doses of the drug and the extracts compared to Ambroxol and Saline. Houttuynia cordata were comparable to Ambroxol at the higher concentration used.
DISCUSSION: Coughing is a normal physiological response to the laryngo-tracheo bronchial system irritation caused by mechanical or chemical stimulation. It may be painful and fatiguing and require suppression by anti-tussive drugs.
The Pharmacognostical, Physiochemical and phytochemical study of the plant were first carried out; the plant's macroscopic and microscopic was evaluated by in which we found unique characters present in the powder microscopy such as raphides of calcium oxalate crystals, vascular bundles, slender shaped fibers, trichomes, lignified vessel, vein islet, oil glands, starch grains, cork cells, epidermal cells, glandular trichomes, phloem elements and acicular crystals, the transverse section of the leaf was studied in this many microscopic character was seen namely the palisade cell, upper and lower epidermis, spongy parenchyma, multicellular and glandular trichomes, vascular bundles, stomata, calcium oxalate crystals and starch grains.
Various physiochemical parameters were also carried out like Ash content, extractive values, moisture content and fluorescence; these parameters are usually checked to judge the purity of the plant, the ash value of total ash was 14.5%, acid insoluble 4.5% and water soluble ash 17.8%; the loss on drying was found to be 0.24%, the florescence analysis of the powder was investigated under daylight, 254nm and 365nm using various chemicals in this investigation it was seen that the powder showed difference fluorescence color at different radiations. The extractive value using water and alcohol was evaluated using same amount of powder and solvent it was found that aqueous extract gave 36% while alcohol gave 20.8% of yield. Extractive yield by various solvent gives the idea about the chemical constituents present in the plant; extraction by carried out by Soxhlation, and different percentage yield of extracts depending on the solvent was obtained: Petroleum ether (10.07%), Benzene (2.27%), Chloroform (1.06%), Acetone (3.34%), Ethanol (6.97 %) and Water (26.21%). The phytochemical investigation gave invaluable information about the different phytoconstituents present in the various extracts of same plant collected. In the phytochemical screening, it was seen that different phytoconstituents such as alkaloids, carbo0hydrates, flavonoids, glycosides, tannins, amino acids, protein, Saponins, and organic acids was found and its strength differs in every extract.
In this study, guinea pigs were used, and cough was induced by chemicals like citric acid and ammonium hydroxide; the anti-tussive activity of Houttuynia cordata was investigated in comparison with a standard anti-tussive drug Ambroxol and saline solution. The Ethanolic and Aqueous extract of H. cordata at 100mg/kg, 250 mg/kg and 400 mg/kg was investigated for pharmacological evaluation against the standard Ambroxol (250 mg/kg); in the investigation using aqueous extract, it was seen that the low dose showed a small difference from the baseline but the high showed remarkably decreased in cough number from the baseline, as for the Ethanolic extract it was seen that the extract showed effective results even at a low dose of the drug when compared with the baseline (saline solution). The high dose showed better results than Ambroxol, which concluded that it is more potent and is a potential anti-tussive drug.
CONCLUSION: Herbal drugs are mostly derived from a natural product, and they are safer as compared to modern medicine, but their utility is reduced as modern medicine has considerably benefited, more active than herbal drugs; however, modern medicine has many adverse effects which lead to the withdrawal from their clinical used. The history of drug development shows that many drugs have been derived from inspiration from traditional medicine. In this present study, leaves of Houttuynia cordata were selected based on their traditional use and literature. The plant was powdered, identified, and authenticated, after which the Pharmacognostical parameter, phytochemical analysis like ash content, loss on drying, florescence analysis was performed. The leaves of the plant were powdered and subjected to extraction using different solvents and phytochemical screening of the extracts obtained was also carried out. After which, the main activity was carried out i.e., anti-tussive activity which showed positive results when compared with a standard anti-tussive drug Ambroxol. Houttuynia cordata showed positive results in this investigation. The plant Houttuynia cordata has not been fully investigated the plant may possess many other activities based on the chemical constituents that were found; future studies on the plant can be evaluated for other activities similarly.
ACKNOWLEDGEMENTS: The Authors would like to thank the Himalayan Pharmacy Institute for providing the facilities to carry out this research.
CONFLICTS OF INTEREST: There is no conflict of interest between authors.
Author(S) Contribution: I.W., responsible for selecting the research work and major data collection by trial and error methods. R.D is responsible for the guidance of the work till the end. Both authors contributed to drafting, designing, formatting, and referencing this research article. R.D. communicated with an esteemed scientific journal with a good scientific reputation. Both authors have read and approved the manuscript.
REFERENCES:
- Satoskar RS, Rege NN and Bhandarkar SD: Pharmacology and Pharmacotherapeutics, Popular Prakashan Pvt. Ltd., Mumbai 2011; 1.
- Anne B and Chang: The physiology of cough, paediatric respiratory reviews Department of Respiratory Medicine, Royal Children’s Hospital, Australia 2006; 7: 2–8, 10. DOI: https://doi.org/10.1016/j.prrv.2005.11.009
- McGarvey LP, Heaney LG, Lawson JT, Johnston BT, Scally CM, Ennis M, Shepherd DR and MacMahon J: Evaluation and outcome of patients with chronic non-productive cough using a comprehensive diagnostic protocol. Thorax 1998; 53: 738-743. DOI: http://dx.doi.org/10.1136/thx.53.9.738
- Alyn H: Morice, Epidemiology of Cough Pulmonary Pharmacology & Therapeutics Academic Department of Medicine 2002; 15: 253-259. DOI: https://doi.org/10.1006/pupt.2002.0352
- Birring SS, Fleming T, Matos S, Raj AA, Evans DH and Pavord ID: The Leicester cough monitor: preliminary validation of an automated cough detection system in chronic cough. Eur Respir J 2008; 31(5): 1013–8.DOI: 10.1183/09031936.00057407
- Jaymin B. Morjaria, Rebecca S. Dickinson and Alyn H. Morice: Novel anti-tussive strategies, Division of Cardiovascular and Respiratory Studies, Hull York Medical School, University of Hull, Castle Hill Hospital, Cottingham HU16 5JQ, UK, April 2013 (18):7/8.DOI: 10.1016/j.drudis.2012.11.004
- Usmani OS, Belvisi MG, Patel HJ, Crispino N, Birrell MA and Korbonits M: Theobromine inhibits sensory nerve activation and cough. FASEB J 2005; 19: 231–3. DOI: 10.1096/fj.04-1990fje
- Dicpinigaitis PV and Alva RV: Safety of capsaicin coughs challenge testing. Chest 2005; 128(1): 196–202. DOI: 10.1378/chest.128.1.196
- Manish Kumar, Satyendra K. Prasad and Damiki Laloo: Pharmacognostical and phytochemical standardization of Houttuynia cordata: A potent medicinal herb of North–Eastern India and China, Department of Pharmaceutics, Indian Institute of Technology, Banaras Hindu University, Varanasi 2014; 6: 1. DOI: 10.5530/pj.2014.1.6
- Rathi RS, Somnath Roy and Singh SK: Ethnobotanical notes on, Houttuynia cordata Thunb in Northeastern region of India, National Bureau of plant genetic resources Regional Station Umiam 2014; 4(4): 432-435.
- Kokate CK, Purohit AP and Gokhale SB: Pharmacognosy, Nirali Prakashan, Pune 2012; 6.3-6.25: 1-A.6.
- Ahmad T, Sing SB and Pandey S: Phytochemical Screening and Physicochemical Parameters of Crude Drugs. Rev Int J Pharma Res & Review 2013; 2(12): 53-60.
- Quality Control Methods for Medicinal Plant Materials (WHO), Geneva A.I.T.B.S. Publishers & Distributors (Regd.), Delhi 2002; 28–30.
- Indian Herbal Pharmacopoeia, Revised edition published by Indian Drug Manufactur 2002; 495–6.
- Prasad SK, Sahu AN and Hemalatha S: Cytomorphological and physicochemical evaluations of Cryptocoryne spiralis. J Herbs Spices Med Plants 2012; 18: 304–317.DOI: 10.1080/10496475.2012.710889
- Chase CR and Pratt R: Fluorescence of powdered vegetable drugs with particular reference to development of a system of identification. J Am Pharm Assoc 1949; 38: 324–331.DOI: 10.1002/jps.3030380612
- Tiwari P, Kumar B, Kaur M, Kaur G and Kaur H: Phytochemical screening and extraction. Review. Int Pharma Sci 2011.
- Khandelwal: Practical Pharmacognosy, Nirali Prakashan, 2005; 149-156.
- Hossein Hosseinzadeh: antitussive effect of thymoquinone, a constituent of Nigella sativa Seeds, in Guinea Pigs, Pharmacologyonline 2008, 2: 480-484
- Jayant N. Dhuley: Anti-tussive effect of Adhatodavasica extract on mechanical or chemical stimulation-induced coughing in animals, Journal of Ethnopharmacology 1999; 16: 361–365.DOI: 10.1016/s0378-8741(99)00074-4
- Kee Jae Song: Expectorant and Anti-tussive Effect of Hedera helix and Rhizomacoptidis Extracts Mixture, Yonsei Med J 2015; 56(3): 819-824. DOI: 10.3349/ymj.2015.56.3.819
How to cite this article:
Warjri I and Das R: Pharmacognostical evaluation, phytochemical screening of Houttuynia cordata thunb leaves and investigation of its anti-tussive effect on guinea pigs. Int J Pharmacognosy 2023; 10(7): 419-30. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.10(7).419-30.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
13
419-430
3520 KB
1042
English
IJP
Ireenia Warjri and Rajat Das *
Department of Pharmacognosy, Himalayan Pharmacy Institute, Majhitar, Rangpo, East Sikkim, India.
rajatdas1992@yahoo.com
17 July 2023
27 July 2023
29 July 2023
10.13040/IJPSR.0975-8232.IJP.10(7).419-30
31 July 2023