EXTRACTION AND COMPREHENSIVE PHYTOCHEMICAL PROFILING OF PEDALIUM MUREX
HTML Full TextEXTRACTION AND COMPREHENSIVE PHYTOCHEMICAL PROFILING OF PEDALIUM MUREX
Balbir Singh *, Pankaj Mishra and Pushpendra Kannojia
BIU College of Pharmacy, Bareilly International University, Bareilly, Uttar Pradesh, India.
ABSTRACT: Medicinal plants are an invaluable source of bioactive compounds with therapeutic potential. Pedalium murex, commonly known as “Gokhru,” has been traditionally used in Ayurveda for managing urological disorders, reproductive health, and inflammatory conditions. This study aimed to perform a comprehensive phytochemical investigation, extraction, and quantification of bioactive constituents in the hydro-alcoholic extract of P. murex leaves. Organoleptic, physicochemical, and preliminary phytochemical evaluations confirmed the presence of carbohydrates, proteins, flavonoids, glycosides, terpenoids, and phytosterols, while saponins, tannins, and alkaloids were absent. The extract yield was 11.87% w/w, and HPLC analysis enabled the identification and quantification of flavonoids, with quercetin and rutin detected at retention times of 2.597 ± 0.08 min and 7.621 ± 0.53 min, respectively. Method validation demonstrated excellent linearity, precision, accuracy, and sensitivity, indicating the reliability of the developed RP-HPLC method for routine quality control. These findings validate the traditional use of P. murex and establish a scientific basis for its standardization, providing a foundation for the development of herbal formulations and nutraceutical applications.
Keywords: Pedalium murex, Phytochemical Investigation, HPLC, Quercetin
INTRODUCTION: Medicinal plants have long been recognized as valuable sources of bioactive compounds with therapeutic potential 1. Pedalium murex, commonly known as “Gokhru,” is a widely used medicinal herb in traditional systems like Ayurveda for the treatment of urological disorders, reproductive health issues, and inflammatory conditions 2, 3. The plant is rich in diverse phytochemicals, including alkaloids, flavonoids, saponins, tannins, and phenolic compounds, which contribute to its pharmacological activities such as antioxidant, antimicrobial, anti-inflammatory, and diuretic effects 4-7.
Phytochemical investigation and extraction studies play a crucial role in identifying these bioactive constituents, understanding their chemical nature, and evaluating their therapeutic potential 8. Systematic extraction methods using solvents of varying polarity allow for the isolation of specific classes of compounds, which can then be subjected to qualitative and quantitative analyses 9. Such studies not only validate the traditional medicinal uses of Pedalium murex but also lay the foundation for the development of novel herbal formulations and drug candidates.
MATERIALS AND METHODS:
Collection and Authentication of Plant Material: The Pedalium murex plant was collected from Village Barkhan, Tehsil Nawabganj, District Bareilly, during the month of June 2023. The collected leaves were dried under shade and crushed into a coarse powder using a mechanical grinder. Plant authentication was the process of identifying the plant species. The plant herbarium was submitted to Dr. Alok Srivastav, Associate Professor, Department of Plant Science, MJP Rohilkhand University, Bareilly. (Plant Voucher Specimen No. MJP/RU/Plant Sc/105).
Drying of the Plant Material: The cleaned and washed collected plant materials were shade dried. After drying, the material packed in polythene bags and bags were closed tightly. Whenever required, the plant materials were taken from these stocks, powdered coarsely and used for extraction 10.
Organoleptic Evaluation of Powdered Crude Drugs: The crude drugs are derived from natural sources like plants, animals and minerals. It is important that they should be properly identified and characterized for their physical and chemical characteristics, So that a control on their quality could be enforced. Organoleptic evaluation of drugs is the evaluation on the basis of morphological and sensory profile of drugs. The powdered crude drugs were evaluated for their organoleptic properties, i.e. color, taste and odor 11.
Physicochemical Evaluation of Selected Plant Drugs and Extraction: Physicochemical evaluation of chosen drugs of Pedalium murex was done to establish their authenticity and purity 12.
The quality assessment of Pedalium murex begins with determining foreign organic matter, which involves spreading 25 g of powdered plant material on a glass plate, examining it with a magnifying glass, and removing impurities. Moisture content, measured as loss on drying (LOD), is critical since excessive water promotes microbial growth and chemical deterioration. Approximately 2 g of sample is weighed, placed in a pre-weighed bottle, dried at 105 °C for 30 minutes, cooled in a desiccator, and reweighed until a constant weight is achieved. Total ash, representing inorganic content and purity, is determined by incinerating 2 g of dried material in a pre-weighed crucible at 450 °C until carbon-free. Acid-insoluble and water-soluble ash fractions are further calculated to assess siliceous and soluble mineral content, respectively, ensuring drug quality and compliance with pharmacopoeial standards 13-15.
The dried, coarsely powdered leaves of Pedalium murex were subjected to Soxhlet extraction to isolate bioactive compounds. The process was performed sequentially using solvents of increasing polarity, starting from non-polar petroleum ether to hydro-alcohol (90%), ensuring complete extraction of phytoconstituents from the plant matrix. Initially, defatting was conducted with petroleum ether at 60–80 °C, followed by filtration and solvent recovery under vacuum. The remaining marc was air-dried and subjected to alcohol extraction under similar conditions. After filtration, the hydro-alcoholic filtrate was concentrated under vacuum to obtain the crude extract. The weight of the extract was recorded, and the percentage yield was calculated (w/w) 16. This method ensures efficient extraction while preserving thermolabile constituents and maintaining reproducibility for further phytochemical and analytical studies.
Qualitative Phytochemical Investigation: Preliminary phytochemical screenings of extract were done as per standard procedure. Small quantities of investigating fractions were dissolved in their parent solvent and were subjected for analysis using specific reagents and the observations were tabulated 17, 18.
TABLE 1: QUALITATIVE PHYTOCHEMICAL OF PEDALIUM MUREX EXTRACT 17, 18
| Class of Compound | Test Name | Procedure / Observation | Indication |
| Carbohydrates | Molisch's test | Purple ring at the junction when α-naphthol is added | Presence of carbohydrates |
| Fehling's test | Heating with Fehling's solution gives brick-red precipitate | Presence of reducing sugars | |
| Glycosides | Baljet test | Reaction with sodium picrate | Yellow to orange color indicates cardiac glycosides |
| Liebermann-Burchard test | Add acetic anhydride + conc. H2SO4, shake, allow to stand | Bluish green lower layer confirms sterols | |
| Alkaloids | Dragendorff's test | Filtrate reacted with potassium bismuth iodide | Orange-brown precipitate indicates alkaloids |
| Mayer's test | Filtrate reacted with potassium mercuric iodide | Cream precipitate confirms alkaloids | |
| Hager's test | Filtrate reacted with saturated picric acid solution | Yellow precipitate indicates alkaloids | |
| Terpenes / Sterols | Salkowski test | Shake with conc. H2SO4 | Golden yellow lower layer indicates terpenes |
| Liebermann-Burchard test | Acetic anhydride + 1 ml conc. H2SO4 | Red color confirms terpenes | |
| Saponins | Foam test | Shake solution vigorously | Formation of stable foam indicates saponins |
| Flavonoids | Shinoda test | Add 5 ml 95% ethanol + few drops conc. HCl + 0.5 g magnesium | Pink color indicates flavonoids |
| Zn/HCl reducing test | Zinc dust + few drops HCl | Magenta red color indicates flavonoids | |
| Tannins | Ferric chloride test | Add ferric chloride solution | Dark color indicates tannins |
| Gelatin test | Treat with gelatin solution | White precipitate confirms tannins | |
| Proteins | Biuret test | Treat with NaOH + dilute CuSO4 | Blue color indicates proteins |
Identification and Quantitative HPLC Analysis of Flavonoids (Quercetin or Rutin) Present in Pedalium murex Extract: The quantification of rutin and quercetin in Pedalium murex extracts was performed using an RP-HPLC-PDA method on a Shimadzu LC-20AD system equipped with a CT0-20A controller, CBM-20A communication bus module, column oven, Rheodyne 7725 injection valve (20 μL loop), and SPD-M20A photodiode array detector, with data acquisition via Lab Solution version 7.1 software. Stock solutions of rutin and quercetin (1 mg/mL) were prepared in methanol and diluted to obtain calibration standards of 20–120 μg/mL. Sample solutions were prepared by dissolving 100 mg of hydro-alcoholic extract in 1 mL methanol, sonicated for 15 min, and filtered. Chromatography was performed on a C18 column (4.6 × 250 mm) using acetonitrile:methanol (80:20, v/v) as the mobile phase, at 1 mL/min flow, 20 μL injection volume, ambient column temperature, and 254 nm detection, with a total run time of 50 min, yielding retention times of 4.65 min for rutin and 8.63 min for quercetin 19-20.
Method Validation: After optimizing the analytical conditions, linearity, precision, accuracy, the limit of quantification and limit of detection were evaluated according to the ICH Q2 (R1) guidelines 21. All the measurements were performed in triplicates.
Calibration Curve and Linearity: To establish the linearity, a calibration plot was constructed using six different concentrations ranges from 12.5 to 200 μg/mL of standard solution (LSS). The linearity of standard solutions was prepared by plotting peak area against standard concentrations. The acceptance criteria for the linearity should be R2 > 0.995 22.
Precision: A stock solution comprising quercetin and rutin was prepared in methanol and injected the repeated samples at six replicates. The % RSD was calculated from the resultant peak areas or retention times using the proposed method to evaluate instrument precision. To study the intraday precision, six samples at three different known concentrations of quercetin and βrutin were analysed on the same day.
Accuracy: In an analytical validation, accuracy defines as the closeness of the agreement between the true value and a measured value. When the true value is identical to the measured value, then the method is highly accurate. Accuracy was performed by adding three different concentration of standard solution i.e., 80, 100 and 120 % in a pre-analysed sample then the spiked samples were analysed by the developed HPLC method in triplicate 24.
Limit of Detection: Limit of detection (LOD) is the measurement of the lowest amount of analyte detected in a sample, which not necessarily quantified.
LOD is determined from the signal/noise ratio and the acceptable limit of S/N ratio for the detection limit is 3:1. LOD was measured by using the formula 25:
LOD = 3.3 × σ / S
Where, σ (sigma) = Standard deviation of the response; S = Slope of the calibration curve
Limit of Quantitation (LOQ): The Limit of Quantitation (LOQ) is the lowest concentration of an analyte that can be quantitatively determined with acceptable precision and accuracy. The permissible limit of S/N ratio for the quantification limit is 10:1. LOQ was determined by using the formula 25:
LOQ = 10 × σ / S
Where, σ (sigma) = Standard deviation of the response; S = Slope of the calibration curve
RESULTS AND DISCUSSION:
Macroscopy Evaluation: The fresh leaves of Pedalium murex were collected and different organoleptic features likecolor, odour, taste, size, shape, type were observed. These parameters are considered useful in the qualitative control of the crude drug. Leaves are simple, opposite, ovate or oblong-obovate, 1-4.5 cm long, irregularly and coarsely crenate-serrate.
FIG. 1: MORPHOLOGICAL STUDY OF PEDALIUM MUREX LEAVES
Organoleptic and Physiochemical Evaluation: The leaves of the Pedalium murex were collected and authenticated. The materials were shade dried and powdered to sieve 80 sizes. To meet the quality standards, the dried powdered materials were subjected to various quality control parameters. They were first evaluated by organoleptic characterization using parameters like appearance, color, taste, odour and texture, followed by physicochemical evaluation. The foreign organic matter was found to be 0.45±0.032%. To determine the moisture contents in the drugs, loss on drying was determined and it was found to be 8.11±0.98% w/w. Further, total ash (7.56±1.98 %w/w for Pedalium murex); acid insoluble ash (1.76±0.032% w/w for Pedalium murex) and water soluble ash (0.99± 0.001% w/w for Pedalium murex) confirm the quality of herbs selected for the study.
TABLE 2: ORGANOLEPTIC EVALUATION OF SELECTED DRUGS IN POWDER FORM OF P. MUREX
| S. no. | Characteristics | P. murex leaves |
| 1 | Colour | Light green or green |
| 2 | Taste | Slightly bitter |
| 3 | Odour | Aromatic |
TABLE 3: PHYSICOCHEMICAL EVALUATION OF P. MUREX LEAVES POWDER
| S. no. | Parameter | Values: % w/w |
| 1 | Foreign organic matter | 0.45±0.032 |
| 2 | Moisture Content | 8.11±0.98 |
| 3 | Total ash | 7.56±1.98 |
| 4 | Acid insoluble ash | 1.76±0.032 |
| 5 | Water soluble ash | 1.94 0.09 |
N=3
Extraction of the Selected Plant Materials: Dried powdered materials of Pedalium murex (100 g) were soxhlated using hydro-alcohol (90% alcohol). The solvent was recovered using recovery unit and traces of solvent were removed under vacuum and percentage yield of extracts were calculated. w/w calculated. Yield of Pedalium murex, leaves with extracts 11.87 was found.
Preliminary Phytochemical Screening of Fractions: The plant extract were subjected to preliminary phytochemical screening for the detection of various plant constituents. Qualitative evaluation of bioactive fractions following to preliminary phytochemical screening extract of revealed that, hydroalocoholic extract of Pedalium murex, contains carbohydrates, protein & amino acid, flavonoids, terpenoids and phytosterols.
TABLE 4: RESULTS OF QUALITATIVE PHYTOCHEMICAL SCREENING OF P. MUREX
| S. no. | Phytochemical | Presence (+)/absence (-) |
| 1 | Flavonoids | + |
| 2 | Glycosides | + |
| 3 | Phytosterols | + |
| 4 | Alkaloids | - |
| 5 | Terpenoids | + |
| 6 | Saponins | - |
| 7 | Tannins | - |
| 8 | Carbohydrate | + |
| 9 | Protein & Amino acid | + |
HPLC Analysis and Quantification of Fractions: HPLC method was also developed to assess quality and standard for Pedalium murex. The solution of plant extracts sample was chromatographed and obtained the concentration of quercetin of extract sample by using the regression equation. In the presently research, the HPLC process become used to study and quantify the flavonoids (quercetin) component from both selected medicinal plants.
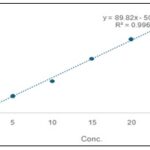
Preparation of the Calibration Curve of the Quercetin: For the preparation of calibration curve, taken standard solutions (quercetin) and injected into the column for 3 times and calculate the mean of peak area of standard drug and plotted the graph against different concentration (5, 10, 15, 20, 25μg/ml) of the drug by using methanol as solvent system. The regression equations were figured out on the basis of the curve. The calibration curve of the standard solution was used for comparing the peak of extracts sample of both plants.
FIG. 2: CALIBRATION CURVE OF THE QUERCETIN
The calibration curve of flavonoids was observed linear relationship over the range of 5, 10, 15, 20, 25μg/ml (R2=0.9967) concentration of standard. The linear regression equation for the curve was Y = 89.827X - 50.765, where y is the ratio of peak area of flavonoids and x is the flavonoid concentration (μg/ml).
Identification of Quercetin in Extract of the Plants: The qualitative and quantitative study of quercetin of selected extracts was performed by the HPLC method. The HPLC chromatogram of hydro-alcoholic extracts of leaves Pedalium murex revealed their highest peaks area for the quercetin as secondary metabolites component. The resulting HPLC chromatogram of standard quercetin and plant extract sample were recorded. The qualitative analysis of phytochemicals was supportive of the quantitative analysis of its. The ongoing study confirmed the presence of flavonoids (quercetin) in medicinal plant as secondary metabolites.
TABLE 5: CHARACTERISTICS OF THE ANALYTICAL METHOD DERIVED FROM THE STANDARD CALIBRATION CURVE
| Compound | Linearity range (μg/ml) | Correlation co-efficient | Slope | Intercept |
| Quercetin | 5-25 | 0.9967 | 89.827 | 50.765 |
FIG. 3: CHROMATOGRAM OF HYDRO-ALCOHOLIC LEAVES EXTRACTS OF P. MUREX
Chromatographic analysis of hydro-alcoholic leaves extract of plants performed by using acetonitrile and methanol 80:20 (V/V) solutions as mobile phase and used a flow rate of 1ml per min and absorbance at 255 nm. It gave good separation of quercetin at RT 2.50min.
A significant amount of quercetin was detected in extract of P. murex. It is reported that flavonoid has good pharmacological, antioxidant and nutraceutical activities. Antioxidant properties of flavonoids are well known to be responsible for human health benefits.
Method Validation: The developed HPLC method was validated for the quantitative estimation of quercetin and rutin in the P. murex extract, and the results are summarized in Table 6. The retention times (Rt) of quercetin and rutin were found to be 2.597 ± 0.08 min and 7.621 ± 0.53 min, respectively, indicating good separation of the compounds within a short analysis time.
The method exhibited excellent linearity over the concentration range of 12.5–200 μg/mL for both compounds, with correlation coefficients (R²) of 0.9998 for quercetin and 0.9997 for rutin, demonstrating a strong linear relationship between peak area and concentration. The sensitivity of the method was evaluated through the limit of detection (LOD) and limit of quantification (LOQ). Quercetin showed an LOD of 0.74 μg/mL and an LOQ of 2.11 μg/mL, whereas rutin had slightly higher LOD and LOQ values of 1.86 μg/mL and 7.87 μg/mL, respectively. These low values indicate that the method is highly sensitive and suitable for detecting even small amounts of the flavonoids in the extract. Overall, the results demonstrate that the HPLC method is precise, accurate, and reproducible for simultaneous estimation of quercetin and rutin, making it a reliable analytical tool for quality control and standardization of P. murex extracts and their phytosome formulations.
TABLE 6: VALIDATION DATA OF QUERCETIN AND RUTIN
| Compound | Concentration Range (μg/mL) | Rt (min) | Regression Equation | R² | LOD (μg/mL) | LOQ (μg/mL) |
| Quercetin | 12.5 – 200 | 2.597 ± 0.08 | y = 5432.1x –365.23 | 0.9998 | 0.74 | 2.11 |
| Rutin | 12.5 – 200 | 7.621 ± 0.53 | y = 5387.9x − 9212.1 | 0.9997 | 1.86 | 7.87 |
Data are represented as mean ± SD (n=6)
Accuracy Studies of Quercetin and Rutin: The accuracy of the developed HPLC method was evaluated by recovery studies at three different concentration levels (80, 100, and 120 μg/mg) for quercetin and rutin, and the results are summarized in Table 7. For quercetin, the recorded amounts ranged from 0.787 ± 0.002 to 1.564 ± 0.005 μg/mg, resulting in percentage recoveries of 99.87 ± 2.94% to 100.44 ± 6.65% with coefficient of variation (% CV) between 1.55 and 1.76. Similarly, rutin showed recorded amounts from 0.842 ± 0.001 to 1.433 ± 0.006 μg/mg, corresponding to recoveries of 99.24 ± 5.87% to 101.31 ± 3.67% with % CV values ranging from 1.24 to 1.43.
These results indicate that the method provides excellent accuracy and precision across the tested concentration range, as the percentage recoveries are close to 100% and the % CV values are low (<2%), demonstrating minimal variability. This confirms that the HPLC method can reliably quantify quercetin and rutin in P. murex extracts and formulations, ensuring reproducibility and suitability for routine quality control and standardization studies. The high accuracy further supports the method’s applicability for precise estimation of these bioactive flavonoids in both raw plant material and prepared phytosome systems.
TABLE 7: ACCURACY STUDIES
| Compound | Amount Added (μg/mg) | Recorded Amount (μg/mg) | % Recovery | % CV |
| Quercetin | 80 | 1.054 ± 0.005 | 100.11 ± 5.76 | 1.55 |
| 100 | 0.787 ± 0.002 | 99.87 ± 2.94 | 1.59 | |
| 120 | 1.564 ± 0.005 | 100.44 ± 6.65 | 1.76 | |
| Rutin | 80 | 1.007 ± 0.003 | 100.65 ± 9.98 | 1.29 |
| 100 | 0.842 ± 0.001 | 99.24 ± 5.87 | 1.24 | |
| 120 | 1.433 ± 0.006 | 101.31 ± 3.67 | 1.43 |
Data are represented as mean ± SD (n=3)
Precision Studies of Quercetin and Rutin: The precision of the developed HPLC method was evaluated by determining both intra-day and inter-day variations for quercetin and rutin. As shown in Table 8, quercetin exhibited intra-day content of 1.1896 ± 0.007% w/w with a relative standard deviation (RSD) of 1.65%, while the inter-day content was 1.1583 ± 0.01% w/w with an RSD of 1.23%. Rutin showed intra-day content of 4.342 ± 0.005% w/w (RSD 1.44%) and inter-day content of 4.632 ± 0.06% w/w (RSD 1.21%). The low RSD values (<2%) for both intra-day and inter-day studies indicate excellent repeatability and reproducibility of the method. These results demonstrate that the HPLC method is highly precise, capable of producing consistent and reliable quantification of quercetin and rutin in P. murex extracts and formulations over time. This precision, along with previously established accuracy, linearity, and sensitivity, confirms that the method is suitable for routine quality control and standardization of the bioactive flavonoids in both raw plant material and prepared phytosome systems.
TABLE 8: PRECISION DATA
| Compound | Intra-day Content (% w/w) | Intra-day RSD (%) | Inter-day Content (% w/w) | Inter-day RSD (%) |
| Quercetin | 1.1896 ± 0.007 | 1.65 | 1.1583 ± 0.01 | 1.23 |
| Rutin | 4.342 ± 0.005 | 1.44 | 4.632 ± 0.06 | 1.21 |
Data are represented as mean ± SD (n=6)
CONCLUSION: The present study demonstrates that Pedalium murex leaves contain a diverse range of bioactive phytochemicals, particularly flavonoids, carbohydrates, proteins, terpenoids, glycosides, and phytosterols. Organoleptic and physicochemical evaluations confirmed the quality and authenticity of the plant material.
The hydro-alcoholic extraction yielded a significant quantity of phytoconstituents, and HPLC analysis successfully identified and quantified quercetin and rutin, two pharmacologically important flavonoids. The validated HPLC method exhibited high precision, accuracy, and sensitivity, making it a reliable tool for standardization and quality control of P. murex extracts. Overall, the study provides scientific support for the traditional therapeutic applications of P. murex and offers a framework for future formulation development and pharmacological evaluation.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Shelke TT, Kothai R, Adkar PP, Bhaskar VH, Juvale KC and Kamble BB: Nephroprotective activity of ethanolic extract of dried fruits of Pedalium murex J Cell Tissue Res 2009; 9(1): 1687–1690.
- Fatima Grace X, Suganya K and Shanmuganathan S: Development of Terminalia chebula loaded ethosomal gel for transdermal drug delivery. Asian J Pharm Clin Res 2018; 11(12): 380–383.
- Ogbeba J: Phytochemical and antibacterial property of finger millet (Eleusine coracana) on some selected clinical bacteria. Access Microbiol 2019; 1(1A). doi: 10.1099/acmi.ac2019.po0037 [DOI] [Google Scholar]
- Chaudhary G and Kaushik N: Phytochemical and pharmacological studies in Pedalium murex Phytochemistry Reviews 2017; 16(5): 921-934.
- Bora Biswa Jyoti and Goswami Dipak Kumar: Pharmacognostic And Phyto-Physicochemical Evaluation of Pedalium murex Linn; International Journal of Applied Ayurved Research 2014; 1(5).
- Banji D, Singh J, Banji OJF and Shanthamurthy M: Scrutinizing the aqueous extract of leaves of Pedalium murex for the antiulcer activity in rats. Pakistan Journal of Pharmaceutical Sciences 2010; 23(3): 295-299.
- Giribabu K, Pushpa S, Latha YJP, Ranganayakulu D, Rajasekhar KK and Haribabu R: Available online through Effect of leaves of Pedalium murex against aspirin induced gastric ulcers in rats 2011; 4(3): 882-883.
- Abirami P and Rajendran A: Evaluation of Antidermatophytic Activity of 2015; 4(3): 1871-1881.64.
- Solanki K and Patel NJ: In-vitro antioxidant activity of aqueous fruit extract of Pedalium murex. International Journal of Preclinical and Pharmaceutical Research 2011; 2(1): 26-29.
- Madhu Babu A, Srinivas P, Venkateshwarulu L and Anil Kumar C: Antioxidant activity of Pedalium murex fruits in carbon tetra chloride- induced hepatopathy in rats. International Journal of Pharma and Bio Sciences 2011; 2(1): 622-628.
- Balamurugan G, Muralidharan P and Polapala S: Aphrodisiac activity and curative effects of Pedalium murex (L.) against ethanol-induced infertility in male rats. Turkish Journal of Biology 2010; 34(2): 153-163.
- Patel DK, Kumar R, Laloo D, Sairam K and Hemalatha S: Aphrodisiac activity of ethanolic extract of Pedalium murex Linn fruit. Asian Pacific Journal of Tropical Biomedicine 2012; 2(3): 1568-1571.
- Balasubramanian MM, Muralidharan P and Balamurugan G: Anti hyperlipidemic activity of Pedalium murex (linn.) fruits on high fat diet fed rats. International Journal of Pharmacology 2008; 4(4): 310-313.65.
- Ahmed S, Hasan MM, Mahmood ZA, Muhammad C, Hasan M and Mahmood A: Antiurolithiatic plants: Multidimensional pharmacology. Journal of Pharmacognosy and Phytochemistry 2016; 5(2): 04-24.
- Shelke TT, Kothai R and Adkar PP: Nephroprotective activity of ethanolic extract of dried fruits of Pedalium murex Journal of Cell and Tissue Research 2009; 9(1): 1687-1690.
- Madan N V., Sahu MS, Jambhulkar YT, Mamidwar AS, Hingawe NT and Maliye AN: A Review on Euphorbiaceae plants for peptic ulcer. Research Journal of Pharmacognosy and Phytochemistry 2018; 10(4): 336.
- Jaideep Singh, Shivalinge Gowda KP, Shrishailappa Badami, Rahul Akuskar and Nagarjan Tukuru: Scrutinizing the fresh juice of leaves of Pedalium murex for the antiulcer activity in rats. Research J.Pharm and Tech 2009; 2(1): 191-194.
- Roy A and Saraf S: Antioxidant and antiulcer activities of an Ethnomedicine: Alternanthera sessilis. Research Journal of Pharm Tech 2008; 1(2): 75-79.
- Sridhar N, Kiran B, Raghavendra M, Prasad M, Kanthal L and Satyavathi K: Comparative anthelmintic evaluation of Glycosmis mauritiana and Pedalium murex roots; Research J. Pharmacology and Pharmacodynamics 2012; 4(3): 185-187.
- Sultana B and Anwar F: Flavonols (Kaempeferol, quercetin, myricetin) contents of selected fruits, vegetables and medicinal plants. Food Chem 2008; 108: 879–884.
- Ou-yang Z, Caoa X, Weia Zhen Ou-yang Y, Zhanga WWY, Zhao M and Duan J: Pharmacokinetic study of rutin and quercetin in rats after oral administration of total flavones of mulberry leaf extract. Revista Brasileira de Farmacognosia 2013; 23(5): 776-782.
- Zhai KF, Duan H, Shi SX, Liu LL, Cao WG, Gao GZ and Shan LL: Synchronised determination of chlorogenic acid and five flavonoids in mulberry leaves using HPLC with photodiode array detection. Quality Assurance and Safety of Crops and Foods 2018; 10(2): 175-82.
- Li J, Bai Y, Zhang P and He J: Simultaneous determination of 5 flavonoids and 7 saponins for quality control of traditional Chinese medicine preparation xinnaoshutong capsule using HPLC-VWD-ELSD. Journal of Analytical Methods in Chemistry 2017; 3190185.
- International conference of harmonization Q2B, Validation of Analytical Procedures—Methodology, US FDA Federal Register, White Oak, MD, USA, 1997.
- International Conference on Harmonisation of Technical requirements for registration of Pharmaceuticals for Human use ICH Harmonised Tripartite Guideline Validation of Analytical Procedures: Text and Methodology Q2(R1).
How to cite this article:
Singh B, Mishra P and Kannojia P: Extraction and comprehensive phytochemical profiling of Pedalium murex. Int J Pharmacognosy 2026; 13(5): 470-77. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(5).470-77.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
11
470-477
720 KB
9
English
IJP
Balbir Singh *, Pankaj Mishra and Pushpendra Kannojia
BIU College of Pharmacy, Bareilly International University, Bareilly, Uttar Pradesh, India.
rkpharma3791@gmail.com
16 April 2026
06 May 2026
12 May 2026
10.13040/IJPSR.0975-8232.IJP.13(5).470-77
15 May 2026