BIOSYNTHESIS AND STRUCTURAL CHARACTERIZATION OF SILVER AND GOLD NANOPARTICLES USING AQUEOUS STEM EXTRACT OF ALHAGI MAURORUM
HTML Full TextBIOSYNTHESIS AND STRUCTURAL CHARACTERIZATION OF SILVER AND GOLD NANOPARTICLES USING AQUEOUS STEM EXTRACT OF ALHAGI MAURORUM
Ashish Malik * 1, Preeti Jain 2, K. D. Sehrawat 3 and Anita Rani Sehrawat 1
Department of Botany 1, Department of Biochemistry 2, Faculty of Life Science, M. D. University, Rohtak - 124001, Haryana, India.
Chaudhary Charan Singh Haryana Agricultural University 3, Hisar - 125004, Haryana, India.
ABSTRACT: Green synthesis is a cheaper, clean, nontoxic route for the synthesis of large scale nanoparticles without the use of high energy, pressure, temperature, and toxic chemicals. With the help of microbial enzyme and plant extract nanoparticles are easily synthesized by utilizing their antioxidant and reducing properties. In the present study, biosynthesis of silver and gold nanoparticles by utilizing Alhagi maurorum stem aqueous extract reducing capabilities was successfully done. Nanoparticles synthesized were characterized with the help of UV - VIS spectroscopy, Transmission Electron Microscopy (TEM) and Fourier Transform Infra-Red spectroscopy (FTIR). UV - VIS spectroscopic analysis showed absorption maxima at 450 nm and 540 nm for silver (AgNP) and gold nanoparticles (AuNP) respectively. TEM study revealed that the silver and gold nanoparticles synthesized were in the range of 26-40 nm and possesses a crystalline structure having a nearly spherical shape. FTIR spectra of both AgNP and AuNP also confirmed the synthesis of nanoparticles and involvement of various reducing agent of plant extract. No change in the intensity of the AgNP and AuNP has been noticed even after 120 days of storage at room temperature. Thus, the synthesis of silver and gold nanoparticles with the help of Alhagi maurorum stem aqueous extract was reducing capabilities seems to be a promising approach for utilizing weed as a source for larger scale production of nanoparticles.
| Keywords: |
Alhagi maurorum, Fourier Transform Infra-Red (FTIR), Gold nanoparticles (AuNP), Silver nanoparticles (AgNP), Transmission Electron Microscopy (TEM)
INTRODUCTION: Particles with spatial dimension less than 100 nm are termed as nanoparticles. They are exhibiting unique and special characteristics which made them be a widely applicable agent in almost all areas including medicines, pharmaceuticals, textile, and food industry 1.
This implies a large scale synthesis of nanoparticles at a faster speed, various physical and chemical synthetic methods have been employed so far, but biological reduction method proves to be an eco - friendly, biocompatible and cost-effective approach in present scenario 2. Biosynthetic methods using micro-organisms (bacteria, fungi, algae) and plants are used as a viable alternative for the synthesis of nanoparticles. Plant parts derived synthesis of nanoparticles seems to be an easier and convenient approach over other biosynthetic methods as the plant cells not only produce chemicals (reducing / oxidizing agent) used for the reduction of ions to nanoparticles but also prevent the formation of aggregates 3.
Thus, the use of plant extract for nanoparticles synthesis is a rapid, cost-effective, eco-friendly and single step method. Among various metal nanoparticles synthesized AgNP and AuNP have been recognized as the one which has wide applicability in almost all fields. Silver nanoparticles applicability has been increased due to its effective and broad antimicrobial property against a large number of pathogens and thus widely applied as a topical ointment to prevent infection against burn and open wounds 4. Along with this, gold nanoparticles are also significant for their wide use in catalysis, optics, optical electronics, microelectronics, bio-diagnostics, imaging, biological and chemical sensing techniques 5. Many reports have been found on the biogenesis of silver nanoparticles using plant extract of various plants as - Azadirachta indica, Argemone mexicana, Medicago sativa, Mangifera indica, Aloe vera, Emblica officinalis, Euphorbia hirta, Rosa hybrid, etc. 6-11
Alhagi maurorum (Syn. Alhagi camelorum, Alhagi pseudalhagi) had antirheumatic, analgesic, antiasthmatic, antipyretics, diuretic, anti-inflammatory, antiulcer activities which were attributed to its plant parts (leaves, stem, flowers, and roots) 12, 13. On consideration of the high medicinal value of the plant, Alhagi maurorum stem aqueous extract was prepared and used for synthesizing AgNP and AuNP. This seems to be a newer novel green chemistry based approach for synthesizing biocompatible, stable, cost-effective and environment-friendly silver and gold nanoparticles as no such efforts have been done previously for this plant.
MATERIALS AND METHODS:
Chemicals: Silver nitrate (AgNO3) and chloroauric acid (HAuCl4) was purchased from Sigma - Aldrich, Banglore, India. All solutions were prepared in double distilled water, and a replica of the experiment has been done.
Preparation of Stem Aqueous Extract: 10 gm of the fresh stem of Alhagi maurorum was washed thoroughly under tap water and surface sterilized with 0.1% mercuric chloride solution by dipping them for 10 min. Shade-dried them for 2 days at room temperature and was grounded into fine powder by surface sterilized pestle and motor. 1.0 g of this powder was suspended in 100 ml of distilled water and boiled for 15 min before finally decanting it. The extract thus formed was filtered through a cheese cloth and the filtrate was stored at 4 °C which was used further as aqueous stem extract for all the experiments.
Biosynthesis of Silver and Gold Nanoparticles: Silver nanoparticles were synthesized by adding 12.0 ml of freshly prepared stem aqueous extract to 88.0 ml of 0.1 mM AgNO3 solution. This reaction mixture was allowed to react at room temperature while the change in the color intensity was noted after a regular interval of 10 min and up to stabilization of color. Similarly, gold nanoparticles were synthesized by the reaction mixture containing 5.0 ml of freshly prepared stem aqueous extract and 25.0 ml of 0.5 mM HAuCl4 solution. The reaction mixture was allowed to react at room temperature by putting them it in an orbital shaker at 35 °C for 15 min. The reduction of Au3+ ions to Au nanoparticles was monitored by observing stable color intensity.
Characterization of AgNP and AuNP: Biosynthesized silver and gold nanoparticles from stem extract after reduction of Ag+ and Au3+ ions has been analyzed by UV - VIS spectroscopy, TEM and FTIR. UV - VIS Spectroscopic analysis was carried out on SPECTRO 20DPLUS spectrophotometer. The measurements were carried out as a function of reaction time at room temperature. The reduction of silver and gold ions to elemental nanoparticles was monitored by measuring the absorbance of the reaction mixture in a range of wavelength from 300 to 600 nm to find the absorbance peak at different time intervals. The TEM images of synthesized AgNP and AuNP were obtained for size and shape determination using Transmission Electron Microscopy (TEM, MorgagniM 268 D, FEI Company, Eindhoven) at Department of Anatomy, All India Institute of Medical Sciences, New Delhi. To identify the bio-molecules present during the synthesis of silver and gold nanoparticles, FTIR analysis was done through Fourier Transmission Infra Red spectroscopy with the help of platinum ATR - IR FTIR Alpha, Bruker, Germany instrument at Department of Genetics, M. D. University, Rohtak. The FTIR was recorded in the range of 400 - 4000 cm-1. The various modes of vibrations were considered and different functional groups involved in the synthesis of silver and gold nanoparticles identified.
Stability Studies of AgNP and AuNP: Storage stability of synthesized silver gold nanoparticles has been evaluated by measuring the decrease in absorption intensity and absorption maxima of the reaction mixture after an interval of 10 days up to 120 days.
RESULT AND DISCUSSION: Silver nanoparticles possess a characteristic pale yellow-brown color that is attributed to their excitation of surface plasmon vibrations. Gradual change in the color intensity of the reaction mixture from initial white color to pale yellow and finally to dark radish brown after 1 h confirmed the formation of silver nanoparticles as shown in Fig. 1a. Only the plant extract having the reducing entities can have the potential of conversion of silver ions into elemental silver 14 and Alhagi maurorum stem extract possesses the same. It seems that among reducing compounds reducing sugars, antioxidants and other compounds as caffeine, eugenols, thiols, etc. play a significant role in bringing reduction process and silver nanoparticles synthesis 15. The reaction mixture of aqueous stem extract and chloroauric acid changed into ruby red color from an initial yellow color within 20 min Fig. 1b. The change was not observed in control sample (without gold salt) postulating the involvement of biomolecules present in the stem extract in synthesizing gold nanoparticles via a simple, non-toxic route and in agreement with previous reports 16.
FIG. 1: A CHANGE IN THE COLOR OF REACTION MIXTURE AFTER SYNTHESIS OF AgNP (A) AND AuNP (B) BY ADDING STEM AQUEOUS EXTRACT OF ALHAGI MAURORUM TO AgNO3 AND HAuCl4 RESPECTIVELY
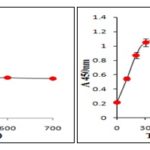
UV - VIS absorption spectrum of synthesized silver nanoparticles shows that the plasmon bond is very broad which indicates the presence of various components in stem extract, but a single peak was observed at 450 nm when absorption at different wavelength had been studied at different time intervals Fig. 2a. In SPR band presence of single peak represented isotropic, spherical shaped nanoparticles 17. With an increase in a number of peaks, anisotropic nature also increases and even after completion of 1 h no considerable change in peak position was noticed thus confirming that once the nanoparticles have been synthesized, there was no effect of time on the size and shape of nanoparticles throughout the course of the reaction Fig. 2b.
FIG. 2: ABSORPTION SPECTRA OF SYNTHESIZED AgNP AT DIFFERENT WAVELENGTH (A) AND AT DIFFERENT TIME INTERVALS (B)
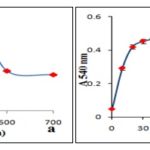
Synthesis of silver nanoparticles from Alhagi maurorum stem extract occurs at a faster speed of 30 minutes of reaction time as compared to other plant-mediated synthesis which at least takes 12 h for completion of reaction 18, 19. Thus, time consumed in the reduction process was several folds lesser than other biosynthetic methods which are attributed to the strong antioxidant capacity of this plant. For gold nanoparticles, a single peak was observed at 540 nm when absorption at different wavelength had been studied at different time intervals which confirmed the synthesis of AuNPs Fig. 3a. Initially, there was no significant peak at 2 min, but after 5 min the building of peak at 540 nm marked the initiation of synthesis of AuNPs Fig. 3b. The subsequent rise in the peak with a maximum at 20 min supported that the reported route of AuNPs synthesis is novel as well as rapid as compared to other plant extract wherein the synthesis was reported to be completed in 2 h 20. The rapidity in the biosynthesis of AuNPs than other biosynthetic methods is attributed to the strong antioxidant capacity of this plant.
FIG. 3: ABSORPTION SPECTRA OF SYNTHESIZED AuNP AT DIFFERENT WAVELENGTH (A) AND AT DIFFERENT TIME INTERVALS (B)
Presence of spherical shaped silver nanoparticles has been confirmed by analyzing the prepared silver nanoparticles with the help of TEM Fig. 4a. TEM study revealed that the spherical shaped silver nanoparticles were synthesized in size range of 16 - 30 nm and possesses crystalline structure. The TEM image in Fig. 4b confirmed the formation of stable, small, spherical shaped gold nanoparticles of size range 12 - 24 nm. It is evident from the TEM study that gold nanoparticles coalesced as nano-clusters, spherical shaped and had a crystalline structure.
FIG. 4: TEM IMAGES OF BIOSYNTHESIZED AgNP (A) AND AuNP (B) AT A MAGNIFICATION SCALE OF 100 nm
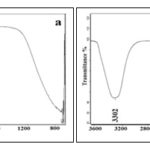
FTIR measurements of synthesized silver nanoparticles were carried out to identify the possible biomolecules present in it. FTIR spectra in Fig. 5a showed variable peaks between 2922-3329 cm−1 for C-H stretching vibrations. The IR spectra for nanoparticles revealed the presence of characteristic bands for Ar-C=C-H stretching between 1509-1637 cm−1 and presence of strong bands especially between 500-800 cm−1 which confirmed the structural and conformational stabilization of silver nanoparticles 21, 22. FTIR absorption spectra of gold nanoparticles showed the presence of characteristic bands for Ar-C=C-H stretching between 1607-1509 cm−1, bending -CH2 and CH3 asymmetrical and symmetrical between 1462-1363 cm−1 and -C-C-O-C- stretching at 1247, 1183 cm−1. The band intensities in different regions of the spectrum are shown in Fig. 5b. The vibration shift around 1647-1557 cm−1 was suggestive of the involvement of aliphatic and aromatic (C–H) plane deformation and vibrations of methyl, methylene and methoxy groups in the reductive process. The peak located at 3299 cm−1 was attributed to the N-H stretching or the CO stretching vibrations. Reduced vibrations of functional groups and the absence of strong bands especially between 800-1000 cm−1 confirmed the synthesis of stable gold nanoparticles by the stem extract 23.
FIG. 5: FTIR SPECTRA OF BIOSYNTHESIZED SILVER (A) AND GOLD NANOPARTICLES (B)
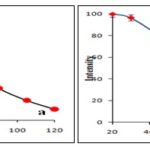
Stability of synthesized silver and gold nanoparticles and efficacy of method has been checked by storing the reaction mixture for 120 days. No precipitation occurs in the reaction mixture of AgNP even after completion of three months and less than 1.76% precipitation occurs in the fourth month. A negligible amount of precipitation occurs in the AuNP solution even after completion of four months. No considerable change in absorption intensity the AgNP and AuNP solution was seen initially but a decrease in absorption intensity after 60 days was noticed as shown in Fig. 6a and 6b respectively. Thus, stable and biocompatible silver and gold nanoparticles have been synthesized by using aqueous stem extract.
FIG. 6: STORAGE STABILITY STUDIES OF BIOSYNTHESIZED AgNP (A) AND AuNP (B)
CONCLUSION: Green synthesis of AgNP and AuNP with the help of Alhagi maurorum stem aqueous extract is reducing capabilities seemed to be a promising approach for utilizing weed as a source for nanobiotechnology. Visual characterization by a change in color of the reaction mixture with broad and stable absorption spectra at 450 and 540 nm confirmed biosynthesis of these AgNP and AuNP respectively. Structural characterization of both nanoparticles by TEM study revealed their small size (10 - 40 nm), spherical shape and crystalline nature. FTIR spectra of these biosynthesized silver and gold nanoparticles postulated the involvement of different biomolecules present in stem extract for the synthesis of these nanoparticles and hence proving green chemistry as a better alternative. Thus, the present study can prove to help synthesize silver and gold nanoparticles at ease via a simple green route for larger scale production with wide applicability.
ACKNOWLEDGEMENT: The author would like to thank Dr. Anita R. Sehrawat, Head of the Department Botany for their encouragement and providing excellent facilities for the above work. We are also grateful to Sophisticated Advanced Instrumentation Facility (SAIF), AIIMS, New Delhi, India for the support and providing the SAIF facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Sobha K, Surendranath K, Meena V, Jwala KT, Swetha N and Latha KSM: Emerging trends in nanobiotechnology. Jrnl of Biotech and Mol Bio Reviews 2010; 5: 01-12.
- Song JY and Kim BS: Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess Biosyst Eng 2009; 32: 79-84.
- Parasharu K, Saxena and Srivastava A: Bioinspired Synthesis of Silver Nanoparticle. Digest Journal of Nanomatrls and Biostrs 2009; 4(1): 159-166.
- Chen X and Schluesener HJ: Nanosilver: A Nanoproduct in Medical Applications. Toxicol Lett 2008; 176: 1-12.
- Chatterjee S, Bandyopadhyay A and Sarkar K: Effect of iron oxide and gold nanoparticles on bacterial growth leading towards biological application. J Nano-biotechnology 2011; 9: 34.
- Chandran SP, Minakshi C, Renu P, Absar A and Murali S: Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotech Progress 2006; 22: 577-83.
- Philip D: Rapid green synthesis of spherical gold nanoparticles using Mangifera indica Spectrochim Acta A Mol Biomol Spectrosc 2010; 77: 807-810.
- Noruzi M, Zare D, Khoshnevisan K and Davoodi D: Rapid green synthesis of gold nanoparticles using Rosa hybrida petal extract at room temperature. Spectrochim Acta A Mol Biomol Spectrosc 2011; 79: 1461-1465.
- Tripathy A, Raichur AM, Chandrasekharan N, Prathana TC and Mukherjee A: Process variables in the biomimetic synthesis of silver nanoparticles by aqueous extract of Azadirachta indica (Neem) leaves. J Nanopart Res 2010; 12: 237-46.
- Khandelwal N, Abhijeet S, Devendra J, Upadhyay MK and Verma HN: Green synthesis of silver nanoparticles using Argimone mexicana leaf extract and evaluation of their antimicrobial activities. Digest Journal of Nanomatrls and Biostrs 2010; 5: 483-489.
- Elumalai EK, Prasad TNVKV, Hemachandran J, Therasa VS, Thirumalai T and David E: Extracellular synthesis of silver nanoparticles using leaves of Euphorbia hirta and their antibacterial activities. J Pharm Sci Res 2010; 2(9): 549-54.
- Nasir E and Ali SI: The Flora of West Pakistan, Department of Botany, University of Karachi 1973; 319.
- Laghari AH, Memon S, Nelofar A, Khan KM, Yasmin A, Syed MN and Aman A: A new flavanenol with urease-inhibition activity isolated from roots of manna plant camelthorn (Alhagi maurorum). J Mol Struct 2010; 965: 65–67
- Sankar KS, Iniyan E, Kumar S and Venugopal S: A green protocol for room temperature synthesis of silver nanoparticles in seconds. Current Sci 2009; 97: 1055 -59.
- Parashar V, Parashar R, Sharma B and Pandey AC: Parthenium leaf extract mediated synthesis of silver nanoparticles: a novel approach towards weed utilization. Dig Jrnl of Nanomatrls and Biostrctrs 2009; 4: 45-50.
- Singh AK, Talat M, Singh DP and Srivastava ON: Biosynthesis of gold and silver nanoparticles by natural precursor clove and their functionalization with amine group. J Nanopart Res 2010; 12: 1667-75.
- Wiley BJ, Sun Y and Xia Y: Synthesis of silver nanostructures with controlled shapes and properties. Accounts of Chemical Res 2007; 40: 1067-1076.
- Ramteke T, Chakrabarti T, Sarangi BK and Pandey RM: Synthesis of silver nanoparticles from the aqueous extract of leaves of Ocimum sanctum for enhanced antibacterial activity. Journal of Chem 2012.
- Leela A and Vivekanandan M: Tapping the unexploited plant resources for the synthesis of silver nanoparticles. African Journal of Biotechn 2008; 7: 3162-3165.
- Andeani JK, Kazemi H, Mohsenzadeh S and Safavi A: Biosynthesis of gold nanoparticles using dried flowers extract of Achillea wilhelmsii Dig Journal Nanomater Bios 2011; 6: 1011-17.
- Sharma VK, Yngard RA and Lin Y: Silver nanoparticles: green synthesis and their antimicrobial activities. Adv in Colloid and Interface Sc 2009; 145(1-2): 83-96.
- Sharma TK, Chopra A, Sapra M, Kumawat D, Pati, SD, Pathania R and Navani NK: Green Synthesis and Antimicrobial Potential of Silver Nanoparticles. Int Journal of Green Nanotech 2012; 4(1): 1-16.
- Kumar V and Yadav SK: Synthesis of Stable, Poly shaped Silver, and Gold Nanoparticles Using Leaf Extract of Lonicera japonica Int Journal of Green Nanotech 2011; 3(4): 281-91
How to cite this article:
Malik A, Jain P, Sehrawat KD and Sehrawat AR: Biosynthesis and structural characterization of silver and gold nanoparticles using aqueous stem extract of Alhagi maurorum. Int J Pharmacognosy 2017; 4(10): 332-37. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(10).332-37.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
332-337
565
1816
English
IJP
A. Malik *, P. Jain, K. D. Sehrawat and A. R. Sehrawat
Department of Botany, Faculty of Life Science, M. D. University, Rohtak, Haryana, India.
anitarsehrawatbotany@gmail.com
25 May 2017
20 August 2017
17 September 2017
10.13040/IJPSR.0975-8232.IJP.4(10).332-37
01 October 2017