TRENDS OF TRADITIONAL MEDICINE RESEARCH AND DRUG DISCOVERY: A REVIEW
HTML Full TextTRENDS OF TRADITIONAL MEDICINE RESEARCH AND DRUG DISCOVERY: A REVIEW
Mebrahtom Gebrelibanos Hiben
Department of Pharmacognosy, School of Pharmacy, College of Health Sciences, Mekelle University, Mekelle-Ethiopia.
ABSTRACT: This review highlights trends in traditional medicine and medicinal plant research and has the purpose of inspiring research focusing on accelerating the integration of traditional medicines into mainstream healthcare systems. Comprehensive literature resources accessible via Google’s search engine were systematically reviewed and described. Traditional medicines in general and traditional African medicine in particular are described. The historical contribution of medicinal plant research to the start of the entire enterprise of drug discovery and development as well as the pharmaceutical industry is highlighted. The decline of traditional medicine during the 19th and 20th centuries and the revival of interest in it towards the end of the 20th century are briefed. The role of conventional new chemical entity-based drug discovery strategies and associated successes and drawbacks related to medicinal plant research plus subsequent advocates foran alternative approach based on traditional medicine inspired by reverse pharmacology by the medicinal plant research community is emphasized. Overall, this review discussed, in addition to applying the well-established and science- and technology-supported drug discovery approach, adopting the concept of traditional medicine-inspired reverse pharmacology may accelerate the integration of traditional medicine into mainstream healthcare.
Keywords: Drug discovery, Healthcare, Reverse pharmacology, Traditional medicine
INTRODUCTION:
Traditional Medicines: Traditional medicine (TM) describes the medical knowledge systems that have developed over centuries within various societies before the era of modern medicine. It is an amorphous concept comprising a range of long-standing and evolving practices that are based on diverse beliefs and theories.
According to the World Health Organization (WHO), traditional medicine refers to “health practices, approaches, knowledge and beliefs incorporating plant, animal and mineral based medicines, spiritual therapies, manual techniques and exercises, applied singularly or in combination to treat, diagnose and prevent illnesses or maintain wellbeing.
It is “the sum total of all knowledge and practices used in the prevention, diagnosis, and elimination of physical, mental or social imbalances and which relies exclusively on practical experience and observation handed down from generation to generation, whether orally or in writing 1-4.
TM is an essential and often undervalued part of healthcare, found worldwide in almost every country, with growing demands for its services. In many cultures, TM functions as an inclusive system of healthcare refined over hundreds or even thousands of years 3-5. Due to the growing availability of Western techno-centered medicine, there is a trend of favoring modern medicine over TM 6. Obviously, Western medicine is today’s most dominant form of health care; however, traditional health care systems remain to play a key role in both rural and urban societies 7 in both developing and developed countries. In high-income countries, most nonconventional medicines are used in a complementary manner, and people who can afford to pay for additional services choose from a broad range of privately delivered treatments. In low- and middle-income countries, many forms of TM are region specific, with the available TM types largely dependent on accessible flora, fauna, and other local resources as well as local traditions 8.
Thus, across the world, TM remains either the mainstay of healthcare delivery or serves as a complement to it. TM with proven quality, safety, and efficacy contributes to ensuring that all people have access to healthcare; currently, many countries recognize the need to develop an organized and integrative approach to health care that allows governments, health practitioners and, most notably, those who use health care services to access TM in a safe, respectful, and cost-effective manner, and the WHO advocates such efforts and actions 3.
Complementary and Alternative Medicine: Complementary and alternative medicine (CAM) is a group of healthcare systems, practices, and products that are not currently considered part of orthodox medicine. CAM is now well popular elsewhere globally and includes TM and modern practices developed outside of indigenous communities. Sometimes the two terms are used synonymously, or TM may be referred to as CAM when it is adopted outside of its traditional culture. The WHO distinguishes between healthcare interventions that are used in a complementary manner, away from their geographical origins, and those rooted in local traditions and culture. According to the WHO, any form of healthcare indigenous to a particular region can be classified as ‘traditional medicine’ within that locality, but outside that region, it may be regarded as ‘complementary/alternative medicine’. CAM systems and therapies can be grouped into broad categories, such as natural products, mind-body medicine, and manipulative and body-based practices 4, 8.
Why should we be Concerned about TM: TM is an important resource for primary healthcare, innovation, and drug discovery 9. There are several reasons and derives to consider TM issues. The four most obvious reasons for studying TM include the following: (a) TM constitutes part of the primary healthcare needs of the majority of rural societies in developing countries; (b) TM can potentially offer novel forms of therapies and medicines; (c) TM is part of the cultural heritage of the society where it is uniquely practiced; and (d) numerous important indigenous knowledge and medicinal plants are being lost. In addition, it can contribute to the nationaleconomy through the export of raw or processed medicinal plants for rendering foreign currency and may provide potential medicines to any host of presently incurable diseases.
TM is part of the Primary Health Care Need for the Majority of Societies: Health is of central value for human health. Hence, most cultures seek to execute a broad range of healing practices. TM is often perceived as more accessible, affordable, and acceptable to people and can therefore represent a tool to help achieve universal health coverage. It is commonly used in many parts of Africa, Asia, and Latin America. Traditional medicines, treatments, and practitioners are the main, and sometimes the only, source of health care for millions of people, often living in rural areas of developing countries 9. For many people living in developing countries, traditional healers and herbal remedies are the only source of available health care. Alternatively, people living in developed countries typically use these approaches as an optional complement to modern medicine, often driven by patient preferences 9, 10. Additionally, it is thought that TM can contribute to addressing a number of global health challenges of the 21st century, particularly in the areas of chronic, no communicable diseases and population aging 9.
TM can Potentially Offer Novel forms of Therapies and Medicines: To find new treatment options, the continued development of therapeutics from natural sources is imperative, and healing traditions can provide insight into such resources 10. TM is a viable resource for innovation and drug discovery. It has already contributed to the development of modern medicines, with as much as one-third to one-half of pharmaceutical drugs being derived from plants 4, 8-10. The vibrant impact of plant-based medicines on current and future generations is undeniable 8. Nearly 25% of all modern medicines are derived from natural products, many of which were first used in a TM context 9. The contribution is even greater for specific classes of drugs. For example, nearly 50% of anti-malarial drugs developed over recent years are either natural products or their derivatives. In addition, several modern therapeutics exploit the ability of plants to synthesize a wide range of small molecules of great variety and complexity, and plant-derived products remain a dominant source of new therapeutic small molecules 10, 11. It is also thought that TM may provide potential medicines to any host of presently incurable diseases.
TM is part of the Cultural Heritage of the Society where it is Uniquely Practiced: Many countries, including Ethiopia, one of the most ancient settlements and civilizations in the world, have considerably rich cultural and architectural heritage. Ethiopia, for example, has rich traditional health practices and widespread use of medicinal and aromatic plants in traditional remedies and religious ceremonies. Written records of herbal medicine are almost absent, even though the country has had written language for over two thousand years 12. Indigenous knowledge is considered a national heritage of a country 13.
Generally, making heritage legible is a desirable goal. This means documenting and preserving traditional knowledge created in the past, which may be on the brink of vanishing 14. TM is historically, culturally, politically and economically intrinsic to societies around the globe. The United Nations Educational Scientific and Cultural Organization (UNESCO), in its definition of intangible cultural heritage (ICH), explicitly included TM in its remit, featuring the terms ‘indigenous knowledge’ and ‘traditional healing systems’ in its domain of ‘knowledge and practices concerning nature and the universe’. ICH and its significance and the continuation of its myriad indicators have reached unique levels of recognition and attention on international and national policy agendas over the past decade, and TM has long been included under the vast umbrella of ICH 15. TM is a form of ICH, as it is part of the practices, knowledge, skills as well as the instruments and objects associated with it that communities, groups, and individuals recognize as part of their cultural heritage. This heritage, passed from generation to generation, is constantly recreated by communities and groups in response to their interaction with nature and provides them with a sense of identity and continuity; thus, promoting respect for cultural diversity and human creativity is essential. The convention for safeguarding ICH is meant to ensure respect for it and to promote awareness at the local, national, and international levels of its importance, providing for international cooperation and assistance.
In this context, safeguarding means not only measures aimed at identification, documentation, research, preservation, and protection but also measures aimed at ensuring the viability of the ICH, including promotion, enhancement, and transmission, particularly via education, as well as the revitalization of the various aspects of such heritage 14. Moreover, in 1992, the United Nations advocated convention on biological diversity for the use of indigenous and traditional knowledge in the conservation of biodiversity, equitable sharing of benefits and sustainable use of natural resources 16, implying that managing threats to plants employed in TM and some species that are on the verge of extinction is advocated.
Numerous Important Indigenous Knowledge and Medicinal Plants are being Lost: TM has been used in the treatment and prevention of common diseases for as long as humans have lived. Long before the advent of mainstream medicine, traditional healers and medicinal plants played important roles in providing healthcare needs. TM, as a knowledge base, has been passed on orally from one generation to another for thousands of years. Currently, developing nations around the world are adopting a Western style of health care and using it alongside indigenous traditional medicine. This trend is on the rise, and the attraction to Western medicine is based on the premise that its benefits outweigh its hitches. As developing countries continue to focus more on Western medicine, much of the knowledge base of TM will slowly become extinct because it is neither practiced as commonly as before, explored, nor citizens encouraged to use TM 5.
Traditional African Medicines: Like other traditional systems of medicine, traditional African medicine (TAM) comprises the oldest form of the healthcare system that has stood the test of time. It is an ancient and culture-bound healing approach that humans have used to cope and deal with various diseases that have threatened their existence and survival 17. Before the evolution of modern medicine, TAM was used as the dominant medical system available to millions of people in Africa as the only source of health care for a greater ratio of population in both rural and urban communities. Several people in African countries have benefited from TAM, and it is still in use by many people not only in Africa but also all over the world. While TAM has been used for centuries for the prevention, treatment, and management of many acute and chronic diseases, it has also evolved and undergone various changes and challenges 17-19.
Traditional healers in native African communities treat patients holistically by reconnecting their social and emotional equilibrium based on community rules and relationships. In many of these communities, the traditional healers often act, in part, as intermediaries between the visible and invisible worlds, between the living and the dead or ancestors, sometimes to determine which spirits are at work and how to bring the sick person back into harmony with the ancestors. Therefore, in TAM, the aetiologies of illnesses are viewed from both natural and supernatural perspectives 17, 18. Most traditional medical theories have a social and religious character and emphasize prevention and holistic features. Traditional medical practices are often characterized by the healer’s personal involvement, secrecy, and reward systems. Instead, biomedical theory and practice show an almost opposite picture: asocial and irreligious with professional fairness. Thus, TAM is assumed to be in contrast with biomedicine 18. It has been argued that discourses on the impact of colonialism in Africa are clouded by a mixture of fortune and agony. Opinions on fortune narratives indicate that the institutionalization of the modern healthcare system marked a significant and rapid innovation in tropical medicine and that the process of modernization in Africa is basically connected with foreign interventions, particularly in the areas of health and democracy.
In contrast, the agony narration considers Western invasion to be a setback in the process of development in Africa, mainly in the ways of knowledge production. Remarks of all forms of Western hegemonies and exploitations, including slavery, capitalism, colonialism and imperialism and neo-colonialism, that have been embedded in these eras have contributed major stumbling blocks in the actualization of indigenous African development. While some critics of colonialism have focused on economic and political impacts, others have focused on the impact associated with indigenous knowledge systems, especially on knowledge of medicine. It is claimed that the arrival of Western medicine and culture gave rise to a cultural-ideological clash and created an unequal power relation that practically undermined and stigmatized the traditional healthcare system in Africa because of its overriding power.
The advent of Europeans marked a major turning point in the history of this tradition and culture. Unfortunately, modern medicine scientists, researchers, and practitioners, including African elites, have strictly criticized its use and practice, and preexisting African systems have been stigmatized and marginalized. In some extreme cases, it has been completely banned. For instance, the South African Medical Association outlawed the traditional medical system in South Africa 1953 17-19. Despite the claim that dominance continued in most African countries even after independence and that passionate ambivalence towards TAM has been noted in Africa, TAM is still in use in modern-day Africa as a major African sociocultural heritage without many reported cases of adverse health effects. Indeed, local efforts were initiated to challenge the condemnation and stigmatization of the TAM in some African communities during and after colonialism. The first protest against the marginalization of TAM in Nigeria dates back to 1922, when a group of native healers insisted that their medicine be legally recognized. In the post independence period, concerted efforts have been made to recognize and hold back TAM as an important aspect of the healthcare delivery system in Africa. Currently, it is accepted that effective health cannot be achieved in Africa through modern medicine alone unless it is complemented with traditional medicines; hence, the new health agenda in Africa focuses on the institutionalization of TAM in parallel with modern medicine into the national health care scheme to move the health agenda forward 17, 18.
The WHO has recognized that the contributions of TM and traditional healers to overall health delivery, particularly in developing countries, can be considered a boosting measure. The WHO has been working with African nations to integrate scientific and medical health models to enhance the potential of TM for the control of endemic diseases. Additionally, the WHO has offered a memorandum to help African member states institutionalize TM in their health systems and challenge different African Research centers on traditional medicine to cure priority diseases in Africa, including malaria, HIV/AIDS, sickle-cell anemia, diabetes, and hypertension 18.
There are several reasons for reconsidering TM as part of the healthcare system, primarily in developing countries, including Africa. Increasing evidence shows that a number of traditional medicines are vital and effective therapeutic regimens for the management of a wide spectrum of diseases, some of which may not be effectively managed using Western medicines. Furthermore, compared to modern medicines, TM is more accessible, affordable, acceptable and at least apparently safe, compatible, and suitable for the treatment of various diseases, chiefly chronic ones. In developed countries, the factors responsible for the widespread use of TM are beyond accessibility, affordability and cultural compatibility. Concerns regarding the adverse effects of chemical drugs, improved access to health information, changing values, and reduced tolerance to paternalism are some of the factors liable for the growing demand for CAM in developed countries. Similarly, an increase in the number of reported cases of chronic diseases, mainly in developed countries, has been attributed to the increasing use of CAM. Although modern treatments are widely available to address these ailments, some patients are convinced that they have not provided satisfactory results; hence, there is a need for alternative or complementary measures 17. Despite its major contribution to primary healthcare in Africa, certain problems and challenges must be overcome to fully achieve the objectives of regulation, standardization, and integration of TM in Africa.
First, the ethnocentric and medicocentric tendencies of the Western hegemonic mentality that are usually reflected by most stakeholders in modern medicine remain a very serious challenge, as it is a general belief in the medical circle that TM is unscientific. It is claimed that the introduction of Western culture, particularly in rural parts of Africa, has had a tremendous negative impact on traditional medicine. As Western education, Christianity, and increased contact with the global community have become integral parts of rural communities, taboos, traditions, and customs have been affected and in some cases abandoned altogether 17, 19.
Again, if integrated, who provides training to medical doctors on the ontology, epistemology and efficacies of African TM given the ethnocentric tendencies in modern medicine and the inherent epistemological and ideological characteristic differences of both medicines. It is part of these difficulties that some scholars suggest that both TM and modern medicine be allowed to operate, develop and flourish independent of one another. Another important challenge to TM is the widespread reported cases of fake healers and healing, albeit this is not limited to TM practice only 17. The indiscriminate or unregulated uses of various herbal medicines and shadow practices may put the health of people mainly in African countries at risk of toxicity, adverse health effects or even death 19. Additionally, over the years, TAM seems to face some ongoing challenges, including lack of usage of information communication technology, lack of adequate funding, lack of sufficient awareness, religion/cultural influences, unethical practices and deforestation of plant medicine 20.
Traditional Medicine and Drug Discovery: Brief Historical View: Throughout history, human beings have searched for remedies to fight diseases, and the human and clinical effects of medicinal and poisonous plants have started the entire enterprise of drug discovery and development as well as the pharmaceutical industry. Medicinal plants have been a valuable source of therapeutic agents for ages, and many of the current drugs are plant-derived natural products or their derivatives. The first written record of the medicinal uses of plants dates back to 2600 BC in Mesopotamia, and the search and use of medicinal plants in different cultures and locations have been reported by several studies worldwide. Medicinal plants, mainly before the 18th C, have only been applied empirically, without mechanistic knowledge of their pharmacological activities or active constituents. During these times, most drugs from medicinal plants were prepared with water- or water/alcohol-based plant extracts such as tinctures, decoctions, infusions, etc., and their chemical identities were unknown 21-23.
In 19thcentury, facilitated by the revolution of chemical and biological sciences and the development of organic chemistry and chemical analysis, the door was opened toward the isolation, chemical and analytical characterization and dosage of the active principles of plant extracts 21. Rational drug discovery from plants began in the early 19th C, when the German apothecary Sertürner succeeded in isolating morphine from the opium in 1806. This was the great discovery that launched the first generation of drugs and that triggered the examination of other medicinal herbs, and during the following decades of the 19thcentury, many bioactive natural products, primarily alkaloids, were isolated from their natural sources 21, 22.
In 1811, the Austrian physician Schmidt introduced pharmacognosy as the oldest branch of pharmacy to describe the study of medicinal plants and their properties 22, 24, 25. Apothecaries, who specialized in the purification of natural compounds, were the progenitors of pharmaceutical companies; German Merck was the first to extract morphine and other alkaloids in 1826 21, 22. Efforts were subsequently carried out to produce natural products by chemical synthesis to facilitate production at higher quality and lower costs. Salicylic acid was the first natural compound produced by chemical synthesis in 1853. After the discovery of penicillin in 1928, an era of drug discovery from microbial sources was initiated in the 1930s, laying the scientific and financial foundation for the modern pharmaceutical industry after World War II. At that time, the therapeutic use of extracts and partly purified natural products was increasingly replaced by the use of pure compounds 22. Therefore, the scientific enlightenment in the first half of the 19thcentury led to the gradual launching of pharmaceutical industries, and in the mid-19th century, pharmacies began to undergo numerous transformations as a consequence of the introduction of new pharmaceutical dosage forms such as gelatin capsules or tablets. The 19thcentury was the golden age for drug discovery, and many drugs that are still in use today were first discovered in this period 21, 26. Broadly speaking, the history of innovation and the discovery of drugs can be divided into two periods: 1820-1935 and 1935 today.
During the first period (beginning of 19th C until 1935), scientific methods aimed to isolate and purify natural products, obtain new compounds by chemical synthesis, and study their physiological properties. Two generations of drugs were introduced during this period: first-generation drugs, such as alkaloids and some inorganic and organic products, introduced between 1820 and 1880, and second-generation drugs, such as vaccines, serums, analgesics, antipyretics, hypnotics, and anti-protozoans, introduced between 1880 and 1935. The second period of the golden age of drug discovery began in 1935–1940, at the dawn of World War II. It started in approximately 1930 with the introduction of third-generation drugs such as vitamins, hormones, sulfonamides, antibiotics, and their derivatives.
This was followed by two more generations: the fourth (1960–1980) and fifth (where we are now). Semisynthetic antibiotics, central nervous system agents (psychopharmacological agents), autonomous nervous system agents (β-blockers), and cardiovascular agents (diuretics and antihypertensive agents), among others, were included in the 4th generation. Finally, in the fifth generation, enzyme inhibitors (cyclooxygenase inhibitors, angiotensin-converting enzyme inhibitors, enzyme inhibitors against viruses and cancer), biotechnologically derived drugs, etc., are included 21.
The Decline and Revival of Traditional Medicine: Although the history of drug discovery is long and complex, a few periods have been particularly fruitful in terms of new drug discovery. The backbone of drug discovery is traditional medicine, which is mainly characterized by the use of medicinal plants. Over history, the use of traditional medicines has experienced several ups and downs as well as notable periods of success globally 21.
Until the beginning of the 19thcentury, all medical practices were what we now call traditional practices. The 19th C scientific revolution introduced scientific materialism to all human activities, notably in the theory and practice of healthcare. Materialism is often allied with reductionism, which intends to break objects or complex phenomena into their component parts and deal with each one in isolation. This approach led to the search for a single cause of disease, and similarly, modern pharmacological investigations were aimed at finding a single active principle that could be isolated from medicinal plants. The introduction of such abstract medicines has proven to provide quick relief from suffering and has won instant admiration and popularity. Backed up by the advances made in physical, biological, chemical, and pharmacological sciences, this system, known as allopathic or modern medicine, made rapid advances during the 19th and 20th centuries and swept other systems of medicine off their feet and, of course, the decline of traditional medicines 21, 27.
Because natural product-based drug discovery is associated with intrinsic difficulties, the pharmaceutical industry has shifted its focus toward pure compound libraries of synthetic or natural origin and high-throughput screening for the discovery of new drug leads. Indeed, the 20th century proved the great success of modern medical sciences, particularly in diagnostics, treatment, and surgical measures, for a number of diseases that were regarded as incurable in the past. Direct intervention through technological and molecular means has become possible, and humankind is harvesting the benefits of the progress of modern medical sciences and technology. However, the results obtained from compound libraries and high-throughput screening did not meet expectations, as evidenced by the declining number of new drugs reaching the market, raising renewed scientific interest in drug discovery from natural sources, despite its known intrinsic challenges. In addition, we are equally aware of the inadequacy of modern medicine in dealing with many metabolic and degenerative disorders and other aliments related to old age.
Moreover, modern medicines have strong side effects, are expensive and are not accessible to the majority of the population but are mainly to the affluent. Instead, growing evidence demonstrates that several traditional medicine systems have satisfactory management and even cure for many such aliments; thus, towards the end of the 20th century, there was a revival of interest in traditional medicine systems, mainly oriental systems 22, 27.
Screening Strategies for New Drug Discovery: It may be recalled that, for millennia, medicines were sourced directly from nature by observing the phenotypic effects of substances on humans or animals, and pharmacological activities were often discovered by testing plant extracts in complex living systems and observing changes in phenotype. At the beginning of the 19th century, a key step towards modern drug discovery was made when it became possible to isolate the pharmacologically active substances responsible for the observed effects. This led to the discovery of many important drugs, and subsequently, it became relatively simple to find small molecules with a given biological effect on a cell or organism 26, 28-30.
Currently, drug discovery has evolved from the identification of active substances in traditional medicines to the direct search for new medicines facilitated by ever-increasing fundamental knowledge and technical knowhow in chemistry, biology, and related disciplines, as well as the power of computers, computational methods, and cognitive science, continuously updated databanks, and useful programs for mining them 31,32. Therefore, over the history of drug discovery, in addition to the shift in the source of drugs (from extracts to pure compounds of natural or synthetic origin), the scientific revolution of the 19thcentury and the subsequent progress of science and technology in the 20thcentury has triggered a shift in the type of screening strategies to identify compounds with the desired therapeutic effect. Accordingly, several strategies have been proposed and implemented for this purpose, and two major strategies for small-molecule drug discovery have been established: target-based and phenotype-based screens 33, 34, both having their respective advantages and limitations. These screening strategies have been well described by Van den Broeck (2015) 33.
Target-based Screening Strategy:
First a Target, then a Drug: The target-based drug screening strategy, which constitutes a more modern approach to drug discovery, involves identifying the target molecule involved in the disease process and then screening a library of compounds to identify those that interact with the target molecule. In the more recent past, the drug discovery trend has changed, and currently, investigating potential drug compounds usually starts with screening large collections of compound libraries against precharacterized disease-relevant targets, aiming to identify hits (compounds with the desired activity) that are then further studied in relevant in-vivo models to validate them 22, 32, 33, 35.
Target identification and validation are two of the most important steps in drug discovery. Target identification, the process of identifying a biological molecule that is involved in a disease process and that could be a potential target for a drug, constitutes the cornerstone of target-based screening. Target validation is the process of confirming that the target is indeed involved in the disease process and that modulating the target would have a therapeutic effect. The term “target” is used frequently to describe the specific molecular entity with which a drug interacts to initiate a biological response. The underlying concept of this strategy is that most drugs function by binding to a specific target at the most fundamental level. Therefore, if it is required to make a truly new drug, the first thing that should be done is to find a new target and then to find small molecules that bind to this target, preferably as specific as possible 32, 33, 35. This concept has been strengthened by the ambition of medicinal chemists to synthesize a “magic bullet,” an ultraspecific compound that would bind only to the target and to nothing else 33. Target-based screens often use in-vitro biochemical assays to search through a library of small molecules and are based on developing assays to detect compounds that interact with a specific molecular entity, which is known or hypothesized from basic research to be involved in processes implicated in a disease of interest 32, 33.
Beginning in the 1980s with a breakthrough in gene technologies, along with the invention of the versatile polymerase chain reaction in 1983 and the publication of the human genome in 2001, pharmaceutical scientists finally received the tools they needed to turn the blind old-fashioned dull drug screening into a highly rational, hypothesis-driven, reductionist, and efficient drug discovery engine. Target-based screening became possible, and the entire industry embraced it, largely replacing phenotypic screens. Even today, the intonation “first a target, then a drug” is still the dominant path for drug discovery. The technological spread of this path is enormous. However, most diseases are still present today, and the only thing that has truly exploded is the cost of discovering and developing new drugs. Target-based screening is now increasingly brought into question.
Although this strategy has certainly led to many successes, it failed more than expected. Often the targets thrown up by this reductionist, the bottom-up approach was wonderful in vitro but disastrous in the clinic owing to the lack of efficacy or unexpected toxicity 33.
Phenotypic Screening Strategy:
First a Drug, then a Target: Phenotypic drug screening strategies, which constitute the historical basis of drug discovery, often employ tests on organisms, organs, tissues, and cell (mammalian, microbial) preparations, followed by in-vitro studies of mechanistic underpinnings to investigate mainly plant-derived substances 22, 32, 33, 35. Phenotypic screens use assays that test whether small molecules or tested substances exert a desirable phenotypic change in the biological material that is utilized 32, 33, 36. The underperformance of target-based screening has raised the question of whether it is possible to develop drugs without knowing the target.
Of course, the answer is yes. For example, aspirin was synthesized in 1897, but its mechanism of action and target were discovered only in 1971 and 1976, respectively. Morphine has been used for age, but its main target was identified in1973, whereas other targets are still under investigation. Targets of many classes of drugs are only gradually emerging. It may also be that the actual declared targets for some drugs are in fact only off-target effects and that the real targets are to be discovered in the future. It can be said that most “first in class drugs” developed before the 1980s were discovered by phenotypic screening: first, an active compound is discovered, and then its target is determined. This is the exact opposite of target-based screening 33.
Indeed, phenotypic screens require a holistic approach most often at the cellular level, although tissues, organs, or even whole organisms can be employed to obtain the desired readouts. It should be recalled that phenotypic screens formed the basis for drug discovery before target-based screens became popular in the 1980s 32, 33 and are now receiving a status of renaissance.
Are Phenotypic and Target-based Screening Strategies Disputing: It has been noted that although there may be different strategies, an accepted aim of drug discovery is to put safe and effective medicines into the hands of physicians treating patients as quickly as possible. However, in recent years, there has been some debate regarding the relative productivity of phenotypic and target-based screening approaches that perhaps should end. Obviously, collective scientific and technological advances have transformed drug discovery from a fundamentally serendipitous activity into today’s high-technology workflows 30 and both phenotypic and target-based screens have made major contributions in this regard. With advances in genomics and molecular biology plus the establishment of large chemical libraries, the more holistic approach of phenotypic screening was largely abandoned in the 1990s in favor of the more reductionist target-based approach. This approach, which was thought to be more rational and efficient, was spurred by the arrival of a new era in which knowledge of the gene sequences of all potential drug targets is available 28, 34, 37. However, it appears that a fierce dispute has arisen between the supporters of phenotypic and target-focused screening regarding which path grants a higher probability of successful drug development 38. Questions about the importance of revealing the specific molecular target and mechanism of action for a new drug and when during the process of drug discovery should such information be obtained are debated. There are two opposing views regarding the need to understand the molecular targets and mechanisms of action of a drug. First, it is unnecessary because there are many beneficial drugs for which the target and mechanism of action remain unknown. The second view is that target identification and mechanism of action should be elucidated very early in the drug discovery process due to the tangible benefits provided by this knowledge 32.
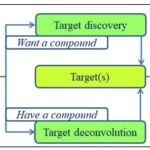
However, it has been reasoned that a chance to reconcile these two views resides in successful target deconvolution 38. Indeed, during drug discovery screening, the identification of relevant targets for screening hits and lead compounds is often the rate-limiting step toward understanding their mechanism of action to gain insights into deleterious on- and off-target adverse effects and thereby avoiding late-stage attrition early on, enabling rational drug design and providing biomarkers for monitoring clinical efficacy as well as enabling efficient structure-activity relationship studies during drug lead optimization 37, 39-41. Targets can be identified in two major ways: target discovery and target deconvolution Fig. 1. Target discovery is a prospective process for identifying and validating new molecular targets, whereas target deconvolution is a retrospective process for identifying and validating molecular target(s) underlying the observed phenotypic responses 33, 37, 39, 42.
The final goal of all target deconvolution strategies is the identification of biological molecules (targets) that directly interact with the active molecules, as well as conformation that modulation of the identified biological target causes functional effects in the phenotypic screen system that was initially used for the identification of the chemical molecule 37, 39. Whatever combination of disease model and read-out measures might have been used for the identification of active compounds, deconvolution is important 37.
In the phenotypic approach, despite the success in identifying an active compound producing a phenotypic effect, exhibiting the target biomolecule on which the hit compound directly acts and the matching between the target biomolecule and the phenotype effect is challenging 38, 43. Alternatively, in the target-based approach, which is meant searching for compounds that act directly on a known target biomolecule, it cannot be applied if the target biomolecule is unknown. It also ignores the concept of polypharmacology. Therefore, target deconvolution is a bottleneck in both phenotypic and target-based approaches to drug discovery 43. Therefore, it has been suggested that the sweet spot might be an integrated paradigm in which phenotypic approaches and target-based approaches are used in conjunction with each other in the discovery of drugs, and this move towards combining the two approaches has been enabled by advances in technology that can more subtly manipulate molecular processes in cells 43, 44.Currently, it is clear that the convergence of phenotype-based and target-based methods is more vital than their “either/or” comparison. The desire and hope are that the combined targeted phenotypic approach integrated with appropriate biological model systems will evolve into more effective strategies and innovative drugs for diseases that are yet to have a cure 44.
FIG. 1: OVERVIEW OF TARGET IDENTIFICATION THROUGH TARGET DISCOVERY AND TARGET DECONVOLUTION
Approaches for New Drug Discovery: New medicines are being researched and developed worldwide. The identification of potential new drugs or medicines is performed through a vigorous process known as drug discovery, which can be divided into two main approaches: classical pharmacology and reverse pharmacology 45.
Forward/Classical/Conventional Pharmacology-based Approach: The forward pharmacology (phenotype-based screening) approach, also known as the classical or conventional approach, has been the primary engine of drug discovery for many years. In this approach, the functional (phenotypic) activity of a medicine is first identified using cellular or animal models. Once the physiological effects of a compound are known, the compound’s ligand is identified and purified, and its derivatives are synthesized. The binding abilities of the ligand and its derivatives to the target receptor were determined via biological assays/screens. The most potent and selective ligand was identified as a new possible drug, and further research is performed with this ligand. The mechanism of action, selectivity for specific tissue receptors, and dose‒response of the ligand are determined by preclinical studies using in-vitro and in-vivo methods. However, as the mechanism of action and dose‒response of the compound/ligand are often not accurately determined, potential secondary effects are not understood and could be risky for human use 22, 45.
Reverse Pharmacology-based Approach: The reverse pharmacology (RP) approach, also known as target-directed drug discovery, begins by identifying a promising pharmacological target against which compounds are screened, and then promising hit compounds are validated in-vivo. While the RP approach has the advantage of reduced animal testing, its drawback is that it often requires a large amount of time and effort for the initial stages without a guarantee of in-vivo efficacy. Additionally, it has been noted that in the medicinal plant research community, the term “reverse pharmacology” is often used to designate “bedside-to-bench” or “field to pharmacy” strategies starting with clinical efficacy data followed by in-vivo and in-vitro mechanistic studies. However, this terminology conflicts with the mainstream understanding existing in the broad drug discovery scientific community that would see the “bedside-to-bench” strategy as a classical forward pharmacology (phenotypic drug discovery), for example, starting with observation of phenotypic changes at the organismal level (e.g., reversal of disease symptoms in patients). Thus, the need for terminology standardization has been suggested to avoid miscommunication between researchers from different scientific disciplines 22.
In this review, an attempt will not be made for terminology standardization, but the source of inspiration will be considered to differentiate them into (1) omics-inspired reverse pharmacology and (2) traditional medicine-inspired reverse pharmacology.
Omics-inspired Reverse Pharmacology: The traditional forward pharmacology approach of drug discovery was carried out mainly in the era before the Human Genome Project and especially before the development of many modern molecular biology techniques 22. It has been indicated that the achievements inomics (genomics, proteomics, metabolomics, etc.) inspired the start of reverse pharmacology-based studies aimed at deorphanization of orphan G protein-coupled receptors (GPCRs). Orphan receptors lack endogenous ligands. Orphan GPCRs are the roots of RP, and the overall strategy for characterizing orphan receptors has often been stated as a `reverse pharmacology' approach to distinguish it from conventional drug discovery approaches 47-49.
Thus, here, this version will be referred to as omics-inspired reverse pharmacologyto distinguish it from traditional medicine-inspired reverse pharmacology. The pharmaceutical industry has embraced genomics to identify new targets for drug discovery. Large-scale DNA sequencing has allowed the identification of a plethora of DNA sequences distantly related to known GPCRs, a super family of receptors that have a proven history of being excellent therapeutic targets 47. Most GPCRs were discovered by sequence similarity and homology screening methods, and the first GPCR was identified in 1987 48. The discovery of new GPCRs by such an approach still suffers from one obvious problem; these receptors lack their pharmacological identities, i.e., their endogenous ligands, and thus orphan GPCRs. The search for endogenous ligands has been a challenge and has given birth to the RP approach, which uses orphan GPCRs as targets to identify endogenous ligands. This approach was successful and led to the deorphanization of approximately 300 GPCRs in a period of two decades. Therefore, the GPCRs that were identified by molecular biological analyses have become the roots of RP, in which receptors have been attempted to be matched to potential transmitters.
Later, when high-throughput screening technology was applied to RP, dozens of orphan GPCRs became deorphanized 47, 48. This version of RP, also called target-based screening, began with the growth of molecular biology and caused a paradigm shift in global drug discovery.
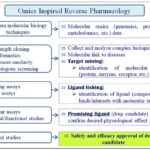
It begins with the identification of a molecular target (protein, enzyme, receptor, etc.) involved in the pathophysiology of a disorder or disease through genomic, proteomic, and metabolomic studies. This process is known as targetmining. Targets can also be identified by the clinical observation of the effects of various drugs, including those used in traditional or alternative medicines. Potential ligands (compounds) were screened through binding and/or other assays, where a highly selective ligand that binds to the molecular target is identified. This process is known as ligand fishing. Then, the biological activities of promising ligands are assessed using cellular assays and functional studies (e.g., animal models) to confirm the desired physiological effects.
This RP-based drug discovery begins by identifying a promising pharmacological target against which compounds are screened, and then promising hit compounds are validated in-vivo. Once the experimental results are consistently significant, clinical studies can be initiated 22, 45, 49. This omics-inspired RP is mainly applied in the search for new chemical entities (NCEs) of natural products or synthetic origin 23, and its general scheme is shown in Fig. 2.
FIG. 2: THE GENERAL SCHEME OF OMICS-INSPIRED REVERSE PHARMACOLOGY IS SHOWN
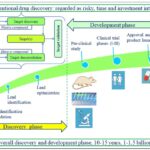
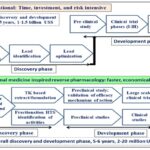
New Chemical Entity-based Drug Discovery and the Concept of Traditional Medicine-inspired Reverse Pharmacology: According to the medicinal plant research community, omics-inspired RP and forward pharmacology approaches are considered conventional NCE-based drug discovery strategies. This conventional NCE-based drug discovery and development path follows a stepwise strategy: molecule ⇾ mice ⇾ man. This process begins with the identification of a new molecule that interacts with a particular target. The molecule is tested for its pharmacological properties, first in-vitro and then in various animal models. Eventually, if all the results were as expected, a set sequence of clinical trials in human volunteers is started 50. There are four core phases (I-IV) of clinical trial studies. Phase I aims to assess the safety of a drug in a small group of healthy people and/or people affected by the target disease. In this phase, the short-term toxicity of a new drug is studied, and the maximum tolerated and effective doses are determined. Phase II is designed to determine the effectiveness of a drug in a larger group (up to 500) of patients with the disease or condition that the drug is intended to treat. When the efficacy and safety of a drug have been demonstrated, the drug will proceed to a phase III trial, which is the most central and gold standard phase of clinical trials that recruits the largest (thousands) patients and aims to compare a drug to the standard treatment for the disease or condition as well as to determine its effectiveness and long-term safety. The results of Phases I-III are used to determine whether the drug should enter the market. In other words, the drug will be approved or disapproved by authorities such as the US Food and Drug Authority (FDA). Thus, phase IV is conducted after the drug has been approved by the authorities, and its goal is to gather more data about the drug’s safety and effectiveness in a larger population over a long period of time 51, 52. The overall scheme of the conventional NCE-based drug discovery strategy is illustrated in Fig. 3. This strategy has been well tested, efficient, and productive and has led to the development of new drugs. Generally, this strategy is unrivalled but not in the area of herbal medicine 51. In addition, the discovery of a new drug candidate via this approach is time-consuming (approximately five years) and costly. The overall discovery and development process often requires a 10-15 year investigation period and investments as much as US $1 to 1.5 billion Fig. 3 and Fig. 4A. This makes it an extremely expensive, riskier and critically inefficient approach, resulting in ‘target rich lead poor’ performance. In other words, conventional drug discovery approaches take too long, are too expensive, have too many clinical failures and have uncertain outcomes 45, 53-55. This situation sought a paradigm shift in approaches to drug discovery 57, especially in the medicinal plant research community. An approach based on TM-inspired RP was proposed to offer a less costly, more efficient and less time-consuming (approximately 2 years) drug discovery process; the overall discovery and development process often requires 5-6 years of research and investments as much as US $2 to 20 million Fig 4B, and the end product is often affordable and available to the poor 45, 53, 54.
FIG. 3: THE OVERALL SCHEME OF THE CONVENTIONAL DRUG DISCOVERY STRATEGY
The concept of TM-inspired R Prelates to reversing the routine ‘laboratory to clinic’ progress of the discovery pipeline to ‘clinic to laboratory’. The classical NCE path: molecule ⇾ mice ⇾ man is reversed in the RP path to man ⇾ mice ⇾ molecule. In this RP process, safety remains the most important starting point, and its efficacy becomes a matter of validation 23, 53, 57. It is argued thattraditional herbal medicines have already been used for many years 50, 58. Thus, they have been field-tested in millions of people for thousands of years. In other words, natural experiments similar to phase IV clinical trials have already taken place; hence, herbal medicines are already “out there”, and they need no classical drug-development strategy to bring them to the market. However, this does not imply that herbal medicines require no further research. A strategy similar to classical drug development, except in reverse order, may be needed. Thus, the conventional path: molecule ⇾ mice ⇾ man is reversed into the reverse pharmacology path: man ⇾ mice ⇾ molecule 23, 27, 50, 57. It should be recalled that a history of traditional use and the extensive use of herbal and other traditional medicines may suggest, but does not assure, that TMs have a favorable risk-benefit ratio 59, 60. In particular, safety is the most crucial issue that needs to be addressed with any largely untested medicine that is used by millions. With herbal medicines, it is claimed that some of the work has already been done through natural field tests of common use. If herbal medicine causes immediate and direct adverse effects, it would be easy to understand. However, there can be delayed adverse effects that cannot be noticed without specific testing 50. The absence of any reported or documented side effects is not an absolute assurance of safety for herbal medicines since it is difficult for traditional practitioners to detect or monitor delayed effects, rare adverse effects, and adverse effects arising from long-term uses 2, 60.Additionally, interactions with synthetic drugs that have not been around for a long time might be encountered. Thus, the need to ensure safety is far too important to be left to speculation 50; however, a full range of toxicological tests may not be necessary 2. Once the safety of the herbal medicine is ensured, it is necessary to determine whether it is effective. In other words, clinical trials are needed to test whether the medicine works better than placebo. If an herbal medicine is found to be effective, the next consideration is the mode of action of the herbal medicine, as it is highly important to understand how a medicine works. However, if a commonly used herbal remedy is ineffective or unsafe, the question of how a medicine works is irrelevant. Thus, it has been proposed that in reverse pharmacology, preclinical research would not precede but rather follow clinical research 23, 50, 57. Once how an herbal medicine works is known, an interest may come in what the pharmacologically active substances are, whether they can be synthesized or even improved, and what receptors they interact with. Therefore, in the reverse pharmacology strategy, basic research comes last. As indicated above, the development of new drugs typically lasts for more than a decade and costs several hundred million dollars. These expenses are prohibitive in the monarchy of herbal medicine. The reverse strategy would answer the most critical questions of safety and efficacy relatively cheaply and quickly. Thus, it can be considered a logical and productive strategy toward filling the enormous gaps that undeniably exist in the current knowledge of commonly used herbal medicines 50.
FIG. 4: COMPARISON OF THE TWO MAIN DRUG DISCOVERY AND DEVELOPMENT APPROACHES: (A) CONVENTIONAL VERSUS (B) REVERSE PHARMACOLOGY APPROACHES
Traditional Medicine Inspired Reverse Pharmacology: A version of traditional medicine-inspired reverse pharmacology has been defined as the science of integrating documented clinical experiences and experiential observations into leads by trans-disciplinary exploratory studies and further developing these leads into drug candidates or formulations through robust preclinical and clinical research 53, 61. This type of RP follows a traditional knowledge-inspired approach and begins with a documented treatment outcome as witnessed by patients, which can offer a smart strategy for new drug candidates to facilitate the discovery process and for the development of rational synergistic botanical formulations. Generally, TM-inspired R Phas often been advocated in India and China, among other places, as a method based on the search for documented therapeutic effects of plants in ancient texts 63.
It is claimed that the concept of RP was first introduced in the development of Ayurvedic medicines in India and suggested in the 1950s for the development of Chinese traditional medicine 64. The Indian Sir Ram Nath Chopra and Gananath Sen laid the foundation for RP in medicinal plants by dogging the clinically documented effects of Ayurvedic drugs. Rawolfia serpentina Benth is mentioned as a major discovery via this approach, and Sen and Bose convincingly verified its antihypertensive and tranquilizing effects in 1931 53, 65. Currently, this approach is widely advocated globally, albeit to different extents in different places.
In the context of thetraditional medicine-inspired RP approach, the first step involves a population-based ethno-medical survey to retrieve patient accounts of recent experiences on the therapeutic effects (cure, worsening or adverse events) of traditional preparations 62. RP works to integrate traditional knowledge, experimental observations and clinical experiences 53, 64, 66. The originality of RP is meant for the integration of traditional remedies with the wisdom of robust documentation of safety and efficacy 23. For example, an RP approach to develop an antimalarial phytomedicine was designed and executed in Mali, resulting in a new standardized herbal antimalarial after six years of research in which the first step was to select a remedy for development through a retrospective study of treatment outcomes. The second step was a dose-escalating clinical trial that showed a dose‒response phenomenon and helped select the safest and most efficacious doses. The third step was a randomized controlled trial to compare phytomedicine with standard first-line treatment. The last step was to identify active compounds that could be used as markers for standardization and quality control. This example verifies that standardized phytomedicine can be developed faster and more cheaply than conventional drugs 67. There are three core stages of TM-inspired RP: experiential, exploratory, and experimental stages. The first experiential phase documents clinical/experiential hits using basal and post intervention data. It includes a literature search, traditional knowledge and well-defined modest observational therapeutic studies with objective response targets.
It involves documentation of robust clinical/experiential observations on the biological effects (responses) of drug candidates or formulations used in folk medicines by careful record keeping. The second exploratory phase aims to develop these hits into leads using relevant trans-disciplinary exploratory studies. It involves studies on tolerability, drug interactions, and dose-range findings in ambulant patients with defined subsets of the disease and preclinical studies using relevant in-vitro and in-vivo models to evaluate target activity. The third experimental phase studies lead to drug candidates or formulations.
This involves well-planned experimental (basic and clinical) studies at several levels of biological organization of positive leads as drug candidates or formulations to identify and validate their safety and efficacy 23, 56, 66, 68. TM-inspired RP-based natural product research begins with a re-examination of ancient/classic ethnic remedies that have been stated to cause the desired physiological effect but have not been scientifically proven or tested. Such remedies are screened against target molecules that are known to play a role in physiological effects to understand their effects and improve their efficacy. Therefore, natural products undergoing RP begin with humans and end with humans, which validates their safety, improves their function and saves time and cost 45. As RP starts from the assumption that the safety and efficacy of a drug material is already established from traditional use track records, pharmaceutical development, safety validation and efficacy studies are undertaken in parallel to controlled clinical studies 57, 66.
Scope of TM-inspired RP: The scope of TM-inspired RP has been reported to be vast, which may include those listed by Raut et al. (2017) 23: (1) clinical evaluation of the evidence of safety, efficacy, and quality of drugs (medicinal plants) used in traditional medicines; (2) discovery of new drugs from natural products already in use by humans; (3) finding new clinical facts and bedside biodynamic phenomena that may lead to new insights in human biology; (4) overcoming the current costly, long, and tiresome process of drug discovery/development; and (5) complementing the existing process using novel phytoactives as chemical scaffolds for new chemical entities. A TM-inspired RP must not merely be a linear process or unimodal in its resourcing. Its origins can also be derived from the diverse and rich big data of traditional/modern literature and ethnobotanical, phytochemical, experimental, clinical, and anecdotal cases 23.
Core Importance of Traditional Medicine-inspired Reverse Pharmacology: The core importance of traditional medicine-inspired reverse pharmacology-based drug discovery is that it offers unique relevance in situations where the pathophysiology of diseases is poorly understood. Its second importance is in situations in which diseases cannot be accurately replicated using the currently available in-vitro bioassays and in-vivo animal models. Third, this approach is suitable to accommodate situations in which multitarget/ multicomponent treatment modalities are needed. Additional importance of the TM-inspired RP approach is that it is suitable for application in developing nations.
It is well known that the pathology of several diseases, especially chronic diseases, is poorly understood 45. The RP approach is believed to be the best strategy for the development of cures for chronic illnesses since it utilizes materials (traditional herbs) with apparently proven safety and clinical efficacy 69. Suitable models and protocols are needed to evaluate the quality, safety, efficacy and clinical advantages of traditional medicine. Most of these medicines are polyherbal, multitargeted, and slow-acting and may have disease-modulating activities rather than direct agonistic or antagonistic activities.
Thus, scientifically validated and technologically standardized botanical products may be derived using the TM-inspired RP approach 70. Additionally, RP can be relevant for new uses of old drugs or for following up on a new unseen indication of a drug candidate 23. In addition, RP can be a bridge for translational medicine from traditional medicine to clinical practice. Such an effort would make healthcare delivery locally relevant and may lead to the global discovery of natural products 56. Likewise, it is well known that many diseases cannot be accurately replicated in animal modelsor through in-vitro bioassays 45. For example, because of the very narrow spectrum of in-vitro tests and limitations of rodent models available for malaria, explanations for the antimalarial clinical effectiveness of Argemone mexicana decoction have not been found from extensive research on its active substances but through RP-based clinical research.
This is not an exceptional situation, as the mechanisms of action of many medicinal plants remain a mystery; of course, this is also true for many synthetic drugs 64. Another core importance of the TM-inspired RP approach is that it adopts principles of systems biology where holistic yet rational analysis is performed to address multiple therapeutic desires 66. Currently, a major shift from single to multitargeted drugs, mainly for polygenic syndromes, is being witnessed 53.
Multiple cell types and diverse pathways contribute to a disease, and a single molecule may not be effective in the modulation of multiple targets; such conditions require combination therapy, witnessing the entry of a new informational paradigm into medicine that is most prominently represented by proteomics and metabolomics sciences 71. Botanical drugs are plant-derivative materials and their complexes that make them unfit for conventional single-target/single-drug development processes and thus have been largely disregarded in the field of medicine. However, it is widely understood in synthetic medicine that the single-drug magic bullet strategy is not adequate for treating especially chronic diseases (e.g., cancers, immune disorders, mental illnesses, lifestyle diseases, cardiovascular diseases) due to their complex pathogenetic mechanisms and that a multitarget/multicomponent approach involving control over a number of target sites is more effective. Traditional herbal medicines, which are a mixture of various components, correspond to the multitarget/multicomponent line, with therapeutic effects that are clinically confirmed, albeit with no analytically defined mechanisms through experience and knowledge accumulated over a long history of treatment of chronic illnesses 69. In fact, drugs of natural origin, especially those in the plant kingdom, are of immense importance in terms of their potential applications in the management of a multiplicity of human disorders 63.
In addition, the TM-inspired RP approach is suitable for application by developing nations, as they are frightened away from the domain by the costly and lengthy process of getting a new drug on the market. The 0.01% market yield from the baseline of 100% NCEs makes one wonder about the need for a paradigm shift in the drug discovery process 56. RP approaches rely primarily on clinical experiences, observations or available data on actual use in patients as a starting point 63. The study designs of RP do not follow the conventional phase I-III that is mandatory for NCEs because in RP, one starts at the bedside first 23: RP reverses the routine classical “laboratory to clinic” process of drug discovery to a “clinic to laboratory” or “bedsides to benches” approach 23, 53, 56, 63, 64. The treatment is given as a standard practice, and the physician in charge of the patient carries out the experiential studies in the regular clinical practice settings. The added elements are the records of the baseline clinical and laboratory findings or markers that defined the therapeutic or adverse response. The RP researcher joins the caretaker physician to ensure that careful attention to the minutest details is a mindful and ceaseless activity 23. Therefore, developing nations can easily apply the principles of TM-inspired RP to scientifically validate and standardize the botanical products used in their respective localities.
CONCLUSION: Traditional medicine (TM) describes medical knowledge systems that have developed over centuries within various societies before the era of modern medicine. It is an amorphous concept comprising a range of long-standing and evolving practices that are based on diverse beliefs and theories. TM is an important and often underestimated part of healthcare found in almost every country in the world, and the demand for its services is growing across the world either as the mainstay of healthcare or as a complement to it. TM is the chief resource for primary healthcare, innovation, and drug discovery. Some reasons why TM deserves attention include the following: (a) it constitutes part of the primary health care for the many rural societies of developing countries; (b) it offers novel forms of therapies and medicines; (c) it is part of the cultural heritage of the society where it is uniquely practiced; and (d) numerous important indigenous knowledge and medicinal plants are being lost. In addition, it can contribute to the national economy and may provide potential medicines for any host of presently incurable diseases.
Traditional African medicine (TAM) is the oldest form of healthcare system that has stood the test of time. It is an ancient culture-bound healing system that humans have used to cope with various diseases that threaten their survival. Traditional healers holistically treat patients in indigenous African communities by reconnecting their social and emotional equilibrium based on community rules and relationships.
Most TAM concepts and medical theories have a social and religious character and emphasize prevention and holistic features. TAM has not undergone the needed scientific advancement and knowledge production, and colonial influence has been blamed as the cause, at least in part. However, it has been argued that discourses on the impact of colonialism in Africa are clouded by a mixture of fortune and agony.However, although passionate ambivalence towards TAM has been noted in Africa, it is still in use in modern-day Africa as a major African sociocultural heritage without many reported cases of adverse effects. Indeed, a trend of institutionalization efforts exists, and the WHO acknowledges and supports such efforts. Although there are several sound reasons to reconsider and strengthen the TAM, there are several challenges to overcome while attempting to advance the TAM.
History recalls that the human and clinical effects of medicinal and poisonous plants have started the entire enterprise of drug discovery and development as well as the pharmaceutical industry. Although the history of drug discovery is long and complex, a few periods have been particularly fruitful in terms of new drug discovery. Traditional medicine, which is mainly characterized by the use of medicinal plants, is the backbone of drug discovery. Indeed, until the beginning of the 19th century, all medical practices were what we now call traditional. Medicinal plants have been a valuable source of therapeutic agents for millennia, and many of the current drugs are plant-derived natural products or their derivatives. Several studies have reported the search and use of medicinal plants in different cultures and locations worldwide. While TM encountered a decline during the 19th and 20th centuries, there was a revival of interest in traditional medicine systems towards the end of the 20th century.
The scientific revolution of the 19th century and subsequent progress in science and technology in the 20th century has triggered a shift in both the source of drugs (from extracts to pure compounds of synthetic or natural origin) and the type of screening strategies to identify compounds with the desired therapeutic effect (from phenotypic to target-based). Supported by considerable scientific and technological advances, the conventional new chemical entity (NCE)-based drug discovery strategy has proven to be a well-tested, efficient, and productive strategy that has led to the development of new drugs. However, despite this success, they are considered extremely expensive, riskier, and critically inefficient, resulting in poor performance. Hence, the medicinal plant research community advocates an alternative approach based on TM-inspired reverse pharmacology, which has been proposed to offer a less costly, more efficient, and less time-consuming drug discovery strategy. The concept of TM-inspired RP relates to reversing the routine ‘laboratory to clinic’ progress of discovery pipeline to ‘clinic to laboratory.’ The classical NCE path: molecule ⇾ mice ⇾ man is reversed in this RP path to man ⇾ mice ⇾ molecule. Safety remains the most important starting point in this RP process, and efficacy is a matter of validation. Therefore, in addition to applying the well-established scientific technology-supported drug discovery approach, adopting the concept of TM-inspired RP may accelerate the integration of traditional medicine in general and traditional African medicine in particular into mainstream healthcare.
Declarations:
Ethical Approval, Consent to Participate and Consent to Publish: This declaration is “not applicable”
Funding: This declaration is “not applicable”
Authors' Contributions: This paper is contributed by one author
Availability of Data and Materials: All data and materials are part of the submitted manuscript and there are no restricted data.
CONFLICTS OF INTEREST: Since the paper is contributed by one author, this declaration is “not applicable”
REFERENCES:
- Hill DM: Traditional Medicine In Contemporary Contexts Protecting and Respecting Indigenous Knowledge and Medicine. National Aboriginal Health Organization, 2003.
- WHO. General Guidelines for Methodologies on Research and Evaluation of Traditional Medicine. WHO/EDM/TRM/2000.1; World Health Organization, Geneva, 2000.
- WHO. WHO traditional medicine strategy: 2014-2023. WHO Library Cataloguing-in-Publication Data. World Health Organization, 2013.
- Abbott R: Documenting Traditional Medical Knowledge. World Intellectual Property Organization 2014.
- Emeagwali G and Dei GJS: African Indigenous Knowledge and the Disciplines. Sense Publishers 2014; 83-93.
- Jensen BA: Importance of Traditional Medicine In the age of technology monitoring with implantable devices. Ochre Media Pvt Ltd 2020.
- Virapongse A and Picheansoonthon C: Researching Traditional Medicine: A Review and Evaluation of Objectives and Methodologies. J R Inst Thail 2005; 30(4): 958-970.
- Chatfield K, Salehi B and Sharifi-Rad J: Applying an Ethical Framework to Herbal Medicine. Evid-Based Complement Altern Med 2018; 2018: 1903629.
- Fan TP, Briggs J and Liu L: The Art and Science of Traditional Medicine Part 1: TCM Today, - A Case for Integration. Sci 2014; 346 (6216): Sxx-Sxx.
- Briggs JP: A global scientific challenge: Learning the right lessons from ancient healing practices. In: Fan TP, Briggs J, Liu L, Lu A, van der Greef J, Xu A (eds). The Art and Science of Traditional Medicine Part 1: TCM Today, - A Case for Integration. Sci 2014; 346 (6216): Sxx-Sxx.
- Howes MJR and Simmonds MSJ: Pharmacognosy in the United Kingdom: Past, present, and future. In: Fan TP, Briggs J, Liu L, Lu A, van der Greef J, Xu A. The Art and Science of Traditional Medicine Part 3: The Global Impact of Traditional Medicine Sci 2015; 350 (6262): Sxx-Sxx.
- Vasisht K and Kumar V: Compendium of Medicinal and Aromatic Plants of Africa - Chapter 3: The Status of Medicinal Plants in East African Countries 2004; 1: 41-66, ICS-UNIDO.
- Padmasiri GR: Traditional methods of knowledge transfer and acquisition with special reference to indigenous knowledge of indigenous medicine in Sri Lanka. In: ICTM. International Conference on Traditional Medicine and Ethnomedical Research. Theme: Healthy Life in Natures Lap Tokyo, Japan 2019; 24-25.
- Vadi V: Intangible heritage: traditional medicine and knowledge governance. J Intellect Prop Law & Practice 2007; 2(10): 682-691.
- Riordan A and Schofield J: Beyond biomedicine: traditional medicine as cultural heritage. Int J Heritage Stud 2015; 21(3): 280-299.
- Chebii WK, Muthee JK and Kiemo K: The governance of traditional medicine and herbal remedies in the selected local markets of Western Kenya. J Ethnobiol Ethnomed 2020; 16: 39.
- Abdullahi AA: Trends and challenges of traditional medicine in Africa. Afr J Tradit Complement Altern Med 2011; 8(S): 115-123.
- Mordeniz C: Introductory Chapter: Traditional and Complementary Medicine Intech Open 2019.
- Oguntibeju OO: African traditional medicine: relevance, regulation, potential challenges and possible remedies. Asian J Agric & Biol 2019; 7(1): 166-175.
- Kingsley A: Traditional Medicine in Contextual African Society: On-Going Challenges. Open Acc J Oncol Med 2018; 1(3): 52-55.
- Ravina E: The Evolution of Drug Discovery: From Traditional Medicines to Modern Drugs. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim ISBN 2011; 978-3-527-32669-3
- Atanasov AG, Waltenberger B and Pferschy-Wenzig EM: Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol Adv 2015; 33(8): 1582-1614.
- Raut AA, Chorghade MS and Vaidya ADB: Chapter 4- Reverse Pharmacology. Innovative Approaches in Drug Discovery, Elsevier Inc 2017.
- Sarker SD: Editorial - Pharmacognosy in modern pharmacy curricula. Pharmacogn Mag 2012; 8(30): 91-92.
- Zahbi M: Scope and Biological Background of Pharmacognosy. Pharm Anal Chem Open Access 2022; 7: 144.
- Dias DA, Urban S and Roessner U: A Historical Overview of Natural Products in Drug Discovery Metabolites 2012; 2: 303-336.
- Pushpangadan P, Govindarajan R and Srivastava S: Scientific Evaluation of Traditional Medicine: Ethnopharmacology, Reverse Pharmacology, System Biology to Metabolomics. In Pushpangadan P, George V, Janardhanan KK (Eds). Ethnopharmacology - Recent Advances; Daya Publishing House, Delhi, 2008.
- Terstappen GC, Schlüpen C and Raggiaschi R: Target deconvolution strategies in drug discovery. Nat Rev/Drug Discov 2007; 6: 891-903.
- Lee J and Bogyo M: Target deconvolution techniques in modern phenotypic profiling. Curr Opin Chem Biol 2013; 17(1): 118–126.
- Bowen W: Is it time to end the target vs phenotypic screening debate? Accessed 0n 08/09/2023 from:https://www.ttp.com/insights/is-it-time-to-end-the-target-vs-phenotypic-screening-debate/
- Nicolaou KC: Advancing the Drug Discovery and Development Process. Angew Chem 2014; 126: 9280 - 9292.
- Davis RL: Mechanism of Action and Target Identification: A Matter of Timing in Drug Discovery. iScience 2020; 23: 101487.
- Van den Broeck WMM: Chapter 3 - Drug Targets, Target Identification, Validation, and Screening. The Practice of Medicinal Chemistry; Elsevier Ltd 2015.
- Kubota K, Funabashi M and Ogura Y: Target deconvolution from phenotype-based drug discovery by using chemical proteomics approaches. Biochim Biophys Acta Proteins Proteom 2019; 1867(1): 22-27.
- Manning AM: Target Identification and Validation. In: Li JJ, Corey EJ (eds). Drug Discovery: Practices, Processes, and Perspectives. John Wiley & Sons, Inc 2013.
- Atanasov AG, Zotchev SB and Dirsch VM: The International Natural Product Sciences Taskforce, Supuran CT. Natural products in drug discovery: advances and opportunities. Nat Rev 2021; 20: 200-216.
- Terstappen GC: Not a dilemma: target deconvolution in drug discovery. Future Med Chem 2009; 1(1): 7-9.
- Heilker R, Lessel U and Bischoff D: The power of combining phenotypic and target-focused drug discovery. Drug Discov Today 2019; 24(2): 526-532.
- Wilkinson IVL, Terstappen GC and Russell AJ: Combining experimental strategies for successful target deconvolution. Drug Discov Today 2020; (DRUDIS 2780): 1–8.
- Ohana RF, Levin S and Hurst R: Streamlined Target Deconvolution Approach Utilizing a Single Photoreactive Chloroalkane Capture Tag. ACS Chem Biol 2021; 16: 404-413.
- Schirle M and Jenkins JL: Chapter 5: Contemporary Techniques for Target Deconvolution and Mode of Action Elucidation. Drug Discovery Series No. 77-Phenotypic Drug Discovery. R Soc Chem., 2021.
- Lansdowne LE: Target Identification & Validation in Drug Discovery. Technology Networks 2023.
- Iwata H, Kojima R and Okuno Y: An in-silico Approach for Integrating Phenotypic and Target-Based Approaches in Drug Discovery. Mol Inf 2019; 38: 1900096.
- Wang S, Wang Z and Fang L: Advances of the Target-Based and Phenotypic Screenings and Strategies in Drug Discovery Int J Drug Discov Pharmacol 2022. https://www.sciltp.com/journals/ijddp
- Arulsamy A, Kumari Y and Shaikh MF: Reverse pharmacology: fast track path of drug discovery. Pharm Pharmacol Int J 2016; 4(3): 358-359.
- Wilson S, Bergsma DJ and Chambers JK: Orphan G-protein-coupled receptors: the next generation of drug targets. Br J Pharmacol 1998; 125: 1387- 1392.
- Civelli O, Saito Y and Wang Z: Orphan GPCRs and their ligands. Pharmacol Therapeut 2006; 110: 525-532.
- Chung S, Funakoshi T and Civelli O: Orphan GPCR research. Br J Pharmacol 2008; 153: 339-346.
- Paul A: Chapter22, Translational and Reverse Pharmacology. In: Raj GM, Raveendran R (eds.) Introduction to Basics of Pharmacology and Toxicology. Volume 1: General and Molecular Pharmacology: Principles of Drug Action. Springer Nature Singapore Pte Ltd 2019.
- Ernst E: Editorial- Reverse Pharmacology. J Diet Supplem 2009; 6(3): 201-203.
- Mahan VL: Clinical Trial Phases. Int J Clin Med 2014; 5: 1374-1383.
- Shultana S, Maraz KM and Ahmed A: Drug design, discovery and development and their safety or efficacy on human body. GSC Adv Res Rev 2021; 17(02): 113-122.
- Patwardhan B and Vaidya ADB: Natural products drug discovery: Accelerating the clinical candidate development using reverse pharmacology approaches. Indian J Exp Biol 2010; 48: 220-227.
- Saxena A, Saxena S and Singh S: Reverse Pharmacology: A new approach to drug development. Innoriginal Int J Sci 2015; 2(2): 7-12.
- Roy A, Patwardhan B and Chaguturu R: Reigniting pharmaceutical innovation trough holistic drug targets. Drug Discov World Summer 2016; 45-55.
- Vaidya ADB: Reverse Pharmacology-A Paradigm Shift for Drug Discovery and Development. Curr Res Drug Discov 2014; 1(2): 39-44.
- Patwardhan B, Vaidya ADB and Chorghade M: Reverse Pharmacology and Systems Approaches for Drug Discovery and Development. Curr Bioact Comp 2008; 4: 201-212.
- Chen S, Song J and Sun C: Herbal genomics: Examining the biology of traditional medicines. In: Fan TP, Briggs J, Liu L, Lu A, van der Greef J, Xu A. The Art and Science of Traditional Medicine Part 2: Multidisciplinary Approaches for Studying Traditional Medicine Sci 2015; 347 (6219): Sxx-Sxx.
- WHO. Operational guidance: Information needed to support clinical trials of herbal products. TDR/GEN/Guidance/05.1. World Health Organization on behalf of the Special Programme for Research and Training in Tropical Diseases 2005.
- Birdi TJ, Brijesh S and Daswani PG: Approaches towards the preclinical testing and standardization of medicinal plants. Report on the South Asian Conference held in Bangalore (India) 2006.
- Dayama A and Patil R: Reverse pharmacology for Ayurveda-based modern medicines. Centre for Society and Policy • IISc • 01C/01/2022 • January 2022
- Graz B, Falquet J and Haouala A: Reverse Pharmacology of Medicinal Plants: Good Luck or Efficient Method? Planta Med 2012; 78-IL31.
- Patwardhan B: Traditional medicine-inspired approaches to drug discovery: can Ayurveda show the way forward? Drug Discov Today 2009; 14(15-16): 804-11.
- Simoes-Pires C, Hostettmann K and Haouala A: Reverse pharmacology for developing an anti-malarial phytomedicine. The example of Argemone mexicana. Int J Parasitol: Drugs and Drug Resistance 2014; 4: 338-346.
- Dutta S, Banerjee S and Kumar S: Reverse Pharmacology: A Science Redefining Drug Discovery. Europ J Biomed Pharmaceut Sci 2019; 6(3): 245-249.
- Surh YJ: Reverse Pharmacology Applicable for Botanical Drug Development – Inspiration from the Legacy of Traditional Wisdom. J Trad Complem Med 2011; 1(1): 5-7.
- Willcox ML, Graz B, Falquet J, Diakite C, Giani S and Diallo D: A “reverse pharmacology” approach for developing an anti-malarial phytomedicine. Malaria J 2011; 10(1): 8.
- Vaidya A: Reverse pharmacological correlates of ayurvedic drug actions. Indian J Pharmacol 2006; 38: 311-5.
- Ahn K: The worldwide trend of using botanical drugs and strategies for developing global drugs. BMB Rep 2017; 50(3): 111-116.
- Patwardhan B: Importance of bioprospecting and new approaches to drug development. Biochem Pharmacol 2013; 2: 4.
- Srinivas M, Kumar PL and Sivaprasad SNV: Reverse Pharmacology and Systems Approaches for Drug Discovery and Development. J Pharmaceut Biol 2013; 3(2): 63-68.
How to cite this article:
Hiben MG: Trends of traditional medicine research and drug discovery: a review. Int J Pharmacognosy 2023; 10(11): 534-53. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.10(11).534-53.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
534-553
950 KB
770
English
IJP
Mebrahtom Gebrelibanos Hiben
Department of Pharmacognosy, School of Pharmacy, College of Health Sciences, Mekelle University, Mekelle-Ethiopia.
mebrahtomgs@yahoo.com
20 October 2023
19 November 2023
29 November 2023
10.13040/IJPSR.0975-8232.IJP.10(11).534-53
30 November 2023