TRANSMISSION BLOCKING STRATEGY FOR MALARIA ERADICATION: THE ROLE OF ANTIMALARIAL AGENTS
HTML Full TextTRANSMISSION BLOCKING STRATEGY FOR MALARIA ERADICATION: THE ROLE OF ANTIMALARIAL AGENTS
T. O. Johnson
Department of Biochemistry, Faculty of Medical Sciences, University of Jos, Nigeria.
ABSTRACT: Elimination and Eradication is an important component of the Global Strategy for combating malaria, and one of the ways of achieving this purpose is blocking the transmission of malaria parasites from humans to mosquito vectors. The ability of Plasmodium to cease asexual replication in the erythrocytes of the human host and commit to the formation of gametes necessary for sexual replication in the gut of the mosquito vector is essential for efficient transmission of malaria. Although a conserved member of the Apicomplexan AP2 family of transcriptional factors acts as the master switch in triggering the transcriptional cascade that initiates gametocytogenesis, some environmental factors including some antimalarial drug treatments, are believed to switch on the gene(s) that determine the commitment to the sexual phase. Antimalarial agents have different effects on gametocyte carriage depending on their mechanisms of action. This review enumerates the role of various classes of antimalarial agents in either promoting or preventing plasmodium gametocytogenesis and points out the need to, in addition to exiting gametocytocidal, design new drugs that are capable of preventing gametocytogenesis and killing mature gametocytes in humans; or preventing gametogenesis and fertilization in the mosquito.
| Keywords: |
Malaria, Gametocytogenesis, Chloroquine, Primaquine, Artemisinin, Antifolates
INTRODUCTION: Malaria is afebrile, mosquito-borne infectious disease of humans, caused by eukaryotic protists of the genus Plasmodium. It is prevalent throughout most of the tropical world. Globally, an estimated 3.2 billion people in 97 countries and territories are at risk of being infected with malaria and developing the disease, and 1.2 billion are at high risk (>1 in 1000 chance of getting malaria in a year). According to the latest estimates, 198 million cases of malaria occurred globally in 2013, and the disease led to 584 000 deaths 1.
Even though these represent a decrease in malaria case incidence and mortality rates of 30% and 47% since 2000, respectively 1, malaria is still one of the ‘big three’ diseases, along with HIV and tuberculosis, affecting the developing world. While it has been eliminated in many regions, it remains a scourge of poorer countries 2, hence the need for continued efforts in the fight against this deadly disease. The early signs of resistance to existing antimalarial drugs shown by the parasites are gradually becoming a major concern, and the World Health Organization (WHO) warns that the public health consequences could be dire 3.
The Malaria Eradication Research agenda initiative, created in 2007 have re-established the long-term goal of malaria eradication 4. One way of achieving this goal is the transmission-blocking strategy aimed at the identification of therapies capable of eliminating Plasmodium gametocytes, the sexual forms of the parasites that are transmitted between humans and Anopheles mosquitoes 5. However, considerable increases in gametocyte prevalence have been observed after widespread use of some antimalarial therapies 6. Drug-induced gametocytogenesis has been investigated in some clinical studies 7, but it has been difficult to draw firm conclusions from these studies. It is, however, clear that gametocytemia is a very sensitive indicator of emerging drug resistance 8, with increasing gametocyte prevalence having been shown to precede measurable changes in parasite clearance or a decrease in cure rates 9. To eradicate malaria, it is highly necessary to monitor the effects of antimalarial chemotherapy on gametocyte carriage and to identify or design new drugs that promote gametocytocidal activity 9.
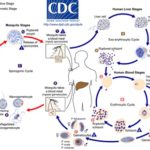
Plasmodium Life Cycle: The malaria parasite exhibits a complex life cycle involving an insect vector (mosquito) and a vertebrate host (human). The major phases of the life cycle are a liver stage, blood stage, sexual stage, and sporogony Fig. 1.
FIG. 1: LIFE CYCLE OF MALARIA PARASITE 10
Sporozoites are inoculated into the human with the bite of a female Anopheles mosquito, they migrate immediately to the liver and invade hepatic cells. Sporozoites undergo an asexual replication known as exoerythrocytic schizogony during a dormant period of approximately two weeks to form schizonts and cause rupture of the hepatocytes. A hepatocyte releases up to 30 000 merozoites, each capable of invading an erythrocyte. However, a proportion of the liver-stage parasites from P. vivax and P. ovale go through a dormant period instead of immediately going through the asexual replication. These hypnozoites reactivate after several weeks to months (or years) after the primary infection and are responsible for relapses.
The invasion of erythrocytes by merozoites begins a period of asexual cycling in the erythrocytes. The invading merozoites undergo a trophic period in which the parasite enlarges. The early trophozoite is often referred to as 'ring form' because of its morphology. Trophozoite enlargement is accompanied by an active metabolism including the ingestion of host cytoplasm and the proteolysis of hemoglobin into amino acids. The parasite avidly ingests and degrades host erythrocyte hemoglobin using a specialized structure called a cytostome11-15, which spans the double membrane between erythrocyte and parasite cytoplasm. Hemoglobin-containing vesicles are pinched off from the cytostome and travel to the digestive vacuole where the hemoglobin is broken down 12, 13, 15-17. The process of hemoglobin degradation releases heme, which is detoxified by polymerization into a crystalline pigment, hemozoin 18, 19.
Multiple rounds of nuclear division manifest the end of the trophic period without cytokinesis resulting in a schizont. Merozoites bud from the mature schizont also called a segmenter, and the merozoites are released following rupture of the infected erythrocyte. Invasion of erythrocytes reinitiates another round of the blood-stage replicative cycle. A single merozoite invading a cell is capable of producing as many as 36 merozoites in a mature multinucleated schizont. On average in P. falciparum, 16 new merozoites are released from an erythrocyte every 48 h, each of which can invade a new cell. The resultant exponential increase in parasitemia is mainly responsible for the onset of clinical symptoms 20.
At a point during the multiple rounds of asexual erythrocytic cycling, a proportion of parasites are stimulated to differentiate into sexual forms known as macro- (female) and micro- (male) gametocytes. The transmission of malaria parasites from man to mosquito depends on the presence of mature Plasmodium gametocytes in the human peripheral blood. P. falciparum gametocytes undergo complex development that is characterized by five morphologically distinct stages (I–V). Only mature stage V gametocytes are observed in the peripheral blood and are accessible to feeding mosquitoes. Once ingested by a feeding female Anopheles mosquito, male and female gametocytes form gametes that fuse to form zygotes that develop into a motile ookinete that can penetrate the peritrophic membrane and traverse the mosquito midgut epithelium to form oocysts. The oocysts enlarge over time and rupture to release sporozoites that migrate to the mosquito salivary glands. Once the sporozoites have migrated into the salivary glands, the mosquito is infectious to humans 21.
Parasite Commitment to Gametocytogenesis: The ability of Plasmodium to cease asexual replication in the erythrocytes of the human host and commit to the formation of gametes necessary for sexual replication in the gut of the mosquito vector is essential for efficient transmission of malaria. Differentiation of some of the parasites in the human bloodstream into sexual forms that can mate inside the mosquito is the key to this commitment. It has been reported that, in P. falciparum, commitment to sexual stage occurs one cycle before gametocytes appear in the blood, such that all merozoites released from a single schizont are already committed for following either a sexual cycle or an asexual one 22, 23. This implies that the trophozoites of the preceding asexual cycle were already committed to follow either a sexual or an asexual cycle 23. Also, all merozoites from a “sexually committed” schizont become either males or females 24, 25. That would indicate that commitment to male or female gametocytes either happens concomitantly or follows the asexual-to-sexual switching 26.
The molecular basis of the developmental switch – the “Holy Grail” for understanding gametocytogenesis has eluded the malaria research community for years. However, recent reports by the teams of Oliver Billker (Wellcome Trust Sanger Institute), Manual Llinás (Princeton and Penn State Universities) and Andy Waters (Glasgow University) provide resounding evidence that a conserved member of the Apicomplexan AP2 family of transcriptional factors acts as the master switch in triggering the transcriptional cascade that initiates gametocytogenesis. The AP2 master switch, termed AP2-G (for gametocytogenesis), was discovered in both human malaria P. falciparum and mouse malaria P. berghei (mouse malaria). A second AP2 transcription factor, termed PbAP2-G2, was also identified in P. berghei as necessary for gametocytogenesis, although not acting in a master switch role 27-29.
Although the molecular switch has been identified, what throws that switch, releasing AP2-G from its silenced state and making some malaria patients into ‘transmitters’ while others are not, is still a mystery. However, in P. falciparum and berghei, there is evidence that the switching mechanism is highly flexible and is responsive to primary signals which include certain environmental factors which are believed to switch on the gene(s) that determine the commitment to the sexual phase 30, 31. The decision to commit to sexual development is, therefore, a multifactorial one, based on an intricate relationship between environmental stimuli and developmental pathways acting on stage-specific genes. Although the mechanisms involved remain poorly understood, it is generally accepted that conversion to a gametocyte occurs when environmental conditions no longer favor asexual growth.
Environmental factors implicated in gametocytogenesis could be grouped as those contributed by the host or the parasite, or could simply include a drug treatment or a signaling mechanism 23. Various host factors contributing to increased commitment to gametocytogenesis include increased immune pressure 32-34, increased steroids and corticosteroids 35, an increased proportion of reticulocytes in blood 36, 37 and anemia 38, 39. Antimalaria drugs were also found to induce gametocytogenesis 40, 23. Many parasite-borne factors have been demonstrated which affect gametocytogenesis. These include the presence of mixed-genotype infections 41, 42, levels of asexual parasitemia 30 and occurrence of lysed parasitized erythrocytes 43. These environmental triggers strongly suggest a signal transduction mechanism through which the parasite receives a cue to sexual commitment. G proteins have been implicated as a signaling mechanism to mediate the switching to sexual development in response to environmental stimulus 30.
Role of Antimalarial Agents in Gametocytogenesis: Various reports have shown the effects of antimalarial agents on the development, density, and intensity of gametocytes. Peatey et al., used transgenic Plasmodium falciparum parasites expressing a green fluorescent protein tag in a fluorescence-activated cell sorting-based assay to measure the effect of 8 antimalarial drugs (chloroquine, quinine, atovaquone, artemisinin, mefloquine, primaquine, piperaquine, and pyronaridine) on gametocyte production in-vitro. Exposure to antimalarial drugs increased the number of gametocytes in test cultures 44. However, Artemisinin derivatives combination therapy 45-47 and primaquine 48 are gametocytocidal and to reduce transmission.
A trend towards the decreased expression of genes involved in glycolysis, protein biosynthesis and hemoglobin catabolism observed in later gametocyte stages 48 have been reported to correspond with the absence or reduced gametocytocidal activity of antimalarial drugs targeting some of these processes. Some inhibitors of nucleic acid synthesis 49 including antifolates50 and Chloroquine 40 have been shown to increase the rate of gametocytogenesis. These suggest that the effect of an antimalarial drugon gametocytogenesis depends of its mechanism of action.
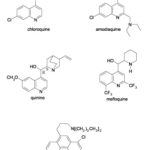
Quinolines: Several studies have indicated that quinolines Fig. 2 such as chloroquine, mefloquine, quinine, and quinidine act against the erythrocytic stage of infection by inhibiting polymerization of the heme that is released during hemoglobin degradation. Primaquine, however, kills intrahepatic forms and gametocytes. Quinine, the first of all antimalarials extracted from the bark of the South American Cinchona spp tree, is reported to be a powerful schizonticide, active only against malaria pigment (hemozoin) producing stages 51. The drug has no activity against the sporozoites and exo-erythrocytic stages of the parasites (liver schizonts) which do not consume hemoglobin. Other blood schizontocidal antimalarial drugs (chloroquine, amodiaquine, mefloquine, halofantrine, and lumefantrine) are suggested to be inhibitors of heme detoxification 52, a property which has been suggested to cause inhibition of asexual growth and subsequent increase in gametocytogenesis 53-58. Buckling et al., demonstrated a 5-fold increase in gametocytogenesis in human malaria, P. falciparum, in-vitro, in response to treatment with chloroquine. In all clones used, gametocytogenesis increased with increasing inhibition of asexual growth by chloroquine 40.
FIG. 2: STRUCTURES OF SOME QUINOLINES 59
Chloroquine-induced gametocytogenesis has been linked with the prevalence of drug resistance. Several studies have found that the proportion of chloroquine-treated patients harboring gametocytes is higher among those presenting therapeutic failures than patients with adequate therapeutic response 60-62. This suggests that resistant parasites are more likely to develop gametocytes after treatment, thereby favoring the spread of drug resistance 63.
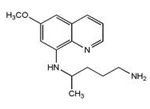
Primaquine, an 8-aminoquinoline, has no sporontocidal effect on P. falciparum 64 but has been known to reduce the prevalence of gametocytes circulating in the peripheral bloodstream of infected individuals and to prevent exflagellation in gametocytes that are present 65.
FIG. 3: STRUCTURE OF PRIMAQUINE 66
Primaquine is metabolized via oxidative deamination at the C-4’ into its primary metabolite carboxyprimaquine which displays much reduced antimalarial activity 67, 68. Other metabolites vary in their antimalarial activity, with 5,6-dihydroxy-8-aminoquinoline showing relatively intense in vitro inhibition of P. berghei exoerythrocytic stages while 6-hydroxy-8-aminoquinoline is even less active than carboxyprimaquine 68. Acylation of the aliphatic side-chains of primaquine derivatives effectively blocks their conversion to carboxyprimaquine, extending their activity. Further development of primaquine derivatives was however discouraged due to diverse interactions between primaquine and G6PD-deficient (G6PDd) individuals. Toxic metabolites of primaquine including 5-hydroxyprimaquine and 6-methoxy-8-aminoquinoline 69 may induce severe hemolytic anemia by oxidizing glutathione to glutathione disulfide which leaches from red cells and leads to denatured, insoluble aggregates of hemoglobin in intact cells that are then preferentially removed from circulation by the spleen and liver 48, 70.
However, WHO has conducted a review of the evidence on the safety and effectiveness of primaquine as gametocytocide of P. falciparum, which indicates that a single 0.25 mg base/kg is effective in blocking transmission and is unlikely to cause serious toxicity in subjects with any of the G6PD variants 71.
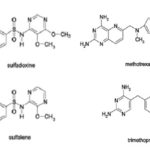
Antifolates: The antifolate drugs used against malaria include various combinations of dihydro- folate-reductase inhibitors (proguanil, chlorpro-guanil, pyrimethamine, and trimethoprim) and sulfa drugs (dapsone, sulfalene, sulfamethoxazole, sulfadoxine, and others) 72 Fig. 4.
FIG. 4: STRUCTURES OF SOME ANTIFOLATES 59
Antifolates are thought to act against the parasite by inhibiting the synthesis of folate, as they do in bacteria, although few studies have been performed to determine whether this is truly their mechanism of action 73. Pyrimethamine and cycloguanil are suggested to target the dihydrofolate reductase (DHFR) activity of the parasite's bifunctional DHFR-thymidylate synthetase (TS) protein, whereas the sulfa drugs affect the dihydropteroate synthetase (DHPS) activity of the bifunctional hydroxymethylpterin pyrophosphokinase (HPPK)-DHPS protein, all of these drugs acting as competitive inhibitors of the natural substrates. DHPS found only in the parasite, participates in the de novo synthesis of essential folate coenzymes. DHFR, present in both host and parasite is required for the maintenance of a constant supply of fully reduced (tetrahydro) forms of folate for essential one-carbon transfer reactions, including the provision of nucleotides for DNA synthesis 74.
The antifolate antimalarial, pyrimethamine-sulfadoxine (Fansidar), has become increasingly used as first-line treatment of malaria in several African countries because of increasing resistance in Plasmodium falciparum to chloroquine. With increased use, resistance in P. falciparum to this drug has also increased 75. It was later suggested that co-trimoxazole, an antifolate antimalarial with relatively short half-lives of its components compared to pyrimethamine-sulfadoxine, may be used as an alternative to the latter for the treatment of uncomplicated falciparum infections in children, because it is equally effective 76, 77.
Nevertheless, however, these drugs have been found to significantly increase the prevalence and intensity of gametocytaemia during acute malaria infections in children from a hyperendemic area in Southwestern Nigeria.
The effect was more pronounced in children treated with pyrimethamine-sulfadoxine than in those treated with co-trimoxazole78. Puta and Manyando reported an enhanced gametocyte production in Plasmodium falciparum malaria patients treated with Fansidar 79. The very high post-treatment prevalence and density of gametocyte carriage following pyrimethamine-sulfadoxine treatment have been reported to contribute to the remarkable spread of its resistance across vast regions 80.
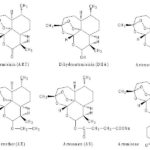
Artemisinin Derivatives: Artemisinin (Qinghaosu), one of the most potent and effective antimalarials to date, discovered by Chinese chemists in the 1970’s; ‘project 523’ 81 are effective not only against multi-resistant strains of P. falciparum, but have broad stage specificity against the Plasmodium life cycle including activity throughout the asexual blood stages 82 and also the sexual gametocyte stages 83. Artemisinin is a sesquiterpene trioxane lactone whose endoperoxide bridge is essential for antimalarial activity. Semisynthetic derivatives of artemisinin include Artesunate, artemether, dihydroartemisinin, artelinic acid and artemoti 84 Fig. 5.
FIG. 5: STRUCTURES OF ARTEMISININ AND DERIVATIVES 85
The cleavage of the peroxide bridge of artemisinin in the presence of ferrous ion (Fe2+) from heme derived from the breakdown of the host cell hemoglobin by the parasites, forms highly reactive free radicals which rapidly rearrange to more stable carbon-centered radicals 86, 87. These artemisinin-derived free radicals have been suggested to chemically modify and inhibit a variety of parasite molecules, resulting in parasite’s death 88, 89. A more recently described alternative is that artemisinins disrupt cellular redox cycling 90.
Artesunate has been shown to potently inhibit the essential Plasmodium falciparum exported protein 1 (EXP1), a membrane glutathione S-transferase 91. Recently Shandilya et al. suggested a free radical mechanism where artemisinin gets activated by iron present in food vacuole which in turn inhibits PfATP6 by closing the phosphorylation, nucleotide binding and actuator domains leading to loss of function of PfATP6 of the parasite and its death 89, 92-95.
Artemisinins act primarily on younger gametocytes 96, 97, inhibiting differentiation to the mature infective stages. ACTs also shorten the typical carriage of gametocytes in the blood from 55.6 days in patients receiving non-ACT therapies to 13.4-28.6 days in those receiving ACTs 98. Certain studies, however, have demonstrated activity against mature gametocytes in-vitro 99, 100, though this effect does not appear to translate to significant clinical impacts against mature gametocytes in infected patients 48.
Single Dose Primaquine with an ACT: As stated earlier, artemisinin is an effective antimalarial, but its activity is limited to asexuals and stage I-III gametocytes. Furthermore, clinical resistance to artemisinin-based combinations (ACTs) has been recently reported in Cambodia 101. Primaquine is the only drug currently available that is effective against stage V gametocytes, and its use as a gametocytocide has great potential to reduce the transmission of falciparum malaria in low transmission settings, and in particular to help contain the spread of artemisinin-resistant falciparum malaria in Southeast Asia. The main limitation to its use has been hemolytic toxicity. The 8-aminoquinoline antimalarials produce dose-dependent acute hemolytic anemia (AHA) in individuals who have G6PD deficiency, an inherited X-linked abnormality.
WHO has conducted a review of the evidence on the safety and effectiveness of primaquine as gametocytocide of P. falciparum. Based on this review, the Malaria Policy Advisory Committee (MPAC) recommends the following: In: (1) areas threatened by artemisinin resistance where single dose primaquine as a gametocytocide for P. falciparum malaria is not being implemented, and (2) elimination areas which have not yet adopted primaquine as a gametocytocide for P. falciparum malaria: A single 0.25 mg base/kg primaquine dose should be given to all patients with parasitologically-confirmed P. falciparum malaria on the first day of treatment in addition to an ACT, except for pregnant women and infants less than 1 year of age 71.
CONCLUSION: A malaria-free world is achievable. Discovery and use of new antimalarials with activities against both the asexual and sexual stages of the parasites could prevent transmission of malaria parasites from humans to mosquito vectors.
AKNOWLEDGEMENT: My special gratitude goes to the main Library, University of Jos, Nigeria, for providing free access to the resource materials used while writing this review, and to the Department of Biochemistry, as well as the African Centre of excellence in Phytomedicine Research and Development (ACEPRD), University of Jos, Nigeria, for providing a good platform for teaching and research.
CONFLICT OF INTEREST: Nil
REFERENCES:
- WHO Global Malaria Programme: World Malaria Report 2014. who.int/malaria. Accessed 26th February 2015.
- Shetty P: The numbers game. Nature. 26th April 2012, 484, S14–S15, www .indiaenvironmentportal.org.in/.../. Assessed 26th February
- Halper M: New Drugs Could Offer Hope for Malaria Patients. Novartis global. Jul 31, 2014, novartis.com/stories/medicines/2014-07-new-malaria-treatment.shtml. Accessed 26 February 2015.
- The Malaria Eradication Research Agenda (malERA). http://malera.tropika.net. Accessed Sept 11, 2010).
- Lelie`vre J, Almela MJ, Lozano S, Migue lC and Franco V: Activity of clinically relevant antimalarial drugs on Plasmodium falciparum mature gametocytesinan ATP bioluminescence ‘‘Transmission Blocking’’ assay. PLoSONE 2012; 7(4):e35019.doi:10.1371/ journal. pone. 0035019 E
- Drakeley C, Sutherland C, Bousema JT, Sauerwein RW and Targett AT: The epidemiology of Plasmodium falciparum gametocytes: weapons of mass dispersion. Trends Parasitol 2006; 22: 424-30.
- Babiker HA, Schneider P and Reece SE: Gametocytes: insights gained during a decade of molecular monitoring. Trends Parasitol 2008; 24: 525-30.
- White NJ: The role of anti‐malarial drugs in eliminating malaria. Malar J 2008; 7(Suppl 1): S8. Google Scholar.
- Greenwood BM: Control to elimination: implications for malaria research. Trends Parasitol 2008; 24: 449-54.
- Centers for Disease Control and Prevention: 2012. http://www.cdc.gov/malaria/about/biology/ Accessed February 25, 2015.
- Rudzinska MA and Trager W: Intracellular phagotrophy by malaria parasites: An electron microscope study of Plasmodium lophurae. Journal of Protozoology 1957; 4: 190-199.
- Rudzinska MA, Trager W and Bray RS: Pinocytotic uptake and the digestion of hemoglobin in malaria parasites. J Protozool 1965; 12: 563-576.
- Aikawa M, Hepler PK, Huff CG, Sprinz H: The feeding mechanism of avian malaria J Cell Biol 1966; 28(2): 355-373.
- Aikawa M: Plasmodium: the fine structure of malarial parasites. Exp Parasitol 1971; 30: 284-320
- Slomianny C, Prensier G and Vivier E: Ultrastructural study of the feeding process of erythrocytic Plasmodium chabaudi Mol Biochem Parasitol 1982; Suppl 695.
- Theakston RDG, Fletcher KA and Maegraith BG: The use of electron microscope autoradiography for examining the uptake and degradation of hemoglobin by Plasmodium falciparum. Ann Trop Med Parasitol 1970; 64: 63-71.
- Yayon A, Timberg R, Friedman S and Ginsburg H: Effects of chloroquine on the feeding mechanism of the intraerythrocytic human malaria parasite Plasmodium falciparum. J Protozool 1984; 31(3): 367-372.
- Sherman LW: Biochemistry of Plasmodium (malarial parasites). Microbiol Rev 1979; 43: 453-494.
- Goldberg, DE, Slater AFG, Cerami A and Henderson GB: Biochemistry Hemoglobin degradation in the malaria parasite Plasmodium falciparum: An ordered process in a unique organelle Proc. Natl. Acad. Sci. USA 1990; 87: 2931-2935.
- Suh K, Kain KC and Keystone JS: "Malaria." Canadian Medical Association Journal 2004; 170(11): 1693-1702.
- Ouédraogo AL, Guelbéogo WM, Cohuet A, Morlais I, King JG, Gonçalves BP et al: A protocol for membrane feeding assays to determine the infectiousness of falciparum naturally infected individuals to Anopheles gambiae. MWJ 2013; 4: 16.
- Bruce MC, Alano P, Duthie S and Carter R: Commitment of the malaria parasite Plasmodium falciparum to sexual and asexual development. Parasitology 1990; 100 (2): 191-200.
- Talman AM, Domarle O, McKenzie FE, Ariey F and Robert V: Gametocytogenesis: the puberty of Plasmodium falciparum. Malaria Journal 2004: 3(1).
- Silvestrini F, Alano P and Williams JL: Commitment to the production of male and female gametocytes in the human malaria parasite Plasmodium falciparum. Parasitology 2000; 121: 465-471.
- Smith TG, Lourenc¸OP, Carter R, Walliker D and Ranford-Cartwright LC: Commitment to sexual differentiation in the human malaria parasite, Plasmodium falciparum. Parasitology 2000; 91: 127-133.
- Smith TG, Walliker D and Ranford-Cartwright LC: Sexual differentiation and sex determination in the apicomplexan. Trends Parasitol 2002; 18(7): 315-323.
- Sinha A: Molecular basis of gametocytogenesis in malaria parasites. D. thesis. The School of Life Sciences College of Medical, Veterinary and Life Sciences University of Glasgow September 2014.
- Sinha A, Hughes KR, Modrzynska KK, Otto TD, Pfander C and Dickens NJ: A cascade of DNA-binding proteins for sexual commitment and development in Plasmodium. Nature 2014; 507; 7491: 253-257.
- Kafsack BF, Rovira-Graells N, Clark TG, Bancells C and Crowley VM and Campino SG: A transcriptional switch underlies a commitment to sexual development in malaria parasites. Nature 2014; 507; 7491: 248-52.
- Dyer M and Day KP: Commitment to gametocytogenesis in Plasmodium falciparum. Parasitol Today 2000; 16: 102-107.
- Alano P: Plasmodium falciparum gametocytes: still many secrets of a hidden life. Mol Microbiol 2007; 66: 291-302.
- Smalley ME and Brown J: Plasmodium falciparum gametocytogenesis stimulated by lymphocytes and serum from infected Gambian children. Trans R Soc Trop Med Hyg 1981; 75: 316-317.
- Ono T, Nakai T and Nakabayashi T: Induction of gametocytogenesis in Plasmodium falciparum by the culture supernatant of hybridoma cells producing anti- Falciparum antibody. Biken J 1986; 29: 7781-7786.
- Buckling A and Read AF: The effect of partial host immunity on the transmission of malaria parasites. Proc R Soc Lond B Biol Sci 2001; 68: 2325-2330.
- Lingnau A, Margos G, Maier WA and Seitz HM: The effects of hormones on the gametocytogenesis of Plasmodium falciparum in-vitro. Appl Parasitol 1993; 34(3): 153-60.
- Trager W and Gill GS: Enhanced gametocyte formation in young erythrocytes by Plasmodium falciparum in-vitro. J Protozool 1992; 39: 429-432.
- Trager W, Gill GS, Lawrence C and Nagel RL: Plasmodium falciparum: enhanced gametocyte formation in-vitro in reticulocyte-rich blood. Exp Parasitol 1999; 91: 115-118.
- Drakeley C, Secka I, Correa S, Greenwood BM and Targett GA: Host hematological factors influencing the transmission of Plasmodium falciparum gametocytes to Anopheles gambiaes. Mosquitoes. Trop Med Int Health. 1999; 4: 131-138.
- Price R, Nosten F, Simpson JA, Luxemburger C, Phaipun L and ter Kuile F: Risk factors for gametocyte carriage in uncomplicated falciparum malaria. Am J Trop Med Hyg 1999; 60: 1019-1023.
- Buckling A, Ranford-Cartwright L, Miles A and Read AF: Chloroquine increases Plasmodium falciparum gametocytogenesis in-vitro. Parasitology 1999; 118: 339-346.
- Taylor LH, Walliker D and Read AF: Mixed-genotype infections of the rodent malaria Plasmodium chabaudi are more infectious to mosquitoes than single-genotype infections. Parasitology. 1997; 115: 121-132.
- Williams JL: Stimulation of Plasmodium falciparum gametocytogenesis by conditioned medium from parasite cultures. Am J Trop Med Hyg 1999; 60: 7-13.
- Schneweis S, Maier WA and Seitz HM: Haemolysis of infected erythrocytes – a trigger for the formation of Plasmodium falciparum gametocytes? Parasitol Res 1991; 77: 458-460.
- Peatey CL, Skinner-Adams TS, Dixon MW, McCarthy JS, Gardiner DL and Trencholme KR: Effect of antimalarial drugs on Plasmodium falciparum J Infect Dis 2009; 200:1518-1521.
- Dutta GP, Bajpai R and Vishwakarma RA: Artemisinin (qinghaosu) - a new gametocytocidal drug for malaria. Chemotherapy 1989; 35: 200-207.
- Drakeley CJ, Jawara M, Targett GA, Walraven G, Obisike U and Coleman R: Addition of artesunate to chloroquine for treatment of Plasmodium falciparum malaria in Gambian children causes a significant but short-lived reduction in infectiousness for mosquitoes. Trop Med Int Health 2004; 9: 53-61.
- Targett G, Drakeley C, Jawara M, von Seidlein L, Coleman R and Deen J: Artesunate reduces but does not prevent posttreatment transmission of Plasmodium falciparum to Anopheles gambiae. J Infect Dis 2001; 183: 1254-1259.
- Kiszewski, AE: Blocking Plasmodium falciparum malaria transmission with Drugs: The gametocytocidal and sporontocidal properties of current and prospective antimalarials. Pharmaceuticals 2011; 4: 44-68.
- Young JA, Fivelman OL, Blair PL, de la Vega P, Le Roch KG and Zhou Y: The Plasmodium falciparum sexual development transcriptome: a microarray analysis using ontology-based pattern identification. Mol Biochem Parasitol. 2005; 143: 67-79.
- Ono T, Ohnishi Y, Nagamune K and Kano M: Gametocytogenesis induction by Berenil in cultured Plasmodium falciparum Exp Parasitol, 1993; 77: 74-78.
- Puta C and Manyando C: Enhanced gametocyte production in Fansidar-treated Plasmodium falciparum malaria patients: implications for malaria transmission control programmes. Trop Med Int Health 1997; 2: 227-229.
- Warhurst DC, Craig JC, Adagu IS, Meyer DJ and Lee SY: The relationship of physicochemical properties and structure of the differential antiplasmodial activity of the cinchona alkaloids. Malar J 2003; 2: 26.
- Hempelmann E and Egan TJ: Pigment biocrystallization in Plasmodium falciparum. Trends Parasitol 2002; 18: 11.
- Hempelmann E: Hemozoin Biocrystallization in Plasmodium falciparum and the antimalarial activity of crystallization inhibitors. Parasitol Res 2007; 100: 671- 676.
- Ezzet F, van Vugt M, Nosten F, Looareesuwan S and White NJ: Pharmacokinetics and pharmacodynamics of lumefantrine (benflumetol) in acute falciparum malaria. Antimicrob Agents Chemother 2000; 44: 697-704.
- Chong CR and Sullivan DJ: Inhibition of heme crystal growth by antimalarials and other compounds: implication for drug discovery. Biochem Pharmacol 2003; 66:2201–2212.
- Tekwani BL and Walker LA: Targeting the hemozoin synthesis pathway for new antimalarial drug discovery: technologies for in vitro β-hematin formation assay. Comb Chem High Throughput Screen 2005; 8: 61-67.
- Sullivan DJ Jr., Gluzman IY, Russell DG and Goldberg DE: On the molecular mechanism of chloroquine’s antimalarial action. Proc Natl Acad Sci USA 1996; 93(21): 11865-11870.
- Delfino RT, Santos-Filho OA and Figueroa-Villar JD: Type 2 antifolates in the chemotherapy of falciparum malaria. J Braz Chem Soc 2002, 13(6): 727-741.
- Robert V, Awono-Ambene H, Le Hesran J and Trape J: Gametocytemia and infectivity to mosquitoes of patients with uncomplicated Plasmodium falciparum malaria attacks treated with chloroquine or sulfadoxine plus pyrimethamine. Am J Trop Med Hyg 2000; 62: 210-216.
- Robert V, Molez J and Trape J: Short report: gametocytes, chloroquine pressure, and the relative parasite survival advantage of resistant strains of falciparum malaria in West Africa. Am J Trop Med Hyg 1996; 55: 350-351.
- von Seidlein L, Jawara M, Coleman R, Doherty T, Walraven G and Targett G: Parasitaemia and gametocytaemia after treatment with chloroquine, pyrimethamine/sulfadoxine combined with artesunate in young Gambians with uncomplicated Trop Med Int Health 2001; 6: 92-98.
- Osorio L, Ferro BE and Castillo CM: Effects of Chloroquine and Sulfadoxine/Pyrimethamine on Gametocytes in Patients with Uncomplicated Plasmodium falciparum Malaria in Colombia. Rio de Janeiro 2002; 97(8): 1221-1223.
- Smalley ME and Sinden RE: Plasmodium falciparum gametocytes: their longevity and infectivity. Parasitology 1977; 74: 1-8.
- Coleman RE, Clavin AM, Schneider I, Song GH, Klein TA and Milhous WK: Transmission-blocking antimalarials prevent sporogony of Plasmodium falciparum and berghei in Anopheles stephensi mosquitoes. Am J Trop Med Hyg 1994; 50: 646-653.
- “Antimalarial Drugs; Primaquine.” http://quod.lib.umich. edu/m/medchem1ic/x-290/ primaquine .tif. University of Michigan Library Digital Collections. Accessed: February 25, 2015.
- Portela MJ, Moreira R, Valente E, Constantino L, Iley J and Pinto J: Dipeptide derivatives of primaquine as transmission-blocking antimalarials: the effect of aliphatic side-chain acylation on the gametocytocidal activity and the formation of carboxyprimaquine in rat liver homogenates. Pharm Res 1999; 16: 949-955.
- Bates MD, Meshnick SR, Sigler CI, Leland P and Hollingdale MR: In-vitro effects of primaquine and primaquine metabolites on exoerythrocytic stages of Plasmodium berghei. Am J Trop Med Hyg 1990; 42: 532-537.
- Vale N, Moreira R and Gomes P: Primaquine revisited six decades after its discovery. Eur J Med Chem 2009; 44: 937-953.
- Bolchoz LJC, Budinsky RA, McMillan DC and Jollow DJ: Primaquine-induced hemolytic anemia: formation and hemotoxicity of the arylhydroxylamine metabolite 6-methoxy-8- hydroxylaminoquinoline. J Pharm Exper Therapy 2001; 297: 509-515.
- World Health Organization, 2012. Global Malaria Programme. Single dose Primaquine as a gametocytocide in Plasmodium. http://www.who.int/malaria/ mpac/sep 2012/primaquine_single_dose_pf_erg_meeting_report_aug2012.pd
- Bloland PB: Drug resistance in malaria. World Health Organization 2001, Malaria Epidemiology Branch Centers for Disease Control and Prevention Chamblee, GA, United States of America WHO/CDS/CSR/DRS/2001.4. Accessed 19th February
- Schlesinger PH, Krogstad DJ and Herwaldt BL: Antimalarial Agents: Mechanisms of Action. Antimicrobial agents and chemotherapy 1988; 32(6): 793-798.
- Müller IB and Hyde JE: Antimalarial drugs: modes of action and mechanisms of parasite resistance. Future Microbiol 2010; 5(12): 1857-73.
- Sibley CH, Hyde JE and Sims PF: Pyrimethamine- sulphadoxine resistance in Plasmodium falciparum: what next? Trends in Parasitology 2001; 17: 582-588.
- Omar SA, Adagu IS, Gump DW, Ndaru NP and Warhurst DC: Plasmodium falciparum in Kenya: high prevalence of drug-resistance-associated polymorphisms in hospital admission with severe malaria in an epidemic area. Annals of Tropical Medicine and Parasitology 2001; 95: 661-669.
- Fehintola FA, Adedeji AA, Tambo E, Fateye BB, Happi TC and Sowunmi A: Cotrimoxazole in the treatment of acute uncomplicated falciparum malaria in Nigerian children: a controlled clinical trial. Clin Drug Invest 2004; 24: 149-155.
- Sowunmi A, FateyeI BA, Adedeji AA, Fehintola FA, Bamgboye AE, BabalolaII CP et: Effects of antifolates— Co-Trimoxazole and pyrimethamine-sulfadoxine — on gametocytes in children with acute, symptomatic, uncomplicated, Plasmodium falciparum malaria. Mem. Inst. Oswaldo Cruz. 2005; 100(4): 451-455.
- Puta C and Manyando C: Enhanced gametocyte production in Fansidar-treated Plasmodium falciparum Malaria Patients: implications for malaria transmission control programmes. Trop Med Int Health 1997; 2: 227-229.
- Barnes KI and White NJ: Population biology and antimalarial resistance: The transmission of antimalarial drug resistance in Plasmodium falciparum. Acta Trop 2005; 94(3): 230-240.
- Zhang JFA: Detailed Chronological Record of Project 523 and the Discovery and Development of Qinghaosu (Artemisinin). Yang Cheng Evening News Publishing Company 2005.
- Terkuile F, White NJ, Holloway P, Pasvol G and Krishna S: Plasmodium falciparum: In-vitro studies of the pharmacodynamic properties of drugs used for the treatment of severe malaria. Exp Parasitol 1993; 76: 85-95.
- O’Neill PM, Barton VE and Ward SA: The Molecular Mechanism of Action of Artemisinin-The Debate Continues. Molecules 2010; 15: 1705-1721.
- Wikipedia 2015. en.m.wikipedia.org/wiki/Artemisinin. Accessed 28th February
- Qigui L, Peter W and Mark H: The use of artemisinin compounds as angiogenesis inhibitors to treat cancer, research directions in tumor angiogenesis, Dr. Jianyuan Chai (Ed.), ISBN: 978-953-51-0963-1, InTech 2013. DOI: 10.5772/54109. Available from: http://www.intechopen. com/books/research-directions-in-tumor-angiogenesis/the-use-of-artemisinin-compounds-as-angiogenesis-inhibitors-to-treat-cancer.
- Meshnick SR, Taylor T and Kamchonwongpaisan S: Artemisinin and the antimalarial endoperoxides: from herbal remedy to targeted chemotherapy. Microbiological reviews 1996: 60: 301-315.
- Posner GH: Further evidence supporting the importance of and the restrictions on a carbon-centered radical for high antimalarial activity of 1,2,4 trioxanes like artemisinin. Journal of medicinal chemistry 1995: 38: 2273-2275.
- Meshnick SR, Thomas A, Ranz A, Xu CM and Pan HZ: Artemisinin (qinghaosu): the role of intracellular hemin in its mechanism of antimalarial action n. Molecular and biochemical parasitology 1991; 49: 181-189.
- Shandilya A, Chacko S, Jayaram B and Ghosh IA: plausible mechanism for the antimalarial activity of artemisinin: A computational approach. Sci Rep 2013; 3: 2513; DOI: 10.1038/srep02513.
- Haynes RK, Cheu KW, Li KY, Tang MMK, Wong HN and Chen MJ: A partial convergence in action of methylene blue and artemisinins: antagonism with chloroquine, a reversal with verapamil, and an insight into the antimalarial activity of chloroquine. Chem MedChem 2011; 6(9): 1603-1615.
- Lisewski AM, Quiros JP, Ng CL, Adikesavan AK, Miura K and Putluri N: Supergenomic Network Compression and the Discovery of EXP1 as a Glutathione Transferase Inhibited by Artesunate. Cell 2014; 158(4): 916-928.
- Doherty JF, Sadiq AD, Bayo L, Alloueche A and Milligan P: A randomized safety and tolerability trial of artesunate plus sulfadoxine-pyrimethamine versus sulfadoxine-pyrimethamine alone for treatment of uncomplicated malaria in Gambian children Trans R Soc Trop Med Hyg 1999; 93: 543-546.
- Drakeley CJ, Jawara M, Targett GA, Walravan G and Obiske U: Addition of artesunate to chloroquine for treatment of Plasmodium falciparum malaria in Gambian children causes a significant but short-lived reduction in infectiousness for mosquitoes. Trop Med Int Health 2004; 9: 53-61.
- Sutherland CJ, Ord R, Dunyo S, Jawara M and Drakeley CJ. Reduction of malaria transmission to Anopheles mosquitoes with a six-dose regimen of co-artemether. PLoS Med 2005; 2: e92. (doi: 10.1371/ journal.pmed.0020092).
- Tangpukdee N, Krudsood S, Srivilairit S, Phophak N, Chonsawat P and Yanpanich W: Gametocyte Clearance in Uncomplicated and Severe Plasmodium falciparum Malaria after Artesunate-Mefloquine Treatment in Thailand. J Parasitol 2008; 46(2): 65-70.
- Pukrittayakamee S, Chotivanich K, Chantra A, Clemens R, Looareesuwan S and White NJ: Activities of artesunate and primaquine against asexual and sexual stage parasites in falciparum malaria. Antimicrob. Agents 2004; 48: 1329-1334.
- White NJ: The role of antimalarial drugs in eliminating malaria. Malaria J. 2008; 7: S8.
- Bousema T, Okell L, Shekalaghe S, Griffin JT, Omar S and Sawa P: Sutherland, C: Revisiting the circulation time of Plasmodium falciparum gametocytes: molecular detection methods to estimate the duration of gametocyte carriage and the effect of gametocytocidal drugs. Malaria J 2010; 9: 136.
- Kumar N and Zheng H: Stage-specific gametocytocidal effect in-vitro of the antimalaria drug qinghaosu on Plasmodium falciparum. Parasitol. Res 1990; 76: 214-218.
- Chotivanich K, Sattabongkot J, Udomsangpetch R, Looareesuwan S, Day NPJ and Coleman RE.: Transmission-blocking activities of quinine, primaquine, and artesunate. Antimicrob. Agents Chemother 2006; 50: 1927-1930.
- Noedl H, Se Y, Schaecher K, Smith BL, Socheat D and Fukuda MM: Evidence of artemisinin-resistant malaria in Western Cambodia. The New England Journal of Medicine 2008; 359(24): 2619-262.
How to cite this article:
Johnson TO: Transmission blocking strategy for malaria eradication: the role of antimalarial agents. Int J Pharmacognosy 2015; 2(10): 484-93. doi: 10.13040/IJPSR.0975-8232.2(10).484-93.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.