THERAPEUTIC POTENTIAL OF NOVEL PHYTO-MEDICINE FROM NATURAL ORIGIN FOR ACCELERATED WOUND HEALING
HTML Full TextTHERAPEUTIC POTENTIAL OF NOVEL PHYTO-MEDICINE FROM NATURAL ORIGIN FOR ACCELERATED WOUND HEALING
Ryan Varghese and Vaibhav Shinde *
Bharati Vidyapeeth Deemed University, Poona College of Pharmacy, Pune - 411038, Maharashtra, India.
ABSTRACT: Nature has always provided a remedy for most ailments that man has ever faced; wounds are no such exception. The wound healing process is complex and consists of overlapping phases, mainly haemostasis, inflammation, proliferation, and remodeling. There is the interplay of various sub-stages, mediators, biomarkers, enzymes, growth factors to form a complex system. Several other conditions like oxygen/blood supply, nutrition, diabetes, infections, aging, obesity, and other diseases make this more perplexing. On this background, faster and better healing of the wound is of paramount importance. Humans have employed different phytomedicines for accelerating the healing of wounds. These phytomedicines are traditionally used natural actives employed in the treatment of wounds as a modality to accelerate wound healing. This article delineates the basics of the wound healing process along with the therapeutic potential of some phytomedicines.
| Keywords: |
Wound healing, Centella asiatica, Anana scomosus, Aloe vera, Comfrey, Ocimum
INTRODUCTION: The healing of wounds is a complex and dynamic process that is still being studied and not yet completely understood 1. The wounds and ulcerations are known to deeply affect the quality of life of the patients suffering from them and, in most common cases, amount up to an annual cost of $25 treatment 2. Plants having therapeutic properties of medicinal plants have been used across the globe for ages as a traditional treatment method for numerous diseases. For instance, there were about 65 herbs included in traditional Persian medicine, which is a holistic system of medicine, which provided valuable insights on the natural remedies 3.
Nearly 80% of the population residing in developed countries, including economic leaders like India and China, still employ traditional medicine as the treatment for a wide spectrum of diseases 4. The natural products derived from these medicinal plants have been proven to be an abundant source of bioactive compounds, of which many have been employed to start the development of new chemicals for the pharmaceutical company 5.
The Continuum of the Skin: The human skin is composed of three distinct layers, namely, the epidermis, dermis, and hypodermis, with a varying level of adeptness within each layer. The epidermis and dermis are well characterized, but not much attention and research have been dedicated to the hypodermis and retinacula of the dermal tissues 6. The basement membrane is a complex assembly, primarily comprising of Type-IV collagen, laminin, nidogen/entactin, and heparin Sulphate proteo-glycans.
Additionally, other proteins, particularly the Anchoring fibril collagen type-VII, help attach the reticular lamina to the basal lamina in the basement membrane, which further promotes epidermis to dermal adhesion 7. The comprehensive proteomic analysis of the micro-dissected skin has discovered that there exist nearly 155 different proteins in the leg dermal samples in contrast to the 174 different proteins found in the breast tissue, and these proteins in collaboration with the collagen (I, II, III, VI, XII, and XIV), and extracellular matrix proteins (major ones include elastin, mimecan, prolargin, decorin, lumican, and periostin), cellular proteins (actin, myosin, vimentin, laminin, tubulin, desmo-plakin, annexins, histones, and 14-3-3 protein) and keratins [Includes both (Type-I cytoskeletal 9, 10, 13, 14, 15 and 16) and (Type-II cytoskeletal1, 2, 5 and 75)] 8.
In the forthcoming years, these targets can provide valuable leads and boost research in the area of wound healing.
The Epidermis: Although the skin as a whole actively participates in the host defense mechanisms, the epidermis is always regarded as more important in preventing and minimizing the loss of water and other components of the body into the external environment (inside-outside barrier) with a combined effect of protecting the body from a variety of environmental stresses (outside-inside barrier) 9.
The raison d’etre of the epidermal layer spans from limiting passive water loss from the body, reducing the absorption of chemicals and radiations from the immediate environment to the prevention of microbial infections. These defensive functions reside primarily in the top stratum of the epidermis, referred to as the stratum corneum (SC), at which they are integrated with the formation of the stratum corneum and its homeostasis. Thus the proper growth, development, and maintenance of homeostasis are linked to the ability of the body or the individual to defend against both chemical and microbiologic attacks as well as dehydration 10. The epidermis is further composed of numerous layers or strata, initiating from the basal layer (basement membrane or stratum basales) just positioned above the dermis proceeding upward layer by layer passing through the prickle layer (stratum spinosum) and the granular layer (stratum granulosum) and clear layer (stratum lucidum) to the uppermost layer the horny layer (stratum corneum) 11. The physical barrier is mainly formed and defined by the integrity of the stratum corneum, although the cellular junctions and cellular proteins and formed cytoskeleton in the subsequent layers may provide further protection and serve the purpose 12. The biochemical basis of the latter consists of a complex integration of numerous lipids, acids, peptides (antimicrobial and structural), and hydrolytic enzymes. The immuno-logic barrier is formed by an immense repertoire composed of both the humoral and cellular constituents (tissue-resident sentinel cells) of the immune system 13.
Dermis: The dermis is a heterogeneous layer that is composed of collagen and elastic fiber and is innervated by a rich vascular network and sensory nerve endings for sensing a tactile response. It also houses numerous sweat glands and hair follicles characterized by specialized epithelial invaginations. The primary functions of the dermis comprise of sweating (eccrine sweat glands innervated by the cholinergic fibers of the sympathetic nervous system), thermal sweating (response to increases in body temperature), emotional sweating (response to pain or emotional stress), and initiation and conduction of a tactile response 14. The mammalian dermis is akin to an archetypal mesenchymal tissue, comprising majorly of type-I and type-III collagen, but also contains several other subtypes of collagen, proteoglycans, and elastin which functions as a structural scaffold, and a basement membrane rich in type-IV collagen 15.
The dermis is histologically bifurcated into a superficial layer, termed as the papillary dermis, which lies immediately at the end of the epidermis, and is known to be comprised of dense but poorly oriented bundles of collagen, which attributes to its structural support delivered to the overlying basement membrane. This layer lies over the reticular dermis, which has some intervening spaces between the directionally oriented and relatively thicker bundles of collagen fibers. The components of the Extracellular Cellular Matrix (ECM) on the ultrastructural level that differentiates between the papillary and reticular dermis. This has further led to many different studies regarding the composition of the Extracellular Matrix (ECM) reflects the differences in the fibroblast identity in the tissue compartments 16.
The Hypodermis: The innermost part of the human skin, the hypodermis is regarded as a subcutaneous fat compartment and thus functions as a natural mechanical shock absorber, and helps in thermoregulation by protecting the body against external heat and cold, and is active in general energy metabolism and storage. It mainly consists of loose connective tissue, which regarding the site, differentiates into gliding layers or large adipose pockets, despite performing the same function. The tissue is particularly abundant in the content of proteoglycans and glycosaminoglycans, which helps in attracting fluid in the tissue, giving the tissue its characteristic mucous-like property 17. The majority of the cells that are conspicuous in the hypodermis include fibroblasts, adipocytes, and macrophages, which have a niche role in adipocyte homeostasis, fat tissue remodeling, and in certain cases, even stimulate thermogenesis of fat in response to cold exposure or exercise 18. This study and the thorough study of these layers of the skin are of paramount importance to unravel the mechanisms involved in increasing the drug transport across the skin and the effect of vesicles on the drug permeation rate, the permeation path-way, and the ultrastructure of the skin perturbations are to be thoroughly analyzed 19.
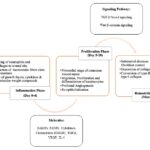
Physiology of Wound Healing: On the occurrence of a skin-deep or superficial injury, an innumerous cascade of systems are activated to primarily exterminate the foreign materials, essentially as the primary protective function of the skin membrane has been compromised, and to reinstate the normal functioning of the cutaneous membrane. It is also evident only to a finite degree, as the wound will never reach the maximum tensile impregnability as that of the unwounded one, and at best, would only make up to near 70% of the latter 20.
Inflammation Phase of Wound Healing: The process of repair and regeneration begins instantly after the injury and is facilitated by the release of a multitude of Growth Factors, Cytokines, and certain other Low-Molecular-Weight compounds from the serum of the affected blood vessels and degranulating platelets. The consecutive derange-ment of blood vessels also paves the way for the formation of blood thrombus explicably comprising cross-linked fibrin protein and proteins of the extracellular matrix such as Fibronectin, Vitronectin, Hyaluronan, and Thrombospondin 21.
However, in cases excluding the hemorrhage, the platelets are impotent to wound healing, so innumerable vasoactive mediators and chemotactic factors are procreated by the coagulation and by the injured parenchymal cells, which facilitate the inflammatory leucocytes to the site of injury 22.
Also, during this phase of hemostasis, the neutrophils and the macrophages traverse to the injured tissue and generate chemical inflammatory mediators, such as Interleukin-4 and Transforming Growth Factor β (TGF-β). Moreover, the ecstatic levels of the Reactive Oxygen Species within the blood components, specifically within the neutron-phils and the macrophages, set to initiate the extrinsic coagulation pathway by invoking the platelets to release coagulation factors which fundamentally construct the hemostatic fibrin clots, which serve as supporting framework or scaffold for the migration of cells into the wounds, thus acting as a temporary reservoir of the cells during wound healing. The inflammatory sequence is marked by termination induced by chemotactic signals including lipoxins and TGF-β 23.
Proliferation Phase of Wound Healing: The proliferative phase is marked by the primordial stages of cutaneous wound repair, where the new stroma is alternatively addressed as the granulation tissue, concerning the granular appearance of the budding tissue when examined microscopically, begins to develop approximately 4-5 days after the injury. This tissue is endowed by the infiltration of the numerous new capillaries alongside its granular appearance. The macrophages still provide a perpetual source of cytokines, which are considered pivotal for the stimulation of fibroplasia and angiogenesis 24.
This and certain other cellular processes of the blood stimulate the traversing of endothelial cells into the injury via the production of Vascular Endothelial Growth Factor (VEGF) and certain other factors, resulting in profound angiogenesis, which facilitates the hypervascular character of the newly formed granulation tissue. The profound re-epithelialization is also marked during this stage, which primarily involves the migration, proliferation, and differentiation of keratinocytes which are acquired from both the injured site and the stem cell populations found in the hair follicular region 25.
Remodeling Phase of Wound Healing: This is the final and the most prolonged stage of wound healing that is characterized by the decrease in the fibroblast content with a simultaneous deposition of collagen, and may usually span anywhere between 3 weeks to 2 years, depending upon a plethora of factors which are known to have a pronounced effect on the healing process. The intact skin comprises both type-I and type-III collagen, in contrast to the granulation tissue, which shows a more type-III collagen content, which is more unorganized and haphazard in its arrangement 26.
This phase shows a steady transition in the conversion of the skin from type-III to a more of type-I collagen content, with the subsequent disposition of the cells from the primitive stages.
This collagen remodeling entails Matrix Metallo-Proteinases (MMPs) and altered collagen synthesis to yield a scar. The tensile strength of the injured tissue increases, thereby regaining about 40% of its original strength by 1 month and approximates to 70% of the latter by the conclusion of 1 year 27.
Notable chronic wounds are a matter of grave concern, primarily for patients ailing with co-morbidities like diabetes mellitus, as most of the wound infections affect the foot wound and especially in neuropathic ulceration. The associated risk factor predominantly stated as for a duration of 1 month and a history of recurrent foot ulcerations, causing it to be the most renowned cause of lower extremity amputation, hence comprising a conspicuous load on both the physicians and the patients 28.
FIG. 1: PHYSIOLOGY OF WOUND HEALING ALONG WITH EMERGING TARGETS
Natural Actives in Wound Healing: Nature has been a consistent source for a multitude of medicinal treatment regimens, and plant-based systems continue to play a key role in primary health care around the globe, with many natural and nature-derived constituents being used in skin and wound care primarily due to their proven anti-inflammatory, anti-microbial and cell-proliferative properties 29. During the last two decades, there has been a phenomenal increase in the number of the plants that have been assessed for phytoconstituents and plant extracts that promote or modulate healing effects; the majority of them were assessed for anti-microbial, anti-oxidant, and angiogenic properties 30. Some of the most widely accepted and studied plants for their active-constituents and active Phytoextracts are enlisted below:
Centella asiatica: The Centella asiatica, popularly known as the Indian Pennywort or Asiatic Pennywort, has been used for ages as a traditional herbal medicine in a multitude of the Asiatic countries in the Asian sub-continent, as a use in the wound healing process, and it has recently gained a lot of popularity in the western countries 31. The plant species are known to find their presence in most tropical and subtropical countries, pertaining to their conducive growth conditions, including the swampy areas. These parts include most regions, including most areas in India, Pakistan, Madagascar, Sri-Lanka and South Africa, and South Pacific and Eastern parts of Europe, to name a few 32. Centella asiatica belongs to the family apiaceae, but the different parts of the plant, where the whole plant extract is used in the assessment methods with animal wound models are applicable in the excision models in the normal as well as the Sprague-Dawley rats, in contrast to where the excision, incision and dead space in Wistar Albino rats are used as the experimental animals in the assessment of the aerial parts of the latter 30. The efficacy of the Centella asiatica in their promotion of the wound healing process was extensively researched both in-vitro and in-vivo and was found to promote incision type wounds and open wounds as represented by a greater collagen content and thickness of the epithelium 33.
A comparison of figures measured after both oral and subcutaneous modalities of administration of the madecassoside, asiaticoside, asiatica acid, and madecassic acid in the experimental studies on rats revealed a varying bioavailability varies between 30% and 50% 34.
The pharmacodynamic effects of the Centella asiatica species have been investigated and, in particular, a wound-healing, ulcer-protective, psy-choneurotic-pharmacological, antimicrobial, and as an antiviral effect of the Centella or asiaticoside extract 35.
In a multitude of experiments, including the in vivo studies on the human fibroblasts, the clinical and mechanical studies have been merged with their chemical and biochemical aspects, where the topical preparations of the Centella extracts are known to be associated with accelerated wound healing, especially in the abnormal conditions of the skin associated with a reduction of granuloma weight, and an increase in the force needed to produce rupture (Rupture-strength) of the wound tissue 36. Furthermore, advanced research has proven that a dose-dependent increase in the synthesis of collagen, intracellular fibronectin content, and combined with the increase in mitotic activity of the germ layer and the enlargement of the kerato-hyaline granules within the scarred tissue 37.
The active constituent named asiaticoside is known to enhance the induction of antioxidant enzymes at an early wound healing phase 38, with an independent increment in hydroxyproline, tensile strength, and total content of the collagen, which leads to better epithelialization 39. The treatment modality formed from the extract was marked by the profound increment of the cellular proliferation and subsequent synthesis of collagen at the wound area and was proved experimentally by the increase in the total content of DNA, proteins, and collagen content of the granulation tissues, which resulted from an increase in the faster epithelialization with a faster wound contraction rate 40. But, the oil-water partition coefficient of the asiaticoside and the marked low solubility of the latter is known to limit its applicability and reduced effect and bioavailability 41, as the asiaticoside is defined as a sparingly ionized pentacyclictriterpene with a notable molecular weight of 959.12 g/mol, which attributes to its lower aqueous solubility 42. A recent induction of the porous microsphere was established as a novel carrier for the sustained released delivery of the poorly soluble asiaticoside, with enhanced absorption and therapeutic efficacy, and is believed to be a promising topical preparation with excellent regenerative effects on the wounds, helping to shape the future of the wound therapy management 43.
Anana scomosus: Pineapple has been a part of traditional folk medicine for ages and continues to be present in many herbal preparations 44. Bromelain has been known as a chemical since 1875, and since then has been applied as a phytotherapeutic medical agent 45. The term bromelain was initially used as a substitution to describe a mixture of proteolytic enzymes and other non-enzymatic substances found in the Bromeliaceae species, particularly in pineapple (Anana scomosus L.) and its various other parts like the stem, fruits, and leaves 46.
Bromelain is a crude extract obtained from the pineapples which contain many natural active components, of which the most conspicuous are a group of numerous closely related proteinases, which both in-vitro and in-vivo portray profound anti-edematous, anti-inflammatory, anti-thrombotic and fibrinolytic effects. Bromelain has gained a lot of acceptance and compliance as a phytotherapeutic drug due to its safety and absence of the undesired side effects or adverse reactions even after oral administration, without any loss of efficacy 47. Recent studies and clinical trials have demonstrated the numerous positive outcomes of Bromelain for the prevention of post-operative and post-procedure ecchymosis and edema 48. Although it has been used as the therapy for many problems and has a multitude of pharmacological effects, the mechanism of its action remains unclear and uncomprehensive. However, many clinical trials have shown its efficacy in the therapy as a treatment regimen in the treatment of some disorders like chronic inflammation and autoimmune disorders, including osteoarthritis and rheumatoid arthritis 49. In-vitro, it has portrayed its ability to help in the modulation of the immune response in allergic conditions and modulating macrophages, Natural-Killer (NK) Cells, and T-cells. Furthermore, it has also been known to increase the secretions of IL-1b, IL-6, and TNF-a 50.
Mainly because of the aforementioned reasons, this is one of the few natural actives that has been approved in Europe for both its topical application and oral administration, particularly for surgical wounds, inflammation due to trauma, surgery, and even the debridement of deep burns 51. There are new researches produced in the last decade which demonstrate the acceleration of healing of the burn wounds, primarily due to the influence of Bromelain 52 primarily due to its antimicrobial and antioxidant properties 53.
Aloe vera: Aloe vera is a tropical herb that is cultivated in hot and dry climates and is distributed in the arid and semi-arid regions of Asia, Africa, and other few tropical zones 54. Aloe vera is a plant that belongs to the Liliaceae family that characteristically grows in hot and arid conditions. The mucilaginous tissue that exists at the center of the leaves in this plant, which is usually deemed as the aloe-gel, is used for numerous cosmetic and medicinal applications 55.
Aloe vera or yellow aloe as it is known, is a herbaceous and perennial plant with thick, succulent, and long leaves. The thistle is also formed, which is a little curl in the margin of its leaves. Aloe vera is endemic to the African regions, and it is also called Desert Lily (Hesperocallis) 56. The oldest usage of the Aloe vera plant as a treatment regimen for wound burnings and infections dates back to the Egyptians. It spread far and wide and was later adopted by the Greeks, Spanish and African populations, to name a few, to treat many more such diseases 57. It has been duly classified as a characteristic hot and dry humor in the classical medicine system of Iran, and its extract is used for medicinal purposes 58. Aloe vera is highly therapeutic, accounting for the presence of 75 potentially active constituents, including vitamins, enzymes, minerals, sugars, lignin, amino acids, saponin, and salicylic acid and their derivatives 59. It also exerts exemplary pain and inflammation relief properties due to its inherent prostaglandin and bradykinin-hydrolyzing enzymes, carboxypeptidase, and bradykinesia 60. It is also abundant in the polysaccharide, named Mannose-6-phosphate, which supports the re-epithelialization process and plays a key role in tissue reorganization, induces the proliferation of fibroblasts, and activates the collagen deposition, and accelerates the overall process of wound healing 61. Another potent polysaccharide, Acemannan, is known for its white-blood-cell activation property, which is of great importance in the wound healing process 62. The multitude of anthraquinones and certain other closely related compounds have profound antibacterial activity 63. Only minimally adverse reactions, like mild pain or a sense of discomfort without any statistical significance, were attributed to the use of Aloe vera both orally and topically 64. Additionally, nearly all of them associated with them were reversible in nature, and Aloe vera is generally very well tolerated 65.
Comfrey: The roots of the Comfrey (Symphytum officinale L.) plant have been native to the European traditional medicine regimen but have also extrapolated to some parts of Asia and South America. In the traditional medicine system, comfrey roots are used topically, particularly for wound treatment, joint disorders, and injuries pertaining to the musculoskeletal system, especially caused due to pyrrolizidine alkaloids that have been linked to liver toxicity (hepatotoxicity) and cancer-formation (carcinogenicity) 66. Several cellular mechanisms have been put forth to explain their mechanism of action, particularly the ones that target different intracellular signaling pathways triggered by NF-kB, AP-1, PPAR, Nrf2, and MAPKs 67. The active constituents have portrayed the key role in comfrey root, which primarily include allantoin, mucilaginous polysaccharides, phenolic compounds such as rosmarinic acid, chlorogenic acid, caffeic acid, and their derivatives and salts, glycopeptides, and triterpenesaponins 68. Comfrey also encompasses pyrrolizidine alkaloids namely, 7-acetylintermedine, 7-acetyllycopsamine, intermidine, lycopsamine, and symphytine which are usually attributed to their inherent hepatotoxic nature, and therefore newer medicinal products use pyrrolizidine alkaloid-depleted or absent extracts as active agents 69.
The recent results were published to shed light on a comparative study between three topical formulations, namely, carbomer gel, glycerol-alcoholic solution, and an O/W emulsion (soft lotion), which led to significant experimental differences, particularly due to its intensity and rate. The results showed that the emulsion induced the largest amount of re-epithelialization and had the furthest repair of damaged tissue. This could be evidenced by the collagen deposition increment from 40% to a humongous 240% from day 4 to 28, with a reduction in the cellular inflammatory infiltrate from 3% to 46%. Furthermore, an 8% emulsion of the prepared has presented the best efficacy 70.
Ocimum: Ocimum is a well-known plant in Indian medicine and has been one of the first plants to be introduced into the treatment regimen in the traditional medicine system. It exhibits different therapeutic effects such as repair properties and cytokine induction 71. The wound healing activity was assessed by the induction of the cold aqueous extract of Ocimum sanctum leaves along with its effect on the Tumour Necrosis Factor-a (TNF-a) using the excision model of wound repair in Wistar Albino rats. After application of the 10% extract of Ocimum sanctum in the petroleum jelly proved to be successful with an increased rate of epithelialization and an increased wound contraction 72. The extracts of Ocimum sanctum may also be efficacious in the treatment of the management of abnormal wound healing such as keloids and hypertrophic scars, owing to its high antioxidant activity 73. Currently, topical formulations, prepared by the incorporation of optimized Silver-Nanoparticles (AgNPs) into the Carbopol gel base, followed by in-vivo evaluation using the rat model of skin wound healing, showed a 96.20% wound healing activity and the inhibitory efficiency of prepared nano gel was akin to the commercial product against the Staphylococcus aureus, E. coli and Pseudomonas aeruginosa 74.
Furthermore, in several other studies, the extract-treated wounds were known to re-epithelialize faster, and the rate of wound contraction had significantly increased, followed by a notable increment in wet and dry granulation tissue weight, granulation tissue breaking strength, and hydroxyproline content in dead space wound model was seen. The extract is known to have greatly decreased the anti-healing activities of dexa-methasone in all wound models 75. The extract helps in the management of abnormal healing and hypertrophic scars as both the doses of alcoholic and aqueous extracts are known to significantly increase wound breaking strength, hydroxyproline, hexuronic acid, hexosamines, superoxide dismutase, catalase, reduced glutathione with the significantly decreased percentage of wound contraction and lipid peroxidation 76.
TABLE 1: NATURAL ACTIVES EMPLOYED IN WOUND HEALING
| S. no. | Botanical Name | Common Name | Family | Mechanism |
| 1 | Centella asiatica L. 71, 77, 78 | Indian Pennywort or Asiatic pennywort | Apiaceae | Procollagen type-III synthesis |
| 2 | Anana scomosus L. 79–81 | Pineapple | Bromeliaceae | Inhibition of pro-inflammatory prostaglandin synthesis |
| 3 | Aloe vera L. 59, 82–85 | Indian Aloe, Chinese Aloe, Burn Aloe | Asphodelaceae | Indirect Collagen synthesis stimulation by stimulation of growth factor receptors on fibroblast |
| 4 | Symphytum officinale L.
68, 70, 71, 86-88 |
Comfrey | Boraginaceae | NF-kB Pathway |
| 5 | Ocimum sanctum L. 71–76, 89, 90 | Holy Basil | Lamiaceae | Upregulation of TNF-a |
Advents and Clinical Trials of Natural Actives in Wound Healing: The list of all the recent clinical trials on the novel therapies of wound healing is appended as follows with reference to and was compiled as “Wound healing” and “extracts” on the official website “clinical trials. gov” and “Pub Med”.
TABLE 2: RECENT ADVANCEMENTS IN NATURAL ACTIVES EMPLOYED IN WOUND HEALING
| S. no. | Name | Condition | Intervention | Status | Reference |
| 1 | Effectiveness and tolerability of Fitostimoline Plus Cream and Gauze vs. Connettivina Bio Plus Cream and Gauze | · Wound Heal
· Wound Infection · Wounds and Injuries |
· Device: Fitostimoline Plus | Recruiting | NCT04596124
|
| 2 | Patch with Onion Extract and Allantoin for C-section Scar | · Cesarean Section;
· Dehiscence · Scar Keloid · Wound Heal |
· Device: Patch | Completed | NCT04046783
|
| 3 | Influencing Wound Healing Through the Application of Hyaluronic Acid with Perfluoro decalin and Physalis angulata extract after split skin removal from the thigh – A study in “Split Wound design” | · Wound Heal | · Drug: Ready Medical post-treatment
· Device: Conventional Treatment
|
Withdrawn | NCT03668665
|
| 4 | Efficacy and Safety of Gynostemma pentaphyllum (Dungkulcha) Extract on Alleviation of Chronic Stress-Induced Strain | · Chronic Stress
· Induced Strain |
· Dietary Supplement: Gynostemma pentaphyllum (Dungkulcha) Extract (400mg/day)
· Dietary Supplement: Placebo |
Completed | NCT03277833
|
| 5 | Effect of Vitisviniferaextract on oxidative stress, Inflammatory biomarkers, and Hormones in high Trained Subjects | · Athletic Injuries | · Dietary Supplements: Grape fruits
· (Vitis vinifera) extract in a capsule with 205 mg |
Unknown (Study has passed its completion date and status has not been verified in more than two years) | NCT01875497
|
| 6 | Efficacy of RPh201 applied topically for the treatment of hard to heal chronic ulcerated wounds | · Chronic Wounds | · Drug: RPh201, Botanical drug product
|
Terminated | NCT01429519
|
| 7 | Effect of Cranberry Extract on Infections in Burn Patients | · Burn | · Dietary Supplement: Cranberry Extract
· Drug: Inert Placebo Capsule |
Withdrawn | NCT01225107
|
The last two decades have witnessed a sea of changes in the field of natural product research 91. As the research in the field of wound healing is under continuous update, there are new targets and new treatment regimes as well. Many phyto-medicines are proving beneficial in wound healing 92, 93. On this background, there is no wonder natural actives are preferred. Because of the immense therapeutic potential of natural actives, they are employed as a part of various treatment modalities through ages.
CONCLUSION: The process of wound healing, being complex in nature, necessitates the use of novel therapeutics approaches for faster and better healing. With mounting evidence, these phyto-medicines are being extensively studied for extracting actives and use them as prologues for the development of numerous chemical moieties of medicinal and therapeutic value. There are many such phytomedicines that have been extensively studied but only a few were selected and elucidated above as these are currently studied for future utilization in medicaments and formulations. A tremendous effort has been put in to demonstrate the potential application and utility of the natural extracts and their isolated actives for developing formulations, which aims at their application in wound therapy and healing. In coming years, one can find the use of these phytomedicines in novel therapeutic forms for better wound healing.
ACKNOWLEDGEMENT: Authors are thankful to Bharati Vidyapeeth Deemed Universty, Poona College of Pharmacy, Pune.
CONFLICTS OF INTEREST: There are no Conflicts of Interest.
REFERENCES:
- Shai A and Maibach HI: Wound healing and ulcers of the skin: Diagnosis and therapy - The practical approach. Wound Healing and Ulcers of the Skin: Diagnosis and Therapy - The Practical Approach 2005.
- Karapanagioti EG and Assimopoulou AN: Naturally occurring wound healing agents: an evidence-based review. Curr Med Chem 2016; 23(29): 3285-21.
- Hosseinkhani A, Falahatzadeh M, Raoofi E and Zarshenas MM: An evidence-based review on wound healing herbal remedies from reports of traditional persian medicine. J Evidence-Based Complement Altern Med 2017; 22(2): 334-43.
- Thangapazham RL, Sharad S and Maheshwari RK: Phytochemicals in Wound Healing. Adv Wound Care. 2016; 5(5): 230-41.
- Georgescu M, Marinas O, Popa M, Stan T, Lazar V and Bertesteanu SV: Natural Compounds for Wound Healing. In: Worldwide Wound Healing - Innovation in Natural and Conventional Methods 2016.
- Wong R, Geyer S, Weninger W, Guimberteau JC and Wong JK: The dynamic anatomy and patterning of skin. Exp Dermatol 2016; 25(2): 92-98.
- Hashmi S and Marinkovich MP: Molecular organization of the basement membrane zone. Clin Dermatol 2011; 29(4): 398-411.
- Mikesh LM, Aramadhaka LR, Moskaluk C, Zigrino P, Mauch C and Fox JW: Proteomic anatomy of human skin. J Proteomics 2013; 84: 190-200.
- Baroni A, Buommino E, De Gregorio V, Ruocco E, Ruocco V and Wolf R: Structure and function of the epidermis related to barrier properties. Clinics in Dermatology 2012; 30(3): 257-262.
- Elias PM and Choi EH: Interactions among stratum corneum defensive functions. Experimental Dermatology 2005; 14(10): 719-26.
- Wickett RR and Visscher MO: Structure and function of the epidermal barrier. Am J Infect Control 2006; 34(10): S98-S110.
- Proksch E, Brandner JM and Jensen JM: The skin: An indispensable barrier. Exp Dermatol 2008; 17(12): 1063-72.
- Richmond JM and Harris JE: Immunology and skin in health and disease. Cold Spring Harb Perspect Med 2014; 4(12): a015339.
- Rutter N: The dermis. Semin Neonatol 2000; 5(4): 297-302.
- Parsonage G, Filer AD, Haworth O, Nash GB, Rainger GE and Salmon M: A stromal address code defined by fibroblasts. Trends in Immunology 2005; 26(3): 150-56.
- Lynch MD and Watt FM: Fibroblast heterogeneity: implications for human disease. Journal of Clinical Investigation 2018; 128(1): 26-35.
- Guimberteau JC, Delage JP, McGrouther DA and Wong JKF: The microvacuolar system: How connective tissue sliding works. J Hand Surg Eur Vol. 2010; 35(8): 614-22.
- KF, BP, HJM, PR, AVD and HSJ: Intrahepatic fat content correlates with soluble CD163 in relation to weight loss induced by Roux-en-Y gastric bypass. Obesity 2015; 23(1): 154-61.
- Bouwstra JA and Honeywell-Nguyen PL: Skin structure and mode of action of vesicles. In: Advanced Drug Delivery Reviews 2002; 54 Suppl 1: S41-S55.
- Han G and Ceilley R: Chronic Wound Healing: A Review of Current Management and Treatments. Advances in Therapy 2017; 34(3): 599-610.
- Burgoyne RD and Morgan A: Secretory granule exocytosis. Physiological Reviews 2003; 83(2): 581-632.
- Singer AJ and Clark RAF: Cutaneous wound healing. New England Journal of Medicine 1999; 341(10): 738-46.
- Zeng R, Lin C, Lin Z, Chen H, Lu W and Lin C: Approaches to cutaneous wound healing: basics and future directions. Cell and Tissue Research 2018; 374(2): 217-32.
- Tonnesen MG, Feng X and Clark RAF: Angiogenesis in wound healing. J Investig Dermatology Symp Proc 2000; 5(1): 40-46.
- Zielins ER, Atashroo DA, Maan ZN, Duscher D, Walmsley GG and Marecic O: Wound healing: An update. Regenerative Medicine 2014; 9(6): 817-30.
- Park JW, Hwang SR and Yoon IS: advanced growth factor delivery systems in wound management and skin regeneration. Molecules 2017; 22(8): 1259.
- Sun BK, Siprashvili Z and Khavari PA: Advances in skin grafting and treatment of cutaneous wounds. Science 2014; 346(6212): 941-45.
- Kaplani K, Koutsi S, Armenis V, Skondra FG, Karantzelis N and Tsaniras SC: Wound healing related agents: Ongoing research and perspectives. Advanced Drug Delivery Reviews 2018; 129: 242-53.
- Saini S, Dhiman A and Nanda S: Traditional Indian Medicinal Plants with Potential Wound Healing activity: A Review. Int J Pharm Sci Res 2016; 7(5): 1809-19.
- Ghosh PK and Gaba A: Phyto-extracts in wound healing. J Pharm Pharm Sci. 2013; 16(5): 760-820.
- Ataide JA, Cefali LC, Croisfelt FM, Shimojo AAM, Oliveira-Nascimento L and Mazzola PG: Natural actives for wound healing: A review. Phytotherapy Research 2018; 32(9): 1664-74.
- Ratz-Lyko A, Arct J, Pytkowska K. Moisturizing and antiinflammatory properties of cosmetic formulations containing Centella asiatica Indian J Pharm Sci. 2016; 78(1):27-33.
- Pytkowska K: Moisturizing and antiinflammatory properties of cosmetic formulations containing Centella asiatica extract. Indian J Pharm Sci 2016; 78(1):27-33.
- Ruszymah BHI, Chowdhury SR, Manan NABA, Fong OS, Adenan MI and Bin SA: Aqueous extract of Centella asiatica promotes corneal epithelium wound healing in-vitro. J Ethnopharmacol. 2012; 140(2): 333-38.
- Vogel HG, De Souza NJ and D’Sa A: Effect of terpenoids isolated from Centella asiatica on granuloma tissue. Acta Ther. 1990; 16(4): 285-98.
- Brinkhaus B, Lindner M, Schuppan D and Hahn EG: Chemical, pharmacological and clinical profile of the East Asian medical plant Centella asiatica. Phytomedicine 2000; 7(5): 427-48.
- Gohil KJ, Patel JA and Gajjar AK: Pharmacological review on Centella asiatica: A potential herbal cure-all. Indian J of Pharmaceutical Sciences 2010; 72(5): 546-56.
- Bylka W, Znajdek-Awizeń P, Studzińska-Sroka E, Dańczak-Pazdrowska A and Brzezińska M: Centella asiatica in dermatology: An overview. Phytotherapy Research 2014; 28(8): 1117-24.
- Shukla A, Rasik AM and Dhawan BN: Asiaticoside-induced elevation of antioxidant levels in healing wounds. Phyther Res 1999; 13(1): 50-54.
- Shukla A, Rasik AM, Jain GK, Shankar R, Kulshrestha DK and Dhawan BN: In-vitro and in-vivo wound healing activity of asiaticoside isolated from Centella asiatica. J Ethnopharmacol 1999; 65(1): 1-11.
- Suguna L, Sivakumar P and Chandrakasan G: Effects of Centella asiatica extract on dermal wound healing in rats. Indian J Exp Biol 1996; 34(12): 1208-11.
- Chan K, Zhang HW and Lin ZX: Treatments used in complementary and alternative medicine. In: Side Effects of Drugs Annual 2014; 31: 745-56.
- Shao W, Cao X, Shen L, Zhang F and Yu B: A Convergent Synthesis of the Triterpene Saponin Asiaticoside. Asian J Org Chem. 2017; 6(9): 1270-76.
- Chan K, Zhang HW and Lin ZX: Treatments used in complementary and alternative medicine. In: Side Effects of Drugs Annual. 2014; 36: 717-24.
- Rathnavelu V, Alitheen NB, Sohila S, Kanagesan S and Ramesh R: Potential role of bromelain in clinical and therapeutic applications (Review). Biomedical Reports 2016; 5(3): 283-88.
- Lourenço CB, Ataide JA, Cefali LC, Novaes LC d. L, Moriel P and Silveira E: Evaluation of the enzymatic activity and stability of commercial bromelain incor-porated in topical formulations. IJCS 2016; 38(5): 535-40.
- Taussig SJ and Batkin S: Bromelain, the enzyme complex of pineapple (Ananas comosus) and its clinical application. An update. J Ethnopharmacol 1988; 22(2): 191-203.
- Maurer HR: Bromelain: Biochemistry, pharmacology and medical use. Cellular and Molecular Life Sciences 2001; 58(9): 1234-45.
- Ho D, Jagdeo J and Waldorf HA: Is there a role for arnica and bromelain in prevention of post-procedure ecchymosis or edema? A systematic review of the literature. Dermatologic Surgery 2016; 42(4): 445-63.
- Akhtar NM, Naseer R, Farooqi AZ, Aziz W and Nazir M: Oral enzyme combination versus diclofenac in the treatment of osteoarthritis of the knee - A double-blind prospective randomized study. Clin Rheumatol 2004; 23(5): 410-15.
- Passali D, Passali GC, Bellussi LM, Sarafoleanu C, Loglisci M and Manea C: Bromelain’s penetration into the blood and sinonasal mucosa in patients with chronic rhinosinusitis. Acta Otorhinolaryngol Ital 2018; 38(3): 225-28.
- Muhammad ZA and Ahmad T: Therapeutic uses of pineapple-extracted bromelain in surgical care - a review. Journal of the Pakistan Medical Association 2017; 67(1): 121-25.
- Krieger Y, Bogdanov-Berezovsky A, Gurfinkel R, Silberstein E, Sagi A and Rosenberg L: Efficacy of enzymatic debridement of deeply burned hands. Burns 2012; 38(1): 108-12.
- Dutta S and Bhattacharyya D: Enzymatic, antimicrobial and toxicity studies of the aqueous extract of Ananas comosus (pineapple) crown leaf. J Ethnopharmacol 2013; 150(2): 451-57.
- Pazyar N, Yaghoobi R, Rafiee E, Mehrabian A and Feily A: Skin wound healing and phytomedicine: A review. Skin Pharmacology and Physiology 2014; 27(6): 303-10.
- Hashemi SA, Madani SA and Abediankenari S: The review on properties of Aloe vera in healing of cutaneous wounds. BioMed Research International 2015; 714216.
- Akinyele BO and Odiyi AC: Comparative study of vegetative morphology and the existing taxonomic status of Aloe vera J Plant Sci 2007; 2(5): 558-63.
- Baroroft A and Myskja A: Aloe vera : Nature’s Silent Healer. Farmakologi dan Terapi. 2010.
- Reynolds T: Aloes: the genus Aloe. Aloes: the genus Aloe. CRC Press, Boca Raton, FL. 2004.
- Burusapat C, Supawan M, Pruksapong C, Pitiseree A and Suwantemee C: Topical Aloe vera gel for accelerated wound healing of split-thickness skin graft donor sites: A double-blind, randomized, controlled trial and systematic review. Plastic and Reconstructive Surgery 2018; 142(1): 217-26.
- Takzare N, Hosseini MJ, Hasanzadeh G, Mortazavi H, Takzare A and Habibi P: Influence of Aloe Vera gel on dermal wound healing process in rat. Toxicol Mech Methods 2009; 19(1): 73-77.
- Steenkamp V and Stewart MJ: Medicinal applications and toxicological activities of Aloe products. Pharm Biol 2007; 45(5): 411-20.
- Boudreau MD and Beland FA: An evaluation of the biological and toxicological properties of Aloe barbadensis (Miller), Aloe vera. Journal of Environmental Science and Health - Part C Environmental Carcinogenesis and Ecotoxicology Reviews 2006; 24(1): 103-54.
- Tamura N, Yoshida T, Miyaji K, Sugita-Konishi Y and Hattori M: Inhibition of infectious diseases by components from Aloe Vera. Biosci Biotechnol Biochem 2009; 73(4): 950-53.
- Visuthikosol V, Chowchuen B, Sukwanarat Y, Sriurairatana S and Boonpucknavig V: Effect of Aloe vera gel to healing of burn wound a clinical and histologic study. J Med Assoc Thai 1995; 78(8): 403-09.
- Vogler BK and Ernst E: Aloe vera: A systematic review of its clinical effectiveness. Br J Gen Pract 1999; 49(447): 823-28.
- Nastić N, Borrás-Linares I, Lozano-Sánchez J, Švarc-Gajić J and Segura-Carretero A: Comparative assessment of phytochemical profiles of comfrey (Symphytum officinale) root extracts obtained by different extraction techniques. Molecules 2020; 25(4): 837.
- Chen L, Teng H, Jia Z, Battino M, Miron A and Yu Z: Intracellular signaling pathways of inflammation modulated by dietary flavonoids: The most recent evidence. Critical Reviews in Food Science and Nutrition 2018; 58(17): 2908-24.
- Seigner J, Junker-Samek M, Plaza A, D’Urso G, Masullo M and Piacente S: A Symphytum officinale root extract exerts anti-inflammatory properties by affecting two distinct steps of NF-κB signaling. Front Pharmacol 2019; 10: 289.
- Brauchli J, Lüthy J, Zweifel U and Schlatter C: Pyrrolizidine alkaloids from Symphytum officinale and their percutaneous absorption in rats. Experientia 1982; 38(9): 1085-87.
- Araújo LU, Reis PG, Barbosa LCO, Saúde-Guimarães DA and Grabe-Guimarães A, Mosqueira VCF: In-vivo wound healing effects of Symphytum officinale L. leaves extract in different topical formulations. Pharmazie 2012; 67(4): 355-60.
- Pazyar N, Yaghoobi R, Rafiee E, Mehrabian A and Feily A: Skin wound healing and phytomedicine: A review. Skin Pharmacology and Physiology 2014; 27(6): 303-10.
- Goel A, Kumar S, Singh DK and Bhatia AK: Wound healing potential of Ocimum sanctum with induction of tumor necrosis-α. Indian J Exp Biol 2010; 48(4): 402-06.
- Shetty S, Udupa S, Udupa L and Somayaji N: Wound healing activity of Ocimum sanctum with supportive role of antioxidant enzymes. Indian J Physiol Pharmacol 2006; 50(2): 163-68.
- Sood R and Chopra DS: Optimization of reaction conditions to fabricate Ocimum sanctum synthesized silver nanoparticles and its application to nano-gel systems for burn wounds. Mater Sci Eng C 2018; 92: 575-89.
- Udupa SL, Shetty S, Udupa AL and Somayaji SN: Effect of Ocimum sanctum on normal and dexamethasone suppressed wound healing. Indian J Exp Biol 2006; 44(1): 49-54.
- Shetty S, Udupa S and Udupa L: Evaluation of antioxidant and wound healing effects of alcoholic and aqueous extract of Ocimum sanctum Linn in rats. Evidence-based Complement Altern Med 2008; 5(1): 95-101.
- Paocharoen V: The efficacy and side effects of oral Centella asiatica extract for wound healing promotion in diabetic wound patients. J Med Assoc Thail 2010; 93(Suppl 7:S166-170.
- Shetty BS, Udupa SL, Udupa AL and Somayaji SN: Effect of Centella asiatica (Umbelliferae) on normal and dexamethasone-suppressed wound healing in Wistar albino rats. Int J Low Extrem Wounds 2006; 5(3): 137-43.
- Soheilifar S, Bidgoli M, Hooshyarfard A, Shahbazi A, Vahdatinia F and Khoshkhooie F: Effect of oral bromelain on wound healing, pain, and bleeding at donor site following free gingival grafting: a clinical trial. J Dent (Tehran) 2018; 15(5): 309-16.
- Wu SY, Hu W, Zhang B, Liu S, Wang JM and Wang AM: Bromelain ameliorates the wound microenvironment and improves the healing of firearm wounds. J Surg Res 2012; 176(2): 503-09.
- MacKay D and Miller AL: Nutritional Support for Wound Healing. Alternative Medicine Review 2003; 8(4): 359-77.
- Teplicki E, Ma Q, Castillo DE, Zarei M, Hustad AP and Chen J: The effects of Aloe vera on wound healing in cell proliferation, migration, and viability. Wounds 2018; 30(9): 263-68.
- Oryan A, Mohammadalipour A, Moshiri A and Tabandeh MR: Topical application of Aloe vera accelerated wound healing, modeling, and remodeling. Ann Plast Surg 2016; 77(1): 37-46.
- Molazem Z, Mohseni F, Younesi M and Keshavarzi S: Aloe vera gel and cesarean wound healing; a randomized controlled clinical trial. Glob J Health Sci 2015; 7(1): 203-9.
- Hekmatpou D, Mehrabi F, Rahzani K and Aminiyan A: The effect of Aloe vera clinical trials on prevention and healing of skin wound: A systematic review. Iranian Journal of Medical Sciences 2019; 44(1): 1-9.
- Hostanska K, Rostock M, Melzer J, Baumgartner S and Saller R: A homeopathic remedy from arnica, marigold, St. John’s wort and comfrey accelerates in vitro wound scratch closure of NIH 3T3 fibroblasts. BMC Complement Altern Med 2012; 12: 100.
- Staiger C: Comfrey: A clinical overview. Phytotherapy Research 2012; 26(10): 1441-48.
- Barna M, Kucera A, Hladíkova M and Kucera M: Randomized double-blind study: Wound-healing effects of a Symphytum herb extract cream (Symphytum uplandicum Nyman) in children. Arzneimittel-Forschung/Drug Res 2012; 62(6): 285-89.
- Freiesleben SH, Soelberg J, Nyberg NT and Jäger AK: Determination of the wound healing potentials of medicinal plants historically used in Ghana. Evidence-based Complement Altern Med. 2017; 9480791.
- Cohen MM: Tulsi - Ocimum sanctum: A herb for all reasons. Journal of Ayurveda and Integrative Medicine. 2014; 5(4): 251-59.
- Shinde V and Dhalwal K: Pharmacognosy: The Changing Scenario. Pharmacognosy Reviews 2007; 1(1): 1-6.
- Murkute AB and Shinde VM: Exploratory studies on diabetic wound healing potential of Cipadessa baccifera (roth.) miq International Journal of Pharmacognosy. 2019; 6(8): 277-86.
- Shinde V, Shende A and Mahadik K: Evaluation of antioxidant and wound healing potential of pomegranate peel gel formulation. International Journal of Pharma-cognosy 2020; 7(1): 23-28.
How to cite this article:
Varghese R and Shinde V: Therapeutic potential of novel phyto-medicine from natural origin for accelerated wound healing. Int J Pharmacognosy 2021; 8(1): 14-24. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(1).14-24.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
14-24
716
1140
English
IJP
R. Varghese and V. Shinde *
Bharati Vidyapeeth Deemed University, Poona College of Pharmacy, Pune, Maharashtra, India.
vaibhavshinde847@gmail.com
18 September 2020
24 January 2021
29 January 2021
10.13040/IJPSR.0975-8232.IJP.8(1).14-24
31 January 2021