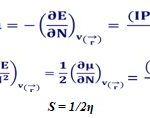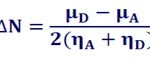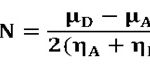THEORETICAL STUDIES OF THE ANTIOXIDANT PROPERTIES OF SOME FLAVONOID CLASS OF COMPOUNDS
HTML Full TextTHEORETICAL STUDIES OF THE ANTIOXIDANT PROPERTIES OF SOME FLAVONOID CLASS OF COMPOUNDS
Amit Kumar
Department of Chemistry, Dyal Singh College, University of Delhi, Lodi Road, New Delhi - 110003, Delhi, India.
ABSTRACT: Antioxidants play an essential role in the inhibition of the oxidative damage of the biomolecules. Flavonoids are known as potent antioxidants and are present in abundance in many of the fruits and vegetables besides being present in a large number of medicinal botanicals. A quantitative analysis of the antioxidant activity of quercetin, luteolin, kaempherol and taxifolin belonging to the flavonoid class of compounds, has been obtained by the determination of the structures, energetics and global reactivity descriptors in both neutral and charged states using Density Functional Theory. A comparison of the antioxidant property of these flavonoids, with those of the vitamins viz. vitamin A and vitamin E molecules, reveals that these flavonoid molecules are better candidates for electronic charge transfer to the ROS and hence possesses greater antioxidant activity. To probe the preferred site of reactivity, the Fukui function; fk- was also evaluated for each of these antioxidant molecules. The trends observed in various reactivity descriptors along with Fukui indices indicates that the substitution of B-ring and the C2-C3 double bond in the flavan structure is very important in determining the antioxidant properties of flavonoid molecules. The results obtained are important guidelines towards the detailed understanding of the antioxidant mechanism of these flavonoids.
| Keywords: |
Free radicals, ROS, Antioxidant, Flavonoids, Density Functional Theory
INTRODUCTION: Free radicals 1, 2 and their reactive metabolites are commonly known as reactive oxygen 1 species (ROS). Reactive oxygen species, produced in an organism, have an irreplaceable role in various biochemical reactions such as hydroxylating, carboxylating, peroxidation reactions and the reduction of ribonucleotides, besides playing important role in the transmission of nerve impulse 3 at fmol concentrations. Thus, under physiological conditions, a certain level of ROS is essential for metabolic processes of an organism.
Excess production of ROS gets triggered in response to certain stimulus. To deal with excess production of ROS, anti-oxidative protection mechanisms 4 are also in place in a healthy organism. Oxidative stress 4, 5 is a condition of imbalance between excessive production of ROS and the endogenous anti-oxidative protection mechanism of an organism. Under oxidative stress condition, the excessively produces ROS such as O2-•, •OH, •OOH, NO•, H2O2, •OONO, HOCl, and 1O2, etc., gets involved in the oxidative damage of important biological constituents, i.e. lipids, proteins and DNA and hence causes severe diseases.
Atherosclerosis and cardiovascular diseases, neurodegenerative diseases such as Alzheimer‘s and Parkinson‘s diseases, diabetes mellitus and metabolic syndrome, skin and tumor diseases, psychic impairments such as schizophrenia or ADHD (attention deficit hyperactivity disease) are some of the examples of diseases resulting from oxidative stress. Moreover, the physiological processes such as aging have also been associated with the action of excess production of ROS.
Thus, at the insufficiency of endogenous anti-oxidative protection mechanism, administration of exogenous antioxidants 6 can be used as an important strategy to counter oxidative stress and its related disorders in an organism. The study of antioxidants 1, 2, 7 is, therefore, an important area of research. An antioxidant is defined as “a substance that when present at low concentration, compared to an oxidizable substrate would significantly delay or prevent oxidation of the oxidizable substrate.” The oxidizable substrate, in a biological system, includes important biomolecules such as lipids, proteins, and DNA, etc. Antioxidants are active molecule and play an important role in the inhibition 8 of the oxidative damage of the biomolecules and are present in abundance in many of the fruits 9 and vegetables besides being present in a large number of medicinal botanical plants.
The antioxidant activity of these molecules is associated with the presence of several aromatic cycles and phenolic hydroxyl groups in their chemical structures. The antioxidant phenolic compounds have been divided into four main classes 8 and include (1) Phenolic acids, (2) Phenolic Diterpenes, (3) Flavonoids and (4) Volatile oils. Among these naturally occurring phenolic antioxidants, the flavonoids are the most important class of compounds. Some experimental studies have established the importance of flavonoids 10 in terms of their antioxidant activity. The antioxidant activity of flavonoids has been ascribed not only due to radical scavenging ability but also due to inhibition 11 of xanthine oxidase, an enzyme responsible for the production of superoxide free radicals.
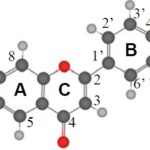
FIG. 1: 15-C FLAVAN STRUCTURE OF FLAVONOIDS COMPRISING RING A, RING B, AND RING C
Flavones, flavonols, dihydroflavonols, flavanols and flavanones represent the subclasses11 within the flavonoid class of compounds. The common characteristic of flavonoids is the presence of 15-Carbon flavan structure as shown in Fig. 1. The flavan structure comprises of three rings; ring A, ring B and the ring C. The level of saturation of the C ring is the basis of the difference between subclasses of flavonoids. Individual compounds within each of these subclasses are characterized by the substitution pattern, i.e., the number and location of phenolic-OH groups on the flavonoid skeleton.
This results in different radical scavenging ability of various flavonoids 11. Thus, a complete understanding of the structure-property relationship is essential to describe the difference in the antioxidant activity of different flavonoids.
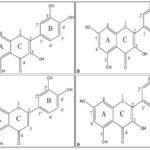
In the present study, the role of substitution pattern of ─OH groups in the three rings of the flavan structure, besides the role of C2-C3 double bond in the B ring makes an important objective of research about understanding the antioxidant activity of flavonoids. Theoretical methods, investigating the molecular geometry, the energy of molecules, chemical reactivity and hence antioxidant activity can be put to use as an apt solution to the current research quandary. With such objectives in mind we have carried out detailed first-principles density functional calculations of the structures and energetics, ionization potentials, electron affinities and various DFT-based global and local chemical reactivity descriptors of interest for quercetin, leuteolin, taxifolin, kaempherol anti-oxidant molecules, in both neutral and charged state. Fig. 2 shows the chemical structures of quercetin, leuteolin, taxifolin, kaempherol antioxidant molecules belonging to the flavonoid class of compounds being studied in the present paper. The results obtained are guidelines to help understand the mechanism of action of these potential flavonoids antioxidant molecules towards their antioxidant activity.
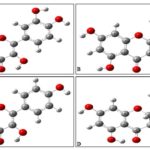
FIG. 2: MOLECULAR-STRUCTURES OF (A) QUERCETIN (B) LEUTEOLIN (C) KAEMPHEROL AND (D) TAXIFOLIN FLAVONOID MOLECULES
Methods: The determination of physical and chemical properties of various chemical species under study is being based on the first principal’s Quantum Mechanical methods. The first-principles quantum-mechanical methods help to obtain an accurate picture of what happens, structurally and dynamically, even at the atomic level.
We worked within the framework of first principles 12 to estimate the electronic structure of the antioxidant molecules by employing Beck’s three parameters exact exchange functional (B3). Combined with nonlocal gradient corrected correlation functional of Lee-Yang-Parr (LYP), denoted as B3LYP and the 6-311G basis sets. The structure of the antioxidant molecules was optimized via direct inversion in iterative subspace (DIIS), as implemented in the Gaussian 09 computational package until the largest component of the ionic forces attained a value 0.00045 au.
With an objective to analyze charge distribution, molecular orbital; MO plots were constructed from Gaussian checkpoint files of the optimized structures with the help of Gauss View 5 software. Natural population analysis (NPA) was performed to determine atomic charges. The ionization potential (IP) and electron affinity (EA) values were calculated using the finite difference approximation, and are given by:
IP = E(N-1) – E(N) & EA= E(N) – E(N+1)
E(N) represents the electronic energy of the N-electron system. The vertical IP (or EA) is calculated as the difference in total energies of the neutral and positively (or negatively) charged molecular species, with the molecular geometry being optimized in the neutral state. Conceptual density functional theory (DFT) is a very successful approach in providing a theoretical basis to obtain the global chemical descriptors such as electronegativity (χ), chemical potential (μ), chemical hardness (η), chemical softness (S) , and electrophilicity index (ω) as well as local chemical descriptors13. The global reactivity descriptors describe the reactivity of the molecule as a whole. The above specified global reactivity descriptors can be calculated as follows
HSAB concept may be employed to assess the reactivity of the antioxidant molecules towards ROS. According to the hard and soft acid-base (HSAB) concept, hard acids prefer to react with hard bases whereas soft acids prefer to interact with soft bases. The charge transfer parameter obtained the global interaction between the Antioxidant (donor) and ROS (acceptor); ΔN. The charge transfer parameter determines the fractional number of electrons, transferred from an antioxidant molecule; A to an acceptor system, i.e. ROS represented as “D,” and is given by:
Where, µ represents the electronic chemical potential and η represents the chemical hardness of the respective chemical species. For electronic charge transfer from the donor to acceptor, the ΔN value should be negative. The reactivity of sites on a different molecule can be studied using local softness. Local softness is related to the global softness S = 1/2η, and the Fukui functions ![]() through the relation,
through the relation,
The condensed Fukui functions may be obtained from local charges using the finite difference method:
Where, qk represents the electronic population of atom k in a molecule and can be obtained using NPA. For investigation of the effect of the environment (solvent effect) on these molecules, the environment was modeled using the Polarizable Continuum Model using the integral equation formalism variant (IEFPCM). The gas-phase structures were reoptimized in the presence of the solvent. While calculating the energy in water, the solvation energy of the electron (1.3 eV) has been subtracted. All the calculations were performed using Gaussian 09 computational package.
RESULTS AND DISCUSSION: The geometry optimization of chemical structures of the flavonoids; quercetin, taxifolin, luteolin and kaempherol, as well as ROS, was carried out in the gas phase at the B3LYP level using the 6-311G basis set. The geometrical parameters such as bond angles corresponding to each of the studied flavonoids are given in Table 1.
The optimized geometries of these quercetin, luteolin, kaempherol and taxifolin flavonoid molecules are shown in Fig. 3. Since, the electronic structure of a molecule is related to the chemical structure which in turn determine the chemical properties, the optimized geometries of these potential antioxidant molecules have been put to use to interpret and understand the electronic structures and hence properties of these molecules. Insight into the electronic and structural variations upon the acquisition of charge can be obtained by examining the molecular orbitals (MOs). Graphical representations of the highest occupied molecular orbitals (HOMO) and the lowest unoccupied molecular orbitals (LUMO) of these molecules are given in Fig. 4.
The vertical ionization potential is the difference in the total energies of the neutral and positively charged molecules, where their molecular geometries were optimized in the neutral state. On the other hand, vertical electron affinity represents the difference in the total energies of the neutral and the negatively charged molecules; here also the molecular geometries were optimized in the neutral state only. The vertical ionization potentials and the electron affinities for each of these potential antioxidant molecules as well as ROS, calculated using 6-311G basis sets, are given in Table 2. The calculated vertical IP values for these flavonoid molecules in the gas phase range from 7.214 to 7.616 eV. The vertical IP values calculated for ROS, •OH, (16.346 eV), and •OOH, (12.628 eV) are higher than the corresponding values for the flavonoid molecules under study.
TABLE 1: STRUCTURAL PARAMETERS; BOND ANGLES IN OPTIMIZED GEOMETRY OF QUERCETIN, LUTEOLIN, TAXIFOLIN AND KAEMPHEROL ANTIOXIDANT MOLECULES
| Molecule
Bond Angle |
Quercetin
(deg) |
Leuteolin
(deg) |
Taxifolin
(deg) |
Kaempherol
(deg) |
| C2− O1−C9 | 119.363 | 116.154 | 112.832 | 117.878 |
| O1− C2−C3 | 120.446 | 123.750 | 111.867 | 121.977 |
| O1− C2−C′1 | 110.253 | 110.152 | 106.350 | 112.450 |
| C3−C2−C′1 | 129.299 | 126.096 | 113.135 | 125.608 |
| C2−C3−C4 | 122.746 | 123.639 | 107.162 | 120.112 |
| C2−C3− O3 | 120.179 | − | 113.406 | 128.946 |
| O3−C3−C4 | 117.073 | − | 107.639 | 110.942 |
| C3−C4−O4 | 119.853 | 123.391 | 117.955 | 116.879 |
| C3−C4−C10 | 115.349 | 112.035 | 118.516 | 118.286 |
| O4−C4−C10 | 124.797 | 124.573 | 123.486 | 124.834 |
| O5−C5−C6 | 114.931 | 122.360 | 122.788 | 124.377 |
| O5−C5−C10 | 124.128 | 116.485 | 118.749 | 118.015 |
| C6−C5−C10 | 120.940 | 121.154 | 118.455 | 117.607 |
| C5−C6−C7 | 119.191 | 119.873 | 118.965 | 119.236 |
| C6−C7− O7 | 122.055 | 122.745 | 122.119 | 121.717 |
| C6−C7−C8 | 121.819 | 120.386 | 122.808 | 123.106 |
| O7−C7−C8 | 116.125 | 116.867 | 115.062 | 115.175 |
| C7−C8−C9 | 117.776 | 118.942 | 118.944 | 118.028 |
| O1−C9−C8 | 114.100 | 113.802 | 118.201 | 118.389 |
| O1−C9−C10 | 123.257 | 123.728 | 122.456 | 121.872 |
| C8−C9−C10 | 122.642 | 122.469 | 119.319 | 119.737 |
| C4−C10−C5 | 123.534 | 122.134 | 117.404 | 117.843 |
| C4−C10−C9 | 118.835 | 120.691 | 121.140 | 119.873 |
| C5−C10−C9 | 117.629 | 117.174 | 121.421 | 122.283 |
| C2−C′1−C′6 | 121.452 | 121.164 | 119.900 | 120.020 |
| C2−C′1−C′2 | 119.113 | − | 119.548 | 121.134 |
| C′2−C′1−C′6 | 119.433 | 119.561 | 120.528 | 118.844 |
| C′1−C′2−C′3 | 120.148 | 121.184 | 119.485 | 120.908 |
| C′2−C′3−C′4 | 119.847 | 119.163 | 119.582 | 119.900 |
| C′2−C′3−O′3 | 117.278 | − | 122.200 | − |
| C′3−C′4−O′4 | 116.324 | 127.414 | 112.007 | 117.072 |
| O′3−C′3−C′4 | 122.873 | − | 118.217 | − |
| C′3−C′4−C′5 | 119.919 | 120.481 | 120.632 | 119.573 |
| C′4−C′5−C′6 | 119.989 | 119.782 | 119.224 | 120.082 |
| C′1−C′6−C′5 | 120.661 | 119.830 | 120.548 | 120.690 |
| C′6−C′5−O′5 | − | 121.947 | − | − |
| C′4−C′5−O′5 | − | 118.269 | − | − |
| C′5−C′4−O′4 | 123.755 | 112.103 | − | 123.353 |
FIG. 3: OPTIMIZED GEOMETRIES STRUCTURES OF (A) QUERCETIN (B) LUTEOLIN (C) KAEMPHEROL AND (D) TAXIFOLIN FLAVONOID MOLECULES
FIG. 4: HOMO AND LUMO ISOSURFACES OF LUTEOLIN, TAXIFOLIN, KAEMPHEROL AND QUERCETIN ANTIOXIDANT MOLECULES IN THEIR NEUTRAL STATE
TABLE 2: CALCULATED IP, EA AND VARIOUS DFT BASED GLOBAL REACTIVITY DESCRIPTORS
| Molecules | Medium | IP(eV) Vert. | EA(eV) Vert. | χ = - μ | η | Ѕ | Ω |
| Quercetin | Gas | 7.214 | 0.653 | 3.933 | 3.280 | 0.152 | 2.358 |
| ε=78.36 | 5.659 | 2.301 | 3.981 | 1.679 | 0.298 | 4.719 | |
| Leuteolin | Gas | 7.573 | 0.308 | 3.941 | 3.633 | 0.138 | 2.138 |
| ε=78.36 | 6.074 | 2.120 | 4.097 | 1.976 | 0.253 | 4.246 | |
| Kaempherol | Gas | 7.837 | 1.046 | 4.217 | 3.170 | 0.158 | 2.804 |
| ε=78.36 | 5.845 | 2.677 | 4.261 | 1.584 | 0.316 | 5.731 | |
| Taxifolin | Gas | 7.616 | 0.909 | 4.263 | 3.353 | 0.149 | 2.709 |
| ε=78.36 | 6.060 | 2.608 | 4.334 | 1.726 | 0.289 | 5.440 | |
| •OH | Gas | 16.328 | 1.771 | 9.050 | 7.279 | 0.069 | 5.651 |
| ε=78.36 | 12.799 | 5.216 | 9.008 | 3.792 | 0.132 | 10.711 | |
| •OOH | Gas | 12.628 | 0.547 | 6.588 | 6.041 | 0.083 | 3.602 |
| ε=78.36 | 9.489 | 3.716 | 6.603 | 2.887 | 0.173 | 7.543 | |
| O2-• | Gas | 2.795 | -7.521 | -2.363 | 5.158 | 0.097 | 0.542 |
| ε=78.36 | 5.877 | 1.434 | 3.656 | 2.222 | 0.225 | 3.007 |
The electron transfer between the antioxidant and the radical can be determined from the IP and EA. A lower IP means a higher probability of losing an electron. It is clear from the trends observed in IP values that all these flavonoid molecules are capable of electronic charge transfer to the neutral ROS. The solvent was found to have an appreciable effect on the energetics of all the chemical species. Although there is an appreciable decrease in the IP in the presence of solvent, the order remains similar to that seen in the gas phase, i.e., Quercetin< Kaempherol < Leuteolin < Taxifolin. For neutral ROS, i.e. •, OH and •OOH, the average decrease in the vertical IP in the aqueous medium is 3.334 and 3.330, respectively.
As the solvent plays a crucial role in deciding the reactivities of the ionic species, the study was done in the aqueous medium for superoxide radial anion; O2-•. Experimental determination of the electron affinities (EA) for chemical systems such as biomolecules is a challenge. To the best of our knowledge, no experimental electron affinity data is available for these antioxidant molecules and ROS. Theoretical calculations of the EA’s become increasingly important as EA values are essential for evaluating reactivity descriptors and thus help in explaining phenomena such as donor–acceptor interaction between the antioxidant molecules and the ROS. The EA, along with the IP, has a crucial influence on the electron transfer between the antioxidant and the radical: a higher EA means a higher probability of gaining an electron. The DFT based methods are the most commonly employed methods for the theoretical determination of EAs. DFT method can be applied to a larger range of atoms and molecules than any other ab initio method. Besides the conventional ab initio methods, other theoretical methods for predicting EAs are Green’s function methods, methods based on the extended Koopmans’ theorem, electron propagator approximations, and calculations using known experimental half-wave reduction potentials. EAs obtained with DFT methods are fairly accurate (within 0.2 eVor less) in most cases. The calculated EA values for these molecules are found to be lower than those for the neutral ROS •OH and •OOH in the same media.
The lower IPs of these flavonoids than those of the neutral ROS and the higher EAs of the neutral ROS compared to those of these molecules support the antioxidant behavior of these flavonoid molecules. It is important to note that the presence of the medium (solvent) significantly influences the EA. The vertical EAs of these molecules are found to be positive in solvent-mediated studies. Interestingly, the EAs of ROS, including O2-•, in an aqueous medium is found to be positive, and thus indicates the possibility of the existence of O2-• in the aqueous medium. Although the EA of O2-• in the aqueous medium is positive, it is, however, found to be lower than those of the flavonoid molecules.
A quantitative analysis of the reactivity of the molecule was performed by determining the global reactivity descriptors using the accurately calculated vertical ionization potentials and electron affinities. The values of varied global reactivity descriptors, calculated for each of these molecules and the reactive oxygen species using the vertical IPs and EAs, are given in Table 2. Hardness (η) parameter is directly correlated with the stability of the molecule, whereas, softness (S) provides a measure of its reactivity. The calculated chemical hardness (η), for each of the chemical species, is found to be lower in the solvent medium than in the gas phase.
The calculated η values of these molecules lies in the range from 3.170 (1.584) for Kaempherol to 3.633 (1.976) for Luteolin in the gas phase (solution phase). The hardness parameter for these molecules follows the order; Leuteolin > Taxifolin > Quercetin, Kaempherol. The above-described trends are also followed in the solvent medium. The effect of solvent, is rationalized in terms of the difference in hardness, is maximum for Luteolin (1.657) and is minimum for Kaempherol (1.586).
The η values of the ROS •OH and •OOH are 7.279 and 6.041 eV, respectively, in the gas phase, which reduces to 3.792 and 2.887 eV in the aqueous medium. Hence, the presence of the solvent increases the reactivities of these flavonoid molecules as well as the ROS. Several mechanisms14 have been proposed in the literature for quenching of ROS by potential antioxidant molecules i.e. hydrogen atom transfer (HAT), single-electron transfer followed by proton transfer (SET-PT), and sequential proton loss electron transfer (SPLET) etc.
The common feature in these mechanisms is charge transfer. For a better understanding of a charge transfer reaction, the chemical reactivity descriptors such as the Mulliken electronegativity (χ) need to be taken into account. Electronegativity (χ), which is the negative of the electronic chemical potential (μ), is a measure of the tendency of the molecule to attract electrons. The calculated χ values for these flavonoid molecules are lower than those for the neutral ROS. The χ of •OH (~ 9 eV) in an aqueous solvent and the gas phase is found to be higher than the χ of •OOH (~ 6.6 eV) in different media.
This suggests that, among the various ROS, ·•OH has the greatest tendency to extract an electron from the antioxidant molecules and this explains its highly reactive nature. These findings are in line with the experimental observations which confirm the high reactivity 15 of the •OH with a very short in-vivo half life 16 of approx, 10-9 s. It is interesting to note that the presence of a solvent and the polarity of the solvent are found to have negligible effects on the electronegativities of these flavonoid molecules and neutral ROS, but they are found to have a considerable effect on the electronegativities of ionic reactive oxygen species such as O2-•.
The magnitude of electronic chemical potential (μ), which is a measure of the tendency of an electron to escape, is found to follow the order Quercetin > Leuteolin> Kaempherol> Taxifolin, in the gas phase as well as in the solvent phase. Electrophilicity index (ω), is another very useful reactivity descriptor and quantifies the tendency of a molecule to soak up the electrons.
Consequently, the higher the electrophilicity index, the greater is the propensity of the ROS to attract electrons from a generic donor molecule. Several studies have shown that this parameter allows quantitative classification of the global electrophilic character of a molecule within the reactivity scale.
The electrophilicity (ω) of ·•OH is 5.651, and 10.711 eV in the gas phase and in an aqueous medium, respectively. On the other hand, electrophilicities for the •OOH as obtained using the 6-311G basis set are 3.602, and 7.543 eV in the gas phase and aqueous medium, respectively. It is important to note that these electrophilicities are higher than those of all the flavonoid. This indicates the high propensity of the ROS to attract electrons from these flavonoid molecules. The high ω values of ·•OH are in line with its highly reactive nature, as mentioned earlier.
Global reactivity parameters, discussed above, are related to the properties of an isolated reactant. To take into account the selectivity vis-à-vis other species, intermolecular parameters must be taken into consideration. The global interaction between the antioxidant molecules and the ROS can be represented by the charge transfer parameter ∆N which determines the fractional number of electrons, transferred from an antioxidant molecule (A) to the reactive oxygen species (ROS) represented by the symbol; D, and is given by:
Where µA, µD and ηA, ηD are the chemical potentials and chemical hardness of a potential antioxidant molecule; A and the ROS; D respectively.
The amount of charge transfer between the antioxidant molecules (Quercetin, Luteolin, Taxifolin, and Kaempherol) and various ROS, as calculated using the ∆N formulae, is given in Table 3. A negative value of ∆N parameter indicates that the molecule under investigation acts as an electron donor to the ROS and hence exhibit antioxidant activity. As a result, the greater the magnitude of the ∆N parameter, the greater is the ability of the molecules to donate electronic charge to the ROS and thus exhibit greater antioxidant activity.
The magnitude of the charge transfer parameter for electronic charge transfer to •OH by these flavonoids follows the order Quercetin > Luteolin > Kaempherol > Taxifolin in both the gas phase and in the aqueous medium.
TABLE 3: CALCULATED CHARGE TRANSFER DESCRIPTORS OF QUERCETIN, LUTEOLIN, TAXIFOLIN AND KAEMPHEROL ANTIOXIDANT MOLECULES
| Gas phase | Solvent medium | |||||
| •OH | •OOH | O2-• | •OH | •OOH | O2-• | |
| Quercetin | -8.869 | -6.384 | -2.138 | -8.652 | -6.181 | -3.168 |
| Leuteolin | -8.864 | -6.377 | -2.129 | -8.644 | -6.167 | -3.146 |
| Kaempherol | -8.849 | -6.361 | -2.110 | -8.615 | -6.133 | -3.107 |
| Taxifolin | -8.848 | -6.359 | -2.109 | -8.611 | -6.126 | -3.096 |
The magnitude of ∆N, ranges from 8.869 for Quercetin to 8.848 for Luteolin in the gas phase, whereas, it varies from 8.652 to 8.611, in the aqueous medium. Similar trends were also observed, for variation in magnitude of ∆N parameter for electron transfer to other ROS, i.e. •OOH and O2-• from these flavonoid molecules. A comparison of the ∆N of these flavonoids shows that these flavonoids possess much more pronounced antioxidant activity. These flavonoids exhibit antioxidant activity also towards the ionic ROS O2-•.
The trends observed in all of these global reactivity descriptors along with charge transfer parameter, unanimously, predict the least antioxidant activity of the Taxifolin, among the four different flavonoids studied presently. This reveals that the C2−C3 double bond in the C ring of the flavan structure is very important for the antioxidant activity, since the C2−C3 double bond is not present in the Taxifolin. The substitution pattern of the phenolic –OH groups in the flavan structure is also an important consideration for the antioxidant activity. The importance of 5 –OH and 7 –OH groups in the A ring of the flavan structure is highlighted in literature 16 for inhibition of xanthine oxidase, an enzyme that catalyze the production of superoxide radical anion; O2-•. The structural parameters (bond angles) reveal the possible existence of hydrogen bonds between the 3΄–OH and 4΄–OH of the quercetin, luteolin and taxifolin molecules.
This observation is in consistency with the observations reported in the literature previously. The existence of hydrogen bonding between 5–OH, O atom of >C=O at C4 site and the 3–OH of Quercetin and Kaempherol molecules is by the structural parameters given in Table 1. The extent of hydrogen bonding in the flavan structure of these flavonoids plays its role in stabilization of the antioxidant radical formed as a result of electronic charge transfer to ROS and thus contributes towards the antioxidant activity of these flavonoids.
To probe the preferred site of reactivity of these flavonoids and the ROS, the Fukui function fk- values were evaluated at the primary hydroxyl group of these antioxidant molecules. These values are given in Table 4. The Fukui function fk-, indicates the decrease in negative charge from the site k (oxygen atom), upon the removal of one electron from the molecule by a ROS. The greater the magnitude of fk-, the greater is the loss in electronic charge on the oxygen atom and consequently the greater the increase in charge on the ROS. A comparing of the magnitude of fk- values at various −OH sites in the Quercetin, it is clear that the 3−OH, followed by 3΄−OH and 4́−OH, group is the preferred site for the electrophilic attack by the respective ROS because of the greater magnitude of fk- function as compared to other sites. The possibility of such an electrophilic attack of ROS on the Quercetin molecule at 3−OH has also been demonstrated as an experimental study on antioxidant properties of Quercetin reported recently. It has also been demonstrated that Quercetin radical species further react with O2-• leading to depside formation17 and favours the initial reactivity at 3−OH site of the Quercetin molecule. The magnitude of fk- for 4́−OH oxygen atom site in the B-ring is found to be greatest for Kaempherol amongst all the four studied flavonoid molecules. This clearly reveals the importance of B-ring substitution pattern of –OH group for antioxidant activity of the flavonoids. In addition, the importance of C2−C3 double bond in the flavan structure of the flavonoids is once again supported by the least value of fk- with respect to 3−OH oxygen atom of Taxifolin.
TABLE 4: FUKUI FUNCTION [fk-] VALUES AT DIFFERENT SITES OF QUERCETIN, TAXIFOLIN AND KAEMPHEROL ANTIOXIDANT MOLECULES
|
Site of Activity |
[f k-] | |
| Gas Phase | Solvent Phase (ε=78.36) | |
| Quercetin | ||
| 3' Oxygen atom | -0.115 | -0.137 |
| 4' Oxygen atom | -0.055 | -0.081 |
| 3 Oxygen atom | -0.067 | -0.070 |
| 5 Oxygen atom | -0.048 | -0.047 |
| 7 Oxygen atom | -0.038 | -0.026 |
| Taxifolin | ||
| 3' Oxygen atom | -0.057 | -0.051 |
| 4' Oxygen atom | -0.051 | -0.053 |
| 3 Oxygen atom | -0.003 | -0.003 |
| 5 Oxygen atom | -0.063 | -0.010 |
| 7 Oxygen atom | -0.021 | -0.005 |
| 3' Oxygen atom | -0.059 | -0.081 |
| Luteolin | ||
| 4' Oxygen atom | -0.055 | -0.073 |
| 3 Oxygen atom | -0.043 | -0.019 |
| 5 Oxygen atom | -0.040 | -0.023 |
| 7 Oxygen atom | -0.038 | -0.026 |
| Kaempherol | ||
| 4' Oxygen atom | -0.078 | -0.084 |
| 3 Oxygen atom | -0.074 | -0.053 |
| 5 Oxygen atom | -0.037 | -0.027 |
| 7 Oxygen atom | -0.066 | -0.037 |
CONCLUSION: We have carried out first principle’s density functional theory based calculations of the structures, energetics, ionization potential, electron affinity and various DFT based global chemical reactivity descriptors of interest for four different flavonoid antioxidants− Quercetin, Luteolin, Kaempherol and Taxifolin in both neutral and charged states. The results of our investigations confirm the rich antioxidant potential of the studied flavonoids. The order of the antioxidant activity, as predicted by the results obtained, i.e. Quercetin > Leuteolin > Kaempherol > Taxifolin, is in agreement with that reported by recent experimental studies 18.
The trends observed in various reactivity descriptors along with Fukui indices clearly indicates that the substitution of B-ring and the C2 C3 double bond in the C-ring of the flavan structure is very important in determining the antioxidant properties of flavonoid molecules. The results obtained are important guidelines towards the detailed understanding of the antioxidant mechanism of these flavonoids.
ACKNOWLEDGEMENT: The author is very grateful to the University of Delhi for financial support. This work was supported by the Innovation Research Project Grant-in-Aid of the University of Delhi (Project Code DS 102).
CONFLICT OF INTEREST: Nil
REFERENCES:
- Carocho M and Ferreira ICF: A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food & Chem Toxi 2013; 51: 15-25.
- Kunwar A and Priyadarsini KI: Free radicals oxidative stress and importance of antioxidants in human health. Journal of Medical and Allied Sciences 2011; 1(2): 53-60.
- Thomas DD, Heinecke JL, Ridnour LA, Cheng R, Kesarwala AH, Switzer CH, McVicar DW, Roberts DD, Glynn S, Fukuto JM, Wink DA and Miranda KM: Signalling and stress: The redox landscape in NOS biology. Free Radical Biology and Medicine 2015.
- Birben E, Sahiner UM, Sackesen C, Erzurum S and Kalayci O: Oxidative stress and antioxidant defense. WAO Journal 2012; 5: 9-19.
- Durackova Z: Some current insights into oxidative stress. Physiological Research 2010; 59: 459-469.
- Benfeito S, Oliveira C, Soares P, Farnandes C, Silva T and Borges F: Antioxidant therapy: Still in search of the magic bullet. Mitochondria 2013; 13: 427-435.
- Hamid AA, Aiyelaagbe OO, Usman LA, Ameen OM and Lawal A: Antioxidants: Its medicinal and pharmacological applications. African Journal of Pure and Applied Chemistry 2010; 4(8): 142-151.
- Brewer MS: Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. 2011; 10 (4): 221-247.
- Fu L, Xu B, Xu X, Gan R, Zhang Y, Xia E and Li H: Antioxidant capacities and total phenolic contents of 62 fruits. Food Chemistry 2011; 129: 345-350.
- Lago JHG, Toledo-Arruda AC, Mernak M, Barrosa KH, Martins MA, Tiberio IF and Prado CM: Structure-activity association of flavonoids in lung diseases. Molecules 2014; 19: 3570-3595.
- Kaur S and Mondal P: Study of total phenolic and flavonoid content, antioxidant activity and antimicrobial properties of medicinal plants. Journal of Microbiology & Experimentation 2014; 1(1): 5-10.
- Kumar V, Kishoe S and Ramaniah LM: First-principles DFT study of cyclic and acyclic nucleoside phosphonates. Structural chemistry 2014; 25 (6): 1725-1732.
- Chattaraj PK, Sarkar U and Roy DR: Electrophilicity index. Chemical Reviews 2006; 106: 2065-2091.
- Sutachan JJ, Casas Z, Albarracin SL, Stab BR, Samudio I, Gonzalez J, Morales L and Barreto GE: Cellular and molecular mechanisms of antioxidants in Parkinson’s disease. Nutritional Neuroscience 2012; 15 (3): 120-126.
- Shen H and Anastasio C: A comparison of hydroxyl radical and hydrogen peroxide generation in ambient particle extracts and laboratory metal solutions. Atmospheric Environment 2012; 46: 665-668.
- Pastor N, Weinstein H, Jamison E and Brenowitz M: A detailed interpretation of OH radical footprints in a TBPDNA complex reveals the role of dynamics in the mechanism of sequence-specific binding. Journal of Molecular Biology 2000; 304: 55–68.
- Trouillas P, Marsal P, Siri D, Lazzaroni R and Duroux Jean-L: A DFT study of the reactivity of OH groups in quercetin and taxifolin antioxidants-The specificity of the 3-OH site. Food Chemistry 2006; 97: 679-688.
- Rong Y, Wang Z, Wua J and Zhao Bo: A theoretical study on cellular antioxidant activity of selected flavonoids. Spectrochimica Acta Part A 2012; 93: 235-239.
How to cite this article:
Kumar A: Theoretical studies of the antioxidant properties of some flavonoid class of compounds. Int J Pharmacognosy 2015; 2(7): 351-61. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(7).351-61.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.