THE MECHANISTIC APPROACH OF THE PROPYLENE GLYCOL AS A CO-SURFACTANT THROUGH “SIX MEMBER CYCLIC HYDROGEN BONDING SYSTEM” FOR THE STABLE AND FLEXIBLE MICROEMULSION DRUG CARRIER
HTML Full TextTHE MECHANISTIC APPROACH OF THE PROPYLENE GLYCOL AS A CO-SURFACTANT THROUGH “SIX MEMBER CYCLIC HYDROGEN BONDING SYSTEM” FOR THE STABLE AND FLEXIBLE MICROEMULSION DRUG CARRIER
Pratiksha Saxena *, Pramod Kumar Sharma and Priyank Purohit
Department of Pharmacy, School of Medical and Allied Sciences, Galgotias University, Greater Noida, Gautam Budh Nagar - 201310, Uttar Pradesh, India.
ABSTRACT: The improvisation of any system has been a challenging task for the scientist in the field of drug delivery. In pursuit of the stable carrier system, here the microemulsion or other meta-state like liquid crystal phase with the specific mechanism, i.e., “six-member stable cyclic system” is highlighted and critically evaluated. The system with co-surfactant (propylene glycol) is examined and concluded with the plausible mechanism of hydrogen bond donor/acceptor felicitator to the surface active molecule by the hydrogen bonding with the surfactant polar head group to increase the fluidity of the system. Through this unique mechanism, the co-surfactant expands the area of existence of microemulsion. The system allows the topical drug with the stable as well as a flexible system with higher drug entrapment. The systems aligned toward the liquid crystal phase than the liquid phase, which makes the system more convenient for the delivery system.
| Keywords: |
Propylene glycol, Hydrogen bonding, Six member ring, Microemulsion
INTRODUCTION: The fundamental concepts of the emulsion 1 signify as a metastable dispersion, which consist of the dispersed phase (droplets of a liquid) and the dispersion medium (continuous phase of another liquid) present in the partially immiscible state 2. The above-mentioned definition gives incomplete sense without using the fascinating word called “surfactant” 3. The surface active player (surfactant) helps in the shearing of two immiscible liquids, results in the formation of stable Meta state dispersion called as an emulsion.
Emulsion exhibit the natural phenomenon of droplet coalescence as the two immiscible liquid are in an unstable state; hence its variability in terms of stability is potentially controlled by the surfactant by reducing the interfacial tension 4, 5. In context with the definition, the surfactant is the surface active agents with the major two essential fragments - a polar head (charged or uncharged) and the non-polar tail makes them an amphiphilic molecules 6.
The surface active molecules localized at the interface between the non-aqueous phases (like oil) and the aqueous phase, results in the reduction of the interfacial tension, due to the amphiphilic character of the agents. The various studies based on the surface active agents reveal the mechanism of the reduction of interfacial tension. The polar head orients towards the aqueous phase and the nonpolar region (tail) orients towards the non- aqueous phase. In the nutshell, the surfactant reduces the interfacial tension, which eventually reduces the interfacial free energy of the system (enthalpy); moreover, it also provides the strength to the interfacial film for the long term stability of the naturally unstable system 7.
As science and research describe a voyage of discovery, a movement from “known to unknown facts” and also to provide a better approach to solve the problem of the current technology. As emulsions are not thermodynamically stable to overcome the stability issues concerning the emulsion, a new concept had been evolved known as ‘Microemulsion”. The pioneering concept on transparent dispersion called oleophilic hydromicelle (W/O) was started in 1943. 8 The word microemulsion was coined by Schulman and co-participants in 1959 9 as clear and novel transparent system or dispersion system. They describe microemulsion as the homogeneous, clear, thermodynamically stable, and isotropic fluid that is optically active. The formation of the microemulsion is a spontaneous process; consist of transparent dispersion of aqueous and nonaqueous (water and oil) phases. This system comprises of spherical droplets of the uniform shape of either oil, in which water is a continuous phase or water droplets in which oil is in the continuous phase.
Every system comprises of essential components that have a positive impact on the property of the system and makes them aesthetically unique and novel from the other system. Microemulsion consists of four important components with the partially biphasic system, i.e. an aqueous phase, non-aqueous phase (oil), surfactant, and the co-surfactant. The surfactant exerts their role by acting at the interface between the oil and water, thus reduces the interfacial tension and governs the primary curvature of the dispersed phase. The fourth component, “a hidden component” called as co-surfactant, has emerged as an interesting component for the new insights/research in the microemulsion system/field 10.
The word co-surfactant itself reflects its potential to act as a facilitator or the initiator of surfactant. The back ground data act as a building block to explore the mechanism and various role of the co-surfactant. In the present experimental work, the co-surfactant has been tried as a microemulsion felicitator. Bending elasticity (BE) 11 is the property of the system or molecule to bend; thus, it provides flexibility to the system as well as it reduces the stiff nature of the system. BE is governed by the two moduli of elasticity termed as bending moduli and the Gaussian moduli. Bending modulus is the parameter that resists the elasticity of the system.
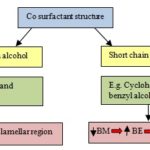
This modulus explains the degree of difficulty or troublesome to deform the layer of surfactant system (amphiphilic layer). In a nutshell, it means that bending modulus and bending elasticity is irreversible with each other. M. Gradzielski et al., (1998), in their literature report, explained the concept of co-surfactant structure with relationship to the bending elasticity and bending moduli. They classified co-surfactant structure into two broad categories, i.e. longer chain alcohols and the shorter chain alcohols. Based on the chain length of co-surfactant, the stiffness or flexibility of the surfactant system was well elaborated by their study described in Fig. 1.
FIG. 1: REPRESENTATION OF CO SURFACTANT SYSTEM AND THEIR EFFECT WITH BENDING ELASTICITY (BE) AND BENDING MODULUS (BM)
Various studies reflect the potential of co-surfactant to form hydrogen bonding with the surfactant head group. The size of the head group plays a vital role in observing the effects related to the hydrogen bonding network. The size of the head group of co-surfactant must be small in relationship with the surfactant bonding. Specifically, the effect was observed in nonionic co-surfactants, they interact with the head group of the surfactant with weak bonding (in comparison with covalent bonding) called hydrogen bonding. For example, fatty alcohols and fatty acids which possess small head group show interaction with the head group of nonionic co-surfactant as an outcome of hydrogen bonding. In relationship to the given statement, methyl acetate (a nonionic co-surfactant) possess bigger head group size shows little effect on the viscosity of the solution. Thus with the concluding remark for efficient hydrogen bonding, the small size of the head group is suitable 12-16.
The co-surfactant modulates the rheological properties of the system through the hydrogen bonding system, which tuned the stability of the system. The interaction with the medium chain co-surfactant affects the rheological properties of the surfactant system influenced by the co-surfactant. Additionally, the co-surfactant also affects the shape and size of the microemulsion system. B. Farago et al., in their study, precisely determined the role of co-surfactant in dynamic shape and size property of microemulsion system 17.
MATERIALS AND METHODS: Etoricoxib (EXB) was received as a gift sample from Kwality Pharmaceutical Pvt. Ltd. Amritsar. Oleic acid, propylene glycol, and polyoxyethylene sorbitan monooleate (Tween 80) from central drug house (P) Ltd. HPLC-grade water from Merck life sciences private limited. All the chemicals used in the experimental work were of analytical reagent grade.
Screening of Oil, Surfactant, and Co-surfactant: Solubility of Etoricoxib was determined in several oils such as castor oil, oleic acid, isopropyl myristate and in surfactants such as tween 80 and tween 40 and co-surfactants such as propylene glycol and PEG 400. Excess amount of drug was added to a fixed amount of different oils, surfactant, and co-surfactant, and the drug was solubilize by using the shake flask method to obtain the equilibrium condition for 72 h. After that, the samples are centrifuged at 3000 rpm for 10 min, and then aliquots are withdrawn, filtered, and diluted suitably. These samples were analyzed using UV-VIS spectrophotometer at 234 nm wavelength, and the solubility of Etoricoxib in each sample was calculated 18. The highest solubility of the drug was found in oleic acid, propylene glycol, and tween 80.
Method of Preparation: The formulation of stable microemulsion (using the different concentration of additives) was prepared by phase titration method. The clear, homogeneous, and transparent isotropic dispersion was prepared by mixing the oily components followed by titration with the aqueous phase 19. The oil (oleic acid) is mixed with the Smix (surfactant + co-surfactant). The mixture of oil with the Smix is considered an oil phase. This oil phase is titrated with the aqueous phase (HPLC-grade distilled water). The titration was carried out until the transparent and clear dispersion was obtained with no sign of phase separation. The resultant phase obtained is the characteristic of the microemulsion. In the experimental work, the emphasis is based on the role of co-surfactant(propylene glycol) in the formation of microemulsion in different ratios from 1:9 to 9:1 and the interpretation was best observed in ratio 1:3 in both the O/W and W/O types.
Experimental Design: It is a big debate that the co-surfactant acts on the surface or it acts out of the surface, to fulfill the requirement, it was decided to design an experiment to see the co-surfactant effect. The concluded remarks were the enhancement of elasticity of surfactant found to be the major role of co-surfactant. It was postulated that the hydroxyl group of propylene glycol might be engaged in the hydrogen bonding phenomenon because it was clear from the below-given results that the propylene glycol does not act on the surface. Because when co-surfactant used alone in o/w system, it is unable to mix the two phases to form a single phase system. The diagrammatic results have been representing in Fig. 2.
FIG. 2: DIAGRAMMATIC REPRESENTATION OF TWO PHASES (OIL AND WATER) REMAINS IMMISCIBLE AFTER ADDING ONLY PROPYLENE GLYCOL (CO-SURFACTANT)
To analyze the effect of sequence or the individual role of the co-surfactant and surfactant, we tried to change the sequential addition of the various components. The first attempt was made to add the surfactant before the co-surfactant that results in the formation of crude emulsion region. This step is followed by the addition of co-surfactant which increases the flow of crude emulsion. The formation of a stiff layer after adding the only surfactant was enough to conclude the role of co surfactant (enhances the elasticity and flow). In the opposite sequences, where the co-surfactant was added first, was showing the macro oil droplets, as we are aware that propylene glycol is soluble in oil not in water Fig. 3 and leads to the macro emulsion visible when seen under the microscope.
FIG. 3: PROPYLENE GLYCOL IN OIL AND WATER AND ITS MICROSCOPIC VIEW
The bending of the surfactant through hydrogen bonding of propylene glycol was found effective to increases the flow of the surfactant layer. The propylene glycol does not act as surface active agents as we are aware from the different literature reports that propylene glycol has two hydroxyl groups closer and having a good capability to make hydrogen bond and it also does not have a long nonpolar chain, which will make the surface active agents. So, it is clear that it does not act on the surface, but it makes hydrogen bond to the polar group through six-member transition state in Fig. 4, which is the driving force to bend the surfactant to reduce the surface tension as well the instability. The six-member transition is a key role player (favorable transition state) to make the system stable for a long time.
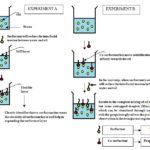
The hypothesis is based on the hydrogen bonding of the co-surfactant through polar head is again justified, with the help of an experiment, when the oil is in the continuous phase, then the polar head inside the droplet where the propylene glycol does not play the role with the effective hydrogen bonding in Fig. 5. Finally, the findings of the experimental results show that the mechanism of action of an individual component (surfactant or co-surfactant) in the microemulsion system shown in Fig. 6. Here the experiment A and experiment B have individual meaning, i.e. in the Experiment A: “First surfactant then co-surfactant was added and in the Experiment B: “First the co-surfactant then surfactant was added” the inference is diagrammatically represented in below given Fig. 6.
FIG. 6: MECHANISTIC VIEW REGARDING THE SEQUENCING FOR ADDITION OF SURFACTANT AND CO-SURFACTANT IN THE MICROEMULSION
W/O Microemulsion: Firstly, the water and oil (1:3 ratios) are mixed with surfactant mixture called Smix (Tween 80/ Propylene glycol, 2:1) to obtain transparent, isotropic, clear and homogeneous phase known as a microemulsion. Secondly, the water and oil (1:3 ratio) are mixed with surfactant only (Tween 80) to obtained transparent, isotropic, clear and homogeneous phase known as microemulsion; The result shows that in both the cases the region of transparent microemulsion Fig. 7 was obtained that highlights that the co-surfactant may not be needed in this case.
FIG. 7: COMPARATIVE REPRESENTATION OF W/O MICROEMULSION IN RATIO 1:3
O/W Microemulsion: In this case, the experimental work was carried at different ratios of Smix and also only with the surfactant to analyze the mechanism of co-surfactant in the system. Firstly the oil and water (1:3 ratio) are mixed with Smix (T/PG-2:1) to obtained transparent, isotropic, clear, and homogeneous phase known as a micro-emulsion. But the experiment shows that a transparent region was not obtained in this case.
FIG. 8: O/W MICROEMULSION IN RATIO 1:3 WITH Smix (T/PG-1:2)
Secondly, the work was proceeding to different Smix (T/PG-1:2), and the result shows that a transparent region called as microemulsion was obtained in this case, Fig. 8. This means that by increasing the amount of co-surfactant as compared to the surfactant favors the formation of a transparent, homogeneous microemulsion system.
In the next part of the work, the Smix (T/PG) was taken in the ratio of 1:1 and initially a transparent gel-like phase was obtained. This arises the susceptibility of the liquid crystal phase (having the property of both liquid and solid), an intermediated metaphase during the formation of the microemulsion system. Heating and cooling cycles were also performed to check the property of this phase shown in Fig. 9.
FIG. 9: HEATING AND COOLING EFFECT ON THE O/W SYSTEM IN RATIO 1:3 in Smix (T/PG-1:1)
Experimental Method to Demonstrate the Effectiveness of the Microemulsion through Effective Drugs: The formulation of microemulsion (using a different concentration of additives) was prepared by the water titration method (low energy emulsification technique).
TABLE 1: COMPOSITION OF INGREDIENT
| S. no. | Ingredients | Composition |
| 1 | Etoricoxib (active agent) | 32 ± 1.8 mg/ml |
| 2 | Oleic acid | 13.09 (% w/w) |
| 3 | Smix (Tween 80 + Propylene Glycol) Ratio: (1:2) | 47.64 (% w/w) |
| 4 | HPLC-grade distilled water | 39.27 (% w/w) |
The microemulsion was prepared by mixing the drug in the oily components followed by titration with the aqueous phase. The given amount of drug is completely solubilized in the oil (oleic acid) in composition 13.09% w/w and mixed with the Smix (surfactant + co-surfactant) in composition 47.64% w/w. The mixture of drug, oil with the Smix is considered an oil phase. This oil phase is titrated with the aqueous phase (HPLC-grade distilled water). Temperature - 37 ± 1 °C and stirring speed- 1000-1200 rpm.
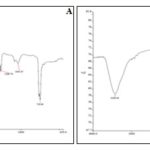
Compatibility Study: The compatibility study of the delivery system was completed with the help of infrared spectroscopy; the inference was the drug was found compatible with the excipients or carrier 20. The fingerprint, as well as the functional group region of the drug IR, was intact, which was the clear indication of the compatibility of the drug in the carrier system, Fig. 10.
FIG. 10: FTIR SPECTRA A) PURE DRUG B) DRUG + EXCIPIENTS IN 1:1 RATIO
Calibration Curve of Drug in the Phosphate Buffer pH 6.8: The UV spectrum of drug Etoricoxib was obtained spectrophotometrically by using UV - spectrophotometer (UV-1800, Shimadzu), 10 mg of drug was dissolved in sufficient quantity of phosphate buffer pH 6.8, and final volume was made up to 10 ml to get the stock solution of 1000 μg/ml. From the stock solution, 1 ml was withdrawn, and the volume was made up to 10 ml with phosphate buffer pH 6.8 to obtain the concentration of 100 μg/ml. From this sub-stock solution 0.2, 0.4, 0.6, 0.8 and 1.0 ml was withdrawn, and the volume was made up to 10 ml with phosphate buffer pH 6.8 to obtain the concentration of 2, 4, 6, 8, 10 μg/ml. The resultant solution was scanned from 200-400 nm, and the spectrum was recorded to obtain maximum wavelength, which helps in checking the purity of the drug.
FIG. 11: CALIBRATION CURVE OF DRUG IN THE PHOSPHATE BUFFER pH 6.8
RESULTS AND DISCUSSION:
Characterization of the Microemulsion:
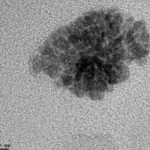
Droplet Size: Based on the HRTEM image through the copper grid, the size of the particle was found in the range between micro & nanoemulsion, 15-22 nm (Dilution of the Microemulsion 100x). This indicates the effectiveness of this co-surfactant based system. In general, the micro-system poses the size greater than the present one, but in the present system, it is smaller in size with the enhanced stability.
FIG. 12: HRTEM IMAGE OF ONE DROPLET OF MICROEMULSION
Drug Entrapment Study: The drug amount was diluted based on the maximum solubility (which was 32 mg in the prepared solution. The concentration was diluted by the phosphate buffer pH 6.8, till the range of UV, to see the value of drug entrapped in the formulation. The calculation of the dilution is given below. The final amount of drug was 5.9 MCG.
| Solution A | 32 mg in 8.5 ml | |||
| Drug + Formulation | ||||
| Conc. 3760 MCG | d = 4.76 | |||
| 10 mg = | 10/4.76 | 2.1 ml | ||
| Solution B | 2.1 ml of solution A in 10 ml PB | |||
| Conc. 652 MCG | ||||
| Solution C | 1 ml of solution B in 10 ml PB | |||
| 59.32 MCG | ||||
| Final dilution - | 10 times further diluted | |||
| 5.9 MCG | ||||
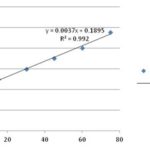
The final concentration of the drug is added into the formulation and after the completion of the formulation; the concentration of the drug was calculated through the calibration curve of the drug in the phosphate buffer pH 6.8 (for topical drug delivery) Fig. 11. The amount of the drug was present in the formulation was calculate using the two variable systems through the nonlinear curve equation, where the R-value was 0.992. The calculated amount of the drug was 5.03 MCG.
Calculation of drug entrapment = The amount of drug present in the formulation / The amount of drug added
5.03 / 5.9 = 0.852
% entrapment is 85.2%. It means approx 85% of the drug is going inside the formulation, which is quite high compared to other patented formulation. If the drug entrapment or drug loading will be more, then the bioavailability of drug will be more to cure the patient. It will be beneficial for the topical delivery system.
Stability of the Microemulsion Carrier: Stability is an essential feature of any system, for the long duration as well as the protection of the ingredients. In the literature, the reason for instability is due to the high energy or kinetic energy of the system. In the proper concentration of co-surfactant, surfactant, oil, and water, which we represented in the present invention (microemulsion) is more stable even after one month from the initial period.
So in the developed formulation, the kinetic energy of the system was quite low due to interstitial space for the moment. The less movement in the system provides energy, so it was justifying our statement for stability. In a nutshell the present ratio was found quite stable for a long time.
FIG. 13: STABILITY OF THE CARRIER
Flexibility or Spreadability of the Micro-emulsion Based Carrier: Stability is an essential feature, with the flow property of the system, because the flow plays an important role in the application of the topical agent. According to the literature, the flow of the excipients (carrier) in the skin enhances the drug bioavailability, so the flow of the excipients imparts a very important role in the topical delivery.
FIG. 14: SPREADABILITY IN MICROEMULSION SYSTEM
During the emulsification process, co-surfactant is defined as a chemical agent that added along with the surfactant to enhance the efficacy of the surface active agent. When the surfactant is added without co-surfactant, the film obtained at the interface was rigid. Hence, the mechanism of action of co-surfactant was explained based on expansion of the interfacial film made by the surfactant makes the amphiphilic monolayer of surfactant film flexible. Thus, they act as a fluidized component in the formation of the system and expand the region of occurrence of a clear, homogenous, isotropic, and transparent part called as a microemulsion.
In the experimental work, the emphasis is based on the role of co-surfactant in the formation of microemulsion in different ratios from 1:9 to 9:1 and the interpretation was best observed in ratio 1:3 in both the O/W and W/O types. In the first case, i.e. W/O microemulsion in ratio 1:3, the two immiscible phases oil and water are mixed with the surfactant mixture (surfactant + co-surfactant) or surfactant only to make them as one macro-molecular homogeneous single phase. In the second case, i.e. O/W microemulsion in ratio 1:3, the homogeneous macromolecular single phase is formed only when the mixture of surfactant and co-surfactant not with the surfactant only as a component. The important point to be considered here is the ratio of Smix; the propylene glycol play an important role when used in a concentration greater than the surfactant (Tween 80). Thus, propylene glycol has a favorable effect in the case of O/W.
TABLE 2: COMPOSITION OF OPTIMIZED BATCHES OF W/O MICROEMULSION (RATIO 1:3)
| Smix | Smix ratio | Oil phase (% w/w) | The aqueous phase (% w/w) | Smix (% w/w) |
| T/Pg | 2:1 | 28.8 | 9.6 | 61.6 |
| Only tween 80 | 33.7 | 11.2 | 55.1 |
T- Tween 80, Pg- Propylene glycol
TABLE 3: COMPOSITION OF OPTIMIZED BATCH OF O/W MICROEMULSION (RATIO 1:3)
| Smix | Smix ratio | Oil phase (% w/w) | The aqueous phase (% w/w) | Smix (% w/w) |
| T/Pg | 1:2 | 11.71 | 35.13 | 53.16 |
T- Tween 80, Pg- Propylene glycol
CONCLUSION: The process for the development of a microemulsion with the specific ratio of the ingredients is designed, and executed to demonstrate the efficacy of system, moreover the study concluded with a plausible mechanism of co-surfactant and surfactant, which is diagrammatically express through a six-member stable cyclic system eventually it converts the system with a hydrogen bond donor/acceptor co-surfactant for the stable and flexible drug carrier system. The many advantages of the invention make this technique useful in the future for the topical application of the all accepted pharmaceutical agents. The flexibility of the system is good enough for the spreadability of a carrier to induce drug in a better quantity to the systemic circulation. The stability with higher flexibility is rarely present due to the higher enthalpy of the system, which makes the system flexible and less stable, however the present invention is stable with excellent spreadability, moreover, the drug entrapment value was found superior than the existing drug delivery system. The delivery system was optimized with the Etoricoxib for the skin delivery stem. In the future, it will be accepted for the skin cancer topical drug delivery system.
ACKNOWLEDGEMENT: Authors are highly thankful to the Department of Pharmacy, School of Medical & Allied Sciences, Galgotias University for providing library facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Seifriz W: Studies in Emulsions. I-II. The Journal of Physical Chemistry 1925; 29(5): 587-00.
- Taylor GI: The formation of emulsions in definable fields of flow. Proc R Soc Lond A 1934; 146(858): 501-23.
- Bibette J and Leal-Calderon F: Surfactant-stabilized emulsions. Current Opinion in Colloid & Interface Science 1996; 1(6): 746-51.
- Mason TG and Bibette J: Emulsification in viscoelastic media. Physical Review Letters 1996; 77(16): 3481.
- Mason TG: New fundamental concepts in emulsion rheology. Current Opinion in Colloid & Interface Science 1999; 4(3): 231-38.
- Wilde PJ: Interfaces: their role in foam and emulsion behavior. Current Opinion in Colloid & Interface Science 2000; 5(3-4): 176-81.
- Opawale FO and Burgess DJ: Influence of interfacial properties of lipophilic surfactants on water-in-oil emulsion stability. Journal of colloid and interface science 1998; 197(1): 142-50.
- Hoar TP and Schulman JH: Transparent water-in-oil dispersions: the oleopathic hydro-micelle. Nature 1943; 152(3847): 102.
- Schulman JH, Stoeckenius W and Prince LM: Mechanism of formation and structure of microemulsions by electron microscopy. The Journal of Physical Chemistry 1959; 63(10): 1677-80.
- Rosano HL, Cavallo JL, Chang DL and Whittam JH: Micro emulsions: a commentary on their preparation. J Soc Cosmet Chem 1988; 39: 201-09.
- Gradzielski M: Effect of the co-surfactant structure on the bending elasticity in nonionic oil-in-water microemulsions. Langmuir 1998; 14(21): 6037-44.
- Mitrinova Z, Tcholakova S, Golemanov K, Denkov N, Vethamuthu M and Ananthapadmanabhan KP: Surface and foam properties of SLES+ CAPB+ fatty acid mixtures: Effect of pH for C12-C16 acids. Colloids and Surfaces A: Physicochemical & Engineering Aspects 2013; 438: 186-98.
- Mitrinova Z, Tcholakova S, Popova Z, Denkov N, Dasgupta BR and Ananthapadmanabhan KP: Efficient control of the rheological and surface properties of surfactant solutions containing C8-C18 fatty acids as co-surfactants. Langmuir 2013; 29(26): 8255-65.
- Tzocheva SS, Danov KD, Kralchevsky PA, Georgieva GS, Post AJ and Ananthapadmanabhan KP: Solubility limits and phase diagrams for fatty alcohols in anionic (SLES) and zwitterionic (CAPB) micellar surfactant solutions. Journal of Colloid and Interface Science 2015; 449: 46-61.
- Tzocheva SS, Kralchevsky PA, Danov KD, Georgieva GS, Post AJ and Ananthapadmanabhan KP: Solubility limits and phase diagrams for fatty acids in anionic (SLES) and zwitterionic (CAPB) micellar surfactant solutions. Journal of Colloid and Interface Science 2012; 369(1): 274-86.
- Mitrinova Z, Tcholakova S and Denkov N: Control of surfactant solution rheology using medium-chain co-surfactants. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2018; 537: 173-84.
- Farago B, Richter D, Huang JS, Safran SA and Milner ST: Shape and size fluctuations of microemulsion droplets: the role of co-surfactant. Physical Review Letters 1990; 65(26): 3348.
- Maheswari DU, Anand T, Manu TM, Khanum F and Sharma RK: Motion sickness induces physiological and neuronal alterations in a mouse model. Int J Pharm Sci & Res 2019; 10(4): 1650-59.
- Lala RR and Awari NG: Nanoemulsion-based gel formulations of COX-2 inhibitors for enhanced efficacy in inflammatory conditions. Appl Nanosci 2014; 4: 143.
- Kaur G, Chandel P and Harikumar SL: Formulation development of self-nanoemulsifying drug delivery system (SNEDDS) of celecoxib for improvement of oral bioavailability. Pharmacophore 2013; 4(4): 120-33.
How to cite this article:
Saxena P, Sharma PK and Purohit P: The mechanistic approach of the propylene glycol as a co-surfactant through “six member cyclic hydrogen bonding system” for the stable and flexible microemulsion drug carrier. Int J Pharmacognosy 2019; 6(5): 172-80. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(5).172-80.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
172-180
960
1084
English
IJP
P. Saxena *, P. K. Sharma and P. Purohit
Department of Pharmacy, School of Medical and Allied Sciences, Galgotias University, Greater Noida, Gautam Budh Nagar, Uttar Pradesh, India.
pratikshascholar.m.pharm@gmail.com
13 April 2019
20 May 2019
26 May 2019
10.13040/IJPSR.0975-8232.IJP.6(5).172-80
31 May 2019