SYNTHESIS, PHYSICAL PROPERTIES OF NEW 4- METHYL COUMARIN DERIVATIVES
HTML Full TextSYNTHESIS, PHYSICAL PROPERTIES OF NEW 4- METHYL COUMARIN DERIVATIVES
Sahar B. Hussein, Redha I. H. Al-Bayati and Shakeeb Majeed Al-Sayed
Department of Chemistry, College of Sciences, Al-Mustansiriya University, Baghdad, Iraq.
ABSTRACT: The aim of the present study, synthesis of new compounds of 4-methyl coumarin derivatives, from starting material (7-dihydroxy-4-methyl coumarin). A variety of chalcones derivative have been synthesized, and the proposed structures were confirmed by physical properties like UV –Visible, FT-IR and ¹ H NMR, Chem Bio Draw was used to evaluated the geometry optimization and total energies as well as dipole moment for all prepared compounds.
| Keywords: |
Coumarin, Starting Material, Physical properties, ¹ H NMR
INTRODUCTION: Coumarin and its derivatives represent one of the most active curriculum of a compound possessing a broad spectrum of biological activity 1, 2, 3. Several synthetic strategies for the synthesis of coumarins have already been developed. Classical routes to coumarins include Pechmann, Knoevenagel, Perkin, Reformatsky, and Wittig condensation reactions 4, 5, 6. To create these typical reactions efficacious, several variations in terms of catalyst and reaction environment have been introduced 7, 8. Chalcones are extensive components in all parts of the plants and are important as flower pigments, growth regulators, phytoalexins, animal toxins 9, 10.
In the present work, we promoted by these observations to synthesize a series of new derivatives of coumarin which probable to have possible biological activity.
EXPERIMENTAL:
Instrumentation: Melting points were determined on GallenKamp(MFB-600) melting point apparatus and are uncorrected. The IR spectra of the compounds were recorded on Shimadzu FT-IR-3800 spectrometer as KBr disk. The UV-Vis spectra were performed on Cintra-5-Gbes scientific equipment. The ¹H-NMR spectra (solvent DMSO) were recorded on Bruker 400 MHz spectro-photometer using TMS as internal.
Synthesis of Compounds:
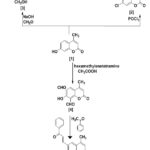
Synthesis of 7-Hydroxy-4-Methyl Coumarin [1]: A mixture of (17.6gm, 0.1mole) m-Hydroxy phenol and (13 gm, 0.1 moles) of Ethyl acetoacetate, was cooled down below 15°C, then (45ml) of conc. Sulfuric acid was added dropwise throughout (30min), allowed the mixture to cool to room temperature. After that, the reaction mixture was poured slowly into ice water with continuous stirring. The precipitate was filtered and recrystallized from Methanol; creamy colored needles were formed. Physical properties are shown in Table 1.
Synthesis of 7-Chloro -4- Methyl coumarin [2]: A mixture of (1.76 gm, 0.01 mole) compound [1] and (25 ml, 0.01 mole) of phosphorus oxychloride, was refluxed for (60 min.), then allowed to cool to room temperature. After that the reaction mixture was poured into crushed ice with vigorous stirring, the precipitate was filtered, washed with distilled water and recrystallized from methanol Table 1 and Scheme 1.
Synthesis of 7-Hydroxy-6,8-bis-hydroxymethyl-4-methyl Coumarin [3]: (1.6 gm, 0.04 mole) of Sodium hydroxide was added to a mixture of (3.7 gm, 0.02mole) compound [1] and (4.1 ml, 0.02 mole) of Formaldehyde, the mixture was refluxed with continuous stirring for 3 hours at 60 °C. The mixture was cooled to room temperature and neutralized with 5% Phosphoric acid the precipitate was filtered off, dried, and recrystallized form ethanol Table 1.
Synthesis of 4-methyl coumarin -6, 8 dicarbaldehyde derivative [4]: To a solution of compound [1] (1.76 gm, 0.01mole) in (50 ml) of acetic acid was added (2.9 g, 0.02mole) hexamethylene tertraamine were added. The mixture was stirred continuously at room temperature until a light brown viscous solution was obtained, and then heated to (70 -90) °C for two hours. The mixture was cooled to room temperature and (10 ml) concentrated H2SO4 was added dropwise and faint yellow precipitate was formed, then collected by filtration and washed thoroughly with cold Methanol and recrystallized from ethanol 1:1. Physical properties can see in Table 1.
Synthesis of (2E,2'Z)-3,3'-(7-hydroxy-4-methyl-coumarin -6,8-diyl)bis(1-phenylprop-2-en-1-one) [5]: To a solution of compound [4] (2.32gm, 0.01 mole) in (30 ml) Ethanol, (2.4 gm, 0.02 mole) from Acetophenone was added in the presence of (4-5) drops of piperidine with continuous stirring for 1 hr. The mixture was refluxed for 8 h, then the reaction mixture was cooled. The product was filtered and recrystallized from ethanol Table 1 and Scheme 1.
SCHEME 1: SYNTHESIS OF COMPOUNDS [4-8] FROM 7-DIHYDROXY-4-METHYL COUMARIN
RESULTS AND DISCUSSION: In this work, the first compound of coumarin derivatives was prepared as starting material for further synthesis Scheme 1. Compound (2) has been prepared by treatment of compound (1) with POCl3. The FTIR spectrum of compound (2) table (2), shows C=C aromatic stretching frequency near (1641cm-1). Other significant bands were observed at (1718) cm-1 being assigned to the C=O stretching frequency, and significant band was observed about (777) cm-1 refer to the C-Cl stretching frequency [13]. The ¹HNMR spectrum of compound (2) showed the following data: (DMSO, 300MHZ) 2.7 ppm(s, 3H, CH3), 6.5-7.4 ppm (m, 4H, Ar-H).
Compound (3) was prepared by a condensation reaction between Compound [1] with formaldehyde in the presence of Sodium Hydroxide as a catalyst. The FTIR Spectrum of compound (3) showed absorption band in the region (3372, 3344 cm-1) which was signal to OH group and absorption band at (2941, 2888 cm-1) due to C-H aliphatic group [3]. The 1H-NMR spectrum of compound [6] shows the following data: 3.1 ppm(s, 3H, CH3), 6.9 ppm (m, 4H, Ar-H). 3.2 (t, 4H, CH2). 3.9 (s, 2H, OH) and 11.3 (s, 1H, OH).
Compound (4) has been prepared by treatment of compound (1) with hexamethylenetetramine. The FTIR spectrum of compound (4) shows the appearance (1708) cm-1 of the C=O carbonyl band of Compound (4) C-H aldehyde appeared in the region (2865, 2783) cm-1 band and O-H exhibited in the region (3327) cm-1 [13]. The 1HNMR spectrum of (4) in DMSO-d6 as a solvent was showed the following data. 3 (s, 3H, CH3), 6.3 (m, 1H, Ar-H), 7 (s, 1H, CH of lactam ring), 11.7 (s, 4H, CHO), 8.04(s, 2H, CHO) and 8.3 (s, 1H, OH).
Chalcone (5) synthesis by condensation of the compound (4) with acetophenone in the presence of piperidine. The FT-IR spectrum of compound (5)show the appearance (1682) cm-1 of the C=O carbonyl band of Compound (12) C-H appeared in the region (2835-2932) cm-1 band and O-H appeared in the region (3423) cm-1 [14]. The 1HNMR spectrum of (5) in DMSO-d6 as a solvent was showed the following data. 2.5ppm (S, 3H, CH3), 6.2-7.0 ppm (m, 8H, Ar-H), 8.6 ppm (s, 2H, CH of lactam ring). 3.6ppm (s, 3H, O=C-CH3), 8.9 ppm (d, 4H, COCH=CH), 8.3ppm (s, 2H, CH ring). 12.3 ppm(s, 1H, OH).
TABLE 1: PHYSICAL PROPERTIES OF THE SYNTHESIZED COMPOUNDS (1-8)
| Comp. | M.P ͦ C | Yield % | Recryst. solvent |
| 1 | 184-186 | 70 | Methanol |
| 2 | 102-104 | 62 | Methanol |
| 3 | 135-137 | 67 | Ethanol |
| 4 | 150-153 | 59 | Ethanol |
| 5 | 164-166 | 73 | Ethanol |
TABLE 2: UV-VIS AND FTIR SPECTRAL DATA FOR NEW COMPOUNDS
| Comp. | UV Ethanol | Characteristic bands of FT-IR (cm⁻¹, KBr) | ||||
| λmax nm | C=O | C=C | C-H ar. | C-H al. | Others | |
| 1 | 226
324 |
1678 | 1597 | 3013 | 2899-2965 | ν(OH ) =3157 |
| 2 | - | 1718 | 1614 | 3086 | - | - |
| 3 | 310
225 |
1708 | 1631 | 3001 | 2865, 2783 | ν(OH )= 3400
ν(C=O )=1699 |
| 4 | 244
340 |
1682 | 1487-1576 | 3070 | 2835-2932 | νOH =3423
ν(C=O )=1650 |
| 5 | 282
205 317 |
1691 | 1607 | 3014 | 2941-2888 | ν(OH) =3372, 3344 |
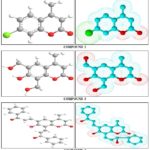
Computational Studies: Compounds 2,3 and 5 with optimized geometries and 3D geometrical structures are given in Figure.
The data showed that the highest atomic charge in compound 2 is at [O(11) -0.723] followed by the next charge value at [C(8) 0.532]and C(2) 0.266]. These data clearly showed that these atoms are most reactive toward the addition substitution reactions. The determined bond angle and twist angle, stretch (0.6230), bend (2.0795), stretch-bend (-0.0445), torsion (-8.5569) and the 3D geometrical structure indicated that this molecule is a non-planar molecule with the stereochemistry at [C(9)-C(7): (Z)].
The data showed that the highest atomic charge in compound 3 is at [C(8) 0.529 ] followed by the next charge value at[O(11)-0.726] and C(6) 0.217]. These data clearly showed that these atoms are most reactive toward the addition substitution reactions. The determined bond angle and twist angle, stretch (1.4221), bend (7.4661), stretch-bend (-0.0556), torsion (-13.0819) and the 3D geometrical structure indicated that this molecule is a non-planar molecule with the stereochemistry at [C(9)-C(7): (Z)].
The data showed that the highest atomic charge in compound 5 is at [C(26) 0.419] followed by the next charge value at[O(27)-0.558] and O(18) -0.552]. These data clearly showed that these atoms are most reactive toward the addition substitution reactions. The determined bond angle and twist angle, stretch (1.3260), bend (6.9459), stretch-bend (0.0180), torsion (-8.8272) and the 3D geometrical structure indicated that this molecule is a non-planar molecule with the stereochemistry at [C(9)-C(7):(Z) ,C(14)-C(15): (E), C(24)-C(25): (E))].
FIG. 1: OPTIMIZED 3D GEOMETRICAL STRUCTURES FOR COMPOUNDS 2, 3 AND 5
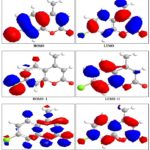
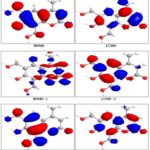
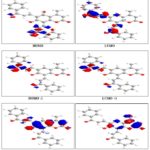
DFT calculations have been performed for compounds 2, 3 and 5. The optimized molecular structure of the most stable form is shown in Fig. 1. The calculated energies are presented in Table 3. Molecular orbital calculations could provide a detailed description of the orbitals including the spatial characteristics, nodal patterns, and individual atom contributions. The contour plots of the frontier orbitals for the ground state of compounds 2, 3 and 5 are shown in Fig. 2, 3 and 4 together with the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO). It is interesting to observe that both orbitals are substantially distributed over the conjugation plane.
TABLE 3: TOTAL ENERGY AND DIPOLE MOMENTS (DEBYE) FOR COMPOUNDS 2, 3 AND 5
| Compound | Total Energy kcal/mol | Dipole Moments |
| 2 | 5.9625 | 5.5958 |
| 3 | 4.8532 | 5.4259 |
| 5 | 17.3705 | 5.8576 |
FIG. 2: HIGHEST OCCUPIED MOLECULAR ORBITAL (HOMO) AND THE LOWEST UNOCCUPIED MOLECULAR ORBITAL (LUMO) OF COMPOUND 2
FIG. 3: HIGHEST OCCUPIED MOLECULAR ORBITAL (HOMO) AND THE LOWEST UNOCCUPIED MOLECULAR ORBITAL (LUMO) OF COMPOUND 3
In addition, it can also be seen from Fig. 2, 3 and 4 that the HOMO orbitals are located on the substituted molecule while LUMO orbitals resemble those obtained for the unsubstituted molecule and therefore the substitution has contributed an influence on the electron donation ability while imposing only a small impact on electron acceptance ability. The orbital energy levels of HOMO and LUMO of compounds 2, 3 and 5 are listed in Table 2. An electronic system with a larger HOMO-LUMO gap should be less reactive compared to the one having a smaller gap. In the present study, the HOMO-LUMO gap values of compound 2 -7.793, -11.775and -13.257eV while for compound 3and 5 are situated at: -7.986, -12.377and -13.604 eV: -5.835,-6.121 and -8.125eV respectively.
The lower value in the HOMO and LUMO energy gap would explain the eventual charge transfer interaction taking place within the molecules. The low values of HOMO for compounds 2, 3 and 5 indicate that these molecules have low ionization energies inferring that they can lose the electrons easily thus indicating that compounds 2, 3 and 5 are potentially good corrosion inhibitors [15]. The dipole moments of compounds 2, 3 and 5 were also calculated and listed in Table 3.
FIG. 4: HIGHEST OCCUPIED MOLECULAR ORBITAL (HOMO) AND THE LOWEST UNOCCUPIED MOLECULAR ORBITAL (LUMO) OF COMPOUND 5
TABLE 4: HOMO AND LUMO ENERGIES (eV) OF COMPOUNDS 2, 3 AND 5
| Comp. | HOMO | LUMO | ΔE | HOMO-1 | LUMO+1 | ΔE | HOMO-2 | LUMO+2 | ΔE |
| 2 | -11.674 | -.881 | -7.793 | -11.769 | 0.006 | -11.775 | -12.082 | 1.175 | -13.257 |
| 3 | -11.825 | -3.839 | -7.986 | -12.045 | 0.332 | -12.377 | -12.277 | 1.327 | -13.604 |
| 5 | -11.571 | -5.736 | -5.835 | -11.626 | -5.505 | -6.121 | -11.919 | -3.794 | -8.125 |
The UV-VIS absorption spectrum of compound 2 was recorded in the ethanol solution. The absorption peaks are observed at 310 and 225 nm and 317,282 and 205 nm for compound 5. It can be deduced that these peaks imply to the n→π* and π→π* transitions. The 3D plots of the HOMO−2, HOMO−1, HOMO, LUMO, LUMO+1, LUMO+2 and the corresponding energy levels for compounds 2, 3 and 5 are shown in Fig. 2, 3 and 4 respectively.
The theoretical electronic transfers (ET) for compound 2 was at -7.793, -11.775, compound 3 was at -7.986,-12.377 and for compound 5 were at -5.835,-6.121 nm corresponding to the UV-VIS spectral absorption peaks and the electronic transfers of HOMO plus LUMO and HOMO-1 plus LUMO+1, respectively. The increased theoretical absorption wavelengths of the compounds have slight blue-shifts when compared to the corresponding experimental absorption wave-lengths. Fig. 2, 3 and 4 shows the six main orbitals that have contributed in the vertical electronic transitions for compounds 2, 3 and 5. These orbitals, namely, HOMO−2, HOMO−1, HOMO, LUMO, LUMO+1, and LUMO+2, represent the three highest occupied orbitals and three lowest unoccupied orbitals in compounds 2, 3 and 5.
Similar spatial distribution of orbitals between HOMO/HOMO−1/HOMO−2 and LUMO/LUMO + 1/LUMO+2 pairs and the population analysis for compound 2 indicates that the electronic transitions and the electron clouds of the HOMO are delocalized on the C=C bond and the carbonyl of the α-β-unsaturated compound (chalcones). However, HOMO−1 is delocalized on C=C- bond, the carbonyl and the phenyl group of the chalcones, while the HOMO−2 is delocalized on the whole molecule. These orbitals are of the π-type bonding orbital.
LUMO is found mainly delocalized on the coumarin and the methyl group that substituted on coumarin and also the carbonyl group between the benzene ring and the five-member ring, while LUMO+1 is delocalized on the pyron ring of the coumarin and also the carbonyl group between the benzene ring and chalcones. For the LUMO+2 it is mainly delocalized on chalcone. For compound 3, the population analysis indicates that the electronic transitions and the electron clouds of the HOMO are delocalized on the C=O-bond and the carbonyl groups of the pyrene, while the HOMO-1 delocalized on the C=O bond, carbonyl bridge and the carbonyl groups of the pyrene but for the HOMO-2 is delocalized on the coumarin ring and carbonyl bridge.
LUMO is found mainly delocalized on pyrene and benzene ring while LUMO+1 coumarin ring and LUMO+2 are delocalized on the whole molecule. For compound 5, the population analysis indicates that the electronic transitions and the electron clouds of the HOMO and HOMO-1 are delocalized on the pyridazine-3,6-dionering, while the HOMO-2 delocalized on the whole molecule. LUMO, LUMO+1 and LUMO+2 are found delocalized on whole molecule.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- El-Agrody A, Abd El-Latif M, El-Hady N, Fakery A and Bedair A: Hetero aromatization with4-ydroxycoumarin Part II: Synthesis of some new pyrano[2,3-d] pyrimidines, [1,2,4]triazolo[1,5-c] pyrimidines and Pyrimido[1,6-b] [1,2,4]triazine derivatives. Molecules 2001; 6: 519–527.
- Rositca DN, Vayssilov GN, Rodios N and Bojilova A: Regio- and Stereoselective [2 + 2]Photodimerization of 3-Substituted 2-Alkoxy-2-oxo-2H-1,2-benzoxaphosphorines. Molecules 2002; 7: 420-432.
- Kadhum AH, Al-Amiery AA, Sikara M and Mohamad A: Synthesis, Structure elucidation and DFT studies of new thiadiazoles. Int J Phys Sci 2011; 6: 6692-6697.
- Milan C, Maja M, Tomislav B, Nela D and Valentina R: Design and synthesis of some thiazolidin-4-ones based on (7- hydroxy- 2- oxo -2h -chromen-4-yl) acetic acid. Molecules 2009, 14, 2501–2513.
- Rajithaa, B, Kumara, V.N, Someshwara, P, Madhava, J.V, Reddy, P.N and Reddy, Y.H. Dipyridine copper chloride catalyzed coumarin synthesis via Pechmann condensation under conventional heating and microwave irradiation. Arkivoc 2006, xii, 23-27.
- Murraya R and Jorge Z: A simple method for differentiating between angular and linear 5-methoxyfuranocoumarins. Phytochemistry 1984; 23: 697–699.
- Chavan S, Shivasankara K, Sivappaa R and Kalea R: Zinc mediated transesterification of β-ketoesters and coumarin synthesis. Tetrahedron Lett 2002; 43: 8583-8586.
- Yavari I, Hekmat-Shoar R and Zonouzi A: A new and efficient route to 4-carboxymethylcoumarinsmediated by vinyl triphenylphosphonium salt. Tetrahedron Lett 1998, 39: 2391-2392.
- Musa AY, Mohamad A, Al-Amiery AA, Kadhum AAH and Tien LT: Galvanic corrosion of aluminium alloy (Al2024) and copper in 1.0 M hydrochloric acid solution. Korean J Chem Eng 2012; 29: 818–822.
- Nakamoto N: Infrared and Raman Spectra of Inorganic and Coordination Compound. John Wiley & Sons, Inc., New Jersey, Edition 6th, 2009.
How to cite this article:
Hussein SB, Al-Bayati RIH and Al-Sayed SM: Synthesis, physical properties of new 4- methyl coumarin derivatives. Int J Pharmacognosy 2016; 3(4): 175-83. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(4).175-83.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
175-183
1,023
1878
English
IJP
S. B. Hussein, R. I. H. Al-Bayati and S. M. Al-Sayed
Department of Chemistry, College of Sciences, Al- Mustansiriya University, Baghdad, Iraq
saharbalkat@gmail.com
07 March 2016
02 April 2016
06 April 2016
10.13040/IJPSR.0975-8232.IJP.3(4).175-183
30 April 2016