SYNTHESIS, CHARACTERIZATION & BIOLOGICAL EVALUATION OF SOME AZETIDINE DERIVATIVES
HTML Full TextSYNTHESIS, CHARACTERIZATION & BIOLOGICAL EVALUATION OF SOME AZETIDINE DERIVATIVES
Pawan Gupta *, Sandeep Jain and Atul Kaushik
Department of Pharmaceutical Chemistry, Institute of Professional Studies, College of Pharmacy, Gwalior - 474001, Madhya Pradesh, India.
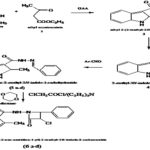
ABSTRACT: New series of N-(3-chloro-2-oxo-4-substituted-azetidine-1-yl) 2-methyl-1H-indole-3-carboxamide derivatives (VIa-d) were prepared and tested for their antibacterial and antifungal activity. The synthesis is based on the condensation of phenylhydrazine and ethyl acetoacetate in the presence of acetic acid to ethyl (2-methyl-1H- indol-3-yl)-2-oxoacetate (III) which on reaction with hydrazine hydrate gives 2-(2-methyl-1H-indol-3-yl)-2-oxoacetohydrazide (IV). Further conden-sation of oxoacetohydrazide and substituted benzaldehyde gave carbohydrazide derivatives (Va-d). Finally addition of triethylamine in dry 1, 4-dioxane and chloroacetyl chloride gave N-(3-chloro-2-oxo-4-substituted-azetidine-1-yl)2-methyl-1H-indole-3-carboxamide derivatives (VIa-d). The structure of newly synthesized 4-Oxoazetidin Substituted derivatives has been established by spectral (IR, 1HNMR) data. These compounds were screened for antibacterial and antifungal activity against various gram-positive and gram-negative strains. All the compounds show significant antibacterial and antifungal activity.
| Keywords: |
Carboxamide, Azetidine, Antibacterial, Antifungal
INTRODUCTION: Synthetic and semi-synthetic antimicrobial agents have been used for a long time against the life-threatening infectious diseases 1. Deaths from bacterial and fungal infection have dropped currently, but still, those are the major cause of death in the world. The treatment of infectious diseases remains an important and challenging problem because of a combination of factors including emerging infectious diseases and the increasing number of multi-drug resistant microbial pathogens with particular relevance for Gram-positive bacteria 2, 3.
Bacterial resistance has currently become a grave concern for physicians 4. Azetidine is a 4 member heterocyclic ring system with nitrogen as heteroatom. 2-Azetidinones are also known as β-lactams, and it is one of the most common heterocyclic rings found in antibiotics. 2-Azetidinones consists of a carbonyl group on the second position 5. The 2-azetidinone (β-lactam) ring system is the common structural feature of some broad-spectrum β-lactam antibiotics, including penicillins, cephalosporins, carbapenems, nocardiosis, monobactams, clavulanic acid, sulbactam, and tazobactam; which have been widely used as chemotherapeutic agents to treat bacterial infections and microbial diseases 6. Literature survey reveals that azetidine has shown various biological activities along with antimicrobial activity 7-15. Given these findings, some azetidine derivatives have been synthesized and evaluated for antibacterial, antifungal activity.
MATERIAL AND METHODS:
Chemistry: In the present study titled compounds (VIa-e) were prepared by the condensation of phenylhydrazine and ethyl acetoacetate in the presence of acetic acid to ethyl (2-methyl-1H- indol-3-yl)-2-oxoacetate (III) which on reaction with hydrazine hydrate gives 2-(2-methyl-1H- indol-3-yl)-2-oxoacetohydrazide (IV). Further condensation of oxoacetohydrazide and substituted benzaldehyde gave carbohydrazide derivatives (Va-d) finally the addition of triethylamine in dry 1, 4-dioxane and chloroacetyl chloride gave N-(3-chloro-2-oxo -4-substituted- azetidine-1-yl) 2-methyl-1H-indole-3-carboxamide derivatives (VIa-d).
Synthesis of ethyl (2-methyl-1H- indol-3-yl)-2-oxoacetate (III): In a 500 ml three-necked flask fitted with a dropping funnel, a sealed stirrer unit, and reflux condenser, place a mixture of 0.1 mol of ethyl acetoacetate (II) and 0.1 mol of acetic acid heat under reflux with stirring and add 0.1 mol of phenylhydrazine (I) during 1 h. Continue the stirring for another one hour. Pour the reaction mixture into a 1-liter beaker and stir vigorously while it solidifies. Cool to 5 ºC and filter at the vacuum pump through Buchner funnel; cool the filtrate in ice and refilter through the same Buchner funnel wash the solid on the filter with 50 ml of water, suck almost dry and then wash with 50 ml of ethanol then keep overnight in room temperature recrystallized from ethanol.
Synthesis of 2-(2-methyl-1H- indol-3-yl)-2-oxoacetohydrazide (IV): A mixture of ethyl (2-methyl-1H- indol-3-yl)-2-oxoacetate (III) and hydrazine hydrate in equimolar portion and 15 ml of ethanol were taken in a round bottom flask and refluxed for 4-6 h. Excess of ethanol was removed by distillation. On cooling crude product was separated. It was filtered, collected and recrystallized from ethanol to obtain silky white crystals.
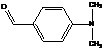
General Procedure for the Synthesis of substituted 2-methyl- 1H- indole- 3-carbohydrazide (Va-d): A mixture of 2-(2-methyl-1H- indol-3-yl)-2-oxoacetohydrazide (IV) (0.01 mol) and substituted benzaldehyde (0.01 mol) was refluxed in ethanol (30 ml) in the presence of catalytic amount of glacial acetic acid for 3 h. The reaction mixture was concentrated, cooled; the solid separated was filtered and recrystallized from aqueous DMF.
General Procedure for the Synthesis of 2-oxo-azetidine derivatives of 2-methyl-1H-indole-3-carboxamide (VIa-d): Substituted 2-methyl-1H-indole-3-carbohydrazide (0.01mol) (Va-f) were dissolved in 1:4 Dioxane (20ml) with constant stirring, triethylamine (0.01 mol) was added followed by dropwise addition of 2-chloro acetyl chloride (0.01 mol). The content was stirred vigorously for 15 minutes and refluxed for 5 h. The mixture was cooled at room temperature, filtered, washed with ice-cooled water, dried and recrystallized from DMF.
Spectral Studies: All melting points were recorded in Digital melting point apparatus and are uncorrected. The IR spectra were recorded on Perkin Elmer FTIR spectrometer using KBr pellets 1HNMR spectra were recorded on Bruker Avance II 400MHz NMR (d in ppm) relative to TMS as an internal standard. The purity of compounds were checked by TLC using precoated silica gel G plate method using ethyl acetate: glacial acetic acid: water and the spots were examined by I2 Vapor or under a UV lamp and Rf value has been reported in Table 1.
TABLE 1: CHARACTERIZATION DATA OF COMPOUNDS VI a-d
Synthesis of N-(4-phenyl- 3-chloro-2-oxo-azetidine-1-yl) 2-methyl -1 H – indole - 3-carboxamide (VIa):
I.R (KBr, cm-1) 3427 (NH), 2923 (-C-H-), 1762 (C=O), 1630 (C=O, CONH), 1354 (C-N), 866 (C-Cl).
1HNMR (Acetone) 6-8.5 (9H, Ar-H), 2.2 (3H, CH3), 0.9 (2H, N-H), 2.4 (1H, CH-Cl), 3 (1H, N-CH).
Synthesis of N-(4-chlorophenyl- 3-chloro-2-oxo-azetidine-1-yl) 2-methyl – 1 H - indole-3-carboxamide (VIb):
I.R (KBr, cm-1) 3427 (NH), 2923 (-C-H-), 1758 (C=O), 1727(C=O, CONH), 1307 C-N), 732 (C-Cl):
1HNMR (Acetone) 6-8.5 (8H, Ar-H), 2.2 (3H, CH3), 0.9 (2H, N-H), 2.4 (1H, CH-Cl), 3(1H, N-CH)
Synthesis of N-(4-(dimethylamino) phenyl - 3-chloro-2-oxo-azetidine-1-yl) 2-methyl-1H-indole-3-carboxamide (VIc):
I.R (KBr, cm-1) 3427 (NH), 2923 (-C-H-), 1756 (C=O), 1638(C=O, CONH), 1313 (C-N), 1065 (C- N 3o):
1HNMR (Acetone) 6-8.5 (7H, Ar-H), 2.2 (3H, CH3), 0.9 (2H, N-H), 2.4 (1H, CH-Cl), 3 (1H, N-CH)
Synthesis of N-(4-(2, 4-dichlorophenyl) - 3-chloro-2-oxo-azetidine-1-yl) 2-methyl-1H-indole-3-carboxamide (VId):
I.R (KBr, cm-1) 3427 (NH), 2923 (-C-H-), 1762 (C=O), 1630 (C=O, CONH), 1354 (C-N), 866 (C-Cl):
1HNMR (Acetone) 6-8.5 (8H, Ar-H), 2.2 (9H, 3CH3), 0.9 (2H, N-H), 2.4 (1H, CH-Cl), 3 (1H, N-CH).
Antimicrobial Activity:
Antibacterial Activity: A novel prepared series of carboxamide derivatives were screened for their antibacterial activity in-vitro in comparison with amoxicillin as a reference drug using the standard agar disc diffusion method against four bacterial species: Staphylococcus aureus (AUMC B71), Bacillus cereus (AUMC B70) represented by Escherichia coli (AUMC B69) and Pseudomonas aeruginosa (AUMC B72).
Nutrient agar medium of the requisite composition viz., peptone (2.5g), beef extract (0.5g), agar-agar (10g) and distilled water (500ml) was prepared, and pH of the medium was adjusted to 6.6.
For the preparation of media, all the above ingredients (except agar-agar) were weighed and dissolved in distilled water (250 ml) by application of gentle heating. After dissolving the ingredients completely, more distilled water and weighed agar-agar added. Then, it was filtered through cotton to obtain a clear solution. The mixture was autoclaved for 30 min at a pressure of 1.5 kg/cm2. All the glass wares were cleaned with chromic acid and then sterilized by keeping in the oven. The medium was cooled to 37.1 °C and the homogenous suspension was prepared by transferring aseptically a loopful of all the corresponding microorganism from a fresh subculture into agar medium followed by vigorous shaking 20 ml of this medium was poured into each sterilized Petri-dish under aseptic conditions and allowed to set. Sterile 5-mm filter paper disk was saturated with 10 µl of the solution of test compound and Ampicillin as a reference drug. Also another disc was impregnated with the solvent DMSO and served as a negative control. The discs were then dried for 1h and placed in each plate. The seeded plates were incubated at 35 ± 2 °C for 24-48 h. The radii of inhibition zones (in mm) of triplicate sets were measured and results are given in Table 2.
TABLE 2: IN-VITRO ANTI-BACTERIAL AND ANTI-FUNGAL ACTIVITY OF NEWLY SYNTHESIZED COMPOUNDS VIa-d
— indicates no activity as representative of gram-positive strains while gram-negative strains were
Antifungal Activity: The synthesized compounds VI a-d were tested for their antifungal activity in-vitro in comparison with fluconazole as a reference drug using the standard agar disc diffusion method against two pathogens namely Candida albicans and Aspergillus niger.
Spore suspension in sterile distilled water was prepared from a 2-5 days old culture of the test fungi grown on Sabourarud agar media. The final spore concentration was nearly 5 × 104 spore mL-1. About 15 mL of growth medium was added to sterilized Petri dishes of 9 cm diameter and inoculated with 1 mL of spore suspension. Plates were shaken gently to homogenize the inocula. Sterile 5-mm filter paper disc was saturated with 10 µL of test compound solution and fluconazole (40 µ mol mL-1 in DMSO). Also, another disc was impregnated with DMSO and served as a negative control. The discs were dried for 1 h and placed in the center of each plate.
The seeded plates were incubated at 28 ± 2 °C for 7 days. The radii of inhibition zones (in mm) of triplicate sets were measured at successive intervals during the incubation period and the results are given in Table 2.
RESULT AND CONLCUSION: Some derivatives of 2-methyl-1H-indole-3-carboxamide (VIa-d) were prepared and tested for their antibacterial and antifungal activity. The synthesis is based on the condensation of phenylhydrazine and ethyl acetoacetate in the presence of acetic acid to ethyl (2-methyl-1H- indol-3-yl)-2-oxoacetate (III) which on reaction with hydrazine hydrate gives 2- (2- methyl- 1H- indol- 3- yl)- 2-oxoacetohydrazide (IV). Further, condensation of oxoacetohydrazide and substituted benzaldehyde gave carbohydrazide derivatives (Va-d) finally the addition of triethylamine in dry 1, 4-dioxane and chloroacetyl chloride gave carboxamide derivatives (VIa-d). All the above reactions are observed summarized in the scheme.
All synthesized compound (VIa-d) was recrystallized from DMF and identified by TLC using ethyl acetate: glacial acetic acid: water. Spots were visualized through the iodine chamber or under a UV lamp and the Rf value was calculated and found to be in the range of 0.61-0.71 cm. The structures of various compounds were assigned on the basis of their melting points, Rf values, IR, 1HNMR spectral data. The compounds were they are evaluated for antibacterial activity using the standard agar disc diffusion method. They showed moderate activity against most of the tested bacterial strains. Among them, compound VI b and VIc showed maximum activity than the other compounds against both Gram-positive and Gram-negative bacteria compared with standard drug Ampicillin. The compounds also evaluated for antifungal activity standard agar disc diffusion method against two pathogens namely Candida albicans and Aspergillus niger. The compound VId shows highly significant activity compared with standard drug fluconazole.
ACKNOWLEDGEMENT: The authors are grateful to the Director for providing necessary facilities to carry out this research work as well as for his valuable advice and constant encouragement during this work. Our sincere thanks are also to the Pharmaceutical chemistry department.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Narendra SNB and Rangappa KS: Design, synthesis, the antibacterial and antitubercular activity of cationic antimicrobial peptides, ovine bactenecin 5. Indian Journal of Chemistry 2008; 47B: 297-304.
- Cohn ML: Epidemiology of Drug Resistance: Implications for a Post Antimicrobial Era. Science 1992; 257: 1050-1055.
- Tenover FC: Mechanisms of Antimicrobial Resistance in Bacteria. A J Med 2006; 119(6A): S3-S10.
- Thomas AB, Nanda RK and Kothapalli LP: Antimicrobial efficacy of n-[3- chloro-(substituted aryl)-4-oxoazetidine- 1-yl] pyridine-4- carboxamides against resistant bacterial strains obtained from clinical isolates 2013; 3(3): 1.
- Zachariah SM, Ramkumar M, George N and Ashif MS: Azetidinones: An overview. Int J Pharm Sci Rev 2015; 30(1): 211-218.
- Seth M and Sah P: Synthesis and antimicrobial activity of 2- azetidinones derived from benzimidazole. Journal of Chemical and Pharmaceutical Research 2012; 4(1): 146-153.
- Melloni P, Dleea Torre A, Meroni M, Ambrosi A and Rossi AC: Azetidine derivatives of tricyclic antidepressant agents, J Med Chem 1979; 22(2): 183-191.
- Dua R and Srivastava SK: synthesis, characterization and anti-microbial activity of 2-(2'-substituted-benzylidene -hydrazino-acetyl)-mercapto-5-methyl-1,3,4-thiadiazoles and 2-[2'-{4-substi-tuted-aryl-3-chloro-2-oxo-zetidine}-acetyl-aminomercapto]-5-methyl-1,3,4-thiadiazoles, International Journal of Pharma and Bio Sciences 2010; V1(2): 11-13
- Sharma MC, Kohli DV, Sahu NK, Sharma S and Chaturvedi SC: 2D-QSAR studies of some 1,3,4-thiadiazole-2yl azetidine 2-one as antimicrobial activity, digest journal of Nanomaterials and Biostructures 2009; 4(2): 339-347.
- Bonde CG, Peepliwal A and Gaikwad NJ: Synthesis and antimycobacterial activity of azetidine-, quinazoline-, and triazolo-thiadiazole-containing pyrazines. Arch Pharm (Weinheim) 2010; 343(4): 228-36.
- Venkateswararao A, Kirubha TSV, Senthamarai R, Sarvani B, Vasuki K, Synthesis and determination of biological activities of new series of azetidinones, Der Pharma Chemica 2011; 3(1): 439-445.
- Aoyama Y, Masaaki U, Makoto K, Mamoru T, Hayasaki-Kajiwara TKY, Noriyuki N and Masatoshi N: Design, synthesis and pharmacological evaluation of 3-benzylazetidine-2-one-based humanchymase inhibitors, Bioorganic & Medicinal Chemistry 2001; 9(11): 3065-3075.
- Manahan-Vaughan D, Reiser M, Pin JP, Wilsch V, Bockaert J, Reymann KG and Riedel G: Physiological and pharmacological profile oftrans-azetidine-2, 4-dicarboxylic acid: Metabotropic glutamate receptor agonism and effects on long-term potentiation, Neuroscience 1986; 72(4): 999-1008.
- Tsai FH, Overberger CG and Zand R: Synthesis and conformation of poly(L -azetidine-2-carboxylic acid- L -proline) and poly([L -proline] 3 - L -azetidine-2-carboxylic acid), Journal of Polymer Science Part A: Polymer Chemistry 1992; 30(4): 551-559.
- Narute AS, Khedekar PB and Bhusari KP: QSAR studies on 4-thiazolidinones and 2-azetidinones bearing benzothiophene nucleus as potential anti-tubercular agents. Indian Journal of Chemistry 2008; 47B: 586-591.
How to cite this article:
Gupta P, Jain S and Kaushik A: Synthesis, characterization & biological evaluation of some azetidine derivatives. Int J Pharmacognosy 2016; 3(1): 30-34. doi: 10.13040/IJPSR.0975-8232.3(1).30-34.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
30-34
575
1350
English
IJP
P. Gupta*, S. Jain and A. Kaushik
Department of Pharmaceutical Chemistry, institute of Professional studies, College of Pharmacy, Gwalior, Madhya Pradesh, India.
prenugupta@yahoo.co.in
20 October 2015
26 November 2015
06 December 2015
10.13040/IJPSR.0975-8232.IJP.3(1).30-34
31 January 2016