SYNTHESIS, CHARACTERIZATION, ANTIOXIDANT AND ANTI-INFLAMMATORY ACTIVITY EVALUATION OF SOME SUBSTITUTED PYRAZOLE DERIVATIVES
HTML Full TextSYNTHESIS, CHARACTERIZATION, ANTIOXIDANT AND ANTI-INFLAMMATORY ACTIVITY EVALUATION OF SOME SUBSTITUTED PYRAZOLE DERIVATIVES
B. Lavanya, B. Nagasudha * and Y. Sivaram Reddy
Department of Chemistry, Creative Educational Society College of Pharmacy, Jawaharlal Nehru Technological University Anantapur, Kurnool - 518218, Andhra Pradesh, India.
ABSTRACT: A series of substituted acetophenones are condensed with hydrazides to the corresponding hydrazones which are subsequently cyclized by using volumizer-Haack reaction to give new pyrazole derivatives. All the compounds 4a, 4b, 4c, 4d, 4e were screened for antioxidant, anti-inflammatory activity. Few of the compounds showing good antioxidant and anti-inflammatory activity.
| Keywords: |
Pyrazoles, Esterification, Hydrazones, Vilsmier-Haack The reaction, Antioxidant, Anti-inflammatory
INTRODUCTION: Pyrazole derivatives have attracted the attention of research scholars on account of their wide range of applications in medicine. The possibility that pyrazole is used directly in the synthesis of the amino acid has been investigated by supplying young melon seedlings with pyrazole alone and in association with various amino acids. The heterocyclic component of the amino acid and that the alanine group was attachedto the ring at nitrogen and not at a carbon atom. The term pyrazole was given to this class of compounds by German Chemist Ludwig Knorr in 1883. In a classical method developed by German chemist Hans von Pechmann in 1898, pyrazole was synthesized from acetylene and diazomethane.
In 1959, the first natural pyrazole, 1-pyrazolyl-alanine, was isolated from seeds of watermelons 1. Pyrazole is the organic compound with the formula C3H3N2H. It is a heterocycle characterized by a 5-membered ring of three carbon atoms and two adjacent nitrogen centers. Pyrazoles are also the class of compounds that have the ring C3N2 with adjacent nitrogen centers. Pyrazoles has a wide range of activities such as antimicrobial 2, 3, anti-inflammatory 4, anticonvulsant 5, antitubercular 6, 7 antiproliferative & antiangiogenic 8.
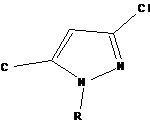
1a-H, 1b-CH3, 1c-C6H6
Several methods have been reported in the literature for the synthesis of pyrazoles. K et al., 9 synthesized a series of 1H-pyrazole derivatives. All the compounds were evaluated for antimicrobial and antiepileptic activities. All the compounds showed mild to moderate antimicrobial activity. 1a, 1b, 1c showed highly significant antiepileptic activity.
MATERIALS AND METHODS: All chemicals used were of LR grade. All the chemicals were obtained from SD fine chemicals India. DPPH, H2O2, ethanol, DMF, ferric chloride, potassium ferricyanide, phosphate buffer and tribromo acetic acid (TBA) trichloroacetic acid (TCA) acetophenones, and other solvents were purchased from molychem, India. Melting point was determined in open capillaries on tempo apparatus; Spectrophotometric determination was recorded on ELICO-SL 164 Double Beam UV-Visible Spectro Photometer. 1HNMR, MASS spectra were obtained from IIT Madras, IR spectra were obtained from BRUKER 300MHZ. The purity of compounds was checked by Thin-Layer Chromatography (TLC) using silica gel-G pre-coated plates, and spots were detected by UV chamber.
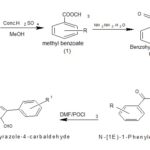
Synthetic Procedure: The general scheme for the synthesis of 1-(Benzoyl)-3-phenyl-1H-pyrazole-4-carbaldehyde derivatives has been represented in Fig. 1.
Step-1 Synthesis of Methyl benzoate: To a 100 ml round bottom flask a mixture of substituted benzoic acid (0.01mol) and methanol (20 ml) were taken with few drops of conc. H2SO4. A condensor was attached to the RB flask refluxed for 3 h at 25-30 °C. The mixture was cooled under running tap water and adds 5% NaHCO3 solution was added. The solid thus separated was filtered. Further purification was done by re-crystallizing with methanol. Melting point measurement was done. The formation of the parent compound was then confirmed by performing TLC using hexane: ethyl acetate (6:4) as the mobile phase. Spots were identified by placing the plates Rf value was calculated.
FIG. 1: SYNTHETIC PROCEDURE
TABLE 1: LIST OF PYRAZOLE DERIVATIVES SYNTHESIZED
| Compounds | R | R1 |
| 4a | C6H5 | C6H5C(O)CH3 |
| 4b | C6H5 | 3-NO2 |
| 4c | 4-NO2 | C6H5C(O)CH3 |
| 4d | 4-NO2 | 4-OCH3 |
| 4e | 4-NO2 | 4-OH |
TABLE 2: PHYSICO CHEMICAL DATA OF COMPOUNDS
| S. no. | Compound | Molecular
Formula |
Melting
Point °C |
Mol.
Wt. |
%
Yield |
Rf
value |
λmax | Color |
| 1 | 4a | C17H12N2O2 | 250-254 | 276 | 68% | 0.4* | 280 | Brown |
| 2 | 4b | C17H11N3O4 | 140-143 | 321 | 57% | 0.57* | 310 | Brown |
| 3 | 4c | C18H14N2O3 | 146-148 | 306 | 65% | 0.8* | 230 | Brown |
| 4 | 4d | C17H11N3O4 | 273-275 | 321 | 69% | 0.7** | 320 | Brown |
| 5 | 4e | C17H10N4O6 | 232-234 | 337 | 71% | 0.5** | 320 | Brown |
TABLE 3: SPECTRAL CHARACTERIZATION OF PYRAZOLE DERIVATIVES (4a-4e)
| Compound Code | UV
(λmax) |
IR
Data |
Mass
Data |
NMR
Data |
| 4a | 280 nm | - | - | - |
| 4b | 310 nm | 1633cm-1(C=O)str, 1529.42cm1(C=N)str,
1348cm-1 (Ar-NO2)str, 712.8cm-1(C-H)ben, 3424.55cm-1(Ar-C-H)str |
- | - |
| 4c | 230 nm | 1175.97cm-1(C-O-C)str, 1603cm-1(C=O)str,
1348.60 cm-1 (C- N)str, 1254.3cm-1(C-O)str ,1529.10cm-1(C=N)str, 2933 cm-1 (C-H) str, 838.50 cm-1(C-H) ben |
306
M+ Peak |
9.7 δ(1H,s,CHO Pyrazole),
7.2-7.7 δ(9H,m,Ar-H), 1.02δ (1H.s, Ar-OCH3), 8.2 δ(1H,s,Pyrazole)
|
| 4d | 320 nm | 3424.61cm-1(Ar-C-H) str , 1604.56cm-1(C=O) str,
1528.28 cm-1(Ar-NO2) str, 1178cm-1(C-N) str, 843.21cm-1(C-H)ben |
- | 9.3 δ(1H,s,CHO Pyrazole),
7.2-7.8 δ(5H,m,Ar-H), 8.2 δ(1H,s,Pyrazole), 8.4 δ(4H,m,Ar-NO2)
|
| 4e | 320 nm | 3130.34 cm-1(OH) str, 1604 cm-1 (C=O) str, 1448.83cm-1(NO2) str, 1203cm-1(C-N) str, 840.2cm-1(Ar-C-H)ben, 1650cm-1 (C=N)Ar-str | - | 9.5 δ(1H,s,CHO-Pyrazole),
6.7-8.4δ(8H,m,Ar-H), 8.2 δ (1H,s,Pyrazole), 5.4 δ(1H,s,Ar-OH)
|
Step-2 Synthesis of Benzohydrazide: Compound 1(0.01mol) and 4ml of hydrazine hydride(99%) were added and refluxed for more than 2 h, the reaction mixture was then cooled and diluted with water and allow to stand for 10 min. Solid was separated and washed with water, dried then re-crystallized with methanol. The solid obtained were dried, and the melting point was determined followed by the TLC using Chloroform: Methanol (7:3) as the mobile phase.
Step-3 Synthesis of N1-(1-phenyl ethylidene) benzohydrazide: A mixture of compound-2 (0.01mol) and acetophenone (0.01mol) in methanol (30ml) containing a drop of glacial acetic acid was refluxed for 30 min. The separated colorless solid was filtered and re-crystallized from ethanol. The solid obtained were dried, and the melting point was determined followed by the TLC using chloroform: methanol (7:3) as the mobile phase.
Step-4 Synthesis of 1-(Benzoyl)-3-phenyl-1H-pyrazole-4-carbaldehyde: To the vilsmeier-Hacck reagent prepared from DMF (10 ml) and PoCl3 (1.1ml, 0.012mol) at 0 °C. Compound III (1.016g, 0.004mol) was added in small aliquots at a time, and the reaction mixture was stirred at 60-65 °C for 4hrs, and the mixture was poured into ice water. The solid separated on neutralization with NaHCO3 was filtered, from washed with water and re-crystallized from methanol. Melting point measurement was done.
The formation of the parent compound was then confirmed by performing TLC using methanol: chloroform (7:3) as the mobile phase. Spots were identified by placing the plates. Rf value was calculated.
RESULTS AND DISCUSSION:
Antioxidant Activity:
DPPH Radical Scavenging Method: The effect of the samples (4a-4e) in addition to the sample stock solution 6 mg of different samples were dissolved in 2 ml of DMSO and volume made up to 10 ml with methanol from the stock solution various dilutions (10, 20, 40, 80, 160, 320 µg/ml) were prepared. To 3 ml of various concentrations of sample solutions, 1 ml of DPPH (2 mg in 50 ml methanol) was added in triplicate manner, i.e. each concentration in 3 series of test tubes.
An equal amount of methanol and DPPH was added and used as a control. Ascorbic acid was used as a standard for comparison, i.e., 6 mg of ascorbic acid was dissolved in 10 ml methanol from the stock solution various dilutions (10, 20, 40, 80, 160, 320 µg/ml) were prepared. After incubation for 20minutes in the dark, absorbance was recorded at 517 nm. The graph was plotted by taking concentration (μg/ml) on x-axis and percentage scavenged/ inhibition on the y-axis.
% scavenging activity was calculated using the formula:
% scavenging = Absorbance of control - Absorbance of test sample / Absorbance of control × 100
Nitric Oxide Scavenging Method: The effect of samples (4a-4e) in addition to 20 mg of different samples was dissolved in 2 ml of DMSO and volume made up to 20 ml with methanol. From the stock solution various dilutions (10, 20, 40, 80, 160, 320 µg/ml) were prepared. To 4ml of various concentrations of sample solutions, 1ml of 25mM Sodium nitroprusside solution (made using phosphate buffer pH 7.4) was added in triplicate manner, i.e. each concentration in 3 series of test tubes. An equal amount of sample solution and 1ml phosphate buffer, pH 7.4 was added and used as the control. Ascorbic acid was used as a standard for comparison. 6 mg of ascorbic acid was dissolved in 10 ml of methanol. From this stock solution various dilutions (10, 20, 40, 80, 160, 320 µg/ml) were prepared using methanol. After incubation for 2 h, 2 ml of the solution was pipette out from each test tube, and 1.2 ml of griess reagent (i.e., 1% Sulfanilamide in 5% H3PO4& 0.1% Naphthyl ethylenediamine dihydrochloride in equal amounts) was added and absorbance was recorded at 570 nm. Graph was plotted by taking concentration (μg/ml) on x-axis and percentage scavenged/ inhibition on the y-axis.
% scavenging activity was calculated using the formula:
% scavenging = [Absorbance of control - Absorbance of test sample / Absorbance of control] × 100
Hydroxyl Radical Scavenging Method: The effect of samples (4a-4e) in addition to 20 mg of different samples was dissolved, and volume made up to 20 ml with methanol. From the stock solution various dilutions (10, 20, 40, 80, 160, 320 µg/ml) were prepared. To 0.4 ml of various concentration of sample solutions, 0.4 ml of 3mM deoxy ribose solution, 0.4 ml of 0.1mM FeCl3 solution, 0.4 ml of 0.1mM EDTA solution, 0.4ml of 20mM H2O2 solution, 0.1mM ascorbic acid solution were added.
Ascorbic acid was used as a standard. 6 mg of ascorbic acid was dissolved in 10 ml of methanol. From this stock solution various dilutions (10, 20, 40, 80, 160, 320 µg/ml) were prepared using methanol. Then these were incubated for 30min. Then 0.4 ml of ice-cold 15% w/v TCA and 1% w/v TBA (except for blank, where TBA was not added) was added to each test tube. These test tubes were kept in boiling water bath for 30 min, cooled and absorbance was recorded at 532 nm. The graph was plotted by taking concentration (μg/ml) on x-axis and percentage scavenged/inhibition on the y-axis.
% scavenging activity was calculated using the formula:
% scavenging = [Absorbance of control - Absorbance of test sample / The ability of the synthesized - Absorbance of control] × 100
Hydrogen Peroxide Method: The following procedure determined compounds to scavenge H2O2. A solution of H2O2 (40mM) was prepared in phosphate buffer (pH 7.4) 0.45 ml (0.136 g) of 30% H2O2 w/v is taken and dissolved in 100 ml of pH 7.4 buffer to give 40mM H2O2 solution.
The concentration of H2O2 was determined by absorption at 230 nm using UV Visible spectrophotometer. Test solutions were added to an H2O2 solution (0.6 ml 40mM). 6 mg test or the standard was weighed and dissolved in 6 ml of methanol with the aid of sonicator.
From the above stock solution 0.12 ml, 0.24 ml, 0.36 ml, 0.48 ml, 0.6 ml, 1.2 ml was taken and the volume is made to 6 ml using methanol which gives the concentrations of 10, 20, 40, 80, 160, 320 µg/ml respectively. The absorbance of H2O2 at 230 nm was determined after 10 min against a blank solution containing phosphate buffer and test compound without H2O2. The control solution was prepared by taking a solution of H2O2 in phosphate buffer (pH 7.4) and its absorbance was measured.
The percentage of H2O2 scavenging by the test and the standard was calculated using the following formula.
% Scavenged = [(A-A1)/A] × 100
Where, A = Absorbance of the control, A1 = absorbance of the test /standard
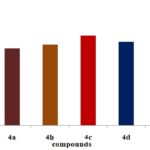
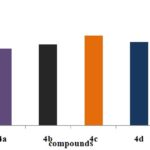
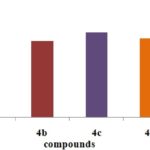
Antioxidant Activity of Pyrazole Derivatives by Different Methods:
FIG. 2: DPPH METHOD
FIG. 3: H2O2 METHOD
FIG. 4: NITRIC OXIDE SCAVENGING METHOD
FIG. 5: HYDROXYL RADICAL SCAVENGING METHOD
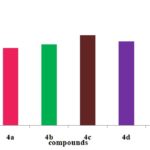
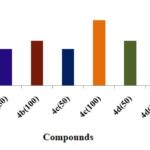
Anti-Inflammatory Activity:
Acute Toxicity Studies: The acute toxicity study was determined in rats. Rats fasted for 12 h were randomly divided into 6 groups of 3 rats per group. Graded doses of the compounds (10, 50, 100, 200,400 and 800 mg/kg p.o.) were separately administered to the rats in each of the groups using a bulbed steel needle.
All the animals were then allowed free access to food and water and observed throughout 48 h for signs of acute toxicity. The number of deaths within this period was recorded. Based on the results of preliminary toxicity test, the doses of 50 mg/kg, 100 mg/kg body weight (according to OECD Guideline 420) of were chosen for further experiments.
Carrageenan-Induced Paw Edema: Albino rats (Wister Strain) of 180-200 g were used for present investigation. They were kept in polypropylene cages in an air-conditioned area at 25 + 20C in 12-12 hr light-dark cycle. They were provided with balanced feed, and aqua guard purified water ad libitum. These animals were randomly divided into 7 equal groups each group consists of 6 animals. Group-I served as control. Group-II served as standard diclofenac sodium 10mg/kg which is a reference drug, while rats in Group-3, 4, 5, 6,7 received 50 mg/ml & 100 mg/ml of test compounds were administered p.o. as a suspension in carboxymethyl cellulose (CMC) (0.5% w/v solution), 1 h prior to carrageenan injection.
The control group received only 0.5% w/v solution of CMC. The left hind paw edema was induced by sub-planter injection of 0.1 ml of 1% carrageenan solution in saline (0.9%). The volume of paw edema (ml) was determined using digital plethysmometer (Aarson Scientific Works) before and after 3 and 4 h of carrageenan injection.
The percentage of edema inhibition was calculated according to the following equation:
The formula calculated the anti-inflammatory activity of the test compounds.
%Inhibition = {1-(A-X/B-Y)} × 100
Where, A=Mean paw volume after administration of the drug, X= Mean paw volume before administration of the drug, B=Mean paw volume of treated control rats, Y=Mean paw volume of treated control rats before administration of carrageenan.
FIG. 6: ANTI INFLAMMATORY ACTIVITY OF DIFFERENT PYRZOLE DERIVATIVES
CONCLUSION: Structures of the synthesized compounds were characterized and confirmed with the help of analytical data such as IR, 1HNMR & MASS SPECTRA. The synthesized compounds were screened for antioxidant activity by DPPH, Nitric oxide, H2O2, hydroxyl radical scavenging method. The compounds have shown good antioxidant activity. Among all the compounds 4c, 4e (4-OCH3, 4-OH) shows considerable activity in comparison to the standard drug (i.e., Ascorbic acid). The synthesized compounds were also screened for anti-inflammatory activity by using carrageenan-induced paw edema method. From all the synthesized compounds 4e, 4c, 4a (4-OH, 4-OCH3, H) shows significant activity comparing to standard (i.e., Diclofenac Sodium). More significantly 4e (4-OH), 4c (4-OCH3) derivative shows nearer action to the standard. Compound with electron donating group at 4th position on the ring exhibit significant action it makes the ring with prompt aromatic to exhibit action. Compounds 4e, 4c possess better anti-inflammatory activity than an antioxidant.
ACKNOWLEDGEMENT: The authors are thankful to Prof. Y. Sivarami Reddy, chairman, and Nagasudha Assistant professor, Department of pharmaceutical chemistry for the support and also I thank SAIF IIT Madras for his kind support to give 1H NMR and MASS spectral data.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Noe FF and Fowden: A heterocyclic amino acid, (-)-.Β-Pyrazole-Lylalanine, was isolated from the seeds of watermelon (Citrullus vulgaris) and its structure confirmed by comparison with synthetic material. Biochemistry Journal 1960; 543-546.
- Aziz SAGA, Ali TES, El-Mahdy KM and Abde-Karim SM: synthesis and antimicrobial activities of some novel bis-pyrazole derivatives containing a hydro phosphoryl unit. European Journal of Chemistry, 2011; 2(1): 25-35.
- Malladi S, Isloor M A, Akhila DS and Hoong-Kun Fun: Synthesis, characterization and antibacterial activity of some new pyrazole schiff bases. Arabian Journal of Chemistry 2013; 6: 335-340.
- Yassin FA: Novel pyrazolyl pyridazine derivatives likely to possess anti-inflammatory activity. Journal of Microbial and Antimicrobials 2010; 2(7): 93-99.
- Abdel-Aziz M, Abuo-Rahma Gel-D and Hassan AA: Synthesis of novel pyrazole Derivatives and evaluation of their antidepressant and anticonvulsant activities. European Journal of Chemistry 2009; 44(9): 3480.
- Pandit U and Dodiya A: Synthesis and anti-tubercular activity of novel pyrazole-quinazolinone hybrid analogs. Springer Link 2013; 22: 3364-3371.
- Pathak RB, Chovatia PT and Parekh HH: Synthesis, antitubercular and antimicrobial evaluation of 3-(4-chlorophenyl) 4-substituted pyrazole derivatives. Bio-organic Medicinal Chemistry Lett 2012; 22: 5129.
- Tzanetou E, Liekens S, Kasiotis KM, Fokialakis N and Haroutounian SA: Novel pyrazole and indazole derivatives: synthesis and evaluation of their anti-proliferative and anti-angiogenic activities. Arch Pharmacy 2012; 345-804.
- Ananda Rajagopal K, Anbu Jeba Sunilson J, Illavarasu A, Thangavelpandian N and Kalirajan R: Antiepileptic and antimicrobial activities of novel 1-(unsubstituted/ substituted)-3, 5-dimethyl-1H-pyrazole derivatives. Inter-national Journal of Chemtech Research 2010; 2: 45-49.
How to cite this article:
Lavanya B, Nagasudha B and Reddy YS: Synthesis, characterization, antioxidant and anti-inflammatory activity evaluation of some substituted pyrazole derivatives. Int J Pharmacognosy 2014; 1(2): 99-06. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(2). 99-06.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
99-106
656
2251
English
IJP
B. Lavanya, B. Nagasudha*, Y. Sivaram Reddy
Department of Chemistry, Creative Educational Society College of Pharmacy, Jawaharlal Nehru Technological University Anantapur, Kurnool, India
lavanyabg19@gmail.com
05 November 2013
21 January 2014
23 January 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.1(2).99-06
01 February 2014