SYNTHESIS AND BIOLOGICAL EVALUATION OF CYCLOPROPANE DERIVATIVE OF COMBRETASTATIN CA-4-PHOSPHATE
HTML Full TextSYNTHESIS AND BIOLOGICAL EVALUATION OF CYCLOPROPANE DERIVATIVE OF COMBRETASTATIN CA-4-PHOSPHATE
Shailendra Kumar * 1, Chandrul Kaushal 1 and Sanjay Kumar Jain 2
Institute of Pharmacy 1, Shri Venkateshwara University, Gajroula, Amroha - 244236, Uttar Pradesh, India.
Institute of Pharmacy 2, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: Combretastatin A-4 (CA-4) is an anti-mitotic agent that is gaining rapid recognition among cancer biologists and clinicians as one of the newer vascular disrupting agents, (VDAs) for cancer therapy. CA-4 belongs to a group of tubulin binding natural products called combretastatin, derived from the African Bush Willow, Combretum caffrum. Due to CA-4's in-vivo efficacy, several analogs of CA-4 have been synthesized to maximize its solubility and bioavailability. Combretastatin A-4 phosphate is a more soluble form of CA-4 that has completed for treatment of acute myelogenous leukemia and relapsed ovarian cancer. This review attempts to highlight the various CA-4 analogs that have been synthesized and their effectiveness in clinical. Combretastatin A-4 phosphate (CA-4-P) is a biologically very active compound by binding to the colchicine binding site which leads to the inhibition of microtubule polymerization as well as showing antiangiogenic and anticancer effects by selectively shutting down the tumor blood flow. To avoid the disadvantage of rather low in-vivo efficacy resulting from the isomerization of the cis-stilbene derivative to the thermodynamically more stable trans-isomer, our research group started the project for CA-4 analogs synthesis. The incorporation of carbocycles with different ring sizes on the connecting carbon-bridge of the natural compound prevents the system from undergoing cis-trans-isomerization. The synthesis of the cyclopropane derivative of CA-4 via the cyclopropanation reaction with diazomethane, and further analogs with incorporated moieties for better water solubility. This is a colorimetric assay that measures the reduction of yellow 3-(4, 5-dimethythiazol-2-yl)-2, 5-diphenyl tetrazolium bromide (MTT) by mitochondrial succinate dehydrogenase. The MTT enters the cells and passes into the mitochondria where it is reduced to an insoluble, colored (dark purple) formazan product. The cells are then solubilized with an organic solvent (e.g., isopropanol) and the released, solubilized formazan reagent is measured spectrophotometrically. Since, the reduction of MTT can only occur in metabolically active cells the level of activity is a measure of the viability of the cells.
| Keywords: |
Cylopropane derivative, Antiangiogenic, Cytotoxicity, Combretastatin A-4-P, Anticancer
INTRODUCTION: Combretastatin A-4 (CA-4) Fig. 1 was isolated from the bark of the South African tree Combretum caffrum in 1989 by Pettit and co-workers 1.
This cis-stilbene strongly inhibits tubulin polymerization by binding to the colchicine site and is found to be a cytotoxic agent against a wide variety of tumor cell lines, including multidrug-resistant lines 2, 3.
The water-soluble sodium phosphate prodrug CA-4P (Zybrestat TM) Fig. 1 is currently in several advanced clinical trials for age-related macular degeneration and anaplastic thyroid cancer based on the vascular shutdown mechanism of action 4, 5. Many efforts have led to the preparation of more soluble analogs 6 for example, combretastatin CA-4 phosphate (CA-4P) 2 or amine 3 pro-drugs 7. They exert antineoplastic activity through disruption of the blood flow within the tumor leading to tumor cell death 8. In particular, the water-soluble CA- 4P has shown great clinical promise. In a phase, I clinical trial, 15 patients with advanced cancer received CA-4P with bevacizumab, and this combination appeared safe and well tolerated. It was shown in this study that CA-4P induced profound vascular changes, which were maintained by the presence of bevacizumab 9.
Combretastatin A-4 phosphate (CA-4-P), a tubulin-depolymerizing agent structurally related to colchicines, is in clinical trials as a vascular disrupting agent. Phase I trials, started in 1998, have established a maximum tolerated dose (MTD) in the range of 52–68mg/m2, and showed significant changes in tumor perfusion in patients by positron emission tomography (PET) and contrast-enhanced proton magnetic resonance imaging (MRI) at these doses 10, 11, 12. CA-4-P has recently entered Phase II trials in combination with conventional radiation and chemotherapy 13.
Combretastatin A-4 was originally isolated from the South African bush willow Combretum caffrum. The phosphate derivative, CA-4-P, was subsequently synthesized as the more soluble prodrug 13. The exact mechanism of action of CA-4-P is not yet understood. In-vitro, CA-4-P causes microtubule depolymerization, which leads to endothelial cell shape changes 14. Other, more rapid effects include activation of the small GTPase Rho A and subsequent reorganization of the actin cytoskeleton, leading to an increase in endothelial monolayer permeability to macromolecules 15.
Further, work in endothelial cells demonstrated that CA-4- P damaged mitotic spindles, arrested cells at metaphase, and led to the death of mitotic cells with characteristic G2/M DNA content, following prolonged drug exposures 16. The mitotic arrest was associated with elevated levels of cyclin B1 protein and increased p34cdc2 activity. In animal models, cyclopropane derivative caused a prolonged and extensive drop in blood flow in tumor vessels, with much less effect in normal tissues 17. A rapid rise in tumor vascular permeability was observed leading to a catastrophic shut down of the established tumor vasculature within minutes of drug exposure, causing necrosis and secondary tumor cell death 18.
CA-4-P treatment has been shown to increase tumor hypoxia (low oxygen) within one hour of drug treatment, as measured by eppendorf oxygen electrode or with the use of the hypoxia marker pimonidazole 19. Severe hypoxia is also known as an inherent characteristic of virtually all solid tumors, including experimental and clinical cancers, but not normal tissues. It has been correlated with poor prognosis, a more aggressive phenotype, increased metastases and resistance to therapy (see review by, 20. Early in-vitro work had analyzed CA-4-P cytotoxicity under 1% oxygen conditions (mild hypoxia), showing an increase in cell kill under hypoxia 21. Beyond that, little is known of the effect of inherent tumor hypoxia on cyclopropane derivative activity, or the effect of CA-4-P on gene expression. Gene regulation under hypoxia is governed by the transcription factor, hypoxia-inducible factor 1 (HIF-1). HIF-1 is a heterodimer consisting of the subunits HIF-1α and HIF-1β/ARNT 22.
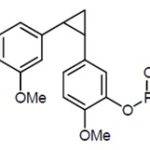
FIG. 1: CYCLOPROPANE DERIVATIVE
The cyclopropanation was followed by the removal of the MOM-protecting group under acidic condition, and the cyclopropyl derivative of CA-4 (1) was isolated. Subsequent derivatization of the free phenolic functionality with dibenzyl phosphate and transformation of the phosphate into the corresponding sodium salt was done to increase water solubility 23. One of the most notable examples of such a modification can be seen in the structure of cyclopropane derivative Fig. 1, in which a disodium phosphate salt is incorporated at the phenolic position 24. The delivery of combretastatin analogs to tumors in-vivo would be challenging without modifications such as these unless harsh organic solvents are used.
These organic solvents pose their health hazards, so the use of water-soluble compounds for treatment is necessary 25. Further, drug development entails the incorporation of bioreductive triggers in the molecular structure. While combretastatins do favor the destruction of tumor vasculature over normal blood vessels, high doses still have damaging side effects 26. Bioreductive triggers address this problem. Unlike other vascular disrupting agents, compounds with bioreductive trigger are introduced into the body in an inactive state. When these compounds come in contact with key bioreductive enzymes, the bioreductive trigger moiety is cleaved, leaving the active form of the compound 27.
As a result, elongated endothelial cells round up, disrupting endothelial cell layer surrounding blood vessel and exposing of the underlying basement membrane. This leads to blood vessel congestion and loss of blood flow, loss of oxygen and nutrient supply to tumor cells. Therefore, tumor cells undergo necrosis. Given strong anticancer/ antivascular activity exhibited by CA-4, we have synthesized some novel combretastatin analogs and tested for their antiangiogenic activity 28.
Synthesis of Compounds:
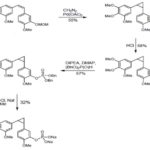
SCHEME 1: SYNTHESIS OF CYCLOPROPANE DERIVATIVE
Chemicals: All the chemicals and reagents used were from CDH, New Delhi. Glassware’s used were from borosil. hydrochloric acid, ceric ammonium nitrate, dimethoxyethane, dimethyl-formamide, dimethyl sulfoxide, diethyl ether, Acetic acid methyl cyanide, etc.
MATERIALS AND METHODS: All compounds were purified by column chromatography, and re-crystallization and the confirmatory establishment of the structure were done by melting point, TLC, UV, IR, and 1H NMR. Column chromatography was performed using silica gel (Qualigens, particle size 60-120mm). TLC was performed on silica gel TLC plates. All melting points were recorded on a DECIBEL digital melting point apparatus. IR spectra were recorded on an 8400S SHIMADZU spectrometer. 1H NMR spectra were recorded on a dpx300 spectrometer (analysis laboratory, CDRI, Lucknow).
Solvents and Chemicals Purification: The used solvents and chemicals were, if necessary, purified and dried according to common procedures as follows. Dry solvents were stored under an argon atmosphere over a molecular sieve (4Ǻ) 21. Methylene chloride was distilled from P2O5, diethyl ether (Et2O) and tetrahydrofuran (THF) were freshly distilled from sodium/benzophenone under argon; diisopropylamine (DIPA), diisopropylethyl-amine (DIPEA) and triethylamine (TEA), acetonitrile (MeCN), hexane and ethyl acetate were distilled from CaH2; toluene was refluxed over sodium and freshly distilled. All other solvents were HPLC grade.
Procedure of Preparation of Compounds:
Sodium 2- methoxy- 5- (2- (3, 4, 5-trimethoxy phenyl) cyclopropyl)-phenyl phosphate: To a mixture of 267 (40mg, 0.07 mmol) and NaI (42mg, 0.28 mmol) in anhydrous acetonitrile was added TMS-Cl (35μl, 0.28 mmol) drop wise at room temperature. The reaction mixture was stirred for 3 hours, quenched with water and extracted with ethyl acetate. The organic layers were combined and the solvent was removed under reduced pressure. The residue was purified by flash chromatography (CH2Cl2/MeOH 5:1, 1% AcOH) affording the free phosphorous acid. The free acid was dissolved in 1 ml MeOH, and sodium methanolate (1.2mg, 0.023 mmol) was added. The reaction mixture was stirred for 5 min, afterward the solvent was removed under reduced pressure affording 236 (10mg, 32%) as a white solid. Rf 0.1 (CH2Cl2/MeOH 3:1, 1% AcOH).
(Z)-2-Methoxy-5- (3,4,5-trimethoxystyryl)phenol (CA-4, 4): To a solution of 258 (0.14 mmol, 50mg) in 2 ml methanol 0.5 ml 3 M HCl were added. The reaction mixture was stirred for 24 h at room temperature. The solution was diluted with CH2Cl2. The organic layer was washed with water (2 × 8 ml), dried over Na2SO4, and the solvent was removed under reduced pressure. The residue was purified by HPLC affording 38mg (95%) of 4. 171 Rf 0.37 (hexanes/ethyl acetate 2:1).
(Z)-2- Methoxy- 5- (3, 4, 5-trimethoxystyryl) aniline: To a solution of catalyst (Pd/CaCO3, 0.032 mmol, 3.4mg) in 2 ml ethyl acetate were added three drops of quinoline and 400 μl of cyclohexene. The solution was stirred for one hour at room temperature. Alkyne 231 (100mg, 0.32 mmol) was dissolved in 1 ml ethyl acetate and was added to the reaction mixture. The solution was exposed to H2 until the total consumption of the starting material (3 h). The reaction mixture was filtered, and the solvent was removed under reduced pressure. The residue was purified by flash chromatography (toluene/ethyl acetate 19:1, 1% NEt3) affording 232 (84mg, 83%) as a yellow oil. Rf 0.27 (toluene/ethyl acetate 5:1).
Tert - butyl 2- methoxy- 5- ((3, 4, 5-trimethoxy phenyl)ethynyl)-phenylcarbamate: To a stirred solution of 293 (300mg, 0.96 mmol) in 10 ml of a solvent mixture THF/H2O in a 1:1 ratio was added K2CO3 (794mg, 5.76 mmol) at room temperature. After 10 min Boc2O was added (838mg, 3.84 mmol). The reaction mixture was stirred for two days at 60 °C. The reaction was quenched with 5% aqueous HCl. The two layers were separated and the aqueous phase was extracted with ethyl acetate (3 × 15ml). The combined organic extracts were washed with water (1 × 20 ml), and brine (1 × 20 ml) and the solvent was removed under reduced pressure. The residue was purified by flash chromatography (hexanes/ethyl acetate 9:1), affording 262 as a colorless foam (356mg, 90%). 172 Rf 0.58 (hexanes / ethyl acetate 2:1).
Dibenzyl- 2- methoxy- 5- (2- (3, 4, 5-trimethoxy phenyl)- cyclopropyl)- phenyl phosphate: A solution of 9 (220mg, 0.67 mmol) and CCl4 (648 μl, 6.7 mmol) in 4 ml acetonitrile was cooled to -10°C and stirred for 10 min. Then DIPEA (228 μl, 1.34 mmol) and DMAP (8mg, 0.067 mmol) were added. After one-minute dibenzyl phosphite (221 μl, 1 mmol) was added slowly, and the mixture was stirred for additional 3 hours at -10 °C. After total consumption of the starting material, the reaction was quenched with 0.5 M aqueous KH2SO4 solution. The two layers were separated, and the aqueous phase was extracted with ethyl acetate (3 × 15 ml). The combined organic extracts were washed with water (1 × 20 ml) and brine (1 × 20 ml), and the solvents were removed under reduced pressure. The residue was purified by column chromatography (hexanes/ethyl acetate 3:1), affording 267 as a colorless oil (265mg, 67%). 91 Rf 0.44 (hexanes / ethyl acetate 1:1).
(Z)-Dibenzyl 2-methoxy- 5- (3, 4, 5-trimethoxy styryl) phenyl phosphate: A solution of 4 (110mg, 0.35 mmol) and CCl4 (338 μl, 3.5 mmol) in 3 ml acetonitrile was cooled to -10 °C and stirred for 10 min. Then DIPEA (118 μl, 0.7 mmol) and DMAP (4mg, 0.035 mmol) were added. After one-minute dibenzyl phosphite (116 μl, 0.53 mmol) was added slowly and the mixture was stirred for an additional 3 hours at - 10 °C. After total consumption of the starting material, the reaction was quenched with 0.5 M aqueous KH2SO4 solution. The two layers were separated and the aqueous phase was extracted with ethyl acetate (3 × 10 ml). The combined organic extracts were washed with water (1 × 15 ml) and brine (1 × 15 ml), and the solvents were removed under reduced pressure. The residue was purified by column chromatography (hexanes/ ethyl acetate 3:1), affording 259 as a colorless oil (115mg, 57%). 91 Rf 0.44 (hexanes/ethyl acetate 1:1).
Sodium (Z)- 2- methoxy- 5- (3, 4, 5-trimethoxy styryl) phenyl phosphate: A mixture of 227 (100 mg, 0.17 mmol) and sodium iodide (99mg, 0.66 mmol) in anhydrous acetonitrile was stirred at 0 °C and TMS-Br (86 μl, 0.66 mmol) was added dropwise. The reaction mixture was stirred for 5 min at that temperature before it was quenched with water and extracted with ethyl acetate. The combined organic layers were dried over Na2SO4, filtered and the solvent was removed under reduced pressure. The residue was purified by flash column chromatography (CH2Cl2/MeOH 3:1, 1% AcOH) affording 30mg (48%) of the free phosphorous acid. The free acid was dissolved in 1mL MeOH, and sodium methanolate (8mg, 0.15 mmol) was added. The reaction mixture was stirred for 5 min, afterward the solvent was removed under reduced pressure affording 14 (33mg, 94%) as a white solid.
Spectral Data:
Sodium 2- methoxy- 5- (2- (3, 4, 5-trimethoxy phenyl)cyclopropyl)-phenyl phosphate: 1H NMR (400 MHz, D20) δ [ppm] 7.39 (s, 1H), 6.82 (d, J=8.56 Hz, 1H), 6.83 (dd, J=8.56, 2,05 Hz, 1H), 6.42 (s, 2H), 3.79 (s, 3H), 3.73 (s, 6H), 3.70 (s, 3H), 2.63-2.53 (m, 1H), 2.52-2.44 (m, 1H), 1.53-1.44 (m, 2H).
(Z)-2-Methoxy-5-(3,4,5-trimethoxystyryl) phenol (CA-4, 4): 1H NMR (400 MHz, CDCl3) δ [ppm] 6.92 (d, J = 2.07 Hz, 1H), 6.80 (dd, J = 8.26, 2.07 Hz, 1 H), 6.73 (d, J = 8.26 Hz, 1H), 6.44 (dd, J = 12.17, 23.67 Hz, 2H), 5.52 (s, 1H), 3.86 (s, 3H), 3.84 (s, 3H), 3.69 (s, 6H); 13C NMR (400 MHz, CDCl3) δ [ppm] 153.01, 145.90, 145.39, 137.34, 132.84, 130,80, 129.62, 129.19, 121.25, 115.19, 110.47, 106.24, 61.06, 56.10, 56.08; IR ν 3752 3422 3152 2837 1419 1274 1005 cm-1.
Z)-2-Methoxy-5-(3,4,5-trimethoxystyryl) aniline: 1H NMR (400 MHz, CDCl3) δ [ppm] 6.93 (d, J = 8.27 Hz, 1H), 6.90 (s, 3H), 6.85 (dd, J = 8.02, 2.02 Hz, 1H), 4.99 (s, 2H), 3.92 (s, 6H), 3.92 (s, 6H), 3.91 (s, 3H), 3.80 (s, 3H); 13C NMR (400 MHz, CDCl3) δ [ppm] 154.14, 148.16, 139.30, 139.05, 121.16, 119.29, 117.11, 115.52, 111.73, 109.75, 90.70, 88.40, 61.35, 57.21, 56.56; IR ν 3469, 3371, 2936, 1613, 1453, 1326, 1028 cm-1.
Tert-butyl 2- methoxy- 5- ((3, 4, 5-trimethoxy phenyl)ethynyl)-phenylcarbamate: 1H NMR (400 MHz, CDCl3) δ [ppm] 8.32 (s, 1H), 7.16 (dd, J = 8.34, 2.02 Hz, 1H), 7.09 (s, 1H), 6.81 (d, J = 8.34 Hz, 1H), 6.75 (s, 2H), 3.89 (s, 3H), 3.88 (s, 6H), 3.86 (s, 3H), 1.54 (s, 9H); 13C NMR (400 MHz, CDCl3) δ [ppm] 153.19, 152.77, 147.69, 138.66, 128.25, 126.02, 121.09, 118.89, 116.03, 109.94, 108.83, 88.94, 88.02, 80.75, 61.11, 56.31, 55.91, 28.51; IR ν 3436, 2938, 2839, 1726, 1527, 1128 cm-1.
Dibenzyl- 2- methoxy- 5- (2- (3, 4, 5-trimethoxy-phenyl)- cyclopropyl)- phenyl phosphate: 1H NMR (400 MHz, CDCl3) δ [ppm] 7.35-7.28 (m, 10H), 7.04 (s, 1H), 6.65 (s, 2H), 6.09 (s, 2H), 5.16-5.05 (m, 4H), 3.72 (s, 3H), 3.68 (s, 3H), 3.62 (s, 6H), 2.41-2.29 (m, 2H), 1.45-1.37 (m, 1H), 1.22-1.14 (m, 1H); 13C NMR (400 MHz, CDCl3) δ [ppm] 152.63, 148.91*, 148.86, 139.29, 139.22, 136.13, 135.95*, 135.87, 134.33, 131.33, 128.63, 128.57, 127.99, 122.83*, 122.81, 112.26, 105.0, 69.91, 69.85, 60.89, 56.05, 24.32, 23.70, 12.18; IR ν 2938, 2838, 1586, 1378, 1234, 1183, 1082 cm-1.
(Z)-Dibenzyl 2- methoxy- 5- (3, 4, 5-trimethoxy styryl)phenyl phosphate: 1H NMR (400 MHz, CDCl3) δ [ppm] 7.38-7.27 (m, 10H), 7.18-7.14 (m, 1H), 7.07 (d, J = 8.32 Hz, 1H), 6.79 (d, J = 8.32, Hz, 1H), 6.49 (s, 2H), 6.43 (dd, J = 18.89, 12.17 Hz, 2H), 5.16-5.10 (m, 4H), 3.80 (s, 3H), 3.77 (s, 3H), 3.68 (s, 6H); 13C NMR (400 MHz, CDCl3) δ [ppm] 153.06, 149.93*, 149.88, 139.62*, 139.55, 137.34, 135.83, 135.76, 132.54, 130.27, 129.76, 128.63, 128.60, 128.55, 127.95, 126.65, 122.30*, 122.27, 112.39, 106.05, 69.87, 69.81, 60.94, 56.05, 56.02; IR ν 2938, 2838, 1579, 1279, 1127, 999 cm-1.
Sodium (Z)- 2- methoxy- 5- (3, 4, 5-trimethoxy styryl) phenyl phosphate: 1H NMR (free phosphorus acid, 400 MHz, MeOD) δ [ppm] 7.27 (s, 1H), 7.01 (d, J = 8.53 Hz, 1H), 6.92 (d, J = 8.53 Hz, 1H), 6.55 (s, 2H), 6.48 (dd, J = 18.45, 12.17 Hz, 2H), 3.82 (s, 3H), 3.74 (s, 3H), 3.66 (s, 6H).
Pharmacology: It shows potent cytotoxicity against M.T.T. Again, the tests were repeated with lower concentrations to determine the IC50 values. Table 1 presents the IC50 concentration for CA-4-P (12, assay # 1107) and in comparison describes the values for the cyclopropane derivative (14, assay # 1111) and the diphosphate analog of CA-4.
Anticancer Activity by MTT Assay Method:
Procedure: Cytotoxicity testing in in-vitro mode will be done by using Woerdenbag et al., method. Incubate 2 × 103 cells/well in the 5% CO2 incubator for 24 h to enable them to adhere properly to the 96 well polystyrene microplates. Add test compounds, dissolved in 100% DMSO, in at least five doses, and left for 6 h. Replace the compound plus media with fresh media and incubate the cells for another 48 h in the CO2 incubator at 37 °C. Then, add 10ml MTT [3-(4, 5-dimethylthiazol-2-yl)-2, 5- diphenyl-tetrazolium bromide, and incubate the plates at 37°C for 4 h. Add 100 μl DMSO to all wells and mixed thoroughly to dissolve the dark blue crystals. After a few minutes at room temperature, to ensure that all crystals are dissolved, read the plates on a Spectra Max 190 Microplate Elisa reader, at 570 nm. Read the plates within 1 h of adding the DMSO. Experiments triplicate and calculate the inhibitory concentration (IC) values as follows:
% inhibition = [1-OD (570 nm) of sample well/ OD (570 nm) of control well] × 100.
IC50 is the concentration μg/ml required for 50% inhibition of cell growth as compared to that of untreated control; OD = Optical density.
The same tests as described for CAM were prepared for the MTT shows the results of the first screening with an assay concentration of 30μM.
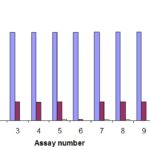
RESULT AND DISCUSSION: The concentration of the assays which were applied to the shell culture was 30 μM. The inhibition of shell proliferation within a certain time is investigated. The bars demonstrate that all compounds with just one exception, the alkyne derivative 261 (assay # 1105) show potent biological activity. When the inhibition of a compound is less than 50% at 30μM, it is considered as inactive. It seems very interesting that also the alkyne derivatives are active at that concentration. For more detailed investigations the test was repeated with a lower concentration to describe the dose-response relationship and the half maximal inhibitory concentration (IC50 value). The graph in Scheme 66 demonstrates the IC50 value for CA-4-P (12, assay # 1107) for CAM cells.
GRAPH 1: IC50 VALUE FOR ALL TESTED COMPOUNDS ON MTT
TABLE 1: IC50 CONCENTRATIONS, (MTT)
| Assay number | Compound number | Calculate IC50 value (μM) MTT |
| 1103 | 260 | 1.24 |
| 1104 | 216 | 20.60 |
| 1105 | 261 | >30 |
| 1106 | 226 | 0.369 |
| 1107 | 12 | 0.0034 |
| 1108 | 237 | 2.85 |
| 1109 | 227 | 1.01 |
| 1110 | 234 | 10.01 |
| 1111 | 14 | 0.202 |
| 1112 | 238 | 09.84 |
| 1113 | 235 | 08.77 |
| 1114 | 236 | 0.397 |
The result is very similar in comparison to CAM, all compounds, except compound 261 (assay # 1105), show potent cytotoxicity against M.T.T. Again, the tests were repeated with lower concentrations to determine the IC50 values. Presents the IC50 concentration for CA-4 (12, assay # 1107) and in comparison describes the values for the cyclopropane derivative (14, assay # 1111) and the diphosphate analog of CA-4 236, assay # 1114). The IC50 values for all tested compounds on MTT are listed in Table 1.
In this study, we found that all compounds with just one exception, the alkyne derivative (261assay # 1105) show potent biological activity. When the inhibition of a compound is less than 50% at 30μM, it is considered as inactive. It seems very interesting that also the alkyne derivatives are active at that concentration.
CONCLUSION: CA-4-P is a biologically very active compound by binding to the colchicine binding site which leads to the inhibition of microtubule polymerization as well as showing the Anticancer effects by selectively shutting down the tumor blood flow. To avoid the disadvantage of rather low in-vivo efficacy resulting from the isomerization of the cis-stilbene derivative to the thermodynamically more stable trans-isomer, our research group started the project for CA-4 analogs synthesis. The incorporation of carbocycles with different ring sizes on the connecting carbon-bridge of the natural compound prevents the system from undergoing cis-trans-isomerization. The synthesis of the cyclopropane derivative of CA-4 (9) via the cyclopropanation reaction with diazomethane, and further analogs with incorporated moieties for better water solubility (268, 273) were achieved within this doctor of philosophy thesis. In cooperation with the Central Drug Research Institute, Lucknow, Uttar Pradesh. I was able to the biological activity of the compounds prepared within this work.
ACKNOWLEDGEMENT: The authors would like to express thanks to the Faculty of CDRI Lucknow for providing laboratory facilities and, innovation center of Bundelkhand University, Jhansi for research work.
CONFLICT OF INTEREST: All authors have no conflict of interest regarding this paper.
REFERENCES:
- Pettit G, Singh S, Hamel E, Lin CM, Alberts D and Garcia-Kendal D: Isolation and structure of the strong cell growth and tubulin inhibitor combretastatin A-4. Experientia 1989; 45(2): 209-11.
- Chaplin D, Pettit G and Hill S: Anti-vascular approaches to solid tumor therapy: evaluation of combretastatin A4 phosphate. Anticancer research 1998; 19(1A): 189-95.
- Dark GG, Hill SA, Prise VE, Tozer GM, Pettit GR and Chaplin DJ: Combretastatin A-4, an agent that displays potent and selective toxicity toward tumor vasculature. Cancer Research 1997; 57(10): 1829-34.
- Siemann DW, Chaplin DJ and Walicke PA: A review and update of the current status of the vasculature-disabling agent combretastatin-A4 phosphate (CA4P). Expert Opin Investig Drugs 2009; 18(2): 189-97.
- Bedford SB, Quarterman CP, Rathbone DL, Slack JA, Griffin RJ and Stevens MFG: Synthesis of water-soluble prodrugs of the cytotoxic agent combretastatin A4. Bioorg Med Chem Lett 1996; 6: 157-160.
- Pettit GR, Temple CJ, Narayanan VL, Varma R, Simpson MJ, Boyd MR, Rener GA and Bansal N: Antineoplastic agents 322. Synthesis of combretastatin A-4 prodrugs. Anticancer Drug Des 1995; 10: 299-309
- Nathan P, Zweifel M, Padhani AR, Koh DM, Ng M, Collins DJ, Harris A, Carden C, Smythe J, Fisher N, Taylor NJ, Stirling JJ, Lu SP, Leach MO, Rustin GJS and Judson I: Phase I trial of combretastatin A4 phosphate (CA4P) in combination with bevacizumab in patients with advanced cancer. Clin Cance Res off J Am Assoc Cancer Res 2012; 18: 3428-3439.
- National Cancer Institute, Understanding Cancer Series. http://www.cancer.gov/cancertopics/understandingcancer.
- Boulet G, Horvath C, Broeck DV, Sahebali S and Bogers J: Human papillomavirus: E6 and E7 oncogenes. The International Journal of Biochemistry and Cell Biology 2011; 39: 2006.
- Galbraith SM, Maxwell RJ, Lodge MA, Tozer GM, Wilson J, Taylor NJ, Stirling JJ, Sena L, Padhani AR and Rustin GJ: Combretastatin A4 phosphate has tumor antivascular activity in rat and man as demonstrated by dynamic magnetic resonance imaging. J Clin Oncol 2003; 21(15): 2831-42.
- Anderson HL, Yap JT, Miller MP, Robbins A, Jones T and Price PM: Assessment of pharmacodynamic vascular response in a phase I trial of combretastatin A4 phosphate. J Clin Oncol 2003; 21(15): 2823-30.
- Rustin GJ, Galbraith SM, Anderson H, Stratford M, Folkes LK, Sena L, Gumbrell L and Price PM: Phase I clinical trial of weekly combretastatin A4 phosphate: clinical and pharmacokinetic results. J Clin Oncol 2009; 21(15): 2815-22.
- Young SL and Chaplin DJ: Combretastatin A4 phosphate: background and current clinical status. Expert Opin Invest Drugs 2004; 13: 1171-1182.
- Galbraith SM, Chaplin DJ, Lee F, Stratford MR, Locke RJ, Vojnovic B and Tozer GM: Effects of combretastatin A4 phosphate on endothelial cell morphology in-vitro and relationship to tumour vascular targeting activity in-vivo. Anticancer Res 2001; 21(1A): 93-102.
- Kanthou C and Tozer GM: The tumor vascular targeting agent combretastatin A-4-phosphate induces reorgani-zation of the actin cytoskeleton and early membrane blebbing in human endothelial cells. Blood 2002; 99(6): 2060-9.
- Kanthou C and Tozer GM: The tumor vascular targeting agent combretastatin A-4-phosphate induces reorgani-zation of the actin cytoskeleton and early membrane blebbing in human endothelial cells. Blood 2004; 99(7): 2060-9.
- Tozer GM, Kanthou C and Baguley BC: Disrupting tumour blood vessels. Nature Reviews 2005; 5: 423-435.
- Voet DJ, Voet JG and Pratt CW: Principles of Biochemistry 2007.
- Horsman MR, Ehrnrooth E, Ladekarl M and Overgaard J: The effect of combretastatin A-4 disodium phosphate in a C3H mouse mammary carcinoma and a variety of murine spontaneous tumors. Int J Radiat Oncol Biol Phys 1998; 42(4): 895-8.
- Dachs GU and Tozer GM: Hypoxia modulated gene expression: angiogenesis, metastasis and therapeutic exploitation. Eur J Cancer 2000; 36(13): 1649-60.
- Dark GG, Hill SA, Prise VE, Tozer GM, Pettit GR and Chaplin DJ: Combretastatin A-4, an agent that displays potent and selective toxicity toward tumor vasculature. Cancer Res 1997; 57(10): 1829-34.
- Pugh CW and Ratcliffe PJ: The von Hippel-Lindau tumor suppressor, hypoxia inducible factor-1 (HIF-1) degradation, and cancer pathogenesis. Semin Cancer Biol 2003; 13(1): 83-9.
- Zhong H, De Marzo AM, Laughner E, Lim M, Hilton DA, Zagzag D, Buechler P, Isaacs WB, Semenza GL and Simons JW: Over-expression of hypoxia-inducible factor 1α in common human cancers and their metastases. Cancer Res 1999; 59(22): 5830-5.
- Boulet G, Horvath C, Broeck DV, Sahebali S and Bogers J: Human papillomavirus: E6 and E7 oncogenes. The International Journal of Biochemistry and Cell Biology 2011; 39: 2006.
- Mason RP, Zhao D, Liu L, Trawick ML and Pinney KG: A perspective on vascular disrupting agents that interact with tubulin: preclinical tumor imaging and biological assessment. Integrative Biology 2011; 3: 375-387.
- Petit GR, Singh SB, Boyd MR, Hamel E, Pettit RK, Schmidt JM and Hogen F: Antineoplastic agents. 291. Isolation and synthesis of combretastatin A- 4, A-5, and A-6. Journal of Medicinal Chemistry 1995; 38: 1666-1672.
- George CS: Targeting the tumor microenvironment through the design and synthesis of potent, small-molecule, anticancer agents. Ph.D. Dissertation, Baylor University, Waco, TX 2012.
- Grubbs RH: Handbook of Metathesis; Ed.; Wiley-VCH: Weinheim, 2003; (b) Cossy J, Arseniyadis S and Meyer C: Metathesis in Natural Product Synthesis; Eds.; Wiley-VCH: Weinheim 2010.
How to cite this article:
Kumar S, Koushal C and Jain SK: Synthesis and biological evaluation of cyclopropane derivative of combretastatin CA-4-Phosphate. Int J Pharmacognosy 2017; 4(8): 273-80. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(8).273-80.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
273-280
679
2066
English
IJP
S. Kumar*, C. Kaushal and S. K. Jain
Institute of Pharmacy, Shri Venkateshwara University, Gajroula, Amroha, Uttar Pradesh, India.
shailowrath@gmail.com
03 April 2017
23 June 2017
28 June 2017
10.13040/IJPSR.0975-8232.IJP.4(8).273-80
01 August 2017