SUBCHRONIC TOXICITY OF TIKONI TEA FROM VITEX MADIENSIS OLIV. LEAVES (LAMIACEAE) AQUEOUS EXTRACT IN THE WISTAR RAT
HTML Full TextSUBCHRONIC TOXICITY OF TIKONI TEA FROM VITEX MADIENSIS OLIV. LEAVES (LAMIACEAE) AQUEOUS EXTRACT IN THE WISTAR RAT
G. F. Nsonde Ntandou *, E. B. Lekounda Yoly, Motondo Eric and G. S. Makemba Nkounkou
Centre d’Etude et de Recherche Médecins d’Afrique (CERMA) 3, B.P. 45, Brazzaville, Congo.
ABSTRACT: Vitex madiensis, consumed daily as a tea named Tikoni, is widely used in traditional medicine in Congo Brazzaville to treat fever, pain, inflammation, malaria, dysentery, female infertility, mental illness and epilepsy. Its aqueous leaf extract does not exhibit acute toxicity. Its pharmacological effects against pain, inflammation, insomnia, oxidative stress, and Plasmodium falciparum are very promising. The objective of this study was to investigate the subchronic toxicity of its aqueous extract at a dose of 200 mg/kg orally in Wistar rats over 42 days. Toxicity assessments were conducted on days 14, 28, and 42. The results show that the Tikoni tea aqueous extract does not significantly reduce body weight gain and the growth of certain vital organs. However, liver and lung weights in males, and liver and spleen weights in females, decreased significantly. This extract decreased platelet counts in both sexes and significantly increased leukocyte counts in males. ALT and ALP activities increased significantly in males, while in females, AST, ALT, and ALP activities decreased significantly, and blood glucose levels increased. No significant changes were observed in cholesterol, urea, triglycerides, creatinine, total protein, direct bilirubin, hemoglobin, hematocrit, MCV, MCH, and MCHC levels.
Keywords: Subchronic toxicity, Vitex madiensis tea, Organs, Biochemical and hematological
INTRODUCTION: Medicinal plants constitute a group of plants of great socio-economic importance because they contain active components used in the treatment of various diseases. However, studies have shown that not all medicinal plants are safe for direct human use. Although the overall number of deaths due to toxic plants is low, they are considered a significant cause of morbidity and mortality, plant toxicity deserves greater attention 1.
Studies conducted on approximately 1,500,000 plants have shown that most of these plants contain toxic substances 18. Vitex madiensi Oliv. is pharmacologically effective and widely used in traditional medicine in Congo in particular and in Africa, Asia, and Latin America in general 5. In Africa, the medicinal properties of the plant are known and vary among different populations 21.
For example, in Cameroon, the roots and aerial parts are used to treat headaches, indigestion, and stomach aches. In the Democratic Republic of Congo, the bark is used to treat hemorrhoids; the leaves for coughs, stomach aches, and the flu; and the entire plant for malaria 20, 25. In the republic of Congo, the bark pulp and the inner part of the root bark are used to treat dysentery-like diarrhea, female infertility, and the mentally ill or epileptics, respectively 16. Some ethnic groups in the Congo drink an aqueous decoction of the bark for spiritual deliverance. The juice of the leaves is used in ear drops to treat ear infections; applied to the gums for toothaches 6. A decoction or fumigation of these leaves relieves headaches. A cold gargle of the leaf decoction is a good remedy for bleeding after a tooth extraction. An ointment made from the roots of Vitex madiensis is used to treat pinworm infections 16. Phytochemical studies of leaf extracts revealed the presence of tannins, anthocyanins, flavones, saponins, mucilage, sterols, and triterpenes. Pharmacological and toxicological studies conducted by our predecessors on the Congo sample showed that the aqueous extract of Vitex madiensis leaves does not exhibit acute or cellular toxicity in rodents and displays interesting analgesic, anti inflammatory, sedative, and antioxidant activities 24, 28. An absence of cytotoxicity in human cells and a promising antiplasmodial effect were demonstrated in the Gabonese sample in the Central African sub-region 32. Despite its medicinal properties, it is also consumed daily as tea by some farmers 24. To determine whether the abundant and continuous use of this species by local populations has long term consequences, this study aimed to investigate chronic toxicity in rats.
MATERIALS AND METHODS:
Tikoni Tea Preparation: Tikoni tea is made with the leaves of Vitex madiensis Oliv. (Lamiaceae). Plant material were collected in the savanna of south of Brazzaville in Ngangalingolo in May 2019. Botanical identification was carried out by Professor Jean Marie MOUTSAMBOTE of the National Institute for Research in Exact and Natural Sciences (IRSEN), and a specimen was deposited in the National Herbarium and registered under number 7658 25. The plant leaves were dried for 7 days at room temperature, approximately 28°C, in the dark. The dried plant material was ground using an IKA-WERKE GmbH-CO-KG, D-79219, Staufen, machine equipped with a 0.25 mm sieve. The obtained powder stocked in a bottle hermatically closed is Tikoni tea.
Animal Material: The animal material consisted of 48 male and female Wistar rats weighing between 150 and 250 g, provided by the animal facility of the National Institute of Health Sciences Research (IRSSA) in Brazzaville, Republic of Congo. These animals were kept under standard conditions (12 hours of light and 12 hours of darkness at an ambient temperature of 26 ± 1°C) and had free access to standard food and drinking water.
Tikoni Tea’s Aqueous Extract Preparation: One hundred (100) g of Tikoni mass were placed in a glass flask containing 1000 mL of distilled water and heated to a boil for thirty (30) minutes on a heating mantle at a temperature of 100°C. The decoction was filtered through absorbent cotton. The resulting filtrate was then placed in an oven at a reduced temperature of 60°C. The dry extract obtained was used to prepare the test solution.
Dose Selection: A dose of 200 mg/kg was chosen based on previous studies 27.
Subchronic Toxicity Study: For the determination of subchronic toxicity, the experimental protocol used was that described by Nsonde Ntandou et al. (2015) 27, 29.
Grouping of Mice 43: 48 rats, male and female were divided into 4 groups of 12 rats each. Groups 1 and 3 (male and female) served as the control group and received distilled water at a dose of 10 mL/kg orally. Groups 2 and 4 (male and female) were treated with an aqueous extract of Tikoni tea at a dose of 200 mg/kg orally. Treatments were administered daily at regular intervals until the evaluation days.
Measurement of Water intake and Food Consumption 31: The volume of water and the amount of food consumed were measured daily for 42 days by weighing the food remaining in the trough and the volume of water remaining in the water bottles.
Study of Rat Weight Gain: The body weight of the rats used in this study was measured before administration of the extract and then every 2 days after administration.
Evaluation of Hematological and Biochemical Parameters 28, 45: Toxicity assessments were performed on days 14, 28, and 42. Rats were fasted overnight before each assessment. On day 0 (before treatment) and on days 14, 28, and 42 (after treatment with Tikoni tea aqueous extract), four rats from each group were anesthetized with ether. Blood was then collected from the jugular vein using EDTA and dry tubes for hematological and biochemical testing respectively. Blood samples collected in tubes containing an anticoagulant (EDTA) were immediately used to determine white blood cell count, red blood cell count, hematocrit, hemoglobin, MCV, MCHC, MCHC, and platelet count. Blood samples in dry tubes were centrifuged at 4000 rpm for 30 minutes after coagulation to separate the serum from the red blood cell pellet. The decanted serum was used for the analysis of direct bilirubin, alkaline phosphatase (ALP), transaminases (ALT; AST), urea, creatinine, triglycerides, total protein, cholesterol, and blood glucose. The measurement of the enzymatic activity of the two transaminases was based on the kinetic enzyme method.
A colorimetric kinetic assay, in accordance with the IFCC (International Federation of Clinical Chemistry and Laboratory Medicine), was used to analyze phosphatase activity. Bilirubin, in the presence of diazero sulfanilic acid and in an alkaline medium, forms a red colored compound. Of the two dyes presented in serum, bilirubin gucuromide and free bilirubin loosely bound to albumin, only bilirubin-gucuromide reacts directly in an aqueous solution (direct bilirubin). Creatinine forms a red complex in a basic picrate solution. The absorbance, at predetermined times during the conversion is proportional to the creatinine concentration in the sample. Triglycerides are enzymatically hydrolyzed into glycerol and free fatty acids.
The released glycerol is first phosphorylated by glycerol kinase and then oxidized by glycerol-3 phosphate oxidase, releasing an equivalent amount of hydrogen peroxide (H₂O₂). The H₂O₂ participates in a Trinder reaction, which leads to the formation of a red quinoneimine dye. The intensity of the color formed is proportional to the triglyceride concentration in the sample. In the presence of glucose oxidase (GOD), β-D-glucose is oxidized to gluconic acid and peroxide. Since glucose exists in both α and β forms in solution, complete conversion of glucose requires mutarotation of α-D-glucose to β-D glucose. This latter reaction is accelerated in the presence of the mutarotase enzyme (MRO). After glucose oxidation, the hydrogen peroxide (H₂O₂) formed is measured by oxidative coupling of 4 aminopantipyrine (AAP) to 4-chlorophenol in the presence of peroxidase (POD), yielding a quinonimine red dye. Cholesterol and its esters are released from lipoproteins by detergents. Cholesterol esterase hydrolyzes the esters, and H₂O₂ is formed in the subsequent enzymatic oxidation of cholesterol by cholesterol oxidase according to the following equations. In the last reaction, a red quinoneimine dye is formed, the intensity of which is proportional to the concentration of cholesterol in the sample. Urea is hydrolyzed to ammonia (NH4+) and CO2. Ammonia reacts with α-ketoglutarate to form L-glutamate and NAD+ from NADH7. This reaction is catalyzed by glutamate dehydrogenase (GLDH). The decrease in NADH is proportional to the urea concentration. Proteins in a basic copper sulfate medium containing tartate (biuret reagent) form a complex colored blue-violet. The intensity of the color formed is proportional to the total protein concentration in the sample.
Organ Collection: After blood sampling at each evaluation, the rats were subsequently sacrificed. The vital organs (liver, heart, lungs, spleen, and kidneys) were collected, weighed, and macroscopically examined. We looked for changes in size, color, and shape, as well as the presence of malformations in the outer layer of the skin 10.
Statistical Analyses: The values presented in the tables correspond to calculations expressed as means plus or minus (±) standard error (MSE). Means were compared using Student's t-test. The significance level was set at p < 0.05. The various statistical analyses of the results were performed using Excel software.
RESULTS:
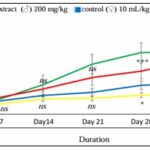
Effect of Tikoni Tea’s Aqueous Extract on Body Weight Gain: The subacute toxicity study yielded results shown in Fig. 1. This figure illustrates the effect of the extract on the weight gain of male and female rats after 42 days of treatment with a single dose of 200 mg/kg and distilled water. A non-significant decrease in body weight was observed in both males and females of the animals treated with the aqueous extract compared to the controls treated with distilled water. On day 28, a highly significant decrease (p < 0.001) in weight was observed in males, and a significant decrease (p < 0.05) in females.
FIG. 1: EFFECT OF AQUEOUS EXTRACT OF TIKONI TEA ON BODY WEIGHT GAIN IN WISTAR RATS. Results are expressed as mean ± standard error, n = 4 rats per group. *p < 0.05; ***p < 0.001 significant difference compared to controls.
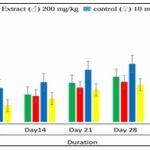
Cumulative Effect of the Aqueous Extract on Food Intake: The Fig. 2 shows the cumulative effect of the aqueous extract of Tikoni tea on the food intake in rats (males and females) subjected to subchronic treatment of 200 mg/kg for 42 days. A decrease in food consumption is observed in the treated rats (males and females) compared to the control group (males and females).
FIG. 2: CUMULATIVE EFFECT OF FOOD INTAKE IN TREATED RATS (MALE AND FEMALE); N = 4 RATS PER GROUP. Results are expressed as mean ± standard error, n = 4 rats per group.
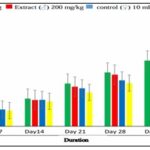
Cumulative Effect of the Aqueous Extract of Tikoni Tea on Water Intake: The Fig. 3 shows the cumulative effect of the aqueous extract of Tikoni tea on the water intake of rats (males and females) subjected to subchronic treatment with 200 mg/kg for 42 days. A decrease in water intake is observed in the treated rats (males and females) compared to the control group (males and females).
FIG. 3: CUMULATIVE EFFECT OF WATER INTAKE IN CONTROL (MALES AND FEMALES) AND TREATED (MALES AND FEMALES) RATS, N = 4 RATS PER GROUP. (E.D. M) DISTILLED WATER (MALE), (EXT. AQ. M) AQUEOUS EXTRACT (MALE), (E.D. F) DISTILLED WATER (FEMALE), (EXT. AQ. F) AQUEOUS EXTRACT (FEMALE)
Effect of the Aqueous Extract of Tikoni Tea on Organ Weight: Table 1 shows the effect of the aqueous extract of Tikoni tea on the organ weight of male rats subjected to subchronic treatment with 200 mg/kg for 42 days. A significant decrease (p<0.01) in liver weight was observed after 42 days of treatment, and a significant decrease (p<0.05) in lung weight was observed after 14, 28, and 42 days of treatment, respectively, compared to controls. Results are expressed as mean ± standard error, n = 4 rats per batch, significant for *p < 0.05; **p < 0.01; ns: not significant compared to control rat.
TABLE 1: EFFECT OF TIKONI TEA’S AQUEOUS EXTRACT ON ORGAN WEIGHT IN MALE RATS
| Organs | Day 0 | Products | Day 14 | Day 28 | Day 42 |
| Control | 6,93±0,05 | 7,51±0,05 | 7,52±0,03 | ||
| Liver | 8,61±0,26 | ||||
| Extract | 6,33±0,10ns | 7,27±0,10ns | 5,52±0,30** | ||
| Control | 1,11±0,02 | 1,03±0,02 | 1,02±0,02 | ||
| Heart | 1,03±0,02 | ||||
| Extract | 0,99±0,07ns | 0,84±0,003ns | 0,95±0,05ns | ||
| Control | 1,31±0,17 | 1,94±0,02 | 1,31±0,17 | ||
| lungs | 1,50±0,02 | ||||
| Extract | 0,93±0,01* | 1,36±0,06* | 0,93±0,005* | ||
| Control | 0,48±0,03 | 0,51±0,005 | 0,67±0,10 | ||
| Spleen | 0,54±0,04 | ||||
| Extract | 0,49±0,02ns | 0,48±0,03ns | 0,40±0,05ns | ||
| Control | 0,77±0,01 | 0,80±0,01 | 0,72±0,01 | ||
| Left kidney | 0,81±0,03 | ||||
| Extract | 0,66±0,04ns | 0,65±0,01ns | 0,61±0,01ns | ||
| Control | 0,82±0,01 | 0,83±0,01 | 0,73±0,01 | ||
| Right kidney | 0,64±0,008 | ||||
| Extract | 0,71±0,06ns | 0,68±0,006ns | 0,64±0,02ns | ||
Subjected to subchronic treatment with 200 mg/kg for 42 days. A significant decrease (p < 0.001) in liver weight was observed after 42 days of treatment, and a significant decrease (p < 0.05) in spleen weight was observed after 14, 28, and 42 days of treatment compared to controls.
TABLE 2: EFFECT OF AQUEOUS EXTRACT OF ON ORGAN WEIGHT IN FEMALE RATS
| Organs | Day 0 | Products | Day 14 | Day 28 | Day 42 |
| Control | 6,04±0,37 | 6,65±0,37 | 5,91±0,01 | ||
| Liver | 5,9±0,12 | ||||
| Extract | 6,75±0,25ns | 6,29±0,25ns | 4,31±0,01*** | ||
| Control | 0,99±0,07 | 0,74±0,02 | 0,72±0,01 | ||
| Heart | 0,77±0,02 | ||||
| Extract | 0,91±0,06ns | 0,71±0,02ns | 0,64±0,01ns | ||
| Lungs | 1,38±0,09 | Control | 0,95±0,02 | 1,33±0,16 | 0,95±0,02 |
| Extract | 1,09±0,15ns | 1,24±0,21ns | 1,09±0,15ns | ||
| Control | 0,97±0,05 | 0,91±0,09 | 0,98±0,09 | ||
| Spleen | 0,5±0,01 | ||||
| Extract | 0,65±0,03* | 0,60±0,06* | 0,45±0,04* | ||
| Control | 0,65±0,05 | 0,59±0,005 | 0,56±0,02 | ||
| Left kidney | 0,44±0,01 | ||||
| Extract | 0,66±0,09ns | 0,52±0,17ns | 0,44±0,01ns | ||
| Control | 0,63±0,03 | 0,65±0,02 | 0,52±0,01 | ||
| Right kidney | 0,48±0,01 | ||||
| Extract | 0,64±0,06ns | 0,55±0,02ns | 0,45±0,03ns | ||
Results are expressed as mean ± standard error, n = 4 rats per group, significant for *p < 0.05; ***p < 0.001; ns : not significant compared to control rats.
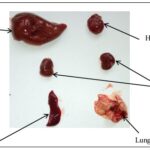
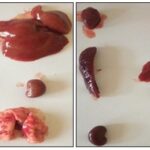
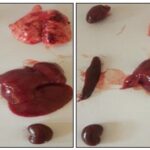
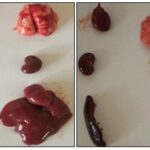
Effect of the Aqueous Extract of Tikoni Tea on Organ Morphology: After macroscopic observation of the different organs collected, their sizes, shapes, and colors were found to be normal Fig. 4 (a, b, c, d). The liver, heart, spleen, and kidneys appear reddish-brown or burgundy, and the lungs are pale pink with variable typical morphologies.
FIG. 4A: PHOTOGRAPH OF THE VITAL ORGANS BEFORE TREATMENT
FIG. 4B: PHOTOGRAPH OF THE TREATED ORGANS WITH CONTROL (LEFT) AND AQUEOUS EXTRACT OF TIKONI (RIGHT) AFTER 14 DAYS OF TREATMENT
FIG. 4C: PHOTOGRAPH OF THE TREATED ORGANS WITH CONTROL (LEFT) AND AQUEOUS EXTRACT OF TIKONI (RIGHT) AFTER 28 DAYS OF TREATMENT
FIG. 4D: PHOTOGRAPH OF THE TREATED ORGANS WITH CONTROL (LEFT) AND AQUEOUS EXTRACT OF TIKONI (RIGHT) AFTER 42 DAYS OF TREATMENT
Effect of the aqueous extract of V. madiensis Oliv leaves on hematological parameters The Table 3 shows the effect of the aqueous extract of Tikoni tea on the hematological parameters of male rats subjected to subchronic treatment with 200 mg/kg for 42 days.
A significant increase (p < 0.01) in white blood cell count was observed after 14, 28, and 42 days of treatment, and a decrease in platelet count (p < 0.01) was observed after 14, 28, and 42 days of treatment compared to controls 15.86 ± 0.40.
TABLE 3: TIKONI TEA’S AQUEOUS EXTRACT EFFECTS ON HEMATOLOGICAL PARAMETERS IN MALE RATS
| HP | Day 0 | Products | Day 14 | Day 28 | Day 42 |
| Control | 8,73±1,42 | 9,45±1,17 | 10,72±0,42 | ||
| WBC 109/l | 13,63±1,24 | ||||
| Extract | 12,17±1,45** | 14,56±4,40** | 15,86±0,40** | ||
| Control | 7,66±0,48 | 8,56±0,08 | 8,56±0,01 | ||
| RBC1012/l | 7,67±0,48 | ||||
| Extract | 7,73±0,43ns | 9,18±0,37ns | 9,6±0,25ns | ||
| Control | 15,1±0,69 | 16,8±0,17 | 15,16±0,14 | ||
| HGBg/dl | 14,56±0,87 | ||||
| Extract | 15,5±0,51ns | 16,95±0,66ns | 16,53±0,14ns | ||
| Control | 48,34±1,88 | 50,28±0,46 | 49,27±0,01 | ||
| HCT % | 42,56±1,34 | ||||
| Extract | 47,96±2,03ns | 53,70±1,86ns | 54,95±1,18ns | ||
| Control | 63,4±1,55 | 59,5±0,42 | 57,66±0,33 | ||
| MCVfl | 63,66±1,58 | ||||
| Extract | 62,2±0,86ns | 58,5±0,28ns | 57±0,57ns | ||
| Control | 17,7±0,17 | 19,73±0,54 | 20,15±0,49 | ||
| MCHpg | 20,46±0,57 | ||||
| Extract | 17,26±0,31ns | 18,4±0,57ns | 19,8±0,34ns | ||
| Control | 30,8±0,28 | 31,4±0,75 | 33,63±0,95 | ||
| MCHCg/dl | 32,46±1,42 | ||||
| Extract | 30,1±0,40ns | 31,23±0,26ns | 32,33±0,31ns | ||
| Control | 821±52,53 | 886±72,74 | 934±10,39 | ||
| PLT.109/l | 859,33±74,02 | ||||
| Extract | 759,6±26,84** | 752±93,21** | 729±5,77** | ||
(HP) Hematological parameters. Results are expressed as mean ± standard error, n = 4 rats per group, significant for *p < 0.05; **p < 0.01; ns: not significant compared to control rats.
The Table 4 shows the effect of the aqueous extract of Tikoni tea on the hematological parameters of female rats subjected to subchronic treatment with 200 mg/kg for 42 days.
A significant decrease in platelets (p < 0.01) was observed after 14, 28, and 42 days of treatment compared to controls.
TABLE 4: TIKONI TEA’S AQUEOUS EXTRACT EFFECTS ON HEMATOLOGICAL PARAMETERS IN FEMALE RATS
| HP | Day 0 | Treatment | Day 14 | Day 28 | Day 42 |
| Control | 9,87±3,49 | 10,55±1,07 | 12,88±0,19 | ||
| WBC 109/l | 11,10±1,19 | ||||
| Extract | 10,39±3,42ns | 10,59±0,32ns | 13,89±0,34ns | ||
| Control | 6,86±0,64 | 7,77±0,30 | 8,42±0,07 | ||
| RBC1012/l | 6,3±0,47 | ||||
| Extract | 7,38±0,25ns | 8,15±0,42ns | 8,48±0,10ns | ||
| Control | 13,82±0,24 | 14,06±0,08 | 14,9±0,15 | ||
| HGBg/dl | 13,03±0,44 | ||||
| Extract | 14,07±0,36ns | 14±0,86ns | 14,83±0,06ns | ||
| Control | 39,62±0,88 | 44,35±1,01 | 47,88±0,22 | ||
| HCT % | 39,90±1,58 | ||||
| Extract | 44,43±1,86ns | 47,85±1,31ns | 47,98±0,68ns | ||
| Control | 46,33±0,88 | 60±0,57 | 62,4±1,27 | ||
| MCVfl | 63,8±2,57 | ||||
| Extract | 40,6±2,59ns | 56,66±1,45ns | 61,5±0,28ns | ||
| Control | 17,9±0,23 | 18,93±0,45 | 21,1±0,28 | ||
| MCHpg | 20,83±0,81 | ||||
| Extract | 17,63±0,20ns | 18,83±0,20ns | 20,9±0,34ns | ||
| Control | 30,93±0,26 | 32,1±0,34 | 32,73±0,37 | ||
| MCHCg/dl | 32,73±0,37 | ||||
| Extract | 30,73±0,66ns | 31,16±0,43ns | 31,83±0,20ns | ||
| Control | 901,33±49,94 | 914±12,70 | 996±46,76 | ||
| PLT.109/l | 850,33±76,17 | ||||
| Extract | 680±97,28** | 777,66±30,31** | 780,33±20,49** | ||
HP: Hematological parameters. Results are expressed as mean ± standard error, n= 4 rats per group, significant for *p < 0.05; **p < 0.01; ns: not significant compared to control rats.
Tikoni Tea’s Aqueous Extract of on Biochemical Parameters: A Table 5 shows the effect of the aqueous extract of Tikoni tea on the biochemical parameters of male rats subjected to subchronic treatment with 200 mg/kg for 42 days. A significant increase (p < 0.05; p < 0.01) in ALT activity was observed after 14, 28, and 42 days, respectively, and a significant increase (p < 0.05; p < 0.01) in ALP activity was observed after 28 and 42 days of treatment, respectively, compared to controls. A Table 6 shows the effect of the aqueous extract of Tikoni tea on the biochemical parameters of male rats subjected to subchronic treatment with 200 mg/kg for 42 days. A significant decrease in AST activity (p<0.05) and ALP activity (p<0.01) was observed after 42 days of treatment, a significant decrease (p < 0.05; p < 0.01) in ALT activity after 14, 28, and 42 days of treatment, and a significant increase (p<0.01) compared to controls.
TABLE 5: EFFECT OF AQUEOUS EXTRACT OF TIKONI TEA ON HEMATOLOGICAL PARAMETERS IN MALE RATS
| BP | Day 0 | Products | Day 14 | Day 28 | Day 2 |
| Control | 138,13±8,96 | 202,70±28,35 | 244,7±53,61 | ||
| AST(U/L) | 273,89±115,33 | ||||
| Extract | 136,80±13,45ns | 254,44±85,95ns | 268,68±74,93ns | ||
| Control | 35,59±4,61 | 40,68±8,01 | 41,32±1,70 | ||
| ALT(U/L) | 65,91±1,30 | ||||
| Extract | 59,24±12,28* | 64,08±18,58** | 70,26±17,37** | ||
| Control | 136,80±13,45 | 173,80±39,42 | 175,28±0,59 | ||
| ALP (U/L) | 207,35±5,46 | ||||
| Extract | 138,13±8,96ns | 237,75±11,64* | 275,70±16,67** | ||
| Control | 0,15±0,04 | 0,22±0,04 | 0,13±0,01 | ||
| Bilirubin (mg/L) | 0,2±0,02 | ||||
| Extract | 0,1±0,03ns | 1,41±0,56ns | 0,18±0,30ns | ||
| Control | 87,81±2,64 | 109,33±4,85 | 108,21±33,44 | ||
| Cholesterol (mg/dL) | 116,61±22,60 | ||||
| Extract | 100,35±20,18ns | 112,10±2,85ns | 113,74±5,17ns | ||
| Control | 40,79±8,02 | 47,63±0,022 | 56,81±21,18 | ||
| Protein(g/L) | 115,33±1,29 | ||||
| Extract | 56,81±21,18ns | 60,66±0,37ns | 82,73±31,70ns | ||
| Control | 0,57±0,07 | 0,61±0,02 | 0,58±0,08 | ||
| Creatinine (mg/dL) | 0,61±0,005 | ||||
| Extract | 0,47±0,03ns | 0,49±0,06ns | 0,57±0,04ns | ||
| Control | 40,8±3,64 | 47,41±0,16 | 62,06±2,39 | ||
| Urea (mg/dL) | 38,78±0,82 | ||||
| Extract | 55,74±1,99ns | 61,10±7,77ns | 61,51±7,29ns | ||
| Control | 58,16±1,75 | 79,65±7,46 | 96,06±7,47 | ||
| Triglicerides (mg/dL) | 81,41±36,53 | ||||
| Extract | 86,39±6,98ns | 108,48±12,06ns | 109,42±23,85ns | ||
| Blood glucose (mg/dL) | 113,82±55,21 | Control | 76,32±24,62 | 88,94±6,55 | 110,31±22,13 |
| Extract | 88,52±3,66ns | 91,64±18,01ns | 104,81±10,67ns | ||
HP: Hematological parameters. Results are expressed as mean ± standard error, n = 4 rats per batch, significant for *p < 0.05; **p < 0.01 ***p < 0.001; ns: not significant compared to control rats.
TABLE 6: BIOCHEMICAL PARAMETERS OF TIKONI TEA'S AQUEOUS EXTRACT IN FEMALE RATS
| BP | Day 0 | Products | Day 14 | Day 28 | Day 42 |
| Control | 144,76±2,80 | 246,92±2,22 | 245,17±13,46 | ||
| AST(U/L) | 292,71±5,65 | ||||
| Extract | 193,67±17,61ns | 257,50±3,49ns | 277,44±7,01* | ||
| Control | 44,77±0,21 | 54,15±1,97 | 55,19±0,60 | ||
| ALT(U/L) | 71,82±3,81 | ||||
| Extract | 59,86±3,43* | 70,08±6,88* | 88,44±0,10* | ||
| Control | 128,44±0,66ns | 130,74±25,30ns | 184,1±10,04 | ||
| APL(U/L) | 164,53±23,77 | ||||
| Extract | 129,1±9,41 | 144,76±2,80 | 193,67±17,61 ** | ||
| Control | 0,51±0,15 | 0,4±0,03 | 0,32±0,40 | ||
| Bilirubin (mg/L) | 0,51±0,44 | ||||
| Extract | 0,27±0,03ns | 0,31±0,005ns | 0,22±0,01ns | ||
| Cholesterol (mg/dL) | 143,65±12,76 | Control | 95,41±2,13 | 112,76±7,29 | 118,7±10,14 |
| Extract | 92,32±16,12ns | 111,9±8,13ns | 111,81±0,47ns | ||
| Control | 83,29±14,57 | 95,40±29,60 | 168,62±12,99 | ||
| Protein (g/L) | 110,15±1,36 | ||||
| Extract | 77,30±3,08ns | 86,95±4,47ns | 167,9±14,21ns | ||
| Control | 0,44±0,07 | 0,51±0,03 | 0,59±0,08 | ||
| Creatinin (mg/dL) | 0,63±0,02 | ||||
| Extract | 0,32±0,08ns | 0,36±0,06ns | 0,56±0,14ns | ||
| Control | 55,86±3,05 | 57,26±2,85 | 63,28±1,48 | ||
| Urea (mg/dL) | 43,77±10,92 | ||||
| Extract | 48,68±0,49ns | 54,01±3,31ns | 59,5±4,14ns | ||
| Control | 83,43±10,27 | 93,70±7,19 | 114,51±20,48 | ||
| Triglicerides (mg/dL) | 111,32±5,12 | ||||
| Extract | 70,21±0,95ns | 81,60±12,26ns | 99,32±2,79ns | ||
| Control | 84,16±3,22 | 100,19±1,68 | 138,83±4,27 | ||
| Blood glucose (mg/dL) | 120,63±17,54 | ||||
| Extract | 87,25±2,52ns | 144,51±2,39** | 161,65±4,47** | ||
BP: Biochemical parameters. Results are expressed as mean ± standard error, n = 4 rats per batch, significant for *p < 0.05; **p < 0.01; ***p < 0.001; ns: not significant compared to control rats.
DISCUSSION: The aim of this study was to investigate the subchronic toxicity of an aqueous extract Tikoni tea from Vitex madiensis Oliv. leaves in Wistar rats. This study is of great importance because this species is widely used in rural areas to treat several ailments, including malaria, inflammation, pain, and fever. It is also the basis of the traditional tea known locally as tikoni. Changes in body weight are used as an indicator of the adverse effects of chemical compounds 17. Weight loss is correlated with the animal's physiological state. The results obtained showed a non-significant decrease in body weight in animals treated with aqueous Tikoni extract, compared to controls treated with distilled water, in both males and females. This decrease, although not significant, could be explained either by a reduction in water and food intake due to decreased appetite, or by malabsorption reducing the amount of food absorbed 31, 35. Food intake is regulated by the hypothalamus and depends on the interaction between two regions: the "appetite center," located laterally in the nuclear region of the medial prosencephalic fasciculus at its junction with the pallidohypothalamic fibers, and a medial "satiety center" located in the ventromedial nucleus. Stimulation of the appetite center triggers feeding behavior in conscious animals, and its destruction in healthy animals induces severe and potentially fatal anorexia. Stimulation of the ventromedial nucleus leads to the cessation of food intake 13.
Our results corroborate those of Ntabaza et al. (2024), who also studied the subacute toxicity of an aqueous extract of Vitex madiensis from the Democratic Republic of Congo after oral administration in another mammalian animal model, the guinea pig, and also observed a significant decrease in weight on day 28 24. The decreased water intake is likely due to stress factors and the husbandry conditions of the rats. Similar findings were reported by Otmani and Yahiaoui 33, who studied the acute and subacute toxicity of Fumaria officinalis in albino Wistar mice. Organ weight is an important indicator of pathology and physiological status in animals 36.
After macroscopic observation of the various organs collected, their sizes, shapes, and colors were found to be normal. However, a significant decrease in liver weight was observed after 42 days of treatment in both sexes, and a significant decrease in lung and spleen weight was also observed during all assessments in male and female rats, respectively. A decrease in spleen weight, or splenic atrophy, may be due to a reduction in demand or function of the organ; it is often a sign of exhaustion or chronic organ distress. The decrease in lung weight, often linked to atrophy or destruction of the lung parenchyma, could be caused by a nutritional deficiency which itself would be due to a decrease in water and food consumption 37. A decrease in liver weight would be due to a poor diet marked by a decrease in water and food consumption or in a liver metabolism dysfionnement 17. Our results are similar to those of Etame et al. (2017), who demonstrated metabolic disturbance of the liver in a study of acute and subacute toxicity of Fumaria officinalis in albino mice 12. The hematopoietic system is one of the most sensitive targets of toxic compounds and an important indicator of physiological and pathological status in humans and animals 23. Changes in the hematopoietic system have a higher predictive value for human toxicity when the data come from animal studies 11, 30. In this regard, bone marrow activity and intravascular effects were monitored by hematological examinations. The results indicate a significant increase in white blood cell (WBC) count after 14, 28, and 42 days of treatment in males but not in females; however, platelet levels in both sexes decreased significantly after 14, 28, and 42 days of treatment.
The significant thrombocytopenia observed could be due to bone marrow and liver disorders. The liver produces thrombopoietin, a hormone that stimulates platelet production in the bone marrow. Liver dysfunction can therefore decrease this production. Thrombocytopenia would thus be linked to liver atrophy, characterized by weight loss, and to liver dysfunction, characterized by the significant elevation of transaminases observed 37. The increase in leukocyte count may be due to an overproduction of hematopoietic regulatory elements such as CSF (colony-stimulating factor), EPO (erythropoietin), and TPO (thrombopoietin) by macrophages and bone marrow stromal cells, thus creating a local environment favorable to hematopoiesis 9, 44.
Hyperleukocytosis may be due either to hematopoietic activity of the aqueous extract of Tikoni tea, as observed with certain plant extracts, or to an inhibitory effect on leukocyte migration out of the blood, as observed with corticosteroids, such as steroidal anti-inflammatory drugs like prednisone 4. The decrease in platelet count in treated rats compared to controls indicates that the aqueous extract affects platelet production or induces thrombocytopenia (a reduction in the number of platelets in the blood). This effect is among the evidence of toxic effects on hematopoiesis. Furthermore, with a decrease in platelet count, there is an increased risk of bleeding 42. These results corroborate those of James et al. (2010), who reported that platelet counts decreased in female rats and mice after injection of the aqueous extract of Polygala fruticosa 19. The liver is the primary target of toxicity and the first organ exposed to everything absorbed in the small intestine; it metabolizes foreign substances into compounds that can be hepatotoxic 38. The liver works in conjunction with the kidneys to remove toxic substances from the blood 43. A study of renal and hepatic function can therefore be useful in assessing the toxic effects of medicinal plants 35.
These tests primarily include the determination of AST, ALT, CREAT, and others, and any necrosis of liver cells leads to a significant increase in AST and ALT enzymes in the blood serum 2, 14. Serum studies performed on rats treated with aqueous extract of Tikoni tea show a significant increase in ALT activity after 14, 28, and 42 days of treatment, an increase in ALP activity after 28 and 42 days in male rats, and a significant decrease in AST and ALP activity after 42 days of treatment, a decrease in ALT activity after 14, 28, and 42 days of treatment, as well as an increase in blood glucose in females. Transaminases (AST, ALT) are plasma enzymes of hepatic origin that allow for the assessment of liver function 22. AST is considered a good indicator of liver function and a biomarker for predicting potential toxicity 17. ALT is located exclusively in the cytoplasm of hepatocytes. It can be released into the bloodstream as a consequence of increased membrane permeability or membrane necrosis, secondary to hepatocyte damage 41.
Elevated levels of this enzyme often indicate hepatocellular injury. Measuring its activity is more specific for liver damage than measuring AST 26. ALP is generally measured to indicate bile duct obstruction. Elevated ALP levels are found in rapidly dividing cells or those in which metabolism is active 37. These cells include the epithelium of the bile ducts and liver, circulating granulocytes, intestinal epithelium, renal proximal tubule, placenta, and lactating mammary glands. ALP elevations occur in primary biliary cirrhosis; in conditions of disorganization of the liver architecture; and in diseases characterized by inflammation, regeneration, and obstruction of the intrahepatic bile canaliculi 39. These results are consistent with those of Ntabaza et al. (2023), who noted a significant increase in ALP with the same plant in Guinea pig toxicity study 24. In our study, the activity of ALP and ALT increased significantly in male rats, suggesting that the aqueous extract of Tikoni tea has direct effects on the liver and kidneys of male rats. A histological study is needed for more precise information on the organ's condition; in addition, a chronic toxicity study is recommended 15. These results are consistent with those of Witthawaskul et al. (2003), who noted a significant increase in ALP activity in rats injected with Schefflera leucantha extract 45.
Analysis of urea and creatinine levels revealed that administration of the extract did not result in any significant changes. Serum urea and creatinine are considered the main markers of nephrotoxicity, although serum urea is often considered a more reliable indicator of renal function than serum creatinine 34. Our results do not agree with those of Ntabaza et al. (2023), who showed a significant increase in urea with the same type of study and extract in guinea pigs. This difference may be explained by the doses or the animal model; therefore, the 200 mg/kg dose is better suited for daily use. In cases of hyperglycemia, the pancreas releases insulin, which stimulates the liver (and muscles) to store glucose as glycogen. In cases of hypoglycemia, the pancreas releases glucagon to stimulate the liver to release glucose and maintain normal blood glucose levels 14. In our context, we lack data to assess pancreatic function, but we know that the liver has shown dysfunction characterized by elevated serum transaminase levels and atrophy. It is likely that the hyperglycemia is linked to impaired liver function and metabolism 40.
The elevated blood glucose level may be due to damage induced by the aqueous extract of Tikoni tea in the liver 8. These results are consistent with those of Baliga et al. (2004), who administered the Alstonia scholaris extract orally to mice and rats 3. Vitex madiensis is very rich in primary and secondary metabolite compounds, mineral elements and vitamins; this constitutes a major advantage and explains the pharmacological and toxicological effects observed with this extract 24, 25, 27, 28.
CONCLUSION: Consumption of aqueous extract of Vitex madiensis as tikoni tea is encouraged in immunocompromised or ill individuals, but prohibited in diabetics.
ACKNOWLEDGEMENTS: The authors would like to thank the Dean of Faculty of Csinces and Technologies and the President of Marien Ngouabi University for funding part of this work, Professor Jean Marie Mountsamboté for the botanical identification and authentication, and the traditional practitioners who contributed to the collection of plant material.
CONFLICTS OF INTEREST: There are no conflicts of interest.
REFERENCES:
- Adel G, Samaneh S, Bamdad R, Anahita AG, Seyed AM and Mahdi B: Plant toxins and acute medicinal plant poisoning in children: A systematic literature review. J Res Med Sci 2018; 23: 26.
- Adeneye AA, Ajagbonna OP, Adeleke TI and Bello SO: Preliminary toxicity and phytochemical studies of the stem bark aqueous extract of Musanga cecropioides in rats. Journal of Ethnopharmacology 2006; 105: 374–379.
- Baliga MS, Jagetia GC, Ulloor JN, Baliga MP, Venkatesh P, Reddy R, Rao KM, Baliga BS, Devi S, Raju SK, Veeresh V, Reddy TK and Bairy KL: The evaluation of the acute toxicity and long term safety of hydroalcoholic extract of Saptha parna (Alstonia scholaris) in mice and rats. Toxicology Letters 2004; 151: 317–326.
- Beaulieu P and Lambert C: Précis de pharmacologie: du fondamentale à la clinique. Les presses de l’Université de Montréal 2010; 873.
- Bossard E: La médecine traditionnelle au centre et à l’ouest de l’Angola. Ministério da ciêciae eda tecnologia. Instituto de recherche scientifique tropical. Lisboa 1996; 531.
- Bouquet A: Féticheurs et médecines traditionnelles du Congo. Mémoire O.R.S.T.O.M. n° 36, Brazzaville – Congo 1969; 34.
- Burtis CA, Ashwood ER, Bruns DE and Saunders WB: Tietz Textbook of Clinical Chemistry and Molecular Diagnostics. 4th Edition 2005; 50-61.
- Burtis CA, Ashwood ER and Tietz NW: Tietz Text Book of clinical Chemistry, Saunders Company, Philadelphia, USA 1994; 2064.
- Chang-Gue S, Seung-Hyun H, Jung-Hyo C, Jang-Woo S, Chin-Ho C, Yeon-Weol L and Chong Kwan C: Induction of hemopoiesis by saenghyuldan, a mixture of Ginseng radix, Paeoniae Radix alba, and Hominis placenta extracts. Acta Pharmacologica Sinica 2003; 24: 120–126.
- Dalmas V, Brémond-Gignac D, Douard R, Dupont S, Latrémouille C, Le Minor JM, Pirro N, Sèbe P, Vacher C and Yiou R: Anatomie générale. Edition Elsevier Masson 2009; 173.
- Damien R: Guide de traitement des déchets. Edition dunod Paris 2002; 3-25.
- Etame LG, Yinyang J, Okalla Ebongue C, Makondo BV, Ngaba GP, Mpondo E and Dibong SD: Étude de la toxicité aigüe et subaigüe de l’extrait au vin des graines de Carica papaya Linn, Journal of Applied Biosciences 2017; 120: 12077-12085.
- Ganong W: Physiologie médicale. France: Amazon France 2005; 849.
- Gaw A, Murphy MJ, Cowan RA, O’Reilly DSt J, Stewart MJ and Shepherd J: Biochimie clinique, Edition Elsevier 2004; 52-57.
- Gazda VE, Gomes-Carneiro MR, Barbi NS and Paumgartten FJR: Toxicological evaluation of an ethanolic extract from Chiococca alba Journal of Ethnopharmacology 2006; 105: 187 195.
- Haxaire C: Phytothérapie et médecine familiale chez Gbaya-Kara (république centrafricaine). Mémoire, université de paris 1979; 5.
- Hilaly JE, Israili ZH and Lyouss B: Acute and chronic toxicological studies of Ajuva Iva in experimental animals. Journal of Ethnopharmacology 2004; 91: 43–50.
- Ishii R, Yoshikawa H, Minakata NT, Komura K and Kada T: Specificity of bioantimutagens in the plant kingdom. Agric Biol Chem 1984; 48: 2587–2591.
- James T, Mukinda P and Eagles FK: Acute and sub-chronic oral toxicity profiles of the aqueous extract of Polygala fruticosa in female mice and rats. Journal of Ethnopharmacology 2010; 128(1): 236-240.
- Kasuku W, Lula F, Paulus J, Ngiefu N & Kaluila D: Contribution à l’inventaire des plantes utilisées pour le traitement du paludisme à Kinshasa (R.D.C.). Revue de médecine et pharmacopées africaines 1999; 13: 95-103.
- Kaur G, Alam M, Jabbar Z, Javed K and Athar M: Evaluation of antioxidant activity of Cassia siamea J Ethnopharmacol 2006; 108, 340-348.
- Morabandza CJ: Etude de la composition chimique et propriétés pharmacologiques des extraits du mésocarpe de Garcinia kola Heckel (Clusiaceae); Thèse de Doctorat, Faculté des Sciences et Techniques, Université Marien NGOUABI Brazzaville 2014; 131.
- Mukinda JT and Syce JA: Acute and chronic toxicity of the aqueous extract of Artemisia afra in rodents. Journal of Ethnopharmacology 2007; 112: 138–144.
- Ntabaza VN, Amuri B, Ompey JMV, Simbi L and Byang K: Acute and sub-acute oral toxicities of aqueous leaf extract of Vitex madiensis in a guinea pig model South African Journal of Botany 2023; 153: 157-162.
- N’goka V, Nsonde Ntandou GF, N’goka V, Boumba SL and Abena AA: Chemical screening, acute toxicity and analgesic effect of the aqueous extracts of Vitex madiensis (Lamiaceae Viticoïdeae) and Phytolacca dodecandra L’hérit. (Phytolaccaceae) Leaves. International Journal of Sciences 2018; 7(1): 2-9.
- Ngalamulume Tshiamuene, Paulus JS, Kabeya M, Nlandu SL & Kizika K: Plantes médicinales à usage domestique cultivées dans deux quartiers de Kinshasa, in: Revue de médecine et pharmacopées africaine 1995; 2: 9-14.
- Nsonde Ntandou GF, Bassoueka DJ, Banzouzi JT, Etou Ossibi AW, Elion Itou RDG, Makambila MC, Ramos S, Benoit-Vical F, Abena AA and Ouamba JM: Assessment of Cassia siamea stem bark extracts toxicity and stability in time of aqueous extract analgesic activity. African Journal of Pharmacy and Pharmacology 2015; 9(41): 988-994.
- Nsonde Ntandou GF, Dianzitoukoulou Matsima LD, Ingabire J, Makemba Nkounkou GS, Nkundineza JC and ABENA AA: Anti-Inflammatory and central analgesic effect of the aqueous extract of the leaves of Vitex madiensis Oliv (Lamiaceae-Viticoïdeae) Scholars International Journal of Traditional and Complementary Medicine 2020; 3(1): 12-17.
- Etude de la toxicité chronique. Ligne directrice de l’OCDE pour les essais des produits chimiques 2009; 4: 16.
- Olson H, Betton G, Robinson D, Thomas K, Monro A, Kolaja G, Lilly P, Sanders J, Sipes G, Bracken W, Dorato M, Deun KV, Smith P, Berger B and Heller A: Concordance of toxicity of pharmaceuticals in humans and in animals. Regulatory Toxicology and Pharmacology 2000; 32: 56–67.
- Research guidelenes for evaluating the safety and efficacy of herbal medecine. Regional office for the western pacifique. Philippines 2002; 216.
- Ondo JP, Lekana-Douki JB, Bongui JB, Zang Edou ES, Zatra R, Toure-Ndouo FS, Elomri A, Lebibi J and Seguin E: In-vitro antiplasmodial activity and cytotoxicity of extracts and fractions of Vitex madiensis, medicinal plant of Gabon. Tropical Medecine and International Health 2012; 17(3): 316-321.
- Otmani A and et Yahiaoui S: Etude de la Toxicité aigüe et Subaiguë des Alcaloïdes de Fumaria officinalis sur des souris albinos Wistar. Mémoire de Master en Pharmacologie Moléculaire. Université Abderrahmane Mira de Béjaia 2016; 94.
- Palani S, Raja R, Kumar P and Jayakumar S: Therapeutic efficacy of Pimpinella tirupatiensis (Apiaceae) on acetaminophen induced nephrotoxicity and oxidative stress in male albino rats. Inter J Pharm Tech 2009; 2: 708-719.
- Philomena G: Concerns regarding the safety and toxicity of medicinal plants - An overview. J Appl Pharmaceut Sci 2011; 01(6): 40-44.
- Raina P, Chandrasekaran CV, Deepak M, Agarwal A and Ruchika KG: Evaluation of subacute toxicity of methanolic/aqueous preparation of aerial parts of sanctum in Wistar rats: Clinical, haematological, biochemical and histopathological studies. J Ethnopharmacol 2015; 175: 509-517.
- Rebecca MA, Ishii-Iwamoto EL, Grespan R, Cuman RKN, Caparroz-Assef SM, Mello JCP and Bersani-Amado CA: Toxicological studies on stryphnodendron adstringens. Journal of Ethnopharmacology 2002; 83: 101–104.
- Rhiouani H, El-Hilaly J, Israili ZH and Badiaa Lyoussi B: Acute and sub-chronic toxicity of an aqueous extract of the leaves of Herniaria glabra in rodents. Journal of Ethnopharmacology 2008; 118: 378–386.
- Sacher RA and McPherson RA: Widmann’s Clinical Interpretation of Laboratory Test. Pennsylvania USA 1991; 416–443.
- Silbernagl S: et Despopoulos A. Nutrition et digestion. Dans Atlas de poche de physiologie. Médecine Sciences Publications, 4ème Edition 2008; 212-216.
- Sillanaukee P and Olsson U: Improved diagnostic classification of alcohol abusers by combining carbohydrate-deficient transferrin and g-glutamyl transferase. Clin Chem 2001; 47: 681-685.
- Slichter SJ: Relationship between platelet count and bleeding risk in thrombocytopenic patients. Transfusion Medicine Reviews 2004; 18: 153–167.
- Tulsawani R: Ninety day repeated gavage administration of Hipphophae rhamnoides extract in rats. Food and Chemical Toxicology 2010; 48: 2483–2489.
- Udut EV, Zhdanov VV, Gur’iantseva LA, Minakova MI and Dygai AM: Mechanisms of the erythropoiesis-stimulating effect of skullcap (Scutellaria baicalensis) extract. Eksperimental’naia i Klinicheskaia Farmakologiia 2005; 68: 43–45.
- Witthawaskul P, Panthong A, Kanjanapothi D and Taesothikul T: Lertprasertsuke NAcute and subacute toxicities of the saponin mixture isolated from Schefflera leucantha Journal of Ethnopharmacology 2003; 89: 115–121.
How to cite this article:
Ntandou GFN, Yoly EBL, Eric M and Nkounkou GSM: Subchronic toxicity of tikoni tea from Vitex madiensis oliv. leaves (lamiaceae) aqueous extract in the Wistar rat. Int J Pharmacognosy 2026; 13(2): 99-11. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(2).99-11.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
99-111
1264 KB
112
English
IJP
G. F. Nsonde Ntandou *, E. B. Lekounda Yoly, Motondo Eric and G. S. Makemba Nkounkou
Centre d’Etude et de Recherche Médecins d’Afrique (CERMA) 3, B.P. 45, Brazzaville, Congo.
nsonde_ntandou@yahoo.fr
31 January 2026
22 February 2026
26 February 2026
10.13040/IJPSR.0975-8232.IJP.13(2).99-11
28 February 2026