STUDY OF PLANTS WITH D-CHIRO-INOSITOL AND ITS DERIVATIVES ON DIABETES
HTML Full TextSTUDY OF PLANTS WITH D-CHIRO-INOSITOL AND ITS DERIVATIVES ON DIABETES
Shahdokht Rastegar 1, 2, Shirin Soltani 3, Azade Roohipoor 4 and Esmaeel Ebrahimi * 1, 2
Department of Biochemistry 1, School of Medicine, Research Committee 2, Department of Pharmacology 3, School of Pharmacy, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
Department of Biochemistry 4, Taft University of Payame-Noor, Yazd, Iran.
ABSTRACT: Diabetes is an endocrine disorder, and it is due to a deficiency in secretion or action of insulin. The increased prevalence of diabetes in recent decades has made it to severe risk for public health. Human always has great consideration to plants for treatment of illnesses, especially diabetes and it is probably due to common ingredients with mammalian cells such as inositol, especially isomer of D-chiro-inositol, which plays a vital role in the signaling pathway of insulin, and its lack in the structure of inositol phosphoglycan leads to insulin resistance. Therefore, the possession of an inexpensive and available source of this ingredient is essential for patients suffering from diabetes. In the current study, we reviewed the effects of D-chiro-inositol and its derivatives and herbs with them on diabetes.
| Keywords: |
Diabetes, Insulin, D-chiro-inositol, Inositol phosphoglycan
INTRODUCTION: Diabetes mellitus is a multi-factorial endocrine disorder with a significant prevalence in worldwide 1, 2. Impairment in insulin secretion and insensitivity of peripheral tissues (skeletal muscle and adipocytes) to insulin are main causes of type 1 and 2 diabetes 3, 4. Study on the prevalence of diabetes mellitus in 219 countries was showed that 382 million people have diabetes; in other words, it has been predicted that the number of people with diabetes about 7% will increase by 2030. 5 Today, diabetes as the fourth main cause of death worldwide is threatening of human health, and according to statistics, every year 6.8% of the world's population die due to complications of diabetes 6.
The increase of blood glucose during diabetes leads to complications such as neuropathy, cardio-myopathy, retinopathy, and nephropathy; however, if this situation gets adjusted, macrovascular and microvascular complications would decrease 7. Improvement of hyperglycemias’ normalizes oxidant-antioxidant imbalance and by reduction of free radicals level modifies any disruptions in diabetes; therefore, blood glucose control is a fundamental strategy for management of diabetes 8. World health organization encourages researches to discover drugs with good efficacy and low side effects to control and treatment of diabetes; and to achieve this goal, plants can be useful due to their rich sources from compounds with biological activity 8.
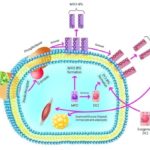
From nine isomer of inositol, D-chiro-inositol (D-CI) has the most important role in insulin signaling pathway Fig. 1; 9 and given that importance of secondary messengers in insulin resistance, focusing on the metabolism of D-chiro-inositol in diabetes is very pivotal 10.
FIG. 1: ROLE OF D-CHIRO-INOSITOL IN INSULIN SIGNALING PATHWAY
One of the events that occur during digestion of glucose is an increase of activation of inositol phosphoglycan with D-chiro-inositol, but this is an interesting point that there is the possibility of their disorder during diabetes so that their levels severely decrease in patients with non-insulin dependent diabetes mellitus while the level of myoinositol increase 11-13. This is due to the reduction of activity of an enzyme that transforms myoinositol to chiro-inositol in very important areas of development of diabetes such as liver, skeletal muscle and adipocytes 14.
Another reason for the abolishment of D-chiro-inositol effect in diabetes is a reduction of hydrolysis of glycophosphatidylinositol to inositol phosphoglycan after induction of diabetes by streptozotocin howbeit; the increase of the urinary level of D-chiro-inositol in this situation indicates that D-chiro-inositol perturbation is along with defect in glycophosphatidylinositol-dependent insulin signaling 15-17.
Therefore, reduction of D-chiro-inositol embedment in the mediators of insulin signaling pathway due to either transformation increase to myo-inositol by epimerase or hydrolysis decrease of glycophosphatidylinositol has a huge impact on the development of diabetes, and its administration can be a reasonable strategy for control and treatment of diabetes 2, 18. Also, Dchiro-inositol is a presence as methyl form (3-O-methyl-D-chiro-inositol or D-pinitol) in plants that have promising effects on the reducing of diabetes-related disorders 9, 19. Our previous studies have confirmed that plants are a suitable source for remedy of diabetes and management of its complications. We investigated insulin resistance 20, dyslipidemia 21, weight of diabetic rats 22, role of pancreatic duct cell in beta cell neogenesis 23, study of association between beverage consumption pattern and lipid profile 24, cardiac and pancreatic biomarkers 25, induction of insulin secretion 1, hematological factors 26, evaluation of sex cells (gametes) and testicular structure 27 and mechanisms of plant ingredients in the treatment of diabetes mellitus 28 in previous study. Here, we reviewed the effects of D-chiro-inositol and its derivatives and plants with them on diabetes.
Review Method: In the recent study, we reviewed the effects of D-chiro inositol and its derivatives on diabetes in conducted studies on diabetic animal models and patients with type 2 diabetes. We also reviewed plants contain these compounds and their effects on diabetes. For conducting this study, we obtained related articles using keywords such as D-chiro-inositol, D-pinitol, and D-chiro-inositol derivatives, diabetes and herbal medicine since 1980 from to now based on databases such as Wiley, Scopus, Science Direct, Springer and PubMed. Then, the articles were categorized and reviewed.
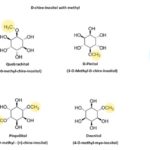
Effects of Administration of D-chiro-inositol and its Derivatives on Diabetes: In Table 1, the beneficial effects of D-chiro-inositol and its derivatives on diabetes were summarized. According to studies, free D-chiro-inositol or derivatives with methyl such as L-quebrachitol, D-pinitol, pinpollitol and D-ononitol and derivatives containing galactosyl such as ciceritol can be find in plants Fig. 2. 29-32 However, many studies have been done on the effects of free form (D-chiro-inositol) or D-3-O-methyl form (D-pinitol). One of the considered suggestions for improvement of diabetes is the use of nutritional supplements that contain D-chiro-inositol 33.
Insulin is effective on the activity of pyruvate dehydrogenase phosphatase through mediators, and this regulatory role of insulin is abolished during insulin resistance. Given that the vital role of D-chiroinositol in this regulatory pathway, its administration can reduces the effects of insulin resistance, so that treatment with D-chiro-inositol improves complications such as hypertension, obesity, cardiovascular disorders and lactic acidosis 34. However, the use of myo-inositol does not have dramatic effects on the level of blood glucose because two week-treatment with myo-inositol could not reduce the increased blood glucose level followed by streptozotocin injection 35.
TABLE 1: EFFECTS OF ADMINISTRATION OF D-CHIRO-INOSITOL AND ITS DERIVATIVES ON DIABETES
| Compound | Subject/animal model | Result(s) | Reference |
| D-chiro-inositol | STZ-diabetic rats | Blood glucose reduction by the administration with insulin after 60 min | 36 |
| STZ-diabetic rats | 47% blood glucose reduction by the administration with manganese chloride after 120 min | 2 | |
| STZ-diabetic rats | Improvement of PPA and CV in sciatic nerves, induction of electrical stimulation in corpus cavernosum and vas deferens | 37 | |
| D-pinitol | STZ-diabetic rats | Normalization of blood glucose and lipid
profile in serum, liver, kidney, heart and brain |
8 |
| STZ-diabetic rats | Activation increase of SOD ،GPx ،GST, improvement of non-defense antioxidant, Reduction of lipid peroxidation and lipid hydro per oxidation | 40 | |
| STZ-diabetic rats | Amelioration of pro-inflammatory factors, insulin serum, and antioxidant-oxidant
a condition in the liver, reduction of lipid peroxidation and lipid hydroperoxidation, protection of the liver structure |
41 | |
| Roman-Cowley model of pressure-natriuresis (P-N) in STZ-diabetic rats | Improvement of parameters related to
kidney function such as RVR, FENa, FF, RBF |
42 | |
| Type 2 diabetic patients | Reduction of blood glucose (fasting and
post prandial) and HbA1c, lack of significant change on adiponectin, leptin, free fatty acid, CRP, lipid profile and fasting c-peptide |
43 | |
| Obese individuals with insulin resistant | Lack of toxic effect by evaluation of whole-body glucose, palmitate and glycerol kinetics under conditions of basal and hyperinsulinemic-euglycemic clamp | 9 | |
| STZ/HDF-diabetic rats | Expression increase of PI3Kp85, PI3Kp110, and kinase B/Akt, glycogen storage amelioration by expression inhibition of
GSK-3β and up-regulation of glycogen synthesis |
31 | |
| STZ-diabetic rats
Obese-diabetic rats L6 rat muscle cells |
Plasma glucose reduction during oral and intraperitoneal administration in STZ-diabetic rats and not obese-diabetic rats, improvement of glucose transport in L6 rat muscle cells | 44 | |
| L-quebrachitol | DB/db Mice | By increase efficacy of pancreas function lead to normalization of serum glucose and insulin | 45 |
| Alloxan-diabetic rats | Improvement of blood glucose | 46 |
PPA: peak-to-peak amplitude, CV: conduction velocity, SOD: superoxide dismutase, GPx: glutathione peroxidase, GST: glutathione s-transferase, FENa: fractional excretion of sodium, FF: filtration fraction, RBF: renal blood flow, RVR: renal vascular resistance, CRP: C-reactive protein, PI3K: phosphatidylinositol-3-kinase, GSK-3β: glycogen synthesis kinase-3β.
FIG. 2: D-CHIRO-INOSITOL AND ITS DERIVATIVES IN PLANTS
In one study, it was showed that concurrent administration of insulin and chiro-inositol glycan could significantly reduce blood glucose during 60 min after administration in induced diabetic rats with streptozotocin while administration of insulin without chiro-inositol glycan led to a significant reduction in blood glucose during 120 min 36.
Moreover, it was well confirmed that D-chiro-inositol at a dose of 15 mg/kg and not 5 mg/kg reduces blood glucose in streptozotocin-diabetic rats about 21% during 120 min after administration and this decreasing effect was up to 47% during treatment with D-chiro-inositol (15 mg/kg) along with manganese chloride (8.3 μg/min) in 120 min after administration. It was also determined that 3-O-methyl-D-chiro-inositol in doses of 5 and 15 mg/kg leads to blood glucose reduction about 6% and 22% respectively followed by 120 min administration; and concurrent treatment of 3-O-methyl-D-chiro-inositol (15mg/kg) with manganese chloride (8.3 μg/min) reduces hyperglycemia about 49% at the same time after administration 2.
There is one study in conjunction with the good effects of D-chiro-inositol on neuropathy caused by diabetes. In this study, it was determined that administration of streptozotocin leads to many disturbances in the function of sciatic nerve including, reduction of peak-to-peak amplitude (PPA) and conduction velocity (CV) and also increase of chronaxy. After treatment with 20 mg/kg/12h of D-chiro-inositol, these problems recovered to normal level. Also, D-chiro-inositol improved electrical stimulation of corpus cavernosum and vas deferens and had a protective effect on injuries related to sciatic nerves 37.
D-pinitol is one of the rich sources of inositol in the herbs, and it has been determined that mimics the effects of insulin 38, 39. D-pinitol can consider as an anti-hyperlipidemic compound because it has been examined that treatment of diabetic rats with D-pinitol leads to the reduction of glucose, total cholesterol, triglyceride, free fatty acids, and phospholipids in serum, liver, kidney, heart and brain. Additionally, reduction in low-density lipoprotein (LDL) and very low -density lipoprotein (VLDL) and an increase of high-density lipoprotein (HDL) level by D-pinitol administration has been seen 8.
In a study, the effects of treatment with 50 mg/kg of D-pinitol on antioxidant status in plasma and pancreas of diabetic rats during one month were examined. According to this study, this dose of D-pinitol resulted in the balance of antioxidant status through its effect on antioxidant enzymes (superoxide dismutase, glutathione peroxidase, and glutathione s-transferase) and non-enzymatic antioxidant compounds (Vitamin E, vitamin C, ceruloplasmin and reduced glutathione). Therefore, to keep antioxidant defense by D-pinitol reduces induced damage on pancreas because lipid peroxidation and hydroperoxidation in the pancreas were significantly decreased after treatment with D-pinitol 40.
In a study, Sivakumar et al., 2010 have shown that hypoglycemic effect of D-pinitol (50 mg/kg b.w.) is parallel to the improvement of inflammatory markers (TNF-α, IL-1β, IL-6, NF-κB p65 unit and nitric oxide) and an increase of insulin level in diabetic rat’s serum. Also, there was a reasonable relationship between oxidant-antioxidant balance (by measurement of enzymatic and non-enzymatic antioxidant defense biomarkers) and reduction of lipid peroxide and hydroperoxide level in liver. Moreover, these results were confirmed after liver histopathologic evaluation 41.
A research group was determined that impairment of kidney function under diabetes condition and treatment with D-pinitol can prevent it. In this study, in order to induction of renal dysfunction, the Roman-Cowley model of pressure-natriuresis (PN) was used and measurement of factors such as fractional excretion of sodium (FENA), filtration fraction (FF), renal blood flow (RBF), renal vascular resistance (RVR) and glomerular filtration rate (GFR) confirmed improvement of kidney function after two month examination in diabetic rats 42. The observation of useful effects in improvement of diabetes during a study on treatment with D-pinitol in animal models encourages researchers to examine its effects on diabetic patients.
On this basis, Kim et al., 2007 conducted a study on the effects of treatment with 20 mg/kg per day of D-pinitol on diabetic patients with poor response to sulfonylurea, metformin and insulin during twelve weeks. According to this study, D-pinitol caused a significant reduction in blood glucose (fasting and after a meal) and HBA1c. Albeit, any significant changes have not seen in adiponectin, leptin, free fatty acids, and c-reactive protein and lipid profile (total cholesterol, triglyceride, and HDL- and LDL-cholesterol) and fasting c-peptide in serum 43. Moreover, determination of whole-body glucose, palmitate, and glycerol kinetics under both basic and hyperinsulinemic-euglycemic clamp conditions was confirmed sensitivity of glucose and lipid metabolism to insulin after daily treatment with 20 mg/kg D-pinitol within 4 weeks in obese patients with type 2 diabetes 9.
Although, D-pinitol is anti-hyperglycemic compound, but has been recently understood its molecular effect mechanism so that in the conducted study by Gao et al., 2015 on streptozotocin-induced type 2 diabetes rats with high-fat diet showed that D-pinitol has effects similar to insulin in liver. It could actives signaling pathway of PI3K/AKT by an increase of expression of phosphatidylinositol-3-kinase (PI3K) P85, PI3KP110 and kinase B/AKT (at ser473). It was also increased glycogen content in the liver by inhibition of expression of glycogen synthesis kinase-3β (GSK-3β) protein and up-regulation of glycogen synthesis 31.
Also, according to a study conducted by Bates et al., 2000 oral and intraperitoneal administration of D-pinitol (100 mg/kg) results in plasma glucose reduction in streptozotocin-diabetic mice while there was not hypoglycemia in obese-diabetic mice. Moreover, this study indicated that D-pinitol improves glucose transport in L6 rat muscle cells 44. In association with other derivatives of D-chiro-inositol, it has been reported that ten-week treatment with sea buckthorn juice enriched with L-quebrachitol (2-0-methyl-chiro-inositol) improves resistance to induced diabetes in db/db mice.
Indeed, L-quebrachitol had an important role in the normalization of glucose and insulin level through improvement of pancreas performance 45. It has also reported by Musalmah et al., 2001 that treatment with L-quebrachitol and L-chiro-inositol recover the level of blood glucose in diabetic rats 46.
Effects of Plants with D-chiro-inositol and its Derivatives on Diabetes: In Table 2, the effects of D-chiro-inositol and its derivatives on diabetes was showed; these plants including:
TABLE 2: EFFECTS OF PLANTS WITH D-CHIRO INOSITOL AND ITS DERIVATIVES ON DIABETES
| Plant | Subject/animal model/cell line/method | Result(s) | Reference |
|
Soybean (Glycine max) |
Type 2 diabetic patients | Reduction of glucose, fructose, HbA1c, insulin, HOMA-IR and lipid profile in blood | 39 |
| Korean type 2 diabetic
patients |
Inhibition of postprandial
hyperglycemia, lack of good effect on blood insulin |
19 | |
| Buckwheat
(Fagopyrum esculentum) |
STZ-diabetic rats | 12 and 19% reduction of hyperglycemia after 90 and 120 min respectively | 16 |
|
Tartary buckwheat (Fagopyrum tataricum) |
KK-Ay mice | Amelioration of serum level of glucose, total cholesterol, TG, C-peptide, glucagon and BUN, reduction of immunologic
reaction in pancreas |
52 |
|
Carob tree (Ceratonia siliqua) |
STZ-diabetic rats | Normalization of serum level
of glucose and lipid profile |
53 |
| STZ-diabetic rats | The protective effect on kidney
Function |
54 | |
| Column chromatography with ion exchange | Confirmation of D-pinitol
Isolation |
55 | |
|
Dragon fruit (Hylocereus polyrhizus) |
STZ-diabetic rats | Improvement of cardiac function and oxidative condition | 56 |
| Fructose-diabetic rats | Amelioration of insulin resistant, hypertriglyceridemia, and atherosclerotic changes | 57 | |
| X-ray crystallography,
HPLC, LC-MS/MS, and NMR |
Confirmation of myo-inositol in
plant |
58 | |
|
Fig leaf gourd (Cucurbita ficifolia) |
STZ-diabetic rats | Improvement of polydipsia and
hyperphagia, hyperglycemia and plasma lipid peroxidation, positive effect on glutathione cycle in liver, pancreas, and kidney |
59 |
| STZ-diabetic rats | Normalization of the level of
hyperglycemia, GTT, total hemoglobin, insulin and liver glycogen |
60 | |
| STZ-diabetic rats | Anti-inflammatory and
antioxidant effects |
61 | |
| 3T3-L1 cell line | Anti-inflammatory and antioxidant effects | 62 | |
| STZ-diabetic rats | Anti-diabetic property except
effect on ATP-sensitive potassium channel |
63 | |
|
Mung Bean (vigna radiata ) |
KK-Ay mice | Level improvement of glucose,
C-peptide, glucagon, total cholesterol, TG and BUN in plasma, reduction of immunologic reaction in the pancreas |
65 |
HOMA-IR: homeostatic model assessment for insulin resistant, STZ: streptozotocin, TG: triglyceride, BUN: blood urea nitrogen, GTT: glucose tolerance test.
Soybean (Glycine max): It has been determined that D-chiro-inositol and its methylated form (D- pinitol) are great part of cyclitols of soybean 47. In a study, it was found that D-pinitol is one of the main ingredients in soybean. According to this study, if soybean leaf (its ethanol extract) place in an aqueous fraction and fully remove lipids through ion exchange resins, ultimately obtain a crystalline product with 85.5% D-pinitol 48. Treatment of thirty patients with type 2 diabetes by isolated D-pinitol from soybean (600 mg twice a day for 13 weeks) resulted in the reduction of blood glucose, fructose, HbA1c, insulin and HOMA-IR (an assessment index of insulin resistance). Also, this study confirms good effects of D-pinitol on lipid profile 39. Remarkable effects of D-pinitol obtained from soybean persuade researchers to use it as a dietary supplement. In one study, fifteen Korean diabetic patients were treated with cooked white rice with 50 g of carbohydrates (with or without soybean D-pinitol). This study was showed that D-pinitol prevents the increase of blood glucose 90 to 120 min after a meal; however, an acceptable effect on insulin level was not observed 19.
Buckwheat (Fagopyrum esculentum): Studies show that Fagopyrum esculentum (buckwheat) is rich in D-chiro-inositol derivatives. Based on a study conducted by Horbowicz et al., 1998 O-α-D-galactopyranosyl- (1→2)- D-chiro-inositol (fagopyritol B1) is the most important carbohydrate in buckwheat seeds 49. It has been reported that seeds of buckwheat have 40% of fagopyritols (mano, dio tri galactosyl derivatives related to D-chiro inositol) in each 6.4 g of total soluble carbohydrates per 100 g of dry weight 50. Also, the extract with D-chiro-inositol can be isolated from buckwheat by thin layer chromatography, sodium periodate and blue benzidine 51. In one study, it has been showed that administration of concentrated buckwheat (including 10 and 20 mg of D-chiro-inositol per kg of body weight) has a significant effect on reduction of blood glucose level in streptozotocin-induced diabetic rats so that a reduction of 12-19% in serum glucose observed after 90 and 120 min administration. Therefore, its administrations as a dietary supplement can seem reasonable for diabetic’s patients 16.
Tartary Buckwheat (Fagopyrum tataricum): Fagopyrum tataricum (tartary buckwheat) is another type of buckwheat. The examination of tartary buckwheat extract rich of D-chiro-inositol on KK-Ay mice was showed that treatment with 20 g/kg its extract had no toxic effects. Additionally, this extract resulted in the reduction of blood glucose, total cholesterol, triglycerides, C-peptide, glucagon, and blood urea nitrogen and also improvement of the induced immune response in the pancreas at the end of period study 52.
Carob Tree (Ceratonia siliqua): Using 600 mg/kg of ethanol extract prepared from carob seeds was obtained promising effects on improvement of glucose and lipid profile in diabetic rats 53. Also, ethanol extracts of carob seeds (600 mg/kg) reduce induced-damages to kidney caused by streptozotocin injection after 16 days of treatment because it could perfectly balance the level of renal biomarkers (creatinine, uric acid, blood urea nitrogen, Na+, K+, and Ca+2) in serum 54. It has also been reported that through chromatography along with ion exchange resin, D-pinitol was separated from this plant 55. According to these studies, it considered that D-pinitol is effective in anti-diabetic features of carob.
Dragon Fruit (Hylocereus polyrhizus): In one study, it was well found that five-week treatment of aqueous extract obtained from fruit pulp of Hylocereus polyrhizus (250 and 500 mg/kg) is very affective in blood glucose normalization. This study was also showed that it has a pivotal effect on kidney function because of improvement of parameters such as pulse wave velocity (leading to a decrease in aortic stiffness), systolic blood pressure, and pulse pressure at the end of the study. Also decrease in plasma level of malondialdehyde, an increase of enzyme activity of superoxide dismutase and total antioxidant capacity reflected a recuperation of oxidative condition in diabetic rats 56. The anti-diabetic effects of Hylocereus polyrhizus can be attributed to the presence of antioxidants and fiber because six-week treatment with its fresh fruit juice rather than use of processed fruits under heat of 95 °C and 105 °C (containing low level of antioxidants and fiber) significantly improved insulin resistance, hypertriglyceridemia and atherosclerotic changes in rats received fructose 57. On the other hand, by use of X-ray crystallography, HPLC, LC-MS/MS and NMR the presence of myo-inositol crystals in H. polyrhizus has been reported 58.
Fig Leaf Gourd (Cucurbita ficifolia): Administration of 200 mg/kg of Cucurbita ficifolia as an aqueous extract for one month reduces symptoms such as polydipsia, and hyperphagia in streptozotocin-induced diabetic rats. It also significantly reduces blood glucose and lipid peroxidation in plasma. The aqueous extract of Cucurbita ficifolia has a positive effect on glutathione cycle in liver, pancreas, and kidney so that levels of reduced and oxidized glutathione, the activity of glutathione peroxidase and glutathione reductase were completely normalized followed by treatment 59. Also, administration of Cucurbita ficifolia (containing 10-20 mg/kg D-chiro-inositol) in terms of normalization of factors such as blood glucose, glucose tolerance test, liver glycogen, total hemoglobin, and plasma insulin is similar to the effects of administration of 20 mg/kg D-chiro-inositol. Thus, this herb as a rich source of D-chiro-inositol can use by diabetic patients 60.
According to a study performed by Ramos et al., 2012 the anti-diabetic characteristic of Cucurbita ficifolia is related to the existence of D-ciro-inositol. AP fraction (fraction containing D-chiro-inosito) obtained from the fruit of Cucurbita ficifolia has 3.31 mg/g of D-chiro-inositol. In this study treatment design was contain first preventive treatment for two weeks and then treatment continued after induction of diabetes with streptozotocin for the 33-day course. In final, it was confirmed that AP fraction has an anti-inflammatory effect and leads to a reduction of TNF-α and also increase of IFN-γ and IL-6 in serum but it increases the anti-inflammatory cytokine level of IL-10. AP fraction was also effective in the increase of glutathione level and reduction of malondialdehyde in liver 61.
Comparing the effects of aqueous extract of Cucurbita ficifolia and synthetic D-chiro-inositol on the oxidative state and level of pre-inflammatory factors in adipocytes (3T3-L1) was showed that both lead to a reduction of hydrogen peroxide and GSH/GSSG ratio and activity increase of glutathione peroxidase. The synthetic D-chiroinositol reduced mRNA level and amount secretion of TNFα, IL-6, and resistin while reduction effects of Cucurbita ficifolia extract was only on resistin level and even increased the level of IL-6 62.
Although, aqueous extract of Cucurbita ficifolia along with high degree of D-chiro-inositol significantly reduced blood sugar and triglyceride level in streptozotocin -induced diabetic rats during a one-month study, but it not inhibited diazoxide-induced relaxation of rat aortic ring precontracted with phenylephrine. Indeed, the glucose-lowering effect of its extract was not through its effect on ATP-dependent potassium channels 63.
Mung Bean (Vigna radiate): Examination of D-chiro-Inositol content in 110 species of mung bean was showed that it is valuable in terms of containing D-chiro-Inositol and can use as a food grain in the diet of individuals with diabetes 64. Besides, it considered as an anti-diabetic herb. The administration 2 and 3 g/kg mung bean sprouts extract, and mung bean seed coat extract have an entirely good effect on the normalization of factors such as blood glucose, plasma C-peptide, glucagon, total cholesterol, triglycerides, and blood urea nitrogen, respectively during five-week treatment in KK-Ay mice and it also balances the immune response in their pancreas 65.
Prospects: It has been suggested that imperfection in D-chiro-inositol metabolism has a vital role in the development of insulin resistance forasmuch as the presence of D-chiro inositol is essential in the building of inositol phosphoglycans in insulin signaling pathway 9, 16. Here, we examined the effects of D-chiro-inositol and its derivatives on diabetes. We also categorized the plants, which contained such compounds. We mention that to possess an available and inexpensive source of D-chiro-inositol is necessary for patients with diabetes and herbs can help us to achieve this goal.
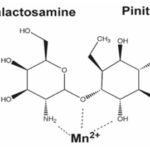
FIG. 3: STRUCTURE OF GALACTOSAMINE-CHIRO-INOSITOL PSEUDO-DISACCHARIDE Mn2+ CHELATE
Thus, world health organization (WHO) persuade researchers in the field of diabetes to more focus on plants and their compounds for remedy of diabetes because by use of plants can obtain impressive treatment with fewer side effects 8. One way to obtain D-chiro-inositol is the use of aqueous solution of kasugamycin fungal by ion exchange resins 66. Additionally, beef liver is another non-plant source of D-chiro-inositol and a compound called galactosamine chiro-inositol pseudo-disaccharide Mn+2 chelate (INS-2, 1) was isolated from it which has like insulin effect and leads to hypoglycemia in an animal model of diabetes Fig. 3. 67
CONCLUSION: Based on our review, D-chiro-inositol is the presence in plants spatially in Fabaceae family. Also, in Cucurbitaceae, D-chiro-inositol was only found in Cucurbita ficifolia; thus, examination of content D-chiro-inositol of both families is valuable owing to their most are comestible.
Finally, we suggest that investigate the effects of herbs such as Lens culinaris (lentil), Cicer arietinum (chickpea), Phaseolus vulgaris (pinto bean), Vicia sativa (vetch) and Vigna unguiculata (black-eyed pea or cowpea) on diabetes. Our suggestion is a comparison between effects of other derivatives of D-chiro inositol such as ciceritol (pinitol digalactoside), D-ononitol (4-O methyl-myo-inositol), pinpollitol(di-O-methyl – (+) - chiro-inositol) to D-chiro-inositol on diabetes.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: The authors declare that there is not any conflict of interest in this study.
REFERENCES:
- Ebrahimi E, Bahramzadeh S, Hashemitabar M, Mohammadzadeh G, Shirali S and Jodat J: Effect of hydroalcoholic leaves extract of Citrullus colocynthis on induction of insulin secretion from isolated rat islets of Langerhans. APJTD 2016; 6(8): 638-41.
- Fonteles M, Almeida M and Larner J: Antihyperglycemic effects of 3-O-methyl-D-chiro-inositol and D-chiro- inositol associated with manganese in streptozotocin diabetic rats. Horm Metab Res 2000; 32(6): 129-32.
- Gardner DGS and Greenspan D: Greenspan's basic & clinical endocrinology. McGraw-Hill, New York, Edition 8th, 2007: 1010.
- Eizirik DL, Colli ML and Ortis F: The role of inflammation in insulitis and β-cell loss in type 1 diabetes. Nat Rev Endocrinol 2009; 5(4): 219-26.
- Guariguata L, Whiting D, Hambleton I, Beagley J, Linnenkamp U and Shaw J: Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 2014; 103(2): 137-49.
- Rawal LB, Tapp RJ, Williams ED, Chan C, Yasin S and Oldenburg B: Prevention of type 2 diabetes and its complications in developing countries: a review. Int J Behav Med 2012; 19(2): 121-33.
- Fazel Nabavi S, Thiagarajan R, Rastrelli L, Daglia M, Sobarzo-Sanchez E and Alinezhad H: Curcumin: a natural product for diabetes and its complications. Curr Top Med Chem 2015; 15(23): 2445-55.
- Geethan P and Prince P: Anti-hyperlipidemic effect of D‐pinitol on streptozotocin‐induced diabetic Wistar rats. J Biochem Mol Toxicol 2008; 22(4): 220-4.
- Davis A, Christiansen M, Horowitz JF, Klein S, Hellerstein MK and Ostlund RE: Effect of pinitol treatment on insulin action in subjects with insulin resistance. Diabetes Care 2000; 23(7): 1000-5.
- Larner J, Brautigan DL and Thorner MO: D-Chiro-lnositol glycans in insulin signaling and insulin resistance. Mol Med 2010; 16(11-12): 543.
- Shashkin P, Shashkina E, Fernqvist-Forbes E, Zhou YP, Grill V and Katz A: Insulin mediators in man: effects of glucose ingestion and insulin resistance. Diabetologia 1997; 40(5): 557-63.
- Kennington AS, Hill CR, Craig J, Bogardus C, Raz I and Ortmeyer HK: Low urinary chiro-inositol excretion in non-insulin-dependent diabetes mellitus. N Engl J Med 1990; 323(6): 373-8.
- Asplin I, Galasko G and Larner J: Chiro-inositol deficiency and insulin resistance: a comparison of the chiro-inositol-and the myo-inositol-containing insulin mediators isolated from urine, hemodialysate, and muscle of control and type II diabetic subjects. Proc Natl Acad Sci 1993; 90(13): 5924-8.
- Sun TH, Heimark DB, Nguygen T, Nadler JL and Larner J: Both myo-inositol to chiro-inositol epimerase activities and chiro-inositol to myo-inositol ratios are decreased in tissues of GK type 2 diabetic rats compared to Wistar controls. Biochem Biophys Res Commun 2002; 293(3): 1092-8.
- Kawa JM, Przybylski R and Taylor CG: Urinary chiro-inositol and myo-inositol excretion is elevated in the diabetic db/db mouse and streptozotocin diabetic rat. Exp Biol Med 2003; 228(8): 907-14.
- Kawa JM, Taylor CG and Przybylski R: Buckwheat concentrate reduces serum glucose in streptozotocin-diabetic rats. J Agric Food Chem 2003; 51(25): 7287-91.
- Sanchez-Arias J, Sanchez-Gutierrez J, Guadano A, Alvarez J, Samper B and Mato J: Impairment of glycosyl-phosphatidylinositol-dependent insulin signaling system in isolated rat hepatocytes by streptozotocin-induced diabetes. Endocrinology 1992; 131(4): 1727-33.
- Ortmeyer HK, Huang LC, Zhang L, Hansen BC and Larner J: Chiroinositol deficiency and insulin resistance. II. Acute effects of D-chiroinositol administration in streptozotocin-diabetic rats, normal rats given a glucose load, and spontaneously insulin-resistant rhesus monkeys. Endocrinology 1993; 132(2): 646-51.
- Kang MJ, Kim JI, Yoon SY, Kim JC and Cha IJ: Pinitol from soybeans reduces postprandial blood glucose in patients with type 2 diabetes mellitus. J Med Food 2006; 9(2): 182-6.
- Shirali S, Zahra Bathaie S and Nakhjavani M: Effect of crocin on the insulin resistance and lipid profile of streptozotocin‐induced diabetic rats. Phytother Res 2013; 27(7): 1042-7.
- Shirali S, Bathaei S, Nakhjavani M and Ashoori M: Effects of saffron (Crocus sativus) aqueous extract on serum biochemical factors in streptozotocin-induced diabetic rats. IJMAPR 2012; 2(56): 293-308.
- Hosseini S, Gorjian M, Rasouli L and Shirali S: A Comparison between the effect of green tea and kombucha prepared from green tea on the weight of diabetic rats. Biosciences Biotechnology Research Asia 2015; 20(1): 141-145.
- Zare M, Rastegar S, Ebrahimi E, Roohipoor A and Shirali S: Role of pancreatic duct cell in beta cell neogenesis: A mini review study. Diabetes & Metabolic Syndrome: Clinical Research & Reviews 2016.
- Mashhadi NS, Saadat S, Afsharmanesh MR and Shirali S: Study of association between beverage consumption pattern and lipid profile in shift workers. Diabetes & Metabolic Syndrome: Clinical Research & Reviews 2016.
- Ebrahimi E, Shirali S and Talaei R: The protective effect of marigold hydroalcoholic extract in induced diabetic rats: evaluation of cardiac and pancreatic biomarkers in the serum. J Bot 2016; 1-6. http://dx.doi.org/10.1155/ 2016/9803928.
- Mansouri E, Kooti W, Bazvand M, Boroon MG, Amirzargar A and Afrisham R: The effect of hydroalcoholic extract of Foeniculum vulgare mill on leukocytes and hematological tests in male rats. Jundishapur J Nat Pharm Prod 2015; 10(1): e18396.
- Kooti W, Mansouri E, Ghasemiboroon M, Harizi M, Ashtary-Larky D and Afrisham R: The Effects of hydroalcoholic extract of Apium graveolens leaf on the number of sexual cells and testicular structure in the rat. Jundishapur J Nat Pharm Prod 2014; 9(4): e17532.
- Bathaie S, Mokarizade N and Shirali S: An overview of the mechanisms of plant ingredients in the treatment of diabetes mellitus. JMP 2012; 11(44): 1-24.
- Dhanasekaran M, Ignacimuthu S and Agastian P: Potential hepatoprotective activity of ononitol monohydrate isolated from tora L. on carbon tetrachloride-induced hepato-toxicity in wistar rats. Phytomedicine 2009; 16(9): 891-5.
- Sureshan KM, Murakami T and Watanabe Y: Total syntheses of cyclitol based natural products from myo-inositol: brahol and pinpollitol. Tetrahedron 2009; 65(20): 3998-4006.
- Gao Y, Zhang M, Wu T, Xu M, Cai H and Zhang Z: Effects of d-Pinitol on insulin resistance through the PI3K/Akt signaling pathway in type 2 diabetes mellitus rats. J Agric Food Chem 2015; 63(26): 6019-26.
- Bernabe M, Fenwick R, Frias J, Jimenez-Barbero J, Price K and Valverde S: Determination, by NMR spectroscopy, of the structure of ciceritol, a pseudodisaccharide isolated from lentils. J Agric Food Chem 1993; 41(6): 870-2.
- Larner J and Kennington A: Administering D-chiroinositol to achieve a reduction in blood glucose level. Google Patents, 1992; Publication number: US 5124360 A.
- Larner J, Kennington A and Huang LC: Administering D-chiro-inositol for insulin-resistant diabetics. Google Patents, 1995; Publication number: US 5428066 A.
- Greene DA, De Jesus Jr PV and Winegrad A: Effects of insulin and dietary myoinositol on impaired peripheral motor nerve conduction velocity in acute streptozotocin diabetes. J Clin Invest 1975; 55(6): 1326.
- Fonteles M, Huang L and Larner J: Infusion of pH 2.0 D-chiro-inositol glycan insulin putative mediator normalizes plasma glucose in streptozotocin-diabetic rats at a dose equivalent to insulin without inducing hypoglycaemia. Diabetologia 1996; 39(6): 731-4.
- Farias V, Macêdo F, Oquendo M, Tomé A, Báo S and Cintra D: Chronic treatment with d‐chiro‐inositol prevents autonomic and somatic neuropathy in STZ‐induced diabetic mice. Diabetes Obes Metab 2011; 13(3): 243-50.
- Larner J: D-chiro-inositol–its functional role in insulin action and its deficit in insulin resistance. J Diabetes Res 2002; 3(1): 47-60.
- Kim JI, Kim J, Kang MJ, Lee MS, Kim JJ and Cha IJ: Effects of pinitol isolated from soybeans on glycaemic control and cardiovascular risk factors in Korean patients with type II diabetes mellitus: a randomized controlled study. Eur J Clin Nutr 2005; 59(3): 456-8.
- Sivakumar S and Subramanian SP: Pancreatic tissue protective nature of D-Pinitol studied in streptozotocin-mediated oxidative stress in experimental diabetic rats. Eur J Pharmacol 2009; 622(1- 3): 65-70.
- Sivakumar S, Palsamy P and Subramanian SP: Attenuation of oxidative stress and alteration of hepatic tissue ultrastructure by D-pinitol in streptozotocin-induced diabetic rats. Free Radic Res 2010; 44(6): 668-78.
- Nascimento N, Cortez LU, Sousa LG, Fonteles M and Santos C: Pinitol ameliorates impaired pressurenatriuresis in experimental diabetes (1063.5). FASEB 2014; 28(1): 1063.5.
- Kim MJ, Yoo KH, Kim JH, Seo YT, Ha BW and Kho JH: Effect of pinitol on glucose metabolism and adipocytokines in uncontrolled type 2 diabetes. Diabetes Res Clin Pract 2007; 77(3): S247-S51.
- Bates SH, Jones RB and Bailey CJ: Insulin‐like effect of pinitol. Br J Pharmacol 2000; 130(8): 1944-8.
- Xue Y, Miao Q, Zhao A, Zheng Y, Zhang Y and Wang P: Effects of sea buckthorn (Hippophaë rhamnoides) juice and L-quebrachitol on Type 2 diabetes mellitus in db/db mice. J Func Foods 2015; 16: 223-33.
- Musalmah M, Elkkairee M, Lau C and Wan Ngah W: Effect of latex quebrachitol and L-chiro-inositol on blood glucose levels in normal and alloxan-induced diabetic rats. MJBMB 2001; 6: 7 11.
- Lee YK, Park PB, Park YI and Chung NH: Comparison of soybean varieties for cyclitol content in soybean curd wastewaters. J Appl Biol Chem 2003; 46(4): 148-51.
- Streeter JG: Simple partial purification of D-pinitol from soybean leaves. Crop Sci 2001; 41(6): 1985-7.
- Horbowicz M, Brenac P and Obendorf RL: Fagopyritol B1, O-α-D-galactopyranosyl-(1→ 2)-D-chiroinositol, a galactosyl cyclitol in maturing buckwheat seeds associated with desiccation tolerance. Planta 1998; 205(1): 1-11.
- Steadman KJ, Burgoon MS, Schuster RL, Lewis BA, Edwardson SE and Obendorf RL: Fagopyritols, D-chiro- inositol, and other soluble carbohydrates in buckwheat seed milling fractions. J Agric Food Chem 2000; 48(7): 2843-7.
- Liu RJ, PIao CH, Yu HS, Peng S, Lu CW and Hu YH: TLC determination method research of D-chiroinositol in buckwheat seed. Food Sci Technol 2006; 8(1): 263-5.
- Yao Y, Shan F, Bian J, Chen F, Wang M and Ren G: D-chiro-inositol-enriched tartary buckwheat bran extract lowers the blood glucose level in KK-Ay mice. J Agric Food Chem 2008; 56(19): 10027-31.
- Mokhtari M, Sharifi E and Tabatabayi M: The effect of the hydroalcoholic extract of Ceratonia siliqua seeds on blood glucose and lipids of male diabetic rats. J Sabzevar Univ Med Sci 2010; 3(57): 148-57.
- Tabatabaee S: The effect of hydroalcoholic seeds extract of Ceratonia siliqua L. on the kidney functional factors and serum electrolytes in diabetic male rats. Yafteh 2011; 13(2): 74-82.
- Camero BM and Merino CS: Isolation of inositol from carob extracts; obtain carob, expose to the chromatographic column, recover inositol. Google Patents; 2004, Publication number: US 20030040609 A1.
- Swarup KRA, Sattar MA, Abdullah NA, Abdulla MH, Salman IM and Rathore HA, et al. Effect of dragon fruit extract on oxidative stress and aortic stiffness in streptozotocin-induced diabetes in rats. Pharmacognosy Res 2010; 2(1): 31-5.
- Omidizadeh A, Yusof RM, Roohinejad S, Ismail A, Bakar MZA and Bekhit AEDA: Anti-diabetic activity of red pitaya (Hylocereus polyrhizus) fruit. RSC Adv 2014; 4(108): 62978-86.
- Rebecca OPS, Boyce AN and Somasundram C: Isolation and identification of myo-inositol crystals from dragon fruit (Hylocereus polyrhizus). Molecules 2012; 17(4): 4583-94.
- Diaz-Flores M, Angeles-Mejia S, Baiza-Gutman L, Medina-Navarro R, Hernandez-Saavedra D and Ortega-Camarillo C: Effect of an aqueous extract of Cucurbita ficifolia Bouché on the glutathione redox cycle in mice with STZ-induced diabetes. J Ethnopharmacol 2012; 144(1): 101 8.
- Xia T and Wang Q: D‐chiro‐Inositol found in Cucurbita ficifolia (Cucurbitaceae) fruit extracts plays the hypoglycaemic role in streptozocin‐diabetic rats. J Pharm Pharmacol 2006; 58(11): 1527-32.
- Roman-Ramos R, Almanza-Perez J, Fortis-Barrera A, Angeles-Mejia S, Banderas-Dorantes T and Zamilpa-Alvarez A: Antioxidant and anti-inflammatory effects of a hypoglycemic fraction from Cucurbita ficifolia Bouché in streptozotocin-induced diabetes mice. Am J Chin Med 2012; 40(1): 97-110.
- Fortis‐Barrera A, Alarcon‐Aguilar FJ, Banderas‐Dorantes T, Diaz‐Flores M, Roman‐Ramos R and Cruz M: Cucurbita ficifolia Bouché (Cucurbitaceae) and D‐chiro‐inositol modulate the redox state and inflammation in 3T3‐L1 adipocytes. J Pharm Pharmacol 2013; 65(10): 1563-76.
- Banderas-Dorantes TR, Roman-Ramos R, Zamilpa A, García-Macedo R, Díaz M and Campos MG: Influence of two hypoglycemic Cucurbitaceae (Cucurbita ficifolia Bouché and Ibervillea sonorae Greene) on ATP-sensitive potassium channels in rat aortic rings. Bol Latinoam Caribe Plantas Med Aromat 2012; 11(6): 510-9.
- Yang Y, Cheng XZ and Ren GX: Contents of D-chiro-Inositol, vitexin, and isovitexin in various varieties of mung bean and its products. ASC 2011; 10(11): 1710-5.
- Yao Y, Chen F, Wang M, Wang J and Ren G: Antidiabetic activity of Mung bean extracts in diabetic KK-Ay mice. J Agric Food Chem 2008; 56(19): 8869-73.
- Sato K, Yoshida M, Kanbe K, Hirazawa K and Takeuchi T. Processes for the preparation of D-chiroinositol. Google Patents; 1998, Publication number: US 5714643 A.
- Larner J, Price JD, Heimark D, Smith L, Rule G and Piccariello T: Isolation, structure, synthesis, and bioactivity of a novel putative insulin mediator. A Galactosamine chiro-Inositol Pseudo-Disaccharide Mn2+ Chelate with Insulin-like Activity. J Med Chem 2003; 46(15): 3283-91.
How to cite this article:
Rastegar S, Soltani S, Roohipoor A and Ebrahimi E: Study of plants with D-chiro-inositol and its derivatives on diabetes. Int J Pharmacognosy 2017; 4(2): 43-53. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(2).43-53.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
43-53
699
1869
English
IJP
S. Rastegar, S. Soltani, A. Roohipoor and E. Ebrahimi *
Department of Biochemistry, School of Pharmacy, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
E.Ebrahimi92@yahoo.com
30 November 2016
22 December 2016
26 December 2016
10.13040/IJPSR.0975-8232.IJP.4(2).43-53
01 February 2017