STUDY OF ANTIARTHRITIC ACTIVITY OF TEPHROSIA PURPUREA L. IN EXPERIMENTAL ANIMALS
HTML Full TextSTUDY OF ANTIARTHRITIC ACTIVITY OF TEPHROSIA PURPUREA L. IN EXPERIMENTAL ANIMALS
Umesh Nigam *, S. C. Mehta and Ranjeet Singh Kushwah
Shri Ramnath Singh Institute of Pharmaceutical Science & Technology, Sitholi, Gwalior, Madhya Pradesh, India.
ABSTRACT: The present study evaluated the antiarthritic activity of the ethanol extract of Tephrosia purpurea (EETP) in experimental animal models. Acute oral toxicity studies revealed that EETP is safe up to 2000 mg/kg, with no mortality or observable toxic effects. Antiarthritic activity was assessed using parameters such as body weight, paw volume, mechanical withdrawal threshold, and thermal withdrawal latency in arthritic rats. EETP treatment showed dose-dependent improvement across all parameters. At 400 mg/kg, EETP significantly restored body weight, reduced paw edema, and improved both mechanical and thermal hyperalgesia, comparable to the standard drug Diclofenac. The findings suggest that EETP possesses significant anti-inflammatory and anti-nociceptive properties, supporting its potential as a therapeutic agent in the management of arthritis.
Keywords: Tephrosia purpurea, Antiarthritic activity, Paw edema, Hyperalgesia, Diclofenac
INTRODUCTION: Rheumatoid arthritis (RA) is a chronic, progressive autoimmune disorder characterized by persistent synovial inflammation, joint swelling, pain, and eventual destruction of cartilage and bone. It not only impairs physical function and quality of life but also imposes a significant socioeconomic burden worldwide 1, 2. The pathogenesis of RA involves complex interactions between genetic, environmental, and immunological factors, leading to the release of pro-inflammatory cytokines, oxidative stress, and immune-mediated joint damage 3. Although conventional treatments such as nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, and disease-modifying antirheumatic drugs (DMARDs)
are commonly used, their long-term use is often associated with adverse effects and limited efficacy in some patients 4, 5. In recent years, there has been increasing interest in exploring plant-based therapies as safer and more cost-effective alternatives for the management of inflammatory disorders, including arthritis 6. Medicinal plants are rich sources of bioactive compounds such as flavonoids, alkaloids, glycosides, and phenolic compounds, which possess significant anti-inflammatory, antioxidant, and immunomodulatory properties 7.
These phytoconstituents may help in reducing inflammation, inhibiting oxidative stress, and modulating immune responses, thereby offering potential therapeutic benefits in the treatment of rheumatoid arthritis 8. Tephrosia purpurea L., commonly known as “Sarapunkha,” is a well-known medicinal plant in traditional systems of medicine such as Ayurveda. It has been widely used for its hepatoprotective, anti-inflammatory, antioxidant, antimicrobial, and immunomodulatory activities 9, 10. The plant contains various phytochemicals including flavonoids (such as rutin and quercetin), rotenoids, and phenolic compounds, which are believed to contribute to its pharmacological effects. Despite its traditional use in inflammatory conditions, scientific validation of its antiarthritic potential through experimental models remains essential 11, 12.
Therefore, the present study aims to evaluate the antiarthritic activity of Tephrosia purpurea leaf extracts using experimental animal models. By assessing parameters such as inflammation, joint swelling, and biochemical markers, this study seeks to provide scientific evidence supporting the traditional use of this plant and to explore its potential as a safer alternative or complementary therapy for the management of arthritis.
MATERIALS AND METHODS:
Collection and Authentication of Plant: Fresh leaves of the Tephrosia purpurea plant were collected from the local area of Gwalior district, Madhya Pradesh, India. The collected leaves were shade-dried and crushed into coarse powder using a mechanical grinder. The powder was stored in an airtight container for subsequent extraction.
Extraction of Plant Materials: The petroleum ether, and ethanolic extracts of the leaves of Tephrosia purpurea were prepared by following methods 13, 14.
- Maceration
- Soxhlet
A weighed quantity (40 g) of air-dried powdered leaves of Tephrosia purpurea was macerated with 250 ml of petroleum ether at room temperature for 7 days with occasional shaking, and the process was repeated using the same volume of solvent. The macerates were pooled, transferred to a previously weighed petridish, and evaporated to dryness at room temperature to obtain the dried petroleum ether extract, the yield of which (PETP) was calculated as a percentage after reweighing the petridish. The marc remaining after petroleum ether extraction was air-dried and subsequently macerated with ethanol following the same procedure to obtain the ethanolic extract (EETP). Both PETP and EETP were immediately transferred into clean, dry, tightly sealed amber-colored glass bottles after solvent evaporation to protect the extracts from light-induced degradation, particularly of photosensitive phytoconstituents such as flavonoids and phenolic compounds. The containers were tightly closed to prevent exposure to air and moisture, thereby minimizing oxidative degradation and contamination, and were stored under refrigerated conditions (2–8°C) to preserve chemical stability, inhibit microbial growth, and maintain pharmacological activity throughout the experimental period.
Phytochemical analysis of PETP and EETP: Phytochemical screening of the leaves of Tephrosia purpurea was performed using following tests. Chemical tests were carried out on the PETP and EETP using standard procedures to identify the constituents. The dry extracts were dissolved in petroleum ether, ethanol and water respectively to obtained solution of respective extract (100 mg/ml) for phytochemical analysis 15.
Pharmacological Study:
Preparation of Dosage form: Dosage forms of individual extracts were prepared as per the following procedures.
Ethanolic Extract: The ethanolic extract of Tephrosia purpurea was suspended with tween 80 (2%) in a glass mortar with gradual addition of distilled water to make up the required volume.
Drugs: Accurately weighed quantity of diclofenac were suspended in distilled water to make volume.
Vehicles: Vehicle was prepared by adding 2% tween 80 into distilled water, without addition of extracts or drugs 16.
Experimental animals: Wistar rats (170–220 g) of either sex were bought from a CPCSEA-approved breeder. The animals were kept in an air-conditioned environment with a 12-hour light/dark cycle, a temperature of 22 ± 2 °C, and a relative humidity of 45 to 55%. Food pellets were freely available to the animals. There was unlimited access to filtered water. The Institutional Animal Ethics Committee (IAEC), which was established in compliance with the regulations of the Committee for the Purpose of Control and Supervision on Experimental Animals (CPCSEA), India, authorised the experimental procedure 17.
Acute Oral Toxicity Study: In acute toxicity investigations, healthy female Wistar rats weighing between 170 and 220 g were employed in accordance with OECD guidelines-425. After an overnight fast, the animals were split up into groups of five rats each. Oral doses of PETP and EETP extracts were given at 5, 50, 300, and 2000 mg/kg body weight. For two hours, the rats' behavioural and autonomic profiles were continually monitored, and for up to 48 hours, they were checked for any indications of toxicity or death (OECD-425) 18.
Antiarthritic Activity 19, 20:
Freund’s Complete Adjuvant Induced Arthritis: On day 0, the right hind paw's subplanter area received an indradermal injection of 0.1 ml of Freund's complete adjuvant (FCA, Sigma) to cause arthritis. The following six groups of six rats each were created from the rats:
TABLE 1: ANIMAL FOR ANTI-ARTHRITIC ACTIVITY
| Group | Animals | Gender | Treatment | Doses | No of Animal | Treatment period |
| Control | Albino rats | Male | 2% Tween 80 | - | 6 | 28 |
| Disease Control | Albino rats | Male | Complete freund’s adjuvant | 0.1 ml | 6 | 28 |
| Standard | Albino rats | Male | Diclofenac sodium | 5 mg/kg | 6 | 28 |
| Test dose 1 | Albino rats | Male | - | 200 mg/kg | 6 | 28 |
| Test Dose 2 | Albino rat | Male | - | 400 mg/kg | 6 | 28 |
The dosing of all the groups was started from day 12, once day. The following parameters were measured at regular intervals (Day 0, 1, 4, 8, 12, 16, 20, 24and 28) body weight, paw volume, joint diameter, tactile allodynia and thermal hyperalgesia.
Quantification of Paw Edema: The volume of the hind paws was measured using a plethysmometer to determine the degree of arthritis. On days 0, 1, 4, 8, 12, 16, 20, 24, and 28 following the development of arthritis, paw volume (ml) was measured. The change in paw volume relative to day 0 paw volume was used to express the data 21.
Joint Diameter: A digital vernier calliper (Model: CD-6 CS, Mitutoyo Corp., Japan) was used to measure the joint diameter before to injection, and adjuvant was then given. On days 1, 4, 8, 12, 16, 20, 24, and 28, the joint diameter was measured once more. The change in joint diameter relative to the joint diameter on day 0 was used to represent the data 22.
Tactile Allodynia: Von Frey Hairs (Model: 2888, Almemo, Germany) of increasing gauge was used to assess the tactile allodynia of the hind paws. Von Frey hairs (0.6 to 12.6g) were put to the planter surface of the rats' hind paws after they had been in the Perspex cage for ten minutes to acclimatise. Each paw received three stimulation for each hair during the course of two to three seconds. The mechanical withdrawal threshold (MWT) was determined by taking the lowest weight of Von Frey hair to cause a withdrawal after three successive applications 23.
Thermal Hyperalgesia: To assess the impact of EEDP, thermal hyperalgesia was measured, and a thermal planter tester (Model: 37360, UGO Basile, Italy) was used to identify a noxious thermal stimulus.
Before beginning the behavioural testing, the rats were, in short, acclimated to the testing area for at least ten minutes. Radiant heat was delivered to the hind paw's planter surface after acclimatisation until the rat raised its paw.
When the animal withdrew its paw, or disrupted the reflected light beam, a photoelectric cell automatically turned off the heat surface. The time at which this happened was noted as the paw withdrawal latency (PWL). 15 seconds was the deadline 24.
Statistical Analysis: The data were analyzed by One-way ANOVA followed by Dunnett’s multiple comparison test using Graphpad 5.0 software. The values were expressed as mean ± SEM. P < 0.05, P < 0.01 and P < 0.001 were considered to be statistically significant.
RESULTS AND DISCUSSION:
Percent Yield and Characteristics of Different Extracts of Tephrosia purpurea: The petroleum ether extract of Tephrosia purpurea (PETP) was pale yellow in color with a semisolid consistency and showed a percentage yield of 4.11%. In contrast, the ethanolic extract (EETP) was dark green, also semisolid in nature, with a higher percentage yield of 5.67%.
Qualitative Phytochemical Analysis of PETP and EETP: As shown in Table 2, the qualitative phytochemical screening of Tephrosia purpurea's petroleum ether extract (PETP) and ethanolic extract (EETP) identified the presence and lack of many classes of phytoconstituents. Proteins, amino acids, lipids and oils, steroids, volatile oils, flavonoids, alkaloids, and triterpenoids were all detected by PETP. But there were no carbohydrates, cardiac glycosides, anthraquinone glycosides, saponin glycosides, cyanogenetic glycosides, tannins, or phenolic substances. Carbohydrates, proteins, amino acids, steroids, cardiac glycosides, anthraquinone glycosides, cyanogenetic glycosides, alkaloids, tannins, phenolic compounds, and triterpenoids were all present in EETP. The ethanolic extract included no fats and oils, volatile oils, saponin glycosides, or flavonoids.
The difference in solvent polarity is the main cause of the change in phytochemical ingredients between PETP and EETP. As a non-polar solvent, petroleum ether primarily removes non-polar substances such fats, oils, steroids, volatile oils, and certain terpenoids. This idea is supported by the fact that PETP contains fats, oils, and volatile oils. On the other hand, a wider range of polar and moderately polar phytochemicals may be extracted using ethanol, a polar solvent. The effectiveness of EETP in extracting polar bioactive elements is demonstrated by the identification of carbohydrates, cardiac glycosides, anthraquinone glycosides, cyanogenetic glycosides, tannins, and phenolic chemicals.
TABLE 2: QUALITATIVE PHYTOCHEMICAL ANALYSIS OF PETP AND EETP
| S. no. | Phytochemical Test | PETP | EETP |
| 1 | Carbohydrates | – | + |
| 2 | Proteins | + | + |
| 3 | Amino acids | + | + |
| 4 | Fats and Oils | + | – |
| 5 | Steroids | + | + |
| 6 | Volatile oil | + | – |
| 7 | Cardiac glycosides | – | + |
| 8 | Anthraquinone glycosides | – | + |
| 9 | Saponin glycosides | – | – |
| 10 | Cyanogenetic glycosides | – | + |
| 11 | Flavonoids | + | – |
| 12 | Alkaloids | + | + |
| 13 | Tannins and Phenolic Compounds | - | + |
| 14 | Triterpenoids | + | + |
(+ present, - absent), PETP- Petroleum ether extract of Tephrosia purpurea, EETP- Ethanolic extract of Tephrosia purpurea.
Pharmacological Activity:
Acute Toxicity Test (AOT): EETP was tested for acute oral toxicity at doses of 5, 30, 50, and 2000 mg/kg body weight (p.o.). Each dosing group employed a total of five animals. During the monitoring period, no death was noted in either of the treated groups. At dosages of 5 mg/kg (0/5), 30 mg/kg (0/5), 50 mg/kg (0/5), and 2000 mg/kg (0/5), every animal lived. Additionally, at whatever dose level, there were no outward indications of toxicity, including altered skin and hair, eyes, mucous membranes, respiratory pattern, autonomic or central nervous system activity, behavioural abnormalities, tremors, convulsions, salivation, diarrhoea, lethargy, sleep, or coma. Since, no mortality occurred even at the highest dose of 2000 mg/kg, the median lethal dose (LD₅₀) of EETP is considered to be greater than 2000 mg/kg body weight.
TABLE 3: ACUTE TOXICITY TEST OF EETP
| Sr. no. | Extract | Dose (mg/kg, p.o.) | No. of Animals (Dead/Survived) |
| 1 | EETP | 5 | 0 / 5 |
| 2 | EETP | 50 | 0 / 5 |
| 3 | EETP | 30 | 0 / 5 |
| 4 | EETP | 2000 | 0 / 5 |
The results of the present acute toxicity study indicate that EETP is relatively safe when administered orally to experimental animals. The absence of mortality and observable toxic signs at doses up to 2000 mg/kg suggests a wide margin of safety.
Antiarthritic Activity:
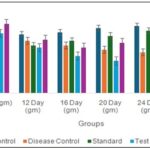
Effect of Oral Administration of Ethanol Extract of on Body Weights in Arthritic Rats: The effect of oral administration of ethanol extract of Tephrosia purpurea (EEDP) on body weight in arthritic rats is presented in Fig. 1. In the vehicle control group, normal progressive weight gain was observed throughout the experimental period, increasing from 190.3 ± 3.66 g on day 0 to 199.5 ± 5.68 g on day 28. In contrast, the disease control group showed a gradual and significant reduction in body weight compared to the vehicle control group. Body weight decreased from 193.11 ± 5.26 g on day 0 to 182.11 ± 5.17 g on day 28, with significant reduction observed from day 16 onward (#). This decrease reflects the systemic impact of arthritis.
The standard-treated group (Diclofenac 5 mg/kg) showed improvement in body weight after initial reduction. Although body weight slightly decreased up to day 12 (187.34 ± 4.11 g), it gradually increased thereafter, reaching 197.31 ± 4.25 g by day 28, approaching normal control values. In the Test 1 group (EEDP 200 mg/kg), body weight initially decreased from 193 ± 3.11 g to 180.11 ± 3.89 g by day 20, followed by gradual recovery to 187.78 ± 3.67 g on day 28. However, the recovery was moderate compared to the standard group. In the Test 2 group (EEDP 400 mg/kg), a better protective effect was observed. Although body weight slightly declined up to day 16 (186.32 ± 6.11 g), a consistent increase was noted thereafter, reaching 199.71 ± 5.25 g on day 28, comparable to the vehicle control and standard-treated groups.
FIG. 1: EFFECT OF ORAL ADMINISTRATION OF ETHANOL EXTRACT OF TEPHROSIA PURPUREA ON BODY WEIGHTS IN ARTHRITIC RATS. Standard-Diclofenac (5 mg/kg); Test 1-EETP (200 mg/kg); Test 2-EETP (400 mg/kg). Data are expressed as mean ± S.E.M.; n=6 rats per group. Two way ANOVA followed by Bonferroni’s post hoc test when compared with arthritic control group *P<0.05, **P<0.01, ***P<0.001 and One way ANOVA followed by Dunnett’s test when arthritic control group compared with vehicle control #P<0.001.
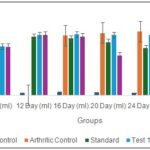
Effect of Oral Administration of Ethanol Extract of Tephrosia purpurea on Right Hind Paw Volume in Arthritic Rats: The effect of oral administration of ethanol extract of Tephrosia purpurea (EETP) on right hind paw volume in arthritic rats is presented in Fig. 2. Data are expressed as mean ± S.E.M. (n = 6). The arthritic control group showed a significant increase in paw volume compared to the vehicle control group from day 1 to day 28 (#P < 0.001). Paw volume progressively increased from 2.571 ± 0.045 ml on day 1 to 3.411 ± 0.033 ml on day 28, indicating successful induction and progression of arthritis. In contrast, the vehicle control group maintained minimal paw volume throughout the study period, confirming the absence of inflammation under normal conditions.
The standard-treated group (Diclofenac 5 mg/kg) showed a significant reduction in paw edema compared to the arthritic control group. The reduction became statistically significant from day 20 (*P < 0.05) and highly significant on days 24 and 28 (***P < 0.001). Paw volume decreased from 3.034 ± 0.053 ml on day 12 to 2.034 ± 0.081 ml on day 28, demonstrating effective suppression of inflammation.
The Test 1 group (EETP 200 mg/kg) showed a moderate reduction in paw volume. Although inflammation persisted during the early phase, a significant reduction was observed on day 28 (*P < 0.05), with paw volume decreasing to 2.321 ± 0.091 ml compared to the arthritic control group. The Test 2 group (EETP 400 mg/kg) demonstrated a more pronounced anti-inflammatory effect. A marked reduction in paw volume was observed from day 20 onward, with significant decreases on days 24 and 28 (***P < 0.001). Paw volume reduced to 2.543 ± 0.44 ml by day 28, indicating dose-dependent efficacy.
FIG. 2: EFFECT OF ORAL ADMINISTRATION OF ETHANOL EXTRACT OF TEPHROSIA PURPUREA ON RIGHT HIND PAW VOLUME IN ARTHRITIC RATS. Standard-Diclofenac (5 mg/kg); Test 1-EETP (200 mg/kg); Test 2-EETP (400 mg/kg). Data are expressed as mean ± S.E.M.; n=6 rats per group. Two way ANOVA followed by Bonferroni’s post hoc test when compared with arthritic control group *P<0.05, **P<0.01, ***P<0.001 and One way ANOVA followed by Dunnett’s test when arthritic control group compared with vehicle control #P<0.001.
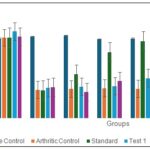
Effect of Oral Administration of Ethanol Extract of Tephrosia purpurea on Right Hind Paw Mechanical Withdrawal Threshold in Arthritic Rats (Tactile allodynia): The effect of oral administration of ethanol extract of Tephrosia purpurea (EETP) on right hind paw mechanical withdrawal threshold in arthritic rats is summarized in Fig. 3.
The arthritic control group showed a significant reduction in paw withdrawal threshold from baseline (65.93 ± 5.47 g) to day 12 (23.26 ± 2.72 g, P<0.001), which persisted throughout the study period, indicating the establishment of mechanical hyperalgesia associated with arthritis. In contrast, the vehicle control group maintained a consistent paw withdrawal threshold throughout the experiment, confirming that changes observed in arthritic rats were due to the disease model. The standard treatment (Diclofenac 5 mg/kg) significantly reversed the mechanical hyperalgesia in arthritic rats.
By day 24, the paw withdrawal threshold increased to 62.92 ± 3.45 g (P<0.001 vs arthritic control), and by day 28, it reached 73.11 ± 5.84 g (P<0.001), demonstrating a strong anti-nociceptive effect. Among the test groups, Test 1 (EETP 200 mg/kg) produced a gradual increase in paw withdrawal threshold from day 16 (25.76 ± 3.85 g) to day 28 (40.11 ± 4.45 g, P<0.01 vs arthritic control), indicating moderate anti-hyperalgesic activity. Test 2 (EETP 400 mg/kg) exhibited a more pronounced effect, with the paw withdrawal threshold increasing to 41.07 ± 2.66 g on day 24 (P<0.01) and 56.98 ± 3.82 g on day 28 (P<0.001), suggesting a dose-dependent improvement in mechanical nociception. Overall, the results indicate that EETP mitigates mechanical hyperalgesia in arthritic rats in a dose-dependent manner, with the higher dose approaching the efficacy of the standard drug Diclofenac.
FIG. 3: EFFECT OF ORAL ADMINISTRATION OF ETHANOL EXTRACT OF TEPHROSIA PURPUREA ON RIGHT HIND PAW MECHANICAL WITHDRAWAL THRESHOLD IN ARTHRITIC RATS. Standard-Diclofenac (5 mg/kg); Test 1-EETP (200 mg/kg); Test 2-EETP (400 mg/kg). Data are expressed as mean ± S.E.M.; n=6 rats per group. Two way ANOVA followed by Bonferroni’s post hoc test when compared with arthritic control group *P<0.05, **P<0.01, ***P<0.001 and One way ANOVA followed by Dunnett’s test when arthritic control group compared with vehicle control #P<0.001.
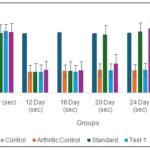
Effect of Oral Administration of Ethanol Extract of Tephrosia purpurea on Right Hind Paw Withdrawal Latency in Arthritic Rats (Thermal hyperalgesia): The effect of oral administration of ethanol extract of Tephrosia purpurea (EETP) on right hind paw withdrawal latency in arthritic rats is presented in Fig. 4.
The vehicle control group maintained a stable withdrawal latency throughout the experimental period (approximately 5 seconds), indicating normal nociceptive response and absence of hyperalgesia.
In contrast, the arthritic control group showed a significant reduction in paw withdrawal latency from baseline (4.88 ± 0.046 sec) to day 12 (1.78 ± 0.032 sec, P<0.001), which persisted up to day 28. This marked decrease confirms the development of thermal hyperalgesia following induction of arthritis. The standard group (Diclofenac 5 mg/kg) did not show improvement at days 12 and 16; however, a significant increase in withdrawal latency was observed from day 20 onward. On day 20, latency increased to 4.86 ± 0.054 sec (P<0.001 vs arthritic control), reaching 5.87 ± 0.045 sec (P<0.001) by day 28, indicating a strong anti-nociceptive effect. Among the test groups, Test 1 (EETP 200 mg/kg) showed minimal improvement until day 24, with a moderate increase in latency observed on day 28 (2.85 ± 0.087 sec), though the effect was less pronounced compared to the standard.
Test 2 (EETP 400 mg/kg) demonstrated a dose-dependent and significant increase in paw withdrawal latency. While early time points showed limited improvement, a marked increase was observed on day 24 (5.38 ± 0.062 sec, P<0.001) and day 28 (5.55 ± 0.38 sec, P<0.001), restoring latency values close to normal control levels. These findings indicate that EETP, particularly at 400 mg/kg, effectively attenuates arthritis-induced thermal hyperalgesia.
FIG. 4: EFFECT OF ORAL ADMINISTRATION OF ETHANOL EXTRACT OF TEPHROSIA PURPUREA ON RIGHT HIND PAW WITHDRAWAL LATENCY IN ARTHRITIC RATS. Standard-Diclofenac (5 mg/kg); Test 1-EETP (200 mg/kg); Test 2-EETP (400 mg/kg). Data are expressed as mean ± S.E.M.; n=6 rats per group. Two way ANOVA followed by Bonferroni’s post hoc test when compared with arthritic control group *P<0.05, **P<0.01.
DISCUSSION: The present study investigated the phytochemical composition, safety profile, and antiarthritic potential of petroleum ether (PETP) and ethanolic extract (EETP) of Tephrosia purpurea. The findings demonstrate that extraction solvent polarity plays a crucial role in determining phytochemical composition, which in turn influences pharmacological activity. The percentage yield of EETP (5.67%) was higher than PETP (4.11%), suggesting that ethanol is more efficient in extracting a broader range of constituents. This is consistent with the principle that polar solvents extract polar and moderately polar compounds, whereas non-polar solvents primarily extract lipophilic substances. PETP contained lipids, oils, steroids, volatile oils, flavonoids, alkaloids, and triterpenoids, confirming its affinity for non-polar phytoconstituents.
In contrast, EETP showed the presence of carbohydrates, glycosides, tannins, phenolic compounds, and alkaloids, indicating its richness in bioactive polar constituents. These phytochemicals, particularly phenolics, tannins, alkaloids, and triterpenoids, are well known for their anti-inflammatory and analgesic properties, which may contribute to the observed pharmacological effects. The acute toxicity study revealed that EETP is safe up to a dose of 2000 mg/kg, with no mortality or observable signs of toxicity. This indicates a wide therapeutic window and supports its potential for safe pharmacological use. The absence of behavioral and physiological abnormalities further confirms its non-toxic nature in experimental animals. In the antiarthritic study, arthritis induction resulted in significant weight loss, increased paw edema, and heightened pain sensitivity (mechanical and thermal hyperalgesia), reflecting systemic inflammation and disease progression. Treatment with EETP showed dose-dependent improvement across all parameters. Body weight analysis demonstrated that EETP, particularly at 400 mg/kg, effectively prevented arthritis-induced weight loss, restoring values close to normal and comparable to the standard drug diclofenac. This suggests an improvement in overall health status and reduction in systemic inflammation. The reduction in paw volume indicates the anti-inflammatory potential of EETP.
The higher dose (400 mg/kg) significantly reduced edema from day 20 onward, approaching the efficacy of diclofenac. This effect may be attributed to inhibition of inflammatory mediators such as prostaglandins, cytokines, and leukotrienes by bioactive constituents like flavonoids, tannins, and triterpenoids. Pain-related parameters further supported the antiarthritic activity. EETP significantly improved mechanical withdrawal threshold and thermal withdrawal latency in a dose-dependent manner. The higher dose showed marked reversal of hyperalgesia, indicating both anti-nociceptive and anti-inflammatory actions. These effects may be mediated through modulation of peripheral and central pain pathways, possibly involving inhibition of inflammatory mediators and oxidative stress. Overall, the study demonstrates that the ethanolic extract of Tephrosia purpurea possesses significant antiarthritic activity, supported by its rich phytochemical profile and safety at high doses.
The observed effects are dose-dependent and comparable, to some extent, with the standard drug diclofenac. The therapeutic potential of EETP may be attributed to the synergistic action of multiple bioactive compounds. However, further studies are needed to isolate and characterize the active constituents, elucidate precise mechanisms of action, and evaluate long-term safety and clinical efficacy.
CONCLUSION: The results of the present study demonstrate that the ethanol extract of Tephrosia purpurea exhibits significant antiarthritic activity in experimental animals. The extract showed a favorable safety profile, with no toxicity observed up to 2000 mg/kg. EETP effectively reduced inflammation, prevented body weight loss, and alleviated both mechanical and thermal hyperalgesia in a dose-dependent manner.
The higher dose (400 mg/kg) produced effects comparable to the standard drug Diclofenac. These findings validate the traditional use of Tephrosia purpurea in inflammatory conditions and suggest its potential as a safe and effective alternative for arthritis management. Further studies are recommended to isolate active constituents and elucidate the underlying mechanisms of action.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Barnes CG and Mason M: Rheumatoid arthritis. In: Mason M, Currey HLF, editors. Introduction to Clinical Rheumatology. Bath: Pitman 1975; 2.
- Möttönen TT: Prediction of erosiveness and rate of development of new erosions in early rheumatoid arthritis. Ann Rheum Dis 1988; 47: 648–653.
- Van der Heijde DM: Joint erosions in patients with early rheumatoid arthritis. British Journal of Rheumatology 1995; 34(8).
- Onions CT, Friedrichsen GWS and Burchfield RW: The Oxford Dictionary of English Etymology. Oxford: Oxford University Press 1996.
- Hunder GG: Atlas of Rheumatology. Philadelphia: Current Medicine 2005.
- Landré-Beauvais AJ: The first description of rheumatoid arthritis (doctoral dissertation, 1800). Joint Bone Spine 2001; 68: 130–143.
- Hussain JK, Qadir IM, Jan A and Hassan GA: Anti-diarrheal activity of methanolic extract of Tephrosia purpurea Pers. APPDR 2013; 70(2): 345–347.
- Pavana P, Sethupathy S, Santha K and Manoharan S: Effects of Tephrosia purpurea Pers. aqueous seeds extract on blood glucose and antioxidant enzymes in streptozotocin-induced diabetic rats. African Journal of Traditional Complementary and Alternative Medicine 2009; 6(1): 78–86.
- Asuntha G, Prasannaraju Y, Sujatha D and Prasad KVSR: Assessment of ethanolic extract of Tephrosia purpurea (L.) Pers. on lithium-pilocarpine induced status epilepticus and oxidative stress in Wistar rats. Braz J Pharmacogn 2010; 20(5): 767–772.
- Gopalakrishnan S, Vadivel E and Dhanalakshmi K: Anti-inflammatory and analgesic activities of Tephrosia purpurea (L.) Pers. aerial and root extracts. J Pharm Res 2010; 3(5): 1103–1106.
- Shah R, Kathad H, Sheth R and Sheth N: In-vitro antioxidant activity of root of Tephrosia purpurea Int J Pharm Pharm Sci 2010; 2(3): 30–33.
- Dalwadi P, Patel J and Patani P: Tephrosia purpurea Linn (Sharpunkha, Wild Indigo): A review on phytochemistry and pharmacological studies. Indian Journal of Pharmaceutical and Biological Research 2014; 2(1): 108–121. Available from: https://ijpbr.in/index.php/IJPBR/article/view/778
- Palbag S, Dey BK and Singh NK: Ethnopharmacology, phytochemistry and pharmacology of Tephrosia purpurea. Chin J Nat Med 2014; 12(1).
- Pundir R, Singh G, Pandey AA and Saraf SA: Demand of herbal hepatoprotective formulations in Lucknow: A survey. Pharma Res 2009; 1.
- Umate JP and Chaudhari SS: Effect Of Sharpunkhamulachurna with Tandulodak in Asrugdara. Ayushdhara 2017; 4(4): 1282-91.
- Kim HS, Bae S, Lim YJ, So KA, Kim TJ and Bae S: Tephrosin suppresses chemoresistance of paclitaxel-resistant ovarian cancer via inhibition of FGFR1 signaling pathway. Biomedicines 2023; 11: 3155.
- Pimple P, Shah J and Singh P: Emerging phytochemical formulations for management of rheumatoid arthritis: A review. Current Drug Delivery 2025; 22(1): 15–40.
- Nikolova-Ganeva KA, Mihaylova NM, Kechidzhieva LA, Ivanova KI and Zarkov AS: Therapeutic potential of phytochemicals in rheumatoid arthritis. Int J Mol Sci 2025; 26(14): 6813.
- Kciuk M, Garg A, Rohilla M, Chaudhary R, Dhankhar S and Dhiman S: Therapeutic potential of plant-derived compounds in rheumatoid arthritis: A comprehensive review. Antioxidants (Basel) 2024; 13(7): 775.
- Gavarkar PS and Chavan RS: A review on selected herbal plants used in treatment of rheumatoid arthritis. World J Adv Res Rev 2024; 23(2): 626–632.
- Patel P, Chaudhari N and Nanera N: Medicinal herbs as potential treatment of rheumatoid arthritis. Res J Pharmacogn Phytochem 2023; 15(1): 77–81.
- Chahal JK, Chaudhary J, Chauhan S, Jain A, Dhingra A and Chopra B: Protective effect of Cordia myxa leaves extract in FCA-induced arthritis in rats. Res J Pharm Technol 2023; 16(9): 4025–4031.
- Shiozawa S, Shiozawa K and Fujita T: Morphologic observations in early phase of cartilage-pannus junction. Arthritis Rheum 1983; 26: 472–478.
- Hazes JM and Van Zeben D: Oral contraception and its possible protection against rheumatoid arthritis. Ann Rheum Dis 1991; 50: 72–74.
How to cite this article:
Nigam U, Mehta SC and Kushwah RS: Study of antiarthritic activity of Tephrosia purpurea L. in experimental animals. Int J Pharmacognosy 2026; 13(4): 311-20. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(4).311-20.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
11
311-320
538 KB
11
English
IJP
Umesh Nigam *, S. C. Mehta and Ranjeet Singh Kushwah
Shri Ramnath Singh Institute of Pharmaceutical Science & Technology, Sitholi, Gwalior, Madhya Pradesh
mspharma8081@gmail.com
21 March 2026
15 April 2026
19 April 2026
10.13040/IJPSR.0975-8232.IJP.13(4).311-20
30 April 2026