STANDARDIZATION OF MARKETED AMRITARISHTA- A HERBAL FORMULATION
HTML Full TextSTANDARDIZATION OF MARKETED AMRITARISHTA - A HERBAL FORMULATION
Kiran A. Wadkar *, Manish S. Kondawar and Sachin G. Lokapure
Department of Pharmacognosy, Appasaheb Birnale College of Pharmacy, South Shivaji Nagar, Sangli- 416416, Maharashtra, India.
ABSTRACT: The present study aims to standardize Amritarishta based upon chromatographic and spectral studies. The spectral data and HPTLC fingerprint of ethanolic extract of Amritarishta could be used as a valuable analytical tool in the routine standardization of Amritarishta to check the batch to bath variation. Fraction, I and II of ethanolic extract of Amritarishta and standard luteolin and apigenin, were compared by TLC, HPTLC, and HPLC analysis to evaluate the presence of luteolin and apigenin is employing toluene: ethyl acetate: glacial acetic acid (5:4:1 v/v/v), as a mobile phase respectively. The Rf values (0.64) and (0.81) for luteolin and apigenin in both sample and reference standard were found comparable under UV light at 366 nm respectively. The High-Performance Thin Layer Chromatography method developed for quantization was simple, accurate and specific. The present standardization provides a specific and accurate tool to develop qualifications for identity, transparency, and reproducibility of biomarkers luteolin and apigenin in a liquid formulation.
| Keywords: |
Standardization, Amritarishta, Markers
INTRODUCTION: Ayurveda is considered by many scientists to be the oldest healing science. In Sanskrit, Ayurveda means “The Science of Life.” Ayurvedic knowledge originated in India more than 5,000 years ago and is often called the “Mother of All Healing” 1. Ayurveda translates into knowledge (Veda) of life (Ayur) and is one of the oldest and still widely practiced medical systems in the Indian subcontinent 2. The concept of Ayurvedic medicine is to promote health, rather than to fight disease, and Ayurveda in daily life aims at maintaining harmony between nature and the “individual” to ensure optimal health 1.
Ayurveda contains 8 branches of sciences and 10 different diagnostic tools based on tridosha theory (three senses of humour of the body). Ayurveda comprises of various types of medicines including the fermented forms namely arishtas (fermented decoctions) and asavas (fermented infusions). These are regarded as valuable therapeutics due to their efficacy and desirable features.
Amritarishta is a polyherbal hydroalcoholic Ayurvedic preparation and is used as an antioxidant and advised as a choice of remedy in mostly all types of fevers 3. The chief ingredient of Amritarishta is guduchi, the dried stem of Tinospora cordifolia. The chemical constituents reported from stems of Tinospora cordifolia belong to different classes such as alkaloids as tinosporin 4, 5, glycosides as cordifoliosides-A and cordifolioside-B 6, 7, steroids as sitosterol, sesquiterpenoid as tinocordifolinand a large amount of phenolic compounds as gallic acid, ellagic acid, catechin and epicatechin 8 and apigenin 7-O-beta rhamno pyranosyl 9. These compounds have many notable medicinal properties as antidiabetic 10, hepatoprotective 11, antioxidant 12, antimalarial 13, immunomodulatory 14 and antineoplastic properties 15.
MATERIAL AND METHODS:
Chemicals: Chloroform, formic acid, ethyl acetate, toluene were purchased from Merck, India. Methanol and ethanol of analytical reagent grade (Merck, Darmstadt, Germany) were used. Apigenin and luteolin reference standard were purchased from Sigma-Aldrich GmbH, Germany. All other solvents and chemicals were of the highest analytical grade.
Apparatus: All the solvents purchased from E. Merck and S.D. Fine Chemicals, Mumbai. All solvents used for extraction, TLC and HPTLC studies were distilled before use. Solvents used for UV and IR studies were of spectroscopy grade. Solvents used for HPLC analysis were of HPLC grade. Precoated silica gel GF-254 plates procured from E. Merck, Mumbai were used for TLC and HPTLC studies. The UV spectra were recorded on a JASCO V 530 spectrophotometer. The FT IR spectra were recorded on JASCO FT IR 410. Atron HPTLC system consisting of Sparylin spotting, elite-miniluminascence photo documentation and CAMAG scanner. The HPLC analyses were done on a TOSOH–CCPM system. All the results are obtained by repetition of each experiment at least three times.
Procurement of Drug: Commercially available brand (M/S Dabar Pharma) of Amritarishta was procured from the local market.
Standardization using Physicochemical Parameters: The sample of Amritarishta was analyzed for various parameters such as pH, specific gravity, total solids content, alcohol content, amount of reducing and non-reducing sugars.
Preparation of Extracts: Dry 50ml of formulation in vacuum to remove the self-generated alcohol. The dried material was extracted with 250ml ethanol in a Soxhlet extractor at a temperature of 45-50 °C for 48 h. The extract obtained was then concentrated under reduced pressure using rotary evaporator which concentrates bulky solution down to small volumes, without bumping, at temperatures between 30 and 40 °C.
Qualitative Chemical Examination: The ethanolic extracts were qualitatively evaluated by chemical tests and TLC studies for the presence of various phytoconstituents like alkaloids, carbohydrates, saponins, phenolic compounds and tannins, phytosterols and anthraquinone glycosides16.
Isolation of Apigenin and Luteolin: Add 50 ml of water to dissolve the extract and partition successively with n-hexane (50×3), chloroform (50×3) and ethyl acetate (50×3) filter and concentrate the ethyl acetate extract under vacuum and weight. Dissolve 20 mg of residue in 1 ml of methanol 17.
TLC Studies: After preliminary chemical testing and isolation process the crude dried extract was obtained. TLC studies of ethanolic extracts were carried out using Silica gel GF-254 as the stationary phase and Toluene: Ethyl acetate: Glacial acetic acid (5:4:1 v/v/v) as the mobile phase. Spots were observed under UV and visible light. Ethanolic extract was found to contain well-resolved fluorescent components. Out of the four components, two components were separated in an appreciable amount by preparative TLC using the same mobile phase. The fractions were separated as isolated fractions I and II, respectively. The Rf value of isolated fractions I and II was calculated.
HPTLC Studies: HPTLC fingerprint of ethanolic extract was recorded at 366 nm. Ethanolic extract was subjected to HPTLC studies to develop fingerprints using the same conditions as used for TLC.
Spectral Studies: UV, IR and fluorescence spectra were recorded for extract. UV spectra were recorded in ethanol. IR spectra were recorded of the neat sample.
HPLC Studies: Isolated fractions I and II indicated the presence of apigenin and luteolin which is reported to be a major active component of Amritarishta formulation. The extract was analyzed by HPLC using the following conditions:
Column: C18 (25 cm×4.6 mm, i.d.), 10πm
Mobile phase: methanol: water (60:40)
Detection: at 254 nm
Flow rate: 1 ml/min
RESULTS AND DISCUSSION:
Physicochemical Parameters: Standardization of Amritarishta as per pharmacopeia was carried out based on the physicochemical parameters 16. The marketed sample of Amritarishta was found to pass all the pharmacopoeial tests Table 1.
TABLE 1: STANDARDIZATION OF AMRITARISHTA-PHYSICOCHEMICAL PARAMETERS
| S.
no. |
Physicochemical
tests |
Standard
values |
Observed
values |
| 1 | Description | Clear Dark brown liquid without frothing and significant sedimentation; with an astringent taste | Clear and Dark brown liquid significant sedimentation and astringent taste |
| 2 | Total Phenolic | 0.080 to 0.103 per cent w/v Appendix 5.1.1 equivalent to tannic acid | 0.96% w/v |
| 3 | Total solids | Not less than 25.0 per cent w/v, | 32% w/v |
| 4 | Specific gravity | 1.05 to 1.20, | 1.154 |
| 5 | Sugars—reducing
Non-reducing |
Not less than 16 percent w/v,
Not more than 0.80 percent w/v, |
18 % w/v
0.342 % w/v |
| 6 | pH | 3.40 to 4.40 | 4.1 |
| 7 | Alcohol content | 5 to 8 per cent v/v | 6 % v/v |
| 8 | Methanol content | Absent | Absent (The formulation does not contain methanol) |
Ethanolic extract of Amritarishta formulation was subjected to qualitative chemical investigation. A summary of the qualitative analysis is given in Table 2.
TABLE 2: QUALITATIVE CHEMICAL EVALUATION OF EXTRACTS
| S.
No. |
Phytoconstituents | Extract |
| Ethanol | ||
| 1 | Alkaloids | + |
| 2 | Steroids | - |
| 3 | Flavonoids | + |
| 4 | Saponins | - |
| 5 | Tannins | + |
| 6 | Cardiac glycoside | - |
| 7 | Anthraquinone glycoside | - |
| 8 | Coumarins glycoside | - |
(+) Indicates presence, (−) indicates absence. The chemical evaluation reported here is for the entire extract and not for the individual spot.
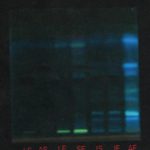
TLC Study: After proper resolution, the spot of apigenin and luteolin was observed in the UV chamber. The developed preparative TLC in UV chamber was shown in Fig. 1. TLC profile and HPTLC fingerprints were developed for ethanolic extract. The TLC profile of ethanolic extract was found to contain four fluorescent components that were well resolved.
The Rf values of the components were 0.43, 0.50, 0.68, and 0.82. Out of the four components, two components were separated in appreciable amounts by preparative TLC using the same mobile phase. The fractions with Rf values of 0.68 and 0.82 were separated as fractions I and II, respectively.
FIG. 1: DEVELOPED PREPARATIVE TLC IN UV CHAMBER
Fractions I and II were evaluated by chemical and spectral methods to study the nature of the components. The color characteristics in UV and visible light revealed the presence of flavones and flavonol in fraction I and isoflavones in fraction II. Table 3. Spectral studies confirmed the results of TLC studies. UV and IR spectra of fraction I and II gave characteristic peaks indicating presence of Flavones. The UV, IR spectra and HPTLC fingerprints of fractions I and II can be used for routine standardization of Amritarishta Table 4.
TABLE 3: TLC STUDIES OF ISOLATED FRACTIONS OF EXTRACT
| S. no. | Isolated fraction | Rf | Color characteristics | Indication |
| 1 | I | 0.68 | Faint Blue | Flavones and /or flavonol |
| 2 | II | 0.82 | Dark Blue | Isoflavones |
TABLE 4: SPECTRAL DATA OF ISOLATED FRACTIONS
| S. no. | Isolated fraction | UV | IR (cm-1) |
| 1 | I | 235, 254, 345 | 3072, 3422 |
| 2 | II | 233, 252 | 1655, 1614 |
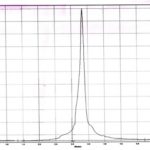
Fingerprint Analysis of Ethanolic Extract of Amritarishta Formulation by HPTLC: HPTLC fingerprint analysis was carried out on ethanolic extract of Amritarishta formulation with solvent system, i.e. toluene: ethyl acetate: glacial acetic acid (5:4:1 v/v/v) using Atron HPTLC system consisting of sparylin spotting, elite-miniluminascence photo documentation and CAMAG scanner. The chromatogram obtained was studied under 366 nm Fig. 2.
FIG. 2: HPTLC FINGERPRINT OF AMRITARISHTA FORMULATION ALONG WITH MARKER COMPOUNDS. 1. LS-Leteolin Standard, 2. AS-Apigenin Standard, 3. LE-Leaf Extract, 4. SE-Stem Extract, 5. IS- Isolated from the formulation, 6. AF- Amritarishta Formulation
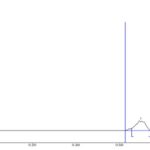
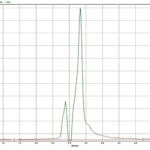
FIG. 3: HPTLC CHROMATOGRAM OF STANDARD APIGENIN
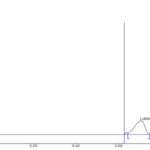
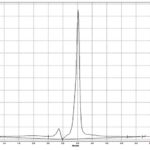
FIG. 4: HPTLC CHROMATOGRAM OF STANDARD LUTEOLIN
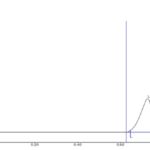
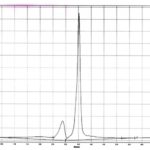
FIG. 5: HPTLC CHROMATOGRAM OF LUTEOLIN AND APIGENIN ISOLATED FROM FORMULATION
The standard HPTLC chromatogram luteolin and apigenin showed two peaks with Rf value 0.64 and 0.81 Fig. 3 and 4. The formulation showed two peaks in HPTLC chromatogram with an Rf value of 0.64 and 0.81 which matched with HPTLC chromatogram of standard luteolin and apigenin Fig. 5.
Characterization of an extract of Amritarishta formulation by HPLC: Fraction, I and II of ethanolic extract of Amritarishta and standard luteolin and apigenin, were compared by HPLC analysis. The HPLC analysis of this standard luteolin and apigenin peaks with retention times of 3.333 and 3.520 min, respectively, Fig. 6 and 7. The HPLC analysis of fraction I and II luteolin and apigenin isolated from formulation under the same conditions as mentioned it gave a peak with RT 3.342 and 3.532 min Fig. 8 and 9.
Thus, it can be said that the fraction I and II may contain luteolin and apigenin. It can be the possible analytical markers for standardization of Amritarishta. After quantitative estimation of luteolin and apigenin, specifications can be stated. This should then serve as a simple, accurate and routine method of analysis for Amritarishtas.
FIG. 6: HPLC PROFILE OF STANDARD LUTEOLIN
FIG. 7: HPLC PROFILE OF STANDARD APIGENIN
FIG. 8: HPLC PROFILE OF LUTEOLIN ISOLATED FROM FORMULATION
FIG. 9: HPLC PROFILE OF APIGENIN ISOLATED FROM FORMULATION
CONCLUSION: The spectral data and HPTLC fingerprint of ethanolic extract of Amritarishta could be used as a valuable analytical tool in the routine standardization of Amritarishta to check the batch to batch variation. Luteolin and apigenin can be used as one of the appropriate analytical markers for standardization of Amritarishta.
ACKNOWLEDGEMENT: This work is financially supported by the University Grants Commission, New Delhi under Major Research Project [Grant No.42-708/2013(SR)]. Authors are also thankful to Principal D. D. Chougule of Appasaheb Birnale College of Pharmacy Sangli for providing the necessary facilities to carry out this work.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Rastogi S: Building bridges between Ayurveda and modern science. International Journal of Ayurveda Research 2001: 1(1): 41-42.
- Valiathan MS and Thatte U: Ayurveda: The time to experiment. International journal of Ayurveda Research 2001; 1(1): 3-4.
- The Ayurvedic Formulary of India. The Controller of Publications, Delhi, Edition 1st, Part-I, 2000: 6.
- Kumar S, Verma NS, Pande D and Srivastava PS: In-vitro regeneration and screening of berberine in Tinospora cordifolia. Journal of Medicinal and Aromatic Plant Science 2000; 22: 61.
- Biset NG and Nwaiwu J: Quaternary alkaloids of Tinospora species. Planta Medica 1983; 48: 275-9.
- Maurya R, Wazir V, Tyagi A and Kapil RS: Cordifoliosides A and B, two new phenylpropene disaccharides from Tinospora cordifolia possessing immunostimulant activity. Natural Product Letter 1996; 8: 7-10.
- Gangan VD, Pradhan P, Sipahimalani AT and Banerji A. Cordifoliosides A, B, C: Norditerpene furan glycosides from Tinospora cordifolia. Phytochemistry 1994; 37: 781-6.
- Dixit SN and Khosa RL: Chemical investigation of Tinospora cordifolia. Indian Journal of Applied Chemistry 1971; 34: 46-7.
- Namita P and Rawat M: Medicinal plants used as antimicrobial agents: Review. International Journal of Research in Pharmacy 2012; 1(3): 31-40.
- Maurya R and Handa SS: Tinocordifolin, a sesquiterpene from cirdifolia. Phytochemistry 1998; 49: 1343-6.
- Kidwai AR, Salooja KC, Sharma VN and Siddiqui S: Chemical examination of Tinospora cordifolia. Journal of Science and Indian Research 1949; 8: 115-8.
- Stanely M, Prince P and Menon VP: Antioxidant action of Tinospora cordifolia root extract in alloxan diabetic rats. Phytotherapy Research 2001; 15: 213-8.
- Mehrotra R, Katiyar CK and Gupta AP: Hepatoprotective compositions and composition for the treatment of conditions related to hepatitis-B and E infection. US Patent 749296. 2000.
- Prince PS and Menon VP: Antioxidant activity of Tinospora cordifolia roots in experimental diabetes. Journal of Ethnopharmacology 1999; 65: 277-81.
- Ikram M, Khattak SG and Gilani SN: Antipyretic studies on some indigenous Pakistani medicinal plants. Journal of Ethnopharmacology 1987; 19: 185-92.
- Kokate CK, Purohit AP and Gokhale SB: Pharmacognosy, Nirali Prakashan, Edition 21st, 2002: 108-109.
- The Ayurvedic Formulary of India: Govt. of India, Ministry of Health and Family Planning, Department of Health, Monograph, Edition 1st, Part-II, 1978: 7.
How to cite this article:
Wadkar KA, Kondawar MS and Lokapure SG: Standardization of marketed Amritarishta- A herbal formulation. Int J Pharmacognosy 2016; 3(9): 392-99. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(9).392-99.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
392-399
748
1833
English
IJP
K. A. Wadkar *, M. S. Kondawar and S. G. Lokapure
Department of Pharmacognosy, Appasaheb Birnale College of Pharmacy, South Shivaji Nagar, Sangli, Maharashtra, India
wadkarka@gmail.com
08 August 2016
15 September 2016
27 September 2016
10.13040/IJPSR.0975-8232.IJP.3(9).392-99
30 September 2016