SERPYLLIFOLIA LEAVES EXTRACT ON STREPTOZOTOCIN-INDUCED DIABETIC RATS
HTML Full TextSERPYLLIFOLIA LEAVES EXTRACT ON STREPTOZOTOCIN-INDUCED DIABETIC RATS
Shyam Sundar Gupta 1, 2, Lubna Azmi 1, Ila Shukla 1, Lalchand Pal 1, P. K. Mohaptra 2 and Ch. V. Rao * 1
Pharmacognosy and Ethnopharmacology Division 1, CSIR-National Botanical Research Institute, Lucknow - 226 001, Uttar Pradesh, India.
Department of Botany 2, Ravenshaw University, Cuttack - 753003, Orissa, India.
ABSTRACT: The purpose of the study was to determine the leaves of Andrographis serpyllifolia for anti-diabetic and antihyperlipidemic effects in streptozotocin-induced diabetic rats. The aqueous, alcoholic and hydroalcoholic extracts of Andrographis serpyllifolia were tested for toxicity up to 4000 mg kg-1 as per OECD-425 guidelines. Anti-diabetic activity was assessed by oral glucose tolerance test and streptozotocin-induced model. In oral glucose tolerance test alcoholic and hydroalcoholic extracts exhibited greater activity compared to aqueous extract. Hence the alcoholic and hydroalcoholic extracts were further screened by the streptozotocin-induced model at 75, 150 and 300mg kg-1 for 15 days. The alcoholic and hydroalcoholic extracts effectively lowered serum glucose, triglycerides, cholesterol, low-density lipoproteins, very low-density lipoproteins, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, urea, creatinine, and elevated high-density lipoprotein levels, body weight and liver glycogen levels. The alcoholic and hydroalcoholic extracts elicited dose-dependent effect, and the effect produced at the lower dose was not considerable. Hydroalcoholic extract (300 mg kg-1) elicited greater anti-diabetic and antihyperlipidemic activity compared to alcoholic extract (300 mg kg-1).
| Keywords: |
Andrographis serpyllifolia, Acanthaceae, Anti-diabetic, Antihyperlipidemic, Streptozotocin, Oral glucose tolerance test
INTRODUCTION: Diabetes mellitus is characterized by raised blood glucose level which in turn is associated with an increase in the risk for microvascular and macrovascular disease 1. The overall worldwide epidemic of diabetes is anticipated to double by 2025 attacking to about 5% of the adult population 2. In the United States diabetes affects for more than 20 million people. According to the worldwide survey, more than 90% of diabetics belong to type II.
The death rate in people with diabetes is twice than that without diabetes 3. According to the worldwide survey, more than 90% of diabetics belong to type II. Medicinal plants are gaining extensive importance, as they are rich sources for natural anti-diabetic and antihyperlipidemic constituents, which lead to minimal side effects and inexpensive cost. By international plant name index (IPNI), Genus Andrographis comprises of 40 species out of them Andrographis serpyllifolia ((Family: Acanthaceae, Syn. Eriathera serpyllifolia, Andrographis orbiculat) is one of the most important plants. Its common names are Round leaf Kariyat, Aaku chandrika, Hasiru chedi, Hasiru havina gida, Kaasina sara, Kirta, kuram aku, Nela ber. This edible, railing and rooting herb is known for its traditional medicinal properties.
The herb is widely distributed throughout China, Deccan and Carnatic region of south India. Only a very few studies are reported in A. serpyllifolia for the presence of chemical constituents like serpyllin, apigenin 7,4’- dimethyl ether and tectochrysin compounds 4. Although their utility on bioactivity is not very clearly known, Andrographolide (AG) has been reported as one of the potential active components and is found to be responsible for several pharmacological and clinical activities 5. A. serpyllifolia is a prostrate growing herb whose plant extract inhibits the growth of Bacillus subtilis, Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Shigella boydii, Shigella flexneri, Salmonella typhimurium, and Salmonella typhi 6-8 and it is used in the treatment of jaundice, digestive problems, snake bites, fever, cancer, inflammation, wound, 9 hypolipidemia 10. This investigation has been designed to evaluate the antihyperlipidemic and anti-diabetic activity of leaves of Andrographis serpyllifolia in streptozotocin-induced diabetic rats.
MATERIALS AND METHODS:
Plant Materials and Extraction: The leaves of Andrographis serpyllifolia were collected from different localities of Tirupati (Chittoor district, Andhra Pradesh, India) and authenticated by Dr. Madhava Chetty, Associate Professor, Department of Botany, Sri Venkateshwara University, Tirupathi-517 502, Andhra Pradesh, India. A voucher specimen ((Herbarium Accession No. 136) was deposited in the college herbarium. The leaves were shade dried, powdered and moved through sieve no. 60. The powdered material was extracted with 60% ethanol at 60 ºC on a water bath for 3 h using soxhlet extractor and filtered, concentrated on rotavapor (Buchi, USA) at 40 ºC to get an aqueous extract and stored in a desiccator.
Phytochemical Screening and HPTLC Analysis: The ethanolic extracts of Andrographis serpyllifolia were analyzed for the occurrence of bioactive phytoconstituents. HPTLC analysis was processed on pre-activated (100 ºC) Aluchrosep silica gel 60F254 HPTLC plates (S.D. fine-chem Ltd., Mumbai, India) together with quercetin and rutin and HPTLC plates were eluted in solvent system toluene: ethyl acetate: formic acid (5:4:1) for phenols. After development, the plates were dried and densitometrically scanned at wavelength 366 nm (Win Cats software, CAMAG, Switzerland).
Animals: Wistar rats (250-300 g) of either sex were procured from the animal house of the National Laboratory Animal Centre, Lucknow, India. The rats were accommodated in polypropylene cages at standard laboratory conditions of temperature (23 ± 2 ºC), humidity (50-55%), light dark cycles (12 h: 12 h), standard rat pellet feed (Hindustan Lever Ltd.,) and water ad libitum. They were acclimatized for 7 days before the experiment. The experimental works were performed by the guide for the care and use of laboratory animals, as approved and promoted by the Institutional Animal Care Committee, CPCSEA, India (Reg. No. 1732/GO/Re/S/13/ CPCSEA).
Acute Oral Toxicity Study: The acute oral toxicity study of plant extract of Andrographis serpyllifolia was performed on 24 h fasted rats by single dose administration each of 2000 and 4000 mg kg-1 (p.o.) according to the OECD guidelines 425. The toxicity signs and symptoms or any abnormalities associated with the extract administration were observed at 0, 30, 60, 120, 180 and 240 min and then once a day for the next 14 days. The number of rats that survived was recorded at the end of the study period.
Oral glucose tolerance test: This test is employed as a preliminary screening model to evaluate anti-diabetic activity. Overnight fasted rats were allocated to six groups possessing five each.
Group I: Normal control administered 1% sodium carboxymethyl cellulose (CMC).
Group II: Diabetic control administered glucose (3 mg kg-1).
Group III: Standard administered glibenclamide (10 mg kg-1).
Group IV, V and VI: Received aqueous, alcoholic, and hydroalcoholic extracts at a dose of 100 mg kg-1 each to assess the effect of extracts on blood glucose levels. Blood samples were withdrawn from retro-orbital plexus at intervals of 0, 30, 90 and 150 min for glucose estimation.
Induction of Diabetes: Streptozotocin (50 mg kg-1) was freshly formulated in 0.01M ice cold citrate buffer (pH 7.4) and administered intraperitoneal to overnight fasted rats. After 72 h rats possessing blood glucose level greater than 200 mg dl-1, were deemed as diabetic and assigned into the experimental study.
Experimental Design: Rats were allocated into nine groups constituting five each.
Group I: Normal control administered 1% sodium carboxymethyl cellulose (CMC).
Group II: Diabetic control administered STZ (50 mg kg-1, i.p.).
Group III: Standard administered glibenclamide (10 mg kg-1).
Group IV V, and VI: Received alcoholic extract of 75, 150 and 300 mg kg-1.
Group VII, VIII, and IX: Received hydroalcoholic extract of 75, 150 and 300 mg kg-1.
Treatment was continued for 15 days and samples were withdrawn from retro-orbital plexus under mild anesthesia on 1st, 5th, 10th and 15th day. Serum separated by centrifugation (2000 rpm, 20 min) was used for estimating the parameters like glucose, triglycerides, cholesterol, HDL, LDL, VLDL, AST, ALT, ALP, creatinine, and urea.
Statistical Analysis: Results were expressed as mean ± SEM. The significance of data was evaluated by graph pad in stat version 3.2. P value of analysis less than 0.05 was considered to be statistically significant.
RESULTS AND DISCUSSION:
Acute Oral Toxicity Study: The extract of Andrographis serpyllifolia found to be safe up to 4000 mg kg-1 (p.o.) with no toxic signs such as anorexia, depression, lethargy, jaundice, dermatitis and also, no mortality happened throughout the examination during the 14 days observation period. So, the extract was safe for long term administration.
Phytochemical Screening: Both alcoholic and hydroalcoholic extract of Andrographis serpyllifolia showed the presence of alkaloids, glycosides, carbohydrates, flavonoids, saponins, tannins, terpenoids, and polyphenols. Aqueous extract showed the only presence of glycosides, carbohydrates, saponins, and terpenoids. HPTLC determination showed the presence of quercetin and rutin in ethanolic extract of Andrographis serpyllifolia Fig. 1.
FIG. 1: HPTLC FINGER PRINT PROFILE OF ETHANOLIC EXTRACT OF ANDROGRAPHIS SERPYLLIFOLIA LEAVES
Oral Glucose Tolerance Test: All the three extracts (aqueous, alcoholic and hydroalcoholic) have shown a reduction in blood glucose levels when compared to the diabetic control group. Among these, hydroalcoholic extract showed a greater decrease (74.40 ± 1.75) then the alcoholic (80.675 ± 2.95) and aqueous extracts (88.84 ± 2.59) except glibenclamide (73.46 ± 2.90) after 150 min Table 1.
TABLE 1: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON ORAL GLUCOSE TOLERANCE TEST IN NORMAL CONTROL RATS (MEAN ± SEM)
| Groups | Treatment
(mg kg-1 b. wt.) |
Serum glucose (mg dl-1) | |||
| Initial | 30 min | 90 min | 150 min | ||
| I | Normal Control | 67.30±2.79 | 66.48±1.36 | 68.98±2.48 | 68.09±3.06 |
| II | Diabetic Control | 69.24±1.23 | 110.81±3.10** | 117.63±4.20** | 131.43±3.22** |
| III | Glibenclamide(10) | 72.32±2.58 | 84.52±2.81** | 78.04±4.39** | 73.46±2.90** |
| IV | Alcoholic (100) | 70.65±2.54 | 86.16±3.51** | 83.55±2.64** | 80.675±2.95** |
| V | Hydroalcoholic (100) | 71.26±2.32 | 86.51±2.15** | 81.56±3.89** | 74.40±1.75** |
| VI | Aqueous (100) | 75.33±1.25 | 93.40±3.04** | 91.56±5.0** | 88.84±2.59** |
n=5; Group II was compared with Group I. Groups III-VI was compared with Group II. *P<0.05. **P<0.01.
Anti-Diabetic Activity: Administration of the vehicle in the normal control group did not elicit a significant change in blood glucose levels. Administration of glibenclamide has shown significant reduction (74.07%) in glucose levels on the 15th day. ASAE and ASHAE at doses of 75, 100 and 300 mg kg-1 produced reduction of (47.14%, 50.64%, 54.14%, 48.31%, 54.10% and 64.42%). Among the groups, ASHAE at a dose of 300 mg kg-1 elicited greater antihyperglycemic activity (64.42%) Table 2.
TABLE 2: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON SERUM GLUCOSE LEVELS IN STZ INDUCED DIABETIC RATS (MEAN ± SEM)
| Groups | Treatment
(mg kg-1 b. wt.) |
Serum glucose (mg dl-1) | ||||
| Initial | 1st Day | 5th Day | 10th Day | 15th Day | ||
| I | Normal Control | 73.2±2.110 | 68.65±2.80 | 70.00±2.30 | 71.70±2.90 | 76.80±3.20 |
| II | Diabetic Control | 74.0±1.20 | 261.97±5.24** | 271.36±5.20** | 282.60±6.20** | 298.50±6.94** |
| III | Glibenclamide (10) | 269.93±0.95 | 245.39±3.30* | 142.30±4.70** | 89.30±3.60** | 70.00±3.20** (74.07 %) |
| IV | ASAE (75) | 272.45±3.15 | 242.05±2.97** | 197.38±2.83** | 188.78±2.61** | 146.73±4.10** (47.14%) |
| V | ASAE (150) | 273.78±2.23 | 243.63±4.90** | 191.70±3.50** | 181.19±3.20** | 135.14±3.63** (50.64%) |
| VI | ASAE (300) | 273.47±8.15 | 260.47±3.11 | 170.40±4.70** | 129.89±2.40** | 125.40±4.20** (54.14%) |
| VII | ASHAE (75) | 274.97±2.73 | 244.75±3.55** | 193.45±3.97** | 181.57±3.11** | 142.23±3.34** (48.31%) |
| VIII | ASHAE (150) | 271.52±3.63 | 246.15±3.09* | 186.20±3.0** | 171.40±3.90** | 124.10±6.80** (54.10%) |
| IX | ASHAE (300) | 272.37±2.45 | 251.00±3.63 | 168.30±3.50** | 103.90±2.70** | 96.90±1.94** (64.42%) |
n=5; Group II was compared with Group I. Groups III-IX was compared with Group II. *P<0.05, **P<0.01
Antihyperlipidemic Activity: Diabetic control rats have shown an elevation in total cholesterol (241.67 ± 4.5), serum triglycerides (148.50 ± 4.2), low-density lipoproteins (181.70 ± 2.4), very low-density lipoproteins (30.50 ± 0.19) and a decline in HDL (31.50 ± 0.3) compared to the normal group. The Alcoholic and hydroalcoholic extracts of An-drographis serpyllifolia (75, 100 and 300 mg kg-1) showed a decrease in triglycerides, total cholesterol, low-density lipoproteins, very low-density lipoproteins and an increase in high density lipoproteins when compared to the diabetic control group. At lower dose, ASAE and ASHAE produced very less effect. ASHAE at a dose of 300 mg kg-1 exhibited greater antihyperlipidemic activity Table 3.
Effect of Extract on AST, ALT & ALP Levels: Increased AST, ALT & ALP levels were found in diabetic control group (44.77%, 38.33%, and 30.70%) when compared to normal control group. The alcoholic and hydroalcoholic extract treated groups (75, 100 and 300 mg kg-1) have shown decrease in the AST levels (12.47%, 22.66%, 28.30%, 14.25%, 17.58%, 35.43%); ALT levels (4.88%, 25.43%, 28.48%, 5.23%, 17.44%, 34.18%); and ALP levels (15.39%, 25.11%, 30.69%, 9.76%, 16.45%, 27.15%) Table 4.
Effect of Extract on Creatinine and Urea Levels: Diabetic control group have shown increase in creatinine (81.79%) and urea levels (48.87%). Glibenclamide, alcoholic and hydroalcoholic extract treated group have shown decrease in creatinine levels (82.08%, 71.68%, 74.86%, 77.17%, 66.47%, 75.43%, 92.49%) and urea levels (49.13%, 20.68%, 31.18%, 42.13%, 23.72%, 36.38%, 47.95%) when compared to diabetic control group Table 5.
TABLE 3: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON SERUM TC, TG, HDL, LDL, VLDL LEVELS ON 15TH DAY
| Groups | Treatment (mg kg-1 b. wt.) | TC (mg dl-1) | TG (mg dl-1) | HDL (mg dl-1) | LDL (mg dl-1) | VLDL (mg dl-1) |
| I | Normal Control | 144.10±3.1 | 75.50±0.8 | 36.95±1.2 | 93.60±1.3 | 15.90±0.11 |
| II | Diabetic Control | 241.67±4.5**
(40.37 %) |
148.50±4.2**
(49.16%) |
31.50±0.3**
(14.75%) |
181.70±2.4**
(47.94%) |
30.50±0.19**
(47.87%) |
| III | Glibenclamide (10) | 146.62±3.0**
(39.33%) |
84.11±1.9**
(43.36%) |
36.70±1.0**
(16.89%) |
94.30±1.7**
(48.10%) |
17.70±0.08**
(41.97%) |
| IV | ASAE (75) | 188.31±4.1**
(22.08%) |
113.15±3.0**
(23.80%) |
31.85±0.27
(4.24%) |
135.53±2.56
(25.41%) |
23.43±0.47**
(23.18%) |
| V | ASAE (150) | 179.89±3.9**
(31.37%) |
94.14±2.3**
(36.61%) |
33.10±0.33
(4.98%) |
128.50±2.0**
(29.28%) |
19.60±0.25**
(35.74%) |
| VI | ASAE (300) | 165.86±3.3**
(20.20%) |
89.80±2.2**
(39.53%) |
34.20±0.4*
(8.52%) |
114.45±1.6**
(37.01%) |
18.70±0.27**
(38.69%) |
| VII | ASHAE (75)
|
179.17±3.9**
(25.86%) |
109.23±3.1**
(26.44%) |
33.41±0.35*
(9.54%) |
123.91±2.61
(31.81%) |
21.85±0.77**
(28.36%) |
| VIII | ASHAE (150) | 160.80±4.0**
(33.46%) |
100.00±2.2**
(32.66%) |
36.20±0.5**
(18.69%) |
122.53±2.3**
(32.56%) |
19.04±0.29**
(37.57%) |
| IX | ASHAE (300) | 150.53±3.3**
(37.71%) |
82.48±2.4**
(44.46%) |
37.80±0.53*
(23.93%) |
96.80±1.7**
(46.73%) |
17.28±0.25**
(43.34%) |
n=5; Group II was compared with Group I. Groups III-IX was compared with Group II. *P<0.05, **P<0.01
TABLE 4: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON AST, ALT, AND ALP
| Groups | Treatment (mg kg-1 b. wt.) | Serum (U/L) | ||
| AST | ALT | ALP | ||
| I | Normal Control | 16.43±0.75 | 14.14±1.19 | 37.29±1.50 |
| II | Diabetic Control | 29.75±2.00** (44.77%) | 22.93±0.90** (38.33%) | 53.81±2.58 ** (30.70%) |
| III | Glibenclamide (10) | 17.38±1.80** (41.58%) | 15.63±0.89** (31.84%) | 38.40±2.70** (28.64%) |
| IV | ASAE (75) | 26.04±1.53 (12.47%) | 21.81±1.70 (4.88%) | 45.53±2.31 (15.39%) |
| V | ASAE (150) | 23.01±1.50 (22.66%) | 17.10±0.83* (25.43%) | 40.30±2.20** (25.11%) |
| VI | ASAE (300) | 21.33±1.70** (28.30%) | 16.40±1.20** (28.48%) | 37.30±1.80** (30.69%) |
| VII | ASHAE (75) | 25.51±1.90 (14.25%) | 21.73±0.96 (5.23%) | 48.56±3.15 (9.76%) |
| VIII | ASHAE (150) | 24.52±1.97 (17.58%) | 18.93±0.20* (17.44% ) | 44.96±1.02* (16.45%) |
| IX | ASHAE (300) | 19.21±0.90** (35.43%) | 15.10±0.70** (34.18%) | 39.20±1.90** (27.15%) |
n=5; Group II was compared with Group I. Groups III-IX was compared with Group II. *P<0.05, **P<0.01
TABLE 5: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON SERUM CREATININE AND SERUM UREA LEVELS ON 15TH DAY
| Groups | Treatment (mg kg-1 b. wt.) | Serum Creatinine (mmol L-1) | Serum Urea (mg dl-1) |
| I | Normal Control | 0.63±0.13 | 32.44±2.40 |
| II | Diabetic Control | 3.46±1.07** (81.79%) | 63.44±4.16** (48.87%) |
| III | Glibenclamide (10) | 0.62±0.13** (82.08%) | 32.27±2.33** (49.13%) |
| IV | ASAE (75) | 0.98±0.14** (71.68%) | 50.32±1.62* (20.68%) |
| V | ASAE (150) | 0.87±0.12* (74.86%) | 43.66±2.75* (31.18%) |
| VI | ASAE (300) | 0.79±0.13* (77.17%) | 36.71±2.18** (42.13%) |
| VII | ASHAE (75) | 1.16±0.13 (66.47%) | 48.39±2.63** (23.72%) |
| VIII | ASHAE (150) | 0.85±0.15* (75.43%) | 40.36±3.03** (36.38%) |
| IX | ASHAE (300) | 0.26±0.14** (92.49%) | 33.02±2.26** (47.95%) |
n=5; Group II was compared with Group I. Groups III-IX were compared with Group II. *P<0.05, **P<0.01
Effect of Extract on Body Weight: Normal rats have shown an increase in body weight on 15th day (9.36%).
A significant decrease in the body weight (6.94%) was observed in the diabetic control group. Alcoholic and hydroalcoholic extract treated groups (150 and 300 mg kg-1) have shown an increase in the body weight (8.73%, 8.00%, 7.52%, and 9.12%) Table 6.
Effect of Extract on Liver Glycogen Levels: Diabetic control group manifested decline (47.94%) in liver glycogen levels on the 15th day when compared to normal control group. Glibenclamide, alcoholic and hydroalcoholic extract treated groups (75,150 and 300 mg kg-1) have shown increase in liver glycogen levels (47.94%, 83.76%, 39.08%, 59.84%, 75.00%, 45.45%, 60.58%, 81.22%) when compared to diabetic control group Table 7.
TABLE 6: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON ANIMAL BODY WEIGHT (MEAN ± SEM)
| Groups | Treatment
(mg kg-1 b. wt.) |
Animal body weight (g) | ||||
| Initial | 5th Day | 10th Day | 15th Day | |||
| I | Normal Control | 276.52±3.34 | 281.40±2.21 | 290.72±5.38 | 302.40±6.69 (9.36%) | |
| II | Diabetic Control | 283.46±5.42 | 277.20±1.27 | 269.09±3.64* | 263.80±4.45** (6.94%) | |
| III | Glibenclamide (10) | 285.64±3.11 | 295.40±3.19* | 299.26±4.80** | 307.20±3.19** (7.55%) | |
| IV | ASAE (75) | 289.45±4.15 | 301.50±3.10** | 297.40±3.10** | 311.50±3.15** (7.62%) | |
| V | ASAE (150) | 286.22±3.12 | 295.00±4.92* | 302.00±5.40* | 311.20±4.92** (8.73%) | |
| VI | ASAE (300) | 288.71±5.50 | 299.20±3.30** | 306.20±3.49** | 311.83±5.59** (8.00%) | |
| VII | ASHAE (75) | 289.67±5.30 | 305.65±3.15** | 310.56±3.11** | 315.32±4.15** (8.85%) | |
| VIII | ASHAE (150) | 291.68±4.86 | 304.20±3.73** | 308.00±3.83** | 313.60±6.96** (7.52%) | |
| IX | ASHAE (300) | 286.28±3.85 | 295.97±4.34* | 303.94±5.97** | 312.40±5.10** (9.12%) | |
n=5; Group II was compared with Group I. Groups III-IX was compared with Group II. *P<0.05, **P<0.01
TABLE 7: EFFECT OF ANDROGRAPHIS SERPYLLIFOLIA ON LIVER GLYCOGEN LEVELS ON 15th DAY (MEAN ± SEM)
| Groups | Treatment (mg kg-1 b. wt.) | Liver glycogen levels (mg gm-1of wet tissue) |
| I | Normal Control | 51.56±2.21 |
| II | Diabetic Control | 26.84±1.31** (47.94%) |
| III | Glibenclamide (10) | 49.32±5.15** (83.76%) |
| IV | ASAE (75) | 37.33±1.20 (39.08%) |
| V | ASAE (150) | 42.90±3.35* (59.84%) |
| VI | ASAE (300) | 46.97±4.20** (75.00%) |
| VII | ASHAE (75) | 39.04±1.56 (45.45%) |
| VIII | ASHAE (150) | 43.10±2.00* (60.58%) |
| IX | ASHAE (300) | 48.64±3.80** (81.22%) |
n=5; Group II was compared with Group I. Groups III-IX were compared with Group II. *P<0.05, **P<0.01
In oral glucose tolerance test aqueous, alcoholic and hydroalcoholic extracts improved glucose tolerance at 90 min and 150 min suggesting peripheral utilization of glucose. ASHAE was more potent when compared to ASAE, and aqueous extract (ASAqE) exhibited decreased glucose tolerance effect at a dose of 100 mg kg-1. Therefore, aqueous extract (ASAqE) is not evaluated for further investigation. Streptozotocin is extensively employed to screen natural products for their insulinomimetic, insulinotropic and other antihyperglycemic activities 11 - streptozotocin-induced hyperglycemia by cytotoxic action on pancreatic beta cells 12.
ASAE and ASHAE elicited antihyperglycemic activity in a dose-dependent manner. ASAE and ASHAE produced the lesser anti-diabetic effect at a lower dose (75 mg kg-1). At a higher dose (300 mg kg-1), ASAE and ASHAE produced greater anti-diabetic effect. Anti-diabetic effect of ASHAE was comparatively more than ASAE at the dose of 300 mg kg-1. Both extracts produced less anti-diabetic activity than glibenclamide and were unable to restore the glucose level to the baseline value.
This indicates Andrographis serpyllifolia should be employed with alternatives like diet or hypoglycemic agents for diabetes control. Glibenclamide, a sulfonylurea derivative, elicited anti-diabetic activity by stimulating β-cells of the pancreas. Mechanisms related to in vitro studies were crucial to assess the mode of action. Hyperglycemia produced is accompanied by an increase in serum triglycerides, total cholesterol, low-density lipoproteins and the decrease in high-density lipoproteins.
Administration of ASHAE and ASAE normalized blood glucose levels along with restoration of serum triglycerides and cholesterol levels. Hence both extracts were considered to possess anti-diabetic activity and antihyperlipidemic activity. Anti-diabetic activity elicited might be related to the existence of active alkaloidal constituents; aporphine and berberine 9. Aporphine produced anti-diabetic effect by inhibiting intestinal glucose uptake 13.
The scientific investigation revealed that berberine modulated glucose and lipid metabolism through a multiple pathway methodologies of AMP-activated protein kinase (AMPK); P38 MAPK-glut4, JNK pathway and PPAR α-pathway 14. Berberine also elevated insulin sensitivity in insulin-resistant rat models 15.
In the diabetic control group, the typical loss of body weight is probably because of impairment in insulin action in the conversion of glucose into glycogen and catabolism of fats 16. Treatment with extracts (ASAE & ASHAE) substantially prevented loss of body weight due to the reversal of gluconeogenesis or release of insulin. Extracts treated groups increased glycogen content might be due to decreased endogenous glucose output from the liver. Derangements in metabolic processes during the progression are frequently associated with alteration in serum enzyme activities. Hence the estimation of serum enzymes has become prominent in diabetes. In diabetes, the increased amino acids; which tend to be active in the absence of insulin were responsible for the increased formation of gluconeogenesis and ketogenesis 17. ASAE & ASHAE have shown a reduction in the level of AST, ALT, and ALP which might be by inhibiting gluconeogenesis process.
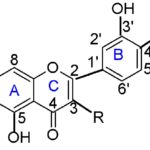
Phytochemical and HPTLC analysis of the extract of Andrographis serpyllifolia was carried out to standardize the extract, using quercetin and rutin as marker components. Quercetin has been reported to prevent and protect against streptozotocin-induced oxidative stress and β-cell damage in rat pancreas 18. Rutin also exhibited protection against streptozotocin-induced diabetes by inhibiting inflammatory cytokines and improving antioxidant 19. Thus, the significant anti-diabetic and antihyperlipidemic activity of the leaves of Andrographis serpyllifolia may be attributed partially due to the presence of quercetin and rutin, although other compounds, as yet unidentified, probably also contribute Fig. 2.
FIG. 2: CHEMICAL STRUCTURE OF FLAVONOIDS (Quercetin: R-OH, Rutin: R-Ogl, gl: Glycosyl)
CONCLUSION: Results of the experimental study reveal that alcoholic and hydroalcoholic extracts of Andrographis serpyllifolia possess promising anti-diabetic and antihyperlipidemic activity in a dose-dependent manner. The hydroalcoholic extract of Andrographis serpyllifolia manifested enhanced anti-diabetic activity as compared to its alcoholic extract. However, investigations are essential to isolate bioactive principles and to illuminate the accurate anti-diabetic mechanism of action.
ACKNOWLEDGEMENT: Authors are thankful to the Director, CSIR-National Botanical Research Institute, Lucknow, India for providing necessary facilities. SSG and LA are grateful to the Department of Science & Technology (DST), Ministry of Science and Technology, New Delhi for providing DST-INSPIRE fellowship.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Krauss RM: Lipids and lipoproteins in patients with type 2 diabetes. Diabetes Care 2004; 27: 1496-1504.
- King H, Aubert RE and Herman WH: Global burden of diabetes. 1995-2025: prevalence, numerical estimates, and projections. Diabetes Care 1998; 21: 1414-1421.
- Harrigan RA, Nathan MS and Beatie P: Oral agents for the treatment of type 2 diabetes mellitus: pharmacology, toxicity and treatment. Ann Emerg Med 2001; 38: 68-78.
- Damu AG, Jayaprakasam B, Gunasekar D, Blond A and Bodo B: Two acylated flavone glucosides from Andrographis serpyllifolia. Phytochem 1999; 52: 147-151.
- Kapil A, Koul IB, Banerjee SK and Gupta BD: Antihepatotoxic effects of major diterpenoid constituents of Andrographis paniculata. Biochem Pharmacol 1993; 46: 182-185.
- Gupta SS, Sharma J, Kumar GR, Pandey G, Mohapatra PK, Rawat AKS and Rao ChV: Effect of Andrographis serpyllifolia leaves extract on experimentally induced typhoid using Salmonella typhi. British J Pharm Res 2014; 3: 230-239.
- Doublet B, Lailler R, Meunier D, Brisabois A, Boyd D, Mulvey MR, Chaslus-Dancla E and Cloeckaert A: Variant Salmonella genomic island 1 antibiotic gene resistance cluster in Salmonella enteric Albany. Emerg Infect Dis 2003; 9: 585-591.
- Pang T, Bhutta ZA, Finlay BB and Altwegg M: Typhoid fever and other salmonellosis: a continuing challenge. Trends Microbiol 1995; 3: 253-255.
- Sekhar J, Penchala PG, Sudarsanam G and Prasad GP: Ethnic information on treatments for snake bites in Kadapa district of Andhra Pradesh. Life Sci leafl 2011; 12: 368-75.
- Chelluboina B and Manga K: Hypolipidemic activity of Andrographis serpyllifolia. J Pharm Res 2010; 3: 769-770.
- Bates SH, Jones RB and Bailey CJ: Insulin-like effect of pinnitol. J Pharmacol 2000; 130: 1944-1948.
- Ho E, Chen G and Bray TM: Alpha-phenyl-tert-butylnitrone (PBN) inhibits NFkappa B activation offering protection against chemically induced diabetes. Free Rad Biol Med 2000; 28: 604-614.
- Lin CJ, Chen CH, Liu FW, Kang JJ, Chen CK, Lee SL and Lee SS: Inhibition of intestinal glucose uptake by aporphines and secoaporphines. Life Sci 2006; 79: 144-153.
- Zhang Q, Xiao X, Feng K, Wang T, Li W, Yuan T, Sun X, Sun Q, Xiang H and Wang H: Beriberine moderates glucose and lipid metabolism through multipath way mechanism. Evid Based Complement Alternat Med 2011; 2011: 1-10.
- Gao CR, Zhang JQ and Huang QL: Experimental Study on the Insulin-Sensitivity-Increasing Effect of Berberine in Insulin Resistant Rat Models. Chin J Integr Med 1997; 3: 282-285.
- Gillespie KM: Type I diabetes: Pathogenesis and prevention. CMAJ 2006; 175: 165-170.
- Mukherjee K, Ghosh NC and Datta T: Coccinia indica as potential hypoglycemic agent. Indian J Exp Biol 1972; 10: 347-349.
- Coskun O, Kanter M, Korkmaz A and Oter S: Quercetin a flavonoid antioxidant, prevents and protects Streptozotocin-induced oxidative stress and beta-cell damage in rat pancreas. Pharmacol Res 2005; 51: 117-123.
- Niture NT, Ansari AA and Naik SR: Anti-hyperglycemic activity of rutin in Streptozotocin-induced diabetic rats: An effect mediated through cytokines, antioxidant and lipid biomarkers. Indian J Exp Biol 2014; 52: 720-727.
How to cite this article:
Gupta SS, Azmi L, Shukla I, Pal L, Mohaptra PK and Rao CV: Serpyllifolia leaves extract on streptozotocin-induced diabetic rats. Int J Pharmacognosy 2016; 3(8): 346-53. doi: 10.13040/IJPSR.0975-8232.3(8).346-53.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
346-353
660
1526
English
IJP
S. S. Gupta, L. Azmi, I. Shukla, L. Pal, P. K. Mohaptra and C. V. Rao *
Pharmacognosy and Ethnopharmacology Division, CSIR-National Botanical Research Institute, Lucknow, Uttar Pradesh, India.
chvrao72@yahoo.com
12 July 2016
19 August 2016
19 August 2016
10.13040/IJPSR.0975-8232.IJP.3(8).346-53
31 August 2016