ROLE OF PLANT-BASED ACTIVE COMPOUNDS IN INFLAMMATORY RESPONSES OF CANCER
HTML Full TextROLE OF PLANT-BASED ACTIVE COMPOUNDS IN INFLAMMATORY RESPONSES OF CANCER
Namrata Kahar * and Rohit Seth
Department of Zoology, Guru Ghasidas Vishwavidyalaya, Koni Bilaspur - 495551, Chhattisgarh, India.
ABSTRACT: Cancer is a non-communicable life-threatening disease and a majority of cancers are caused due to inflammatory responses and association with precursor lesions termed as proliferative inflammatory factors. Inflammatory response arises because of dysfunction of anti-oxidative proteins due to DNA damage and may increase oxidative stress. This review mainly focuses on the inflammatory factors which trigger specific pathways inside the cell and create a tumour microenvironment that subsequently leads to cancer induction and progression. We are providing information about the role of various plant derived active compound and their role in treatment of cancer via induction of chronic inflammation and altering the level of reactive oxygen species (ROS). In this review, we will also document currently available treatments and their possible therapeutic implications in ameliorating the ailments that arise due to cancer. In the later section of this review, we have added notes on the challenges that need to be addressed to improve the efficiency of drugs derived from medicinal plants. It appears that herbal therapies are effective for targeting the cellular compartment and effectively correcting the tumour microenvironment and plays a major role inmodulating the inflammatory responses.
| Keywords: |
Inflammation, Cytokines, Tumour microenvironment, Cancer, Herbal therapy, Inflammatory factor
INTRODUCTION: Cancer is one of the leading causes of death and poses a threat to mankind 1. It is the second leading cause of death after cardio-vascular disease 2. The majority of cancers present some form of inflammation in the beginning that progress and leads to the formation of a tumor. An inflammatory response is the part of the immune system which aids in the survival and proliferation of cancer cells. The cell and inflammatory mediators are the main creators for tumour micro-environment which regulates the pathogenesis of tumour generating cells (tumour growth).
Researchers have explored the role of inflammation, the immune tumour microenviron-ment and potential immune biomarkers in the progression of cancer where some inflammation is relative to putative cancer precursor lesions, termed as proliferative inflammatory atrophy 3.
It is generated inside the cell because of the dysfunction of anti-oxidative proteins or because of DNA damage that might increase oxidative stress. Together these factors might serve as a drive to carcinogenesis via oxidative stress mechanisms and generation of ROS species induced mutagenesis 4. This sort of damage in biomacromolecules may form a ferocious cycle of oxidative stress, resulting in cancer development 5. Where cyclooxygenase has an important role in the induction of inflammation 6-8. Furthermore, genomic instability, an appropriate phenomenon and alternation in the epigenetic characters such as DNA methylation or micro RNA dysregulation play a vital role in carcinogenesis, especially in inflammation-related cancer. DNA methylation and his tone post-translation modifications are the most common forms of Epigenetic modifications 9, 10. Oncogenic changes in cells lead to induction of inflammatory pathways in pre-malignant and malignant cells, which promotes chronic inflammation caused by infections, exposure to irritants, or some other factors, including suffering from autoimmune disease in almost 20% of human cancers 11. In this review, we will address some of the plant active compounds that can have an effect on inflammation and induce the pathway of the inflammatory response in cancer cells. These inflammatory mediators generated by cancer cell may serve as potential targets for cancer therapy. Furthermore, the mechanism by which inflammation is dysregulated in cancer also provides tumor-promoting signals that may offer new therapeutic opportunities in cancer therapy.
Inflammation and Cancer: Inflammatory responses and cancer generally go hand in hand. Inflammation is a self-activated and controlled mechanism of the immune system to eliminate infections from the body and repairing itself. While dysregulation induces inflammatory disorder it is important to understand inflammation progression from early stage to late stage of cancer.
Here we have elucidated the different inflammation stages in terms of cancer generation. As inflammation progression takes place, and is exemplified by cell injury (bacterial infection, obesity or smoking etc) it induces the activation of tumour micro-environment (TME) which maintain early stage of tumour generation.
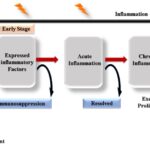
Enhanced inflammatory response causes immuno-suppression Certainly the anti-tumour stimulation therapy is beneficial for immunosuppression in an early stage. Several studies have shown that chronic inflammation is observed during the last stage of the excessive proliferation of cancer cells in patients. Different stages of inflammation during the progression of cancer is illustrated in the Fig. 1.
FIG. 1: DIFFERENT STAGES OF THE INFLAMMATION. Any type of injury or cell damage may cause inflammation. These are maintained by the tumour microenvironment, and infiltrative cells play an important role in the early to the late stage of inflammation progression followed by cancer generation. The persistent excessive proliferation causes acute inflammation drives the cell transformation in the late stage if not resolved. At the same time, chronic inflammation accelerates tumorigenesis followed by tumour metastasis. Where chronic inflammation contributes to all forms of human cancer.
Inflammation Inducing Factors:
Do Inflammatory Factors Support or Limit Tumour Growth: As an anti-inflammatory mediator, prostaglandin plays a crucial role in inflammation, whereas cytokines are involved in inflammation. An up-regulated prostaglandin response has been observed in colon carcinoma 12. Although, there are five primary prostaglandins; prostaglandins D2, prostaglandins E2, prostaglandins F2a, prostaglandins 12, and thromboxane A2. They are biosynthesized by arachidonic acid metabolism 13-18. After the subsequent conversion of the proteinoids by specific synthases or isomers, it develops the biological activities to regulate immune function like inflammation 19. Cytokines are important mediators of communication between cells in the inflammatory tumour microenvironment. In the neoplastic cells generally, over-expression of pro-inflammatory mediators, including proteases, eicosanoids, cytokines, and chemokinesis observed 20.
Several cytokines have been linked with both human and experimental cancer, which can either stimulate or inhibit tumour development such as macrophage migratory inhibitory factor (MIF), Tumor necrosis factor (TNF-α), Interleukins (IL)-6, IL-17, IL-12, IL-23, IL-10 and transforming growth factor (TGF- β). MIF is a major cytokine in many cancers, and there is evidence that this cytokine is produced by both malignant cell and infiltrative leukocytes. Inflammatory processes can promote, or maybe even initiate, or cause malignant disease 21. While the tumor-suppressor proteins can function as a positive regulator, it stimulates the production of inflammatory mediators. TNF-α signalling can promote cell survival, invasion, and angiogenesis 22.
Its production is associated with increased risk of multiple myeloma, bladder cancer, hepatocellular carcinoma, gastric cancer, and breast cancer, as well as poor prognosis in various hematological malignancies 23. Tumour-promoting inflammation is considered one of the permissive characteristics of cancer development. Following the initial success of immune therapies that modulate the adaptive immune system, based on preclinical and epidemiological data, it is reported that inflammation and innate immunity are important targets in patients with cancer 24. Epidemiological studies have suggested some beneficial effects of anti-inflammatory medicines such as aspirin or non-steroidal drugs in protecting against several forms of cancer, especially that of the colon 25. Aspirin is used as a powerful chemopreventive drug 26-28, as it blocks the prostaglandin production as well as induces the production of endogenous anti-inflammatory mediators 29.
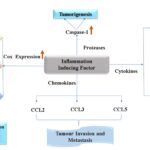
FIG. 2: POTENTIAL INFLAMMATORY FACTORS AND THEIR ROLES IN CANCER GENERATION. Overexpression of different inflammatory factor COX proteins 47, 48, 49, protease, chemokines, and cytokines are responsible for tumour cell progression, tumorigenesis, metastasis and angiogenesis, etc. Although these inflammatory factors regulate the immune system. TNF-α 23, IL-6, IL-17, IL-12, IL-23, IL-10 these are cytokines that are responsible for the cancer cell proliferation induces tumour cell invasion, metastasis, and angiogenesis.
In every aspect of carcinogenesis, tumour microenvironment contributes to the most promising target for cancer therapy. Gao F. et al. defined tumour microenvironment and also discussed their role in the initiation and progression of cancer 30. As inflammation is the first line of defense in the immune system thus, not only can inflammation cause cancer, but cancer also leads to inflammation. Inflammatory factors are important for tumour growth Fig. 2. Their role depends on the nature of the tumour and inflammatory cell interaction 20, which may produce growth factors for tumour proliferation which in turn creates a tumour microenvironment and provide factors for antitumor immune responses. There are various types of immune cells in filtering cancer and are amajor source of inflammation. This includes natural killer cells, Cytotoxic CD8+ Cells (CTL), IL-1-producing T helper cells (TH1), TH17, macrophages, monocytes, dendritic cells produce cytokines like IL-1, 6, 17, 23, and TNF. Pro-tumorigenic cytokines include interleukins IL-6, 11, 21, 22 that activates the signal transducer and activator of transcription 3 (STAT3), TNFα, IL-1, 18 that activates Nuclear Factor (NF-κB) and the IL-23 to 17 that activates both STAT3 and NF-κB in tumour cells 31, 32. NF-κB and STAT3 are essential for inflammation-promoted cancer development 33-36. Tumour environment is maintained by the infiltrating immune cells and their secreting inflammatory factors. The production of cytokines, chemokines, and extracellular enzymes, infiltrating immune cells may serve as a tumour promoter by supporting the proliferation of tumour cells and inhibiting programmed cell death Fig. 3. Tumour infiltrating Macrophages and neutrophils produce IL-6, IL-23, IL-1, and TNFα and activation of Th17 lymphocytes for the production of IL-17, IL-21, IL-22. In contrast, the dendritic cells facilitate T-cell mediated immunity against cancer by IL-12 production and antigen presentation. Th-1 cells further activate the natural killer cell (NK) and cytotoxic T- lymphocytes (CTLs) by secreting IFN-γ. NK cells and CTLs target cancer cells for destruction. Moreover, regulatory T cells (Treg) inhibit pro and anti-cancer immunity by producing IL-10 and TGF-β through the contact depend and independent mechanism 37. Targeting any of these cellular components of the tumour has emerged as a promising approach and constitutes the basis for anti-angiogenic and anti-inflammatory therapies applied to various types of human cancer 38, 39.
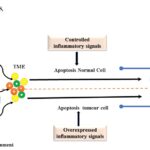
FIG. 3: THE RELATION BETWEEN INFLAMMATION-INDUCED APOPTOSIS AND TUMOUR GROWTH. Reactive oxygen species (ROS).chemotherapeutic agents and other drugs used in cancer therapy can trigger the inflammation signals inside the cell and genesis of tumour microenvironment where the controlled or regulated inflammation induces apoptosis however in the tumour cells apoptosis is inhibited by overexpression of inflammatory signals leading to cell survival.
The Role of Cyclooxygenase In Inflammation: One of the foremost important events of inflammation is the induction of its form (cyclooxygenase, a rate-limiting enzyme), by several stimuli associated with cell activation and inflammation made applicability of finding disease. COX-1 and COX-2 are the two isoforms of cyclooxygenase responsible for the generation of prostaglandins. Although COX-1 is constitutively expressed in all cell types relentlessly, the expression of that protein is substantially higher in tumour than in any of the normal tissues 40-42, and COX-2 has been demonstrated to be active only under the influence of certain stimulating growth factors and cytokines 43- 45. Immunoblot analysis revealed an extremely high level of COX-2 protein in two tumour samples of colon tumour and breast tumour 46. Some experiments suggested that the overexpression of COX-2 may not be unique to colon cancer and may be a feature common to other epithelial tumours 46. For instance, the inducible form of COX-2 is overexpressed in colon tumour tissues 6-8 and very often overexpressed in early, malignant tissues and several cancer cells including lung, liver, skin, breast, and colon 47-49. Similarly, prostaglandin is produced more in the human malignant breast cancer tissue than the normal breast tissues 50. The activity of COX-2-PGE2 signal pathway can suppress Dendritic cells, NKC, T cells, type-1 immunity excluding type-2 immunity which promote tumor immune evasion. Aromatase which is an important enzyme involved in estrogen biosynthesis is another major proliferator of carcinomas in particular the breast cancer.
Reactive Oxygen Species (ROS) and Inflammation: By product of oxygen consumption and cellular metabolism are generally considered as reactive oxygen species (ROS) which are generated by the partial reduction of molecular oxygen 51, 52. They are continuously generated inside living organisms by different mechanisms 53. ROS has long been associated with cancer, where different types of tumour cells have shown to produce an elevated level of ROS compared to their normal counterparts. Elevated levels of ROS are thought to be oncogenic, causing damage to DNA, proteins, and lipids, thereby promoting genetic instability and tumorigenesis 54-58. Oxidative stress caused by chemotherapy is added to the oxidative stress that is inherent in the tumour cell. Tumour cells have an altered redox balance when compared with their normal counterparts 59. However, investigation and development of novel phototherapeutic agents to detect anti-tumour and free radical scavenging activities become an unmet need. Hence, identifying altered ROS activity as a potential target for cancer therapies 59. See enclosed Table 1 for details of plants with their active ingredients and ROS activity.
TABLE 1: DETAILS OF PLANT WITH THEIR ACTIVE PRINCIPLES AND THEIR EFFECTIVE TARGETS
| S. no. | Common Name | Botanical Name | Active principles | Effect | ROS Activity | Cancer | Refe |
| 1. | Giant Voodoo Lily | Sauromatumgiganteum (Engl.) Cusimano & Hett (Typhoniumgiganteum)
(Arecaceae). |
Beta-sitosterol, Campesterol, n-hexadecenoic acid, octadecanoic acid | Induces apoptosis | ROS level increased | Gastric carcinoma and liver cancer, hepatocellular carcinoma. | 93,94, 95, 96, 105 |
| 2. | Giant Voodoo Lily / Typhonium Tuber | RhizomaTyphonii
(Arecaceae). |
Beta-sitosterol, Campesterol, n-hexadecenoic acid, octadecanoic acid | Induces apoptosis | ROS level increased | hepatocellular carcinoma. | 96, 105 |
| 3. | Garden Stock/ Bromton Stock | Mathiolaincana
(Bracaecaeae) |
Luteolin (LUT) | Induce pro apoptosis
Inflammatory markers expression |
Antioxidant enzyme activity increases | Renal cell | 98, 135 |
| 4. | Gukulakanta/
Tamilakanha |
Hygrophila spinosa
(Acanthaceae). |
Apigenin and LUT | Induced Cytotoxicity | Antioxidant activity | ovarian and breast cancer cell (MCF-7, MDA MB-231, SKOV-3 cell line) | 99 |
| 5. | Star anise
|
Illicium verum
(Schisandraceae)
|
Induces apoptosis /Downregulate antiapoptotic genes | _ | 100 | ||
| 6. | Mulethi (Root)
|
Glycyrrhiza glabra
(Fabaceae)
|
Induces apoptosis/ Downregulate antiapoptotic genes | _ | 100 | ||
| 7. | Leenseed or Flax | Linumusita tissimum
(Linaceae) |
Induces apoptosis / Downregulate antiapoptotic genes | _ | 100 | ||
| 8. | Glossy Buckthron | Rhamnus Frangula
(Ramnaceae) |
Downregulate antiapoptotic genes | _ | 100 | ||
| 9. | Voodoo lily | Sauromatum venosum
(Araceae) |
Lectin | Induces apoptosis | _ | Murine cancer cell lines | 104 |
| 10. | Kariyat, Creat | Andrographis paniculata
(Acanthaceae) |
Andrographolide | Inhibit the proliferation of Tumour cells, enhances
TNF- α and induces Apoptosis, increase proliferation of IL-2 |
_ | Colon cancer (HT-29), B16F0 melanoma | 106, 107,108 |
| 11. | Turmeric | Curcuma longa
(Zingiberaceae) |
Curcumin | Regulates inflammatorycytokines, Induces apoptosis,
Inhibits NF- kappa β |
Regulates Oxidative stress and activates oxidative enzymes | Carcinoma cancer cell, leukaemia, colon cancer cell | 89, 136 |
| 12. | Kokam | Garcinia indica
(Guttiferae) |
Garcinol, Isgarcinol and
Cyanidin-3- glucoside |
Induces apoptosis | Inhibit NO radical generation | Human leukaemia (HL-60) | 124, 125
|
| 13. | Rodent tuber | Typhoniumflagelliform
(Araceae) |
Pheophorbide-a, and methy l- pyropheophor- bide-A | Induces apoptosis,
Antiproliferative activity |
Antioxidant activity | Lungs cancer (NCI-H23), Breast cancer (HS578T) | 126 |
| 14 | Creeping saxifrage, strawberry saxifrage, creeping rockfoil,
mother of thousands, roving sailo |
Saxifragasto lonifera
(Saxifragaceae) |
Quercetin | Induce apoptosis DNA fragmentation | _ | BGC-823 cells. | 127 |
| 15. | Basil, Sweet basil | Ocimumbasilicum
(Lamiaceae) |
Ursolic acid | Anti-inflammatory, anticancer activity | Antioxidant scavenging activity | Colorectal Adenocarcinoma cells | 128, 129 |
| 16.. | KutkiKardi | Picrorhizakurroa
(Scrophulariacea) |
Apocynin (Iridoid glycoside) | Induces apoptosis, induce cytotoxicity | Inhibit lipid peroxidation,
Antioxidant scavenging activity |
Human Breast Carcinoma (MDA-MB-435S), Human Hepatocellular Carcinoma (Hep3B), Prostate cancer cell lines (PC-3) | 130 |
| 17. | Anemone clematis, Indian virgin’s bower garol, Geor Bel, Kanguli, Kaunie- Bali | Clematis montana (Ranunculaceae) | Lectin | Induces apoptosis | Antioxidant scavenging activity | L929, HeLa, MCF7 and HepG2 cells | 131 |
| 18. | Ram Tulsi, Wild Basil, African Basil. | Ocimumviride
(Lamiaceae) |
Thymol,
Ursolic acid |
Induces apoptosis | Antioxidant scavenging activity | Human colorectal adenocarcinoma cells COLO 205 cells | 128, 129 |
| 19. | Velame’ and Velamebranco | Macrosiphonialongiflora (Des)
(Apocynaceae) |
Ellagic acid | Anti-inflammatory
inhibition of IL-1β, IL-10 |
Nitric oxide releases
|
RAW 264.7 cells | 132 |
| 20. | Red oil palm | Elaeisguineensis
(Arecaceae). |
Tocopherols, tocotrienols, their isomers and carotenoids | Inhibitory action | Scavenging free radicals or ROS, effect on lipid peroxidation | Human breast cancer cells | 133 |
| 21. | Black Seed | Nigella sativa | Melanin | Inhibits MAPK pathway through TLR4 | Increased ROS and decreased and Glutathione levels | Colorectal, Adenocarcinoma HT 29 and mCRC SW620 cell lines | 134 |
Cancer and Inflammatory Pathways: Several signalling pathways are involved in the regulation of inflammatory actions during cancer. One such pathway is the TGF-β pathway, TGF-β is very efficient in regulating fundamental cellular processes such as growth, differentiation, or apoptosis. Although sporadic mutations in TGF-β and its pathway are not very common in colorectal cancer, pancreatic cancer, and their degree is different in other types of cancers 60. Hence, it is not suggested as a significant prognostic marker 61. The transcription factor, NF-κB, has a role in proliferation, cell survival, and regulation of the cell cycle and in the development of resistance to drug therapies 62-64. Researchers have indicated that the microenvironment of the tumour, especially fibroblasts, secretes a Want family member Wnt-16B through the NF-κB-dependent pathway, thereby mediating the reduction of chemotherapy-induced programmed cell death in the prostate cancer 65 cell and is also associated with pancreatic cancer 66-68 and up-regulated in lung 69. Recently, a form of programmed necrosis has also been identified 70. Activation of the death receptor can lead to caspases- independent cell death termed necroptosis. While the necroptosis is induced by the TNF-family of cytokines and mediated by the kinases RIP1 and RIP3 in the absence of the caspases-8 activation, RIP1 forms a complex with the key regulator of necroptosis, RIP3, and its downstream substrate, MLKL 71. TNFα was frequently detected in human cancer (produced either by epithelial tumour cells, as in, for instance, ovarian and renal cancer) or the stromal cell (as in Breast cancer) 72. Not only the intracellular or extracellular protein elevates the inflammatory response, but intracellular ions also contribute. It is also reported that inflammatory responses are inhibited by magnesium through downregulation of TLR4/NF-kB, activation of phosphoinositide 3-kinase (PI3K)/Akt pathway or the inhibition of HMGB1 secretion 73-75. These key molecules may serve as potential markers for prognosis of various cancers.
Currently Available Treatments: Despite the intense investigation /trial of the potential anti-cancer drugs, current therapies are not effective in treating most forms of cancer. The major reason for this potential failure is the occurrence of drug resistance. Increasing evidence suggests that the tumour microenvironment plays a critical role in the resistance of tumour cells to both targeted therapy and DNA damaging chemotherapy 76. At present, several treatment options are available depending on the cancer stages, such as targeted therapies, immune therapies, cancer vaccines, chemotherapy, radiation therapy, and surgery. If we focus just on chemotherapy, this is the process through which synthetic drugs used for curing cancer are introduced into the patient, and it is a very painful procedure. Recently, WHO studies reported that about 55% of patients suffer pain, and this experience is very unpleasant and also affects emotional health during, before, and after treatment. A lot of cancer victims would preferably use other treatment methods that are less painful but are as effective. The cost of treating cancer in most of hospitals is discouraging to patients and making cancer victims more traumatized. Modern technology for cancer detection is more efficient and precise than the past methods used, which are time-consuming and too general. Majority of cancer patients still rely on the use of chemotherapeutics.
Chemotherapy drugs like Gemcitabine and Folfirinox (oxaliplatin, irinotecan, fluorouracil, and leucovorin) are widely used for the treatment of metastatic pancreatic cancer (MPC). It has been recently studied that nab-paclitaxel plus gemcitabine (nab-p/gem) significantly improved overall survival, progression-free survival, and response rate in patients with metastatic pancreatic adenocarcinoma, and as compared with that, FOLFIRINOX WITH NAB-P/GEM treatment has been observed with an increased survival advantage and toxicity 77. In contrast, Ruxolitinib is also a potent JAK1/JAK2 inhibitor that has shown clinical benefit in patients with myelofibrosis, a myeloproliferative neoplasm characterized by cachexia, weight loss, elevated pro-inflammatory cytokines, and dysregulated JAK/STAT signaling 78-80.
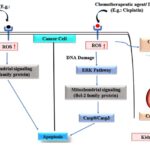
Among them, cisplatin is also a potent chemotherapeutic drug which has been used over the last 25 year. However, it causes nephrotoxicity in many ways 81. Acute kidney injury (AKI) is one of the most serious and more common complications Fig. 4. Which occurs in 20-30% of patients. It has been studied that cisplatin induces increased expression of several chemokines and cytokines responsible for the inflammation. For example, the expression of IL-1β, IL-18, CX3CL1, and IL-6 are increased as a result of cisplatin nephrotoxicity 82, 83. While in-vivo deletion of caspase 1 is responsible for the formation of active IL-1β, IL-18 reduces cisplatin kidney injury and neutrophil infiltration 84-86. Cisplatin nephrotoxicity also contributes to significant upregulation of IFN-γ expression, but neutralizing antibodies to IFN-γ provided no protection against renal injury 87, 88. Doxorubicin is another potent drug that is used to treat many cancers but patient receiving this drug are reported to develop heart attack 89-91.
FIG. 4: UNDERLYING MECHANISMS OF DIFFERENT PATHWAY SUTLILIZED BY CHEMOTHERAPEUTIC DRUGS AND NATURAL PLANT DERIVED COMPOUNDS. Both chemotherapeutic drugs and plant derived compounds trigger subsequent cytoplasmic signals leading to induction of apoptosis in the cancer cell. Chemotherapeutic agents or drugs (e.g. cisplatin) inhibits proliferation of cancer as well as normal cell whereas the plant active compound inhibits cancer proliferation. Comparison of plant based compounds and chemotherapeutic drugs suggest multiple side effects of the chemotherapeutic drugs (for example Cisplatin) like AKI Acute kidney injuryand increased expression of some specific inflammatory marker (INF, IL- 18. IL-6 etc.) even in normal cells apart from cancer cells.There are also chances of other harmful effects of these treatments viz. second cancers after chemotherapy 92
Active Herbal Compounds Mediating Inflammatory Responses: We are currently in that era where chemotherapeutic drugs are used for the treatment of cancer and despite several contraindications, most of the patients largely rely on chemotherapy. Active herbal compounds hold promise for the treatment of malignancies, although the plant constituents have become a promising source of active natural products which differ widely in terms of their biological and structural properties. These compounds are less exploited in terms of treatment for various types of cancers. Herbal drugs are also known to have good immune-modulatory properties which act by stimulating both non-specific and specific immunity 93.
In recent years, the prevention of non-communicable diseases such as cancer and many disorders is found to be associated with the ingestion of fresh fruits, vegetables, tea, or plant beverages that are rich in natural antioxidants 94, 95. The therapeutic potential of plant products is due to the presence of several compounds having distinct mechanisms of action; some of them are enzymes and proteins while others are low molecular weight compounds such as vitamins, anthocyanins, carotenoids, flavonoids and other phenolic compounds. The reason for investigations on the medicinal plant is the ethnobotanical knowledge which still exists in remote parts of the world 96 and these plants are used by regional healers for ameliorating the ailments.
Several plant active compounds have shown immunomodulatory activity through their interaction with specific surface receptors and show specific mechanism of action. These potential mechanisms trigger the secretion of inflammatory factors causing the death of cancer cell by a mechanism called apoptosis.
Therapeutic Implications of Plant-Derived Active Compounds in Cancer: Once a better insight about the action of the plant products is achieved at the cellular and biochemical level, it could be possible to make a better assessment of what other agents (e.g., synthetic compounds) may interact with plant active compounds in a synergistic and antagonistic manner. This provides a better understanding and a good prediction as to which combination would be successful. For appropriate understanding, both cell lines and animal models should be employed. Several natural products are effective in accelerating wound healing by stimulating the different growth factors and cytokines. They may have fewer side effects and represent a cost-effective substitute 97, 98 to synthetic drugs. Sauromatum giganteum (Engl.) Cusimano and Hetttuber have been used to treat many kinds of cancer, such as gastric carcinoma and liver cancer. Experimental studies suggest that SFE-CO2 extract from Sauromatum giganteum (Engl.) Cusimano and Hetttuber could induce apoptosis in tumour cells 99.
The extract showed significant increases in reactive oxygen species on SMMC-7721 cell lines, where the caspases-3 & caspases -9 have risen in Western Blotting analysis. Ultimately the SFE-Co2 extract from Sauromatum giganteum or Typhonium giganteum induced the apoptosis 99. The up-regulation of TRAIL/ TRAIL-R1 and TRAIL-R2 by lignans of Rhizoma Typhonii could be involved in the induction of apoptosis 100. But it is unclear whether decoction or Vinum had a better curative effect 101. Recently, Albarakatim et al. (2020) reported that luteolin (LUT), a plant-derived compound, provide significant protection against lead acetate (PbAc)-induced renal injury in rats. PbAc significantly lowers the level of antioxidant enzyme activity and expression (SOD, CAT, GPx, GR, and MDA). PbAc exposure down-regulated Nfe 212 and Homx1 mRNA expression and significantly increased inflammatory marker (TNF-α, IL-1β, and NO) levels in renal tissue. It is also reported that down-regulated Nfe212 and Homx1 mRNA expression significantly increase levels of inflammatory markers (TNF-α, IL-1β, and NO) in the renal tissue. All these factors together up-regulated the synthesis of pro-apoptotic proteins and down-regulated anti-apoptotic protein expression. Notably, LUT pre-treatment of PbAc -treated rats provides significant protection against PbAc intoxication via anti-inflammatory, anti-oxidant, and anti-apoptotic activities by triggering the activities of the Nrf2/ARE signalling pathway 102. It is also reported that antioxidant-enriched fraction (AEF) of H. spinosa from the whole plant is rich in the presence of apigenin and LUT, which exerted dose-dependent anticancer potential against both ovarian and breast cancer cell lines with a low level of IC50 (43 μg/ml) in SKOV-3 cell line 103. Illicium verum, Glycyrrhiza glabra, Rhamnus Frangula, and Linum usitatissimum extracts have shown reduced in-vitro tumour cell proliferation significantly and using MTT assay and RT-PCR analysis suggested that these extracts could be employed as chemotherapeutical adjuvants for different cancer treatments 104.
Hence, it is very important to identify the component of plant extract causing cell cytotoxicity and inhibiting the cell viability leading to apoptosis. So, it is very clear that we need to take one more step out of this current treatment scenario and investigate new diagnostic, therapeutic improvement, thereby subsiding the side effects of chemotherapy that are often difficult to ameliorate and can significantly impair a cancer patient’s quality of life, in both early-stage and late stage of cancer.
Several studies described here and elsewhere provide convincing evidence regarding the antitumor and anticancer activities of plant products like lectins. These scientific findings, together with evidence for their therapeutic activity against cancer 105, 106. Singh Bain and colleges have been reported that SVL (Sauromatum venosum lectin) showed significant inhibition toward the various cancer cell lines 107. Based on the results they suggested that this plant lectin has antiproliferative response-ability which may help in the identification of new lectin probe for detection and study of certain types of cancer. The effect of aqueous extract of Typhonium giganteum in hepatocellular carcinoma SMMC-7721 cells gene expression is reported by Wang SQ 108. The Elevated level of IL-2 estimated using DUOSET-ELISA kit has shown in cancer cell line from dichromethen fraction of Adrographispaniculata extract 109–111. In general medical practice, phytopharmaceuticals play an important role for the treatment of diseases of the cardiac and vascular system 112, nervous system 113, and immune system 114 and a huge number of herbal drugs have suggested prophylactic effect besides their therapeutic use for disease 115, 116. Although, the annual report of CDC, NCI stated that overall cancer death rates declined for women, men, children and young one and researchers look forward to treat common cancers of lung, prostate, breast and colorectal cancer. List of some important plant-derived materials used for treatment of different cancers are mentioned in Table 1.
Major Challenges to Improve the Anticancer Properties of Plant Active Candidates: Several studies have reported that anticancer drugs act through the introduction of apoptosis and induce inflammation in human cancer in vitro; not only they exhibit the cancer cell death; they exhibit the anti-angiogenesis 117, 118, anti-metastasis 119, 120 and differentiation 117, 121, 122. Plant lectins have been extensively utilized for the specific targeting of immunotoxins 123. The environment factors (temperature, humidity), harvest and postharvest procedure also impact on the quality of plant extracted compound employing various extraction methods (using different extraction solvents, their concentration and ratio) 124. This is a very essential process in establishing the quality of drug obtained from the plant extract. Moreover, one must bear in mind that not only experiments evaluating the toxicity levels but also most regulatory parameters should be considered before establishing the new idea of the therapeutic process. Any other type of toxic effect, that might result from interactions of various compounds in the plant extract and their effects on different types of cells and tissues should also be examined. Appropriate control groups should be incorporated in most of the toxicity testing studies. Another major challenge that one may encounter is due to the short half-life of many herbal compounds of therapeutic importance. One may enhance the potential of plant extracts by identifying the active compounds and creating a nanoparticle to increase specificity and sensitivity of the compound.
CONCLUSION AND FUTURE PERSPECTIVE: The herbal medicine is a rational medicine among them some of the herbal medicines are in the clinical trials and have shown very good efficacy. Investigating the mechanism of action of herbal medicine in killing the cancer cell without compromising the normal cells is of interest to most of the researchers working in this area. Several studies provide convincing evidence regarding the antitumor and anticancer activities of different plant products. Most of the modern therapies including immunotherapy, radiation therapy, CART cells and combination therapy are widely practised and represent one thing in common that is “inflammatory responses”. As we understand how the inflammation and cancer go hand in hand there is the vast role of TME (tumour microenvironment) to create a strong correlation between inflammation and cancer generation. Tumour environment is maintained by the infiltrating immune cells and their secreting inflammatory factors. Which activates different essential signal transducers and transcriptional factors to generate inflammation promoted cancer.
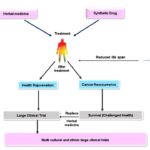
To promote the use of herbal plant medicine, multicentric large scale clinical trials should be promoted Fig. 5. It is essential to identify the right stage of cancer and treatment should be designed accordingly. If clinicians can provide herbal treatment /supplement at an early stage of cancer, treatment will be more efficacious when compared with the late stage of cancer. There are several instances of cancer reoccurrence even after using a chemotherapeutic drug hence herbal supplements may be provided to the patients. Providing herbal medicines may improve the quality as well as the life span of patients. It is essential to dig deep into the ancients wisdom that is available in the literature and also contact regional healers that are located in different parts of the country to learn the use of herbal medicines. Newer researches should focus on increasing the life span and quality of life of patients suffering from cancer. Use of active biomolecules such as nanoparticle may revolutionize the use of herbal medicines. It is evidenced by the published work that such nanoparticles may improve the efficacy of the drug and also subside complications like nephrotoxicity 102, 125, 126. It is a high time to develop methods and technology to improve the use of plant active compounds in the treatment as well as improving the quality of life of patients suffering from cancer. In this review, we have tried to understand the use of plant-derived active compounds and their effect on cancer and inflammation. We need to employ multicentric clinical trials for plant medicines to achieve this aim. More researches should be done and funding from governmental and non-governmental agencies should pour in to support the cause.
FIG. 5: FUTURE PROSPECTS OF HERBAL MEDICINE IN THE TREATMENT OF CANCER. Using herbal medicine as a treatment option can be considered farely safe in comparison to conventional synthetic drug treatments. Nowadays, several techniques are available to improve the half life of herbal medicines and increasing the shelf life. The improvement of method of analysis and quality control of herbal drugs and medicine along with advancement in clinical trials will certainly increase their efficacy and safety bringing the value of life and health rejuvenation of cancer patients.
ACKNOWLEDGEMENT: Thanks to University and Central Council for Research in Ayurvedic Science (CCRAS) for supporting the research work.
Funding: Guru Ghasidas central Vishwavidyalaya (GGV) Bilaspur (C.G.), Central Council for Research in Ayurvedic Science (CCRAS).
CONFLICTS OF INTEREST: For all the author, there is no potential conflict of interest.
Author’s Contribution:
Namrata Kahar: Literature review, manuscript drafting, preparation of figure, concept and design the study.
Rohit Seth: Literature review, manuscript editing and approval of the final version of the manuscript.
REFERENCES:
- Alawode TT: An overview of the anti-cancer properties of some plants used in traditional medicine in Nigeria. Int Res J Bio Chem Bio Inform 2013; 3(1): 7-14.
- Kutluk T and Kars A: General knowledge about cancer Ankara. Turkey Cancer Investigation and Fight Society Publications 1998; 7-15.
- Lee B and Gibbs P: Inflammation biomarkers and immuno-oncology pathways in pancreatic cancer. Journal of Personalized Medicine 2019; 9(2): 20.
- Sfanos KS, Yegnasubramanian S, Nelson WG and De Marzo AM: The inflammatory micro environment and microbiome in prostate cancer development. Nature Reviews Urology 2018; 15(1): 11-24.
- Murata M: Inflammation and cancer. Environmental Health and Preventive Medicine 2018; 23(1): 1-8.
- Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S and Du Bois RN: Up-regulation of. Cyclooxygenase 2 1183-8.
- Sano H, Kawahito Y, Wilder RL, Hashiramoto A, Mukai S, Asai K, Kimura S, Kato H, Kondo M and Hla T: Expression of cyclooxygenase-1 and-2 in human colorectal cancer. Cancer Research 1995; 55(17): 3785-9.
- Kutchera W, Jones DA, Matsunami N, Groden J, McIntyre TM, Zimmerman GA, White RL and Prescott SM: Prostaglandin H synthase 2 is expressed abnormally in human colon cancer evidence for a transcriptional effect. Proceedings of the National Academy of Sciences 1996; 93(10): 4816-20.
- Paradise BD, Barham W and Fernandez-Zapico ME: Targeting epigenetic aberrations in pancreatic cancer, a new path to improve patient outcomes. Cancers 2018; 10(5): 128.
- Kwon HM, Kang EJ, Kang K, Kim SD, Yang K and Yi JM: Combinatorial effects of an epigenetic inhibitor and ionizing radiation contribute to targeted elimination of pancreatic cancer stem cell. Oncotarget 2017; 8(51): 89005.
- Kraus S and Arber N: Inflammation and colorectal cancer. Current Opinion in Pharmacology 2009; 9(4): 405-10.
- Karpisheh V, Nikkhoo A, Hojjat-Farsangi M, Namdar A, Azizi G, Ghalamfarsa G, Sabz G, Yousefi M, Yousefi B and Jadidi-Niaragh F: Prostaglandin E2 as a potent therapeutic target for treatment of colon cancer. Prostaglandins & Other Lipid Mediators 2019; 144: 106338.
- Hara S, Miyata A, Yokoyama C, Inoue H, Brugger R, Lottspeich F, Ullrich V and Tanabe T: Isolation and molecular cloning of prostacyclin synthase from bovine endothelial cells. Journal of Biological Chemistry 1994; 269(31): 19897-03.
- Kuwamoto S, Inoue H, Tone Y, Izumi Y and Tanabe T: Inverse gene expression of prostacyclin and thromboxane synthases in resident and activated peritoneal macrophages. FEBS Letters 1997; 409(2): 242-6.
- Suzuki T, Watanabe K, Kanaoka Y, Sato T and Hayaishi O: Induction of hematopoietic prostaglandin D synthase in human megakaryocytic cells by phorbol ester. Biochemical and Biophysical Research Communications 1997; 241(2): 288-93.
- Forsberg L, Leeb L, Thorén S, Morgenstern R and Jakobsson PJ: Human glutathione dependent prostaglandin E synthase gene structure and regulation. FEBS letters 2000; 471(1): 78-82.
- Bonvalet JP, Pradelles P and Farman N: Segmental synthesis and actions of prostaglandins along the nephron. American Journal of Physiology-Renal Physiology 1987; 253(3): F377-87.
- Smith WL: Prostanoid biosynthesis and mechanisms of action. American Journal of Physiology-Renal Physiology 1992; 263(2): 181-91.
- Narumiya S, Sugimoto Y and Ushikubi F: Prostanoid receptors structures properties and functions. Physiological Reviews 1999; 79(4): 1193-226.
- Mantovani A, Allavena P, Sica A and Balkwill F: Cancer-related inflammation. Nature 2008; 454(7203): 436-44.
- Balkwill F, Charles KA and Mantovani A: Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell 2005; 7(3): 211-7.
- Kulbe H, Thompson R, Wilson JL, Robinson S, Hagemann T, Fatah R, Gould D, Ayhan A and Balkwill F: The inflammatory cytokine tumor necrosis factor-α generates an autocrine tumor-promoting network in epithelial ovarian cancer cells. Cancer Research 2007; 67(2): 585-92.
- Mocellin S, Rossi CR, Pilati P and Nitti D: Tumor necrosis factor cancer and anticancer therapy. Cytokine & Growth Factor Reviews 2005; 16(1): 35-53.
- Crusz SM and Balkwill FR: Inflammation and cancer advances and new agents. Nature Reviews Clinical Oncology 2015; 12(10): 584-96.
- Thun MJ, Namboodiri MM, Heath JR and CW JR: Aspirin use and reduced risk of fatal colon cancer. N Engl J Med 325: 1593-6.
- Drew DA, Cao Y and Chan AT: Aspirin and colorectal cancer the promise of precision chemoprevention. Nature Reviews Cancer 2016; 16(3): 173.
- Chan AT, Arber N, Burn J, Chia WK, Elwood P, Hull MA, Logan RF, Rothwell PM, Schrör K and Baron JA: Aspirin in the chemoprevention of colorectal neoplasia an overview. Cancer Prevention Research 2012; 5(2): 164-78.
- Flossmann Ea and Rothwell PM: Effect of aspirin on long-term risk of colorectal cancer consistent evidence from randomised and observational studies. The Lancet 2007; 369(9573): 1603-13.
- Gilligan MM, Gartung A, Sulciner ML, Norris PC, Sukhatme VP, Bielenberg DR, Huang S, Kieran MW, Serhan CN and Panigrahy D: Aspirin-triggered proresolving mediators stimulate resolution in cancer. Proceedings of the National Academy of Sciences 2019; 116(13): 6292-7.
- Gao F, Liang B, Reddy TS, Farias-Eisner R and Su X: Role of inflammation-associated micro environment in tumorigenesis and metastasis. Current Cancer Drug Targets 2014; 14(1): 30-45.
- Wang K and Karin M: Tumor-elicited inflammation and colorectal cancer. Advances in Cancer Research 2015; 128: 173-96.
- Elinav E, Nowarski R, Thaiss CA, Hu B, Jin C and Flavell RA: Inflammation-induced cancer crosstalk between tumours, immune cells and microorganisms. Nature Reviews Cancer 2013; 13(11): 759-71.
- Baldwin AS Regulation of cell death and autophagy by IKK and NF‐κB critical mechanisms in immune function and cancer. Immunological Reviews 2012; 246(1): 327-45.
- Yu H, Kortylewski M and Pardoll D: Crosstalk between cancer and immune cells role of STAT3 in the tumour micro environment. Nature Reviews Immunology 2007; 7(1): 41-51.
- NF HG: KB and STAT3 key players in 1iver inflammation and cancer. Cell Res 2011; 21(1): 159.
- Grivennikov SI and Karin M: Dangerous liaisons STAT3 and NF-κB collaboration and crosstalk in cancer. Cytokine & Growth Factor Reviews 2010; 21(1): 11-9.
- Tang L and Wang K: Chronic inflammation in skin malignancies. Journal of Molecular Signaling 2016; 11.
- Hanahan D and Coussens LM: Best of Supplement-Cancer Cell Best of 2012.
- Ebos JM and Kerbel RS: Antiangiogenic therapy impact on invasion disease progression and metastasis. Nature Reviews Clinical Oncology 2011; 8(4): 210-21.
- Bakhle YS and Botting RM: Cyclooxygenase-2 and its regulation in inflammation. Mediators of Inflammation. 1996; 5(5): 305-23.
- Botting RM: Vane’s discovery of the mechanism of action of aspirin changed our understanding of its clinical pharmacology. Pharma Reports 2010; 62(3): 518-25.
- Jouzeau JY, Terlain B, Abid A, Nédélec E and Netter P. Cyclo-oxygenase isoenzymes. Drugs 1997; 53(4): 563-82.
- Menter DG, Schilsky RL and Du Bois RN: Cyclooxygenase-2 and cancer treatment: understanding the risk should be worth the reward. Clinical Cancer Research 2010; 16(5): 1384-90.
- Nakanishi M, Gokhale V, Meuillet EJ and Rosenberg DW: mPGES-1 as a target for cancer suppression A comprehensive invited review Phospholipase A2 and lipid mediators. Bio Chem 2010; 92(6): 660-4.
- Wang MT, Honn KV and Nie D: Cyclooxygenases, prostanoids and tumor progression. Cancer and Metastasis Reviews 2007; 26(3): 525-34.
- Hwang D, Byrne J, Scollard D and Levine E: Expression of cyclooxygenase-1 and cyclooxygenase-2 in human breast cancer. JNCI Journal of the National Cancer Institute 1998; 90(6): 455-60.
- Hashemi Goradel N, Najafi M, Salehi E, Farhood B and Mortezaee K: Cyclooxygenase‐2 in cancer. A Review Journal of Cellular Physiology 2019; 234(5): 5683-99.
- Kundu N, Yang Q, Dorsey R and Fulton AM: Increased cyclooxygenase‐2 (cox‐2) expression and activity in a murine model of metastatic breast cancer. International Journal of Cancer 2001; 93(5): 681-6.
- Voutsadakis IA: Pathogenesis of colorectal carcinoma and therapeutic implications the roles of the ubiquitin-proteasome system and Cox‐2. Journal of Cellular and Molecular Medicine 2007; 11(2): 252-85.
- Bennett A, Berstock DA, Carroll MA, Stamford IF and Wilson AJ: Breast cancer its recurrence and patient survival in relation to tumor prostaglandins. Advances in Prostaglandin, Thromboxane, and Leukotriene Research 1983; 12: 299-02.
- Giorgio M, Trinei M, Migliaccio E and Pelicci PG: Hydrogen peroxide a metabolic by-product or a common mediator of ageing signals. Nature Reviews Molecular Cell Biology 2007; 8(9): 722-8.
- Zorov DB, Juhaszova M and Sollott SJ: Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiological Reviews 2014; 94(3): 909-50.
- Liu S and Huang H: Assessments of antioxidant effect of black tea extract and its rationals by erythrocyte haemolysis assay plasma oxidation assay and cellular antioxidant activity (CAA) assay. Journal of Functional Foods 2015; 18: 1095-105.
- Storz P: Reactive oxygen species in tumor progression. Front Bio Sci 2005; 10(1-3): 1881-96.
- Roy K, Wu Y, Meitzler JL, Juhasz A, Liu H, Jiang G, Lu J, Antony S and Doroshow JH: NADPH oxidases and cancer. Clinical Science 2015; 128(12): 863-75.
- Stanicka J, Russell EG, Woolley JF and Cotter TG: NADPH oxidase-generated hydrogen peroxide induces DNA damage in mutant FLT3-expressing leukemia cells. Journal of Biological Chemistry 2015; 290(15): 9348-61.
- Ames BN, Shigenaga MK and Hagen TM: Oxidants antioxidants and the degenerative diseases of aging. Proceedings of the National Academy of Sciences 1993; 90(17): 7915-22.
- Szatrowski TP and Nathan CF: Production of large amounts of hydrogen peroxide by human tumor cells. Cancer Research 1991; 51(3): 794-8.
- Moloney JN and Cotter TG: ROS signalling in the biology of cancer. In Seminars In cell & Developmental Biology Academic Press 2018; 82: 50-64.
- Zhao M, Mishra L and Deng CX: The role of TGF-β/SMAD4 signaling in cancer. International Journal of Biological Sciences 2018; 14(2): 111.
- Herzig DO and Tsikitis VL: Molecular markers for colon diagnosis prognosis and targeted therapy. Journal of Surgical Oncology 2015; 111(1): 96-102.
- Ahmed KM, Cao N and Li JJ: HER-2 and NF-κB as the targets for therapy-resistant breast cancer. Anticancer Research 2006; 26(6B): 4235-43.
- Bourguignon LY, Spevak CC, Wong G, Xia W and Gilad E: Hyaluronan-CD44 interaction with protein kinase cϵ promotes oncogenic signaling by the stem cell marker nanog and the production of microrna-21 leading to down-regulation of the tumor suppressor protein pdcd4, anti-apoptosis and chemotherapy resistance in breast tumor cells. Journal of Biological Chemistry 2009; 284(39): 26533-46.
- Duffey DC, Crowl-Bancroft CV, Chen Z, Ondrey FG, Nejad-Sattari M, Dong G and Van Waes C: Inhibition of transcription factor nuclear factor-κB by a mutant inhibitor-κBα attenuates resistance of human head and neck squamous cell carcinoma to TNF-α caspase-mediated cell death. British Journal of Cancer 2000; 83(10): 1367-74.
- Sun Y, Campisi J, Higano C, Beer TM, Porter P, Coleman I, True L and Nelson PS: Treatment-induced damage to the tumor microenvironment promotes prostate cancer therapy resistance through WNT16B. Nature Medicine 2012; 18(9): 1359-68.
- Prabhu L, Mundade R, Korc M, Loehrer PJ and Lu T: Critical role of NF-κB in pancreatic cancer. Oncotarget 2014; 5(22): 10969.
- Greer JB and Whitcomb DC: Inflammation and pancreatic cancer an evidence-based review. Current Opinion in Pharmacology 2009; 9(4): 411-8.
- Fujioka S, Sclabas GM, Schmidt C, Frederick WA, Dong QG, Abbruzzese JL, Evans DB, Baker C and Chiao PJ: Function of nuclear factor κB in pancreatic cancer metastasis. Clinical Cancer Research 2003; 9(1): 346-54.
- Tang X, Liu D, Shishodia S, Ozburn N, Behrens C, Lee JJ, Hong WK, Aggarwal BB and Wistuba II: Nuclear factor‐κB (nf‐κB) is frequently expressed in lung cancer and preneoplastic lesions. Cancer Interdisciplinary International Journal of the American Cancer Society 2006; 107(11): 2637-46.
- Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, Alnemri ES, Altucci L, Amelio I, Andrews DW and Annicchiarico-Petruzzelli M: Molecular mechanisms of cell death recommendations of the. Nomenclature Committee on Cell Death 2018.
- Dickens LS, Powley IR, Hughes MA and MacFarlane M: The complexities of life and death receptor signalling platforms. Experimental Cell Research 2012; 318(11): 1269-77.
- Balkwill F: Tumor necrosis factor or tumor promoting factor. Cytokine & Growth Factor Reviews 2002; 13(2): 135-41.
- Xia J, Chen H, Yan J, Wu H, Wang H, Guo J, Zhang X, Zhang S, Zhao C and Chen Y: High-Purity magnesium staples suppress inflammatory response in rectal anastomoses. ACS Applied Materials & Interfaces 2017; 9(11): 9506-15.
- Su NY, Peng TC, Tsai PS and Huang CJ: Phosphoinositide 3-kinase/Akt pathway is involved in mediating the anti-inflammation effects of magnesium sulfate. Journal of Surgical Research 2013; 185(2): 726-32.
- Liu Z, Chang Y, Zhang J, Huang X, Jiang J, Li S and Wang Z: Magnesium deficiency promotes secretion of high-mobility group box 1 protein from lipopolysaccharide-activated macrophages in-vitro. Journal of Surgical Research 2013; 180(2): 310-6.
- Östman A: The tumor microenvironment controls drug sensitivity. Nature Medicine 2012; 18(9): 1332-4.
- Conroy T, Desseigne F and Ychou M: On behalf of the Groupe tumeurs digestives of unicancer and the prodige intergroup. folfirinox versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011; 364: 1817-25.
- Verstovsek S, Kantarjian H, Mesa RA, Pardanani AD, Cortes-Franco J, Thomas DA, Estrov Z, Fridman JS, Bradley EC, Erickson-Viitanen S and Vaddi K: Safety and efficacy of INCB018424 a JAK1 and JAK2 inhibitor in myelofibrosis. New England Journal of Medicine 2010; 363(12): 1117-27.
- Verstovsek S, Mesa RA, Gotlib J, Levy RS, Gupta V, DiPersio JF, Catalano JV, Deininger M, Miller C, Silver RT and Talpaz M: A double-blind placebo-controlled trial of ruxolitinib for myelofibrosis. New England Journal of Medicine 2012; 366(9): 799-07.
- Harrison C, Kiladjian JJ, Al-Ali HK, Gisslinger H, Waltzman R, Stalbovskaya V, McQuitty M, Hunter DS, Levy R, Knoops L and Cervantes F: JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. New England Journal of Medicine 2012; 366(9): 787-98.
- Miller RP, Tadagavadi RK, Ramesh G and Reeves WB: Mechanisms of cisplatin nephrotoxicity. Toxins 2010; 2(11): 2490-518.
- Faubel S, Lewis EC, Reznikov L, Ljubanovic D, Hoke TS, Somerset H, Oh DJ, Lu L, Klein CL, Dinarello CA and Edelstein CL: Cisplatin-induced acute renal failure is associated with an increase in the cytokines interleukin (IL)-1β, IL-18, IL-6 and neutrophil infiltration in the kidney. Journal of Pharmacology and Experimental Therapeutics 2007; 322(1): 8-15.
- Lu LH, Oh DJ, Dursun B, He Z, Hoke TS, Faubel S and Edelstein CL: Increased macrophage infiltration and fractalkine expression in cisplatin-induced acute renal failure in mice. Journal of Pharmacology and Experimental Therapeutics 2008; 324(1): 111-7.
- Faubel S, Ljubanovic D, Reznikov L, Somerset H, Dinarello CA and Edelstein CL: Caspase-1–deficient mice are protected against cisplatin-induced apoptosis and acute tubular necrosis. Kidney International 2004; 66(6): 2202-13.
- Renu K, VG A, Picchia PBT and Arunachalam S: Molecular mechanism of doxorubic ininduced cardio. myopathy-an update 2018; 818: 241-53.
- Zhang YW, Shi J, Li YJ and Wei L: Cardiomyocyte death in doxorubicin-induced cardiotoxicity. Archivum immunologiae Et the Rapiae Exp2009; 57(6): 435-45.
- Moslehi J, Amgalan D and Kitsis RN: Grounding cardio-oncology in basic and clinical science. Circulation 2017; 136(1): 5.
- Amgalan D, Garner TP, Pekson R, Jia XF, Yanamandala M, Paulino V, Liang FG, Corbalan JJ, Lee J, Chen Y and Karagiannis GS: A small-molecule allosteric inhibitor of BAX protects against doxorubicin-induced cardio myopathy. Nature Cancer 2020; 1(3): 315-28.
- Kumar S, Jawaid T and Dubey SD: Therapeutic plants of Ayurveda a review on anticancer. Pharmacognosy Journal 2011; 3(23): 1-1.
- Koch W: Dietary polyphenols-important non-nutrients in the prevention of chronic noncommunicable diseases. A Systematic Review Nutrients 2019; 11(5): 1039.
- Grosso G: Dietary antioxidants and prevention of non-communicable diseases.
- Khan MA, Shahwar D, Ahmad N and Ajaib M: Chemical constituents of Carissa opaca extracts and their evaluation as antioxidants and preservatives in edible oils. Asian Journal of Chemistry 2010; 22(1): 379.
- Karunanidhi A, Ghaznavi-Rad E, Jeevajothi Nathan J, Abba Y, Belkum AV and Neela V: Allium stipitatum extract exhibits in-vivo antibacterial activity against methicillin-resistant Staphylococcus aureus and accelerates burn wound healing in a full-thickness murine burn model. Evidence-Based Complementary and Alternative Medicine 2017; 2017.
- Said A, Wahid F, Bashir K, Rasheed HM, Khan T, Hussain Z and Siraj S: Sauromatum guttatum extract promotes wound healing and tissue regeneration in a burn mouse model via up-regulation of growth factors. Pharmaceutical Biology 2019; 57(1): 736-43.
- Li Q, Jiang C, Zu Y, Song Z, Zhang B, Meng X, Qiu W and Zhang L: SFE-CO2 extract from Typhoniumgi ganteum Engl tubers induces apoptosis in human hepatoma SMMC-7721 cells involvement of a ROS-mediated mitochondrial pathway. Molecules 2011; 16(10): 8228-43.
- Ma YL, Li YF, Tang LP and Ai FW: Study of Lignins of rhizoma typhonii on the expression of TRAIL and its receptors in gastric carcinoma cell line SGC-7901. Chin J. New Drugs 2010; 19: 225-8.
- Shi-Yong G: 2014.
- Albarakati AJ, Baty RS, Aljoudi AM, Habotta OA, Elmahallawy EK, Kassab RB and Moneim AE: Luteolin protects against lead acetate-induced nephrotoxicity through antioxidant, anti-inflammatory anti-apoptotic, and Nrf2/HO-1 signaling pathways. Molecular biology reports 2020: 1-3.
- Satpathy S, Patra A, Hussain MD and Ahirwar B: Amelioration of postmenopausal osteoporosis and anticancer properties of an antioxidant enriched fraction from Hygrophila spinosa Anders South African Journal of Botany 2018; 117: 247-55.
- Lombardi VR, Carrera I, Cacabelos R. In-vitro screening for cytotoxic activity of herbal extracts, evidence-based complement. Altern Med 2017; 8.
- Chanchal DK, Alok S, Rashi S, Bijauliya RK, Yadav RD and Sabharwal M: Various medicinal plants used in the treatment of anticancer activity. Int J Pharm Sci Res 2017; 9(4): 1424-9.
- Ohsawa M, Naka N, Tomita Y, Kawamori D, Kanno H and Aozasa K: Use of immune his to chemical procedures in diagnosing angiosarcoma evaluation of 98 cases. Cancer 1995; 75(12): 2867-74.
- Lenartz D, Stoffel B, Menzel J and Beuth J: Immuno protective activity of the galactoside-specific lectin from mistletoe after tumor destructive therapy in glioma patients. Anticancer Research 1996; 16(6B): 3799-802.
- SinghBains J, Singh J, Kamboj SS, Nijjar KK, Agrewala JN, Kumar V, Kumar A and Saxena AK: Mitogenic and anti-proliferative activity of a lectin from the tubers of voodoo lily Sauromatum venosum. Biochimica Biophysica Acta (BBA)-General Subjects 2005; 1723(1-3): 163-74.
- Wang SQ, Ni H, Cheng H, Wang GL, Wang TS and Chen L: Detection of differentially expressed genes in hepato cellular carcinoma cells SMMC-7721 treated with Typhoniumgiganteum extract by mRNA differential display. Zhongguo Zhong yao za zhi Zhongguozhongyaozazhi China Journal of Chinese Materia Medica 2004; 29(10): 974-7.
- Kumar RA, Sridevi K, Kumar NV, Nanduri S and Rajagopal S: Anticancer and immune stimulatory compounds from Andrographis paniculata. Journal of Ethno Pharmacology 2004; 92(2-3): 291-5.
- Mishra SK, Sangwan NS and Sangwan RS: Phcog rev plant review Andrographis paniculata (Kalmegh): a review. Pharmacognosy Reviews 2007; 1(2): 283-98.
- Rajagopal S, kumar AR, Deevi D, Satyanarayana C and Rajagopalan R: and rographolide, a potential cancer therapeutic agent isolated from Andrographis paniculata. J Exp Ther Oncol 2003; 3: 147-58.
- Baharvand-Ahmadi B, Bahmani M, Tajeddini P, Rafieian-Kopaei M and Naghdi N: An ethno botanical study of medicinal plants administered for the treatment of hypertension. Journal of Renal Injury Prevention 2016; 5(3): 123.
- Srivastava P and Yadav RS: Efficacy of natural compounds in neurodegenerative disorders. In The Benefits of Natural Products for Neurodegenerative Diseases 2016; 107-23.
- Ford CT, Richardson S, McArdle F, Lotito SB, Crozier A, McArdle A and Jackson MJ: Identification of (poly) phenol treatments that modulate the release of pro-inflammatory cytokines by human lymphocytes. British Journal of Nutrition 2016; 115(10): 1699-710.
- Kim YS, Park HJ, Kim TK, Moon DE and Lee HJ: The effects of Ginkgo biloba extract EGb 761 on mechanical and cold allodynia in a rat model of neuropathic pain. Anesthesia & Analgesia 2009; 108(6): 1958-63.
- Amirghofran Z, Bahmani M, Azadmehr A, Javidnia K and Miri R: Immuno modulatory activities of various medicinal plant extracts effects on human lymphocytes apoptosis. Immunological Investigations 2009; 38(2): 181-92.
- Bhutia SK, Behera B, Nandini Das D, Mukhopadhyay S, Sinha N, Panda PK, Naik PP, Patra SK, Mandal M, Sarkar S and Menezes ME: Abrus agglutinin is a potent anti‐proliferative and anti‐angiogenic agent in human breast cancer. International Journal of Cancer 2016; 139(2): 457-66.
- Park WB, Lyu SY, Kim JH, Choi SH, Chung HK, Ahn SH, Hong SY, Yoon TJ and Choi MJ: Inhibition of tumor growth and metastasis by Korean mistletoe lectin is associated with apoptosis and anti-angiogenesis. Cancer Biotherapy and Rad Pharmaceuticals 2001; 16(5): 439-47.
- Sinha N, Meher BR, Naik PP, Panda PK, Mukhapadhyay S, Maiti TK and Bhutia SK: P73 induction by Abrus agglutinin facilitates Snail ubiquitination to inhibit epithelial to mesenchymal transition in oral cancer. Phytomedicine 2019; 55: 179-90.
- Zhou SM, Cheng L, Guo SJ, Wang Y, Czajkowsky DM, Gao H, Hu XF and Tao SC: Lectin RCA-I specifically binds to metastasis-associated cell surface glycans in triple-negative breast cancer. Breast Cancer Research 2015; 17(1): 1-4.
- Jordinson M, El-Hariry I, Calnan DA, Calam J and Pignatelli M: Vicia faba agglutinin the lectin present in broad beans stimulates differentiation of un differentiated colon cancer cells Gut 1999; 44(5): 709-14.
- Panda PK, Naik PP, Praharaj PP, Meher BR, Gupta PK, Verma RS, Maiti TK, Shanmugam MK, Chinnathambi A, Alharbi SA and Sethi G: Abrus agglutinin stimulates BMP‐2‐dependent differentiation through autophagic degradation of β‐catenin in colon cancer stem cells. Molecular Carcinogenesis 2018; 57(5): 664-77.
- Jiao P, Zhang J, Dong Y, Wei D and Ren Y: Construction and characterization of the recombinant immune toxin RTA-4D5-KDEL targeting HER2/neu-positive cancer cells and locating the endoplasmic reticulum. Applied Microbiology and Bio Technology 2018; 102(22): 9585-94.
- Bezerra AG, Negri G, Duarte-Almeida JM, Smaili SS and Carlini EA: Phytochemical analysis of hydroethanolic extract of Turneradiffusa Willd and evaluation of its effects on astrocyte cell death. Einstein São Paulo 2016; 14(1): 56-63.
- Baj T and Seth R: Role of curcumin in regulation of TNF-α mediated brain inflammatory responses. Recent patents on Inflammation & Allergy Drug Discovery 2018; 12(1): 69-77.
- Padhye S, Ahmad A, Oswal N and Sarkar FH: Emerging role of Garcinol, the antioxidant chalcone from Garcinia indica Choisy and its synthetic analogs. Journal of Hematology & Oncology 2009; 2(1): 1-3.
- Baliga MS, Bhat HP, Pai RJ, Boloor R and Palatty PL: The chemistry and medicinal uses of the underutilized Indian fruit tree Garcinia indica Choisy kokum a review. Food Research International 2011; 44(7): 1790-9.
- Lai CS, Mas RH, Nair NK, Mansor SM and Navaratnam V: Chemical constituents and in vitro anticancer activity of Typhonium flagella forme Araceae. Journal of Ethno Pharmacology 2010; 27(2): 486-94.
- Chen Z, Liu YM, Yang S, Song BA, Xu GF, Bhadury PS, Jin LH, Hu DY, Liu F, Xue W and Zhou X: Studies on the chemical constituents and anticancer activity of Saxifraga stolonifera (L) Meeb. Bioorganic & Medicinal Chemistry 2008; 16(3): 1337-44.
- Sharma M, Agrawal SK, Sharma PR, Chadha BS, Khosla MK and Saxena AK: Cytotoxic and apoptotic activity of essential oil from Ocimumviride towards COLO 205 cells. Food and Chemical Toxicology 2010; 48(1): 336-44.
- Silva MG, Vieira ÍG, Mendes FN, Albuquerque IL, Dos Santos RN, Silva FO and Morais SM: Variation of ursolic acid content in eight Ocimum species from north eastern. Brazil Molecules 2008; 13(10): 2482-7.
- Rajkumar V, Guha G and Kumar RA: Antioxidant and anti-neoplastic activities of Picrorhizakurroa extracts. Food and Chemical Toxicology 2011; 49(2): 363-9.
- Peng H, Lv H, Wang Y, Liu YH, Li CY, Meng L, Chen F and Bao JK: Clematis montana lectin, a novel mannose-binding lectin from traditional Chinese medicine with antiviral and apoptosis-inducing activities. Peptides 2009; 30(10): 1805-15.
- da Silva AO, Alves AD, de Almeida DA, Balogun SO, de Oliveira RG, Aguiar AA, Soares IM, Marson-Ascêncio PG, Ascêncio SD and de Oliveira Martins DT: Evaluation of anti-inflammatory and mechanism of action of extract of macrosiphonia longiflora desf müll Arg. Journal of Ethno Pharmacology 2014; 154(2): 319-29.
- Obahiagbon FI. A review: aspects of the African oil palm (Elaeisguineesis Jacq.) and the implications of its bioactives in human health. American Journal of Biochemistry and Molecular Biology 2012; 2(3): 106-19.
- Al-Obeed O, El-Obeid AS, Matou-Nasri S, Vaali-Mohammed MA, AlHaidan Y, Elwatidy M, Al Dosary H, Alehaideb Z, Alkhayal K, Haseeb A and McKerrow J: Herbal melanin inhibits colorectal cancer cell proliferation by altering redox balance, inducing apoptosis and modu-lating MAPK signaling. Cancer cell Int 2020; 20: 1-7.
- Miceli N, Cavò E, Ragusa S, Cacciola F, Dugo P, Mondello L, Marino A, Cincotta F, Condurso C and Taviano MF: Phytochemical characterization and biological activities of a hydroalcoholic extract obtained from the aerial parts of Matthiolaincana R Br subsp incana Brassicaceae growing wild in Sicily (Italy). Chemistry & Biodiversity 2019; 16(4): 1800677.
- Aggarwal BB, Sundaram C and Malani N: Curcumin the Indian solid gold. Adv Exp Med Biol 2007; 595: 1-75.
How to cite this article:
Kahar N and Seth R: Role of plant based active compounds in inflammatory responses of cancer. Int J Pharmacognosy 2021; 8(3): 89-103. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(3).89-103.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.