REVIEW ON ANTI-MICROBIAL PROPERTIES OF MEDICINAL PLANTS IN ETHIOPIA
HTML Full TextREVIEW ON ANTI-MICROBIAL PROPERTIES OF MEDICINAL PLANTS IN ETHIOPIA
Tafere Mulaw Belete
Department of Pharmacology, College of Medicine and Health Sciences, University of Gondar, P.O. Box 197, Gondar, Ethiopia.
ABSTRACT: Infectious diseases are one of the major problems in the world. After the discovery of penicillin, many classes of the antimicrobial agent were introduced and most infectious diseases were brought under control. However, the increased use of these antimicrobial agents in clinical practice was soon followed by the emergence of antibiotic resistance. Because of multiple drug resistance and adverse effects of these antimicrobial agents on the host, there is the need to discover and develop natural antimicrobials that are safe and with better therapeutic effects. Medicinal plants have been used as an alternative source of medicine to treat infectious diseases due to their rich antimicrobial activity. These Plants contain bioactive compounds such as quinines, phytoalexins, phenols, alkaloids, flavonoids, terpenoids, steroids, tannins, glucosinolates and pathogenesis-related proteins, which exert antimicrobial activity by different mechanisms, such as destruction of the cell membrane, inhibition of protein synthesis and enzymes impregnated on the cell membrane. There are various extracts of medicinal plants with minimum inhibitory concentration against pathogenic microorganisms that are safe and effective as therapeutic agents. In this review, data on 91 medicinal plants from Ethiopia distributed in 49 families against 27 species of bacteria, 11 species of fungi were assessed, to provide information, and opportunity for further analysis of medicinal plant extracts for the development of effective antimicrobial agents.
| Keywords: |
Medicinal Plants, Anti-microbial Activity, Anti-bacterial Activity, Microorganism
INTRODUCTION: In spite of the tremendous progress in drug development, infectious diseases remain a major public health problem, and the impact of infectious diseases is serious in sub-Saharan African countries 1. There is a great concern about antimicrobials currently in use for the treatment of infectious diseases because of increasing costs of the synthetic anti-microbials, side effects, and rapidly emerging resistance of pathogenic microorganisms to these antimicrobials.
Plant-derived anti-microbials offer a great therapeutic benefit and are less expensive treatment options 2, 5. Natural products such as medicinal plants represent a major reservoir of novel compounds for new drug identification. Several medicinal plant-derived bioactive compounds are used for the management of a variety of chronic disorders and infectious diseases.
The healing effects of medicinal plant extracts had long been discovered and widely used before pathogenic microbes recognized as a causative agent of infectious diseases 2, 3. Bioactive compounds derived from these plants have continued to play crucial roles in the management of many human ailments since ancient times 3. These bioactive substances, which are non-nutritive, have been found to protect the plant from pathogenic microbes. It is, therefore, crucial to screen a variety of medicinal plants for their bioactive substances as the basis for further research into the discovery of new antimicrobials. With technological advances in areas of analytics, metabolomics, biotechnology, proteomics, and genomics it is now possible to isolate and purify numerous bioactive substances from different medicinal plants and used as essential synthetically improved therapeutic agents. Among these bioactive substances are alkaloids, terpenoids, glycosides, flavonoids, phenols, and tannins, which are important raw materials for the production of life- saving antimicrobials 4 - 6.
Today, more than 50% of drugs released into the world market, and a great number of novel pharmaceutical agents derived from natural products and their derivatives that used in herbal medicine 3, 4, 7, 8. Of these drugs, 25% derived from higher plants. Due to this reason, today pharmaceutical companies pay more attention to research on safer bioactive substances isolated from medicinal plants, which have elsewhere been used in traditional folklore medicine with acceptable safety for the development of new drugs with a novel mechanism of action 5, 6.
Globally, between 250,000 to 500,000 species of plants are known of which 35,000 to 50,000 are medicinal plant species. Out of these, 4000 and 6000 have been introduced into the pharmaceutical markets, but only about one hundred medicinal plant species have been used as a source of a drug as shown in Table 1 7.
TABLE 1: MOST COMMONLY CLINICAL USED PLANTS DERIVED DRUGS
| Drug name | Bioactive compound | Name of the plant [family] | Therapeutic use |
| Atropine | Alkaloid | Atropa belladonna [solanaceae] | Abdominal colics, bradyarrhythmias, mydriatics |
| Artemisinin | Sesquiterpene | Artemisia annua [compositae] | Anti-malaria agent |
| Apomorphine | Alkaloid | Papaver somniferum [papaveraceae] | Parkinson's disease |
| Arecoline | Alkaloids | Areca calechu [Polmoae] | Parasympathomimetic |
| Cannabidiol, Dronabinol | Terpenoids | Cannabis sativa [cannabaceae] | Anti-pain, glaucoma |
| Capsaicin | alkaloid | Capsicum annuum [solanaceae] | Anti pain |
| Caffeine | Alkaloid | Camellia sinensis [rubiaceae] | CNS stimulant. |
| Colchicine | Alkaloid | colchicum autumnale [iridaceae] | Anti-gout |
| Camphor | terpenoide | Cinnamomum camphora [Lauraceae] | Rubefacient |
| Camptothecin | Alkaloid | Camptotheca acuminate [nyssaceae] | Anti-cancer |
| Cocaine | Alkaloid | Erytroxylum coca [erythroxylaceae] | Local anesthetic. |
| Coumarin | Coumarin glycosides | Dipteryx odorata [leguminosae] | Anti-coagulants |
| Crofelemer | condensed tannins | Croton lechleri [euphorbiaceae] | Anti diarhea |
| Danthron | anthraquinon | Cassia species | Laxative |
| Digitoxin, digoxin,
digitalin |
Steroidal-glycoside
|
Digitalis purpurea
[plantaginaceae] |
Heart failure, atrial flutter, atrial fibrillation, paroxysmal atrial tachycardia |
| Demecolcine | Colchicum autumnale [iridaceae] | Anti-tumour agent | |
| Elliptinium | Bleekeria vitensis [Apocynaceae] | Anti-tumour agent | |
| Emetine | Alkaloid | Hedera helix [araliaceae)]
Cephaelis ipecacuanha [rubiaceae] |
Anti-amebic , Emesis |
| Ephedrine | Alkaloid | Ephedra sinica [ephedraceae] | Sympathomimetic |
| Etoposide | Lignans | Podophyllum peltatum [berberidaceae] | germ-cell testicular tumours
Small-cell lung carcinoma |
| Galanthamine | Alkaloide | Leucojum oestivum [Amoryllidoceoe] | Anti-Alzheimer's disease. |
| Glycyrrhizin | Triterpenoid | Glycyrrhiza glabra [Leguminosae] | Sweetener, |
| Gossypol | Sesquiterpen | Cotton plant [malvaceae] | Male contraceptive |
| homoharringtonine | cephalotaxine alkaloid | Cephalotaxus harringtonia
[Cephalotaxaceae] |
Anti-cancer |
| Hyoscyamine | Alkaloid | Hyoscyamus niger
[solanaceae] |
Peptic ulcers and GIT disorder
Caused by spasm |
| Irinotecan | Alkaloid | Camptotheca acuminate [nyssaceae] | Colorectal cancer |
| Lanatosides A, B | cardiac glycoside | Digitalis lanata [Scrophulariaceae] | Cardiotonic |
| L-dopa | Alkaloid | Mucuna pruriens [fabaceae] | Anti-parkinson |
| Morphine, codeine
Noscapine |
Alkaloid | Papaver Somniferum [papaveraceae] | Narcotic analgesics |
| mycophenolate | immunosuppression | ||
| galegine | Alkaloid | Galega officinalis [fabaceae] | Type 2 diabetes mellitus |
| Nicotine | Alkaloid | Nicotiana tabacum [solanaceae] | Insecticide |
| Ouabain | Steroid glycosides | Strophanthus gratus [apocynaceae] | Heart failure |
| Paclitaxel | Diterpenoid | Taxus brevifolia [taxaceae] | Ovarian cancer and metastatic
Breast cancer |
| Papavarine | Alkaloid | Papaver somniferum [papaveraceae] | Smooth muscle relaxant |
| Physostigmine | Alkaloid | Physostigma venenosum [leguminosae] | Increases intestinal and bladder motility, Glaucoma, esotropia |
| Podophyllotoxin | Terpenoids | Podophyllum peltatum [berberidaceae] | Wart, anti-cancer |
| Pilocarpine | Alkaloid | Pilocarpus jaborandi [rutaceae] | Glaucoma, xerostomia |
| Picrotoxin | Lactone | Anamirta cocculus [menispermaceae] | Analeptic |
| Psoralen | Furanocoumarin glycosides | Psoralea corylifolia [leguminosae] | |
| Quinidin | Alkaloid | Cinchona succirubra [rubiaceae] | Anti-arrhythmic |
| Quinine | Alkaloid | Cinchona ledgeriana [rubiaceae] | Malaria |
| Reserpine | Alkaloid | Rauvolfia serpentine [apocynaceae] | Anti-hypertensive |
| Rutin | Flavonol glycosides | Fagopyrum esculentum [polygonaceae] | Reduce capillary bleeding |
| Scopolamine | Alkaloid | Datura stramonium [solanaceae] | Motion sickness, antispasmodic Preanaesthetic |
Salicin (aspirin) |
Phenolic glycosides |
Salix alba [salicaceae] | Anti Pain anti inflammatory,
and anti-clotting agent |
| Strychnine | Alkaloid | Strychnos nux-vomica [loganiaceae] | Cns stimulant |
| Tiotropium | Alkaloid | Atropa belladonna [solanaceae] | Anti-asthma |
| Teniposide | polyphenol | Podophyllum peltatum [berberidaceae] | Anti-cancer |
| Theophylline | Alkaloids | Camellia sinensis [theaceae] | Bronchodilator |
| Tetrahydrocannabinl | Terpinoid | Cannabis sativa [cannabaceae] | Anti-emetic, decreases occular tension |
| Tubocurarine | Alkaloid | Strychnos castelnaei [loganiaceae] | Skeletal muscles relaxant |
| Vinblastine vincristine | Alkaloid | Catharanthus roseus [apocynaceae] | Anti-cancer |
| Yohimbine | Alkaloid | Pausinystalia yohimbe [rubiaceae] | Aphrodisiac |
Ethiopia, a tropical country known for its rich flora and fauna biodiversity, is home to more than 7,000 species of higher plants, out of which 12% are indigenous to the country, making it one of the most diverse floristic regions in the world 7, 8. Being a land of diverse climatic and edaphic potentials, more than 800 species of medicinal plants are distributed in different parts of the country. The knowledge and the therapeutic use of these medicinal plants have played a significant role, as an alternative source of medicinal drugs to treat a variety of human and livestock diseases, in supporting the fragile health care system of the country 8, 9.
There are written and oral pharmacopeias about the therapeutic use of medicinal plants in the country as the therapeutic uses of plant-based remedies have been practiced for a very long time. At present, more than 80% of the human population in Ethiopia and 90% of livestock population depend on traditional remedies that mainly (95%) involves the therapeutic use of medicinal plants 10. The demand for medicinal plants increase in the future due to the trust the communities have in the medical values of the medicinal plants, affordability, safety of the plants and the readily available traditional healers in the communities in contrast to the modern orthodox healthcare systems which are expensive, with few trained health care professionals, and limited number of healthcare facilities 9, 10. The scientific researches on medicinal plants in Ethiopia have given evidence-based alternative medicines, which has formed the basis of the herbal drug industry and isolation of drug targets in the pharmaceutical industry 11. In the last few years, several medicinal plants have been evaluated in Ethiopia for their antimicrobial activities against different pathogenic micro-organisms 12-52. Many of them were found to be effective against resistant microbial strains 13, 14, 39, 40, 52. To the best of our knowledge, there are no reviews on medicinal plants with antimicrobial effects in Ethiopia. This review provides up-to-date information on medicinal plants, with highlights on the need for experimental research that will analyze the extracts of these plants for the future development of effective antimicrobial agents.
Methodology:
Systematic Search: A systematic literature review was conducted on pre-reviewed papers on herbal medicines and their phytochemical antimicrobial activity shown on the scientific database; Medline, Pub Med, Science Direct, Springer Link, Embase, Hinari and Google Scholar. To identify the recent studies not published, we also searched abstracts presented at conferences.
The search was restricted to studies with abstracts published in English. Literature Database search was performed using the following keywords; Ethiopia / Ethiopian plants / Ethiopian medicinal plants/Ethiopian anti-microbial plants / Traditional knowledge / Traditional Medicine / Traditional medicinal plants, Medicinal Plants / Medicinal herbs, Plants / Herbal / Medicine / Remedies, Folk Medicine / Folk remedies / Home remedies / Herbal remedies, Ethnobotanical. However, the search excluded the papers on the antimicrobial activity of derivatives from bioactive compounds.
Collection of Studies and Abstraction of Data: The author assessed studies for eligibility and extracted details of the medicinal plants from each article by using a structured abstraction forms, which contains the botanical and local name, the plant part, the solvent used for extraction, activity against microorganism and their minimum inhibitory concentration.
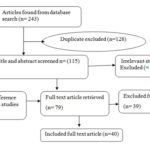
Selection of Articles: A total of 243 articles were obtained from the search. After accounting for duplicates, 128 articles were excluded leaving 115 articles. Of these, 42 articles were further excluded because they did not satisfy the inclusion criteria following review of their abstracts and were therefore considered irrelevant. Six additional articles were however selected by searching the references of the retrieved articles. A total of 79 articles were reviewed in detail but 39 were further excluded after the detailed review because they did not contain the required information. Eventually, only 40 articles were reviewed for this paper.
FIG. 1: FLOW DIAGRAM OF PUBLISHED ARTICLE SEARCH RESULTS
DISCUSSION:
Historical Development of Medicinal Plants: The use of medicinal plants as drug dated back to prehistoric times. The oldest known medical record is a Sumerian clay tablet that mentioned the use of plants about the 3rd millennium BC. In the time of the Egyptian civilization, the therapeutic use of medicinal plants was documented in the Papyrus Ebers in 1500 BC 53. In China Pun-Tsao, a pharmacopeia that was written around 1600, which list thousands of medicinal plants that helped to the works of Shen-Nung, China’s emperor and father of Chinese medicine lived over 4500 years ago 54. Hippocrates (460 - 377 BC), the father of medicine, described 400 medicinal plants. Theophrastus (371-286 BC) who wrote the book Historia Plantarum that mentioned more than 500 medicinal plants (55). Dioscorides (78 AD), a Roman physician wrote: "De Materia Medica” which described more than 600 medicinal plants 56.
Claudius Galen (135 - 201 AD) was a pharmacist in Rome who wrote about 400 works covering all aspects of medicine. He prepares drugs in a complex mixture of more than 30 ingredients called ‘‘galenicals’’. In the fourth century, after the fall of the Roman Empire, the cultural center shifted to the Byzantine Empire, and then to the Arab world 57. Ebne Sina, known in the West as Avicenna (980-1037) a famous scientific figure of the Arabian world who wrote the Canon Medicinae compendium, which describes the therapeutic use of 750 medicines 58. Drugs used before 1800 were prepared with water plant extracts as infusions, tinctures, decoctions, but their chemical structure was unknown. In the nineteenth century, with the emergence of organic chemistry and chemical analysis the isolation of bioactive compounds from plant extract was opened. The first discovery was the isolation of morphine from opium in 1806 by Serturner 57. Between 1820 and 1850, several compounds were isolated and evaluated for medicinal applications. In Paris, Pierre Joseph Pelletier and his collogues isolated emetine from ipecacuanha root and strychnine from the seed of Strychnos nux vomica, quinine sulfate from cinchona bark 57, 58. Chinese researchers identified Artemisinins in 1972 from Artemisia annua L 59.
Determination of Antimicrobial Efficacy of Medicinal Plant Extracts: Anti-microbial susceptibility testing methods are vital tools used to determine the antimicrobial efficacy of medicinal plant extracts against different microorganisms through the determination of the Minimum Inhibitory Concentration (MIC) and minimum bactericidal concentration (MBC) and comparing with available antimicrobial agents as control. Antimicrobial susceptibility testing methods can be categorized into dilution (broth microdilution, broth macro dilution) methods and diffusion (disc diffusion, agar diffusion) methods 60.
Diffusion Methods: These are common and simple methods for determining the antibacterial activity of medicinal plant extracts against microorganisms. In the agar diffusion method, it involves the use of wells cut into the agar plates that have been streaked with a standardized inoculum of bacteria. A known concentration or volume of the extracts is poured into the well and allowed to diffuse out to inhibit bacterial growth after incubating inappropriate atmosphere. The zone of inhibition (in mm) around the well is then measured and estimated as the antibacterial activity of the extracts when compared with the zone of inhibition of a known antibiotic or a standard. The agar diffusion method was replaced by paper discs impregnated with extracts or antibiotics (disc diffusion method), but the lack of standardization of this method resulted in unreliable and non-reproducible results 60, 61. Therefore, disc diffusion method is not a suitable method for determining the antibacterial activity of medicinal plant extracts.
Dilution Methods: Dilution methods are assays used to determine the minimum concentration of antimicrobial agents or extracts that result in either killing (minimum bactericidal concentration, MBC) or inhibition (minimum inhibitory concentration, MIC) of the growth of microorganisms. Dilution tests can be broth microdilution, broth macro dilution (tube dilution), or agar dilution methods. The final volume of the test defines whether the method is macro dilution, i.e. if done using 2 ml tube or microdilution when performed using ≤500 μl per well. In both the agar and broth dilution techniques, the lowest amount of the antimicrobial agent or extract that inhibits the visible growth of microorganisms under known conditions is termed the MIC. The advantages of dilution over diffusion methods include increased sensitivity for smaller extracts, quantitative analysis, and differentiation of bacteriostatic and bactericidal effects of the extracts or anti-microbial agents. Dilution methods considered the reference methods for in-vitro susceptibility testing and used to evaluate the performance of other techniques 60-62.
TABLE 2: ETHIOPIAN MEDICINAL PLANT EXTRACTS WITH THEIR MINIMUM INHIBITORY CONCENTRATION (MIC) AGAINST DIFFERENT MICROORGANISMS
| Botanical name [Family] | local name
(Parts used) |
Extract | MIC | Activity Against microorganism | Ref |
| Curcuma longa [Zingiberaceae] | ERD
(Rhizome) |
Oil | >25 mg/ml | E. coli | [12] |
| 25 mg/ml | S. aureus, P. aeruginosa | ||||
| Aframomum corrorima
[Zingiberaceae] |
Kororima
(Fruit) |
Ac | >25 mg/ml | S. aureus, E. coli, | |
| 25 mg/ml | P. aeruginosa | ||||
| Asparagus africanus [Asparagaceae] | Yefurded
(Leaf) |
ETOH | .512 mg/ml | E. faecalis | [13] |
| Guizoti aschimperi [Asteraceae] | Mocho (Leaf) | ETOH | .128 mg/ml | S. aureus, E. Faecalis | |
| .512 mg/ml | C. albicans | ||||
| Lippia adoensis [Verbenaceae] | Kessie
(Leaf) |
ETOH | .256 mg/ml | S. aureus, E. faecalis | |
| .512 mg/ml | C. albicans | ||||
| Premna schimperi [Lamiaceae] | Teqoqi
(Leaf) |
ETOH | .512 mg/ml | S. aureus, E. faecalis | |
| Rhamnus prinoides
[Rhamnaceae] |
Gesho
(Leaf) |
MEOH | 2.03 mg/ml | S. aureus | [14] |
| 4.06 mg/ml | S. pyogen, S. typhi | ||||
| 8.13 mg/ml | P. aeruginosa, E. coli, | ||||
| Ranunculus multifidus
[Ranunculaceae] |
Gubducaffee (Leaf) | MEOH
CHCl3 |
16.5 mg/ml | P. aeruginosa, S. typhi,
E. coli, S. aureus |
[15] |
| Leucasmartinicensis [Lamiaceae] | Mata burusa (Leaf) | MEOH | 16.5 mg/ml | S. typhi, E. coli,
P. aeruginosa |
|
| CHCl3 | 5.6 mg/ml | S. aureus | |||
| Kosteletzkya begonifolia [Malvaceae] | Ingiccee (Leaf) | MeOH
CHCl3 |
16.5 mg/ml | S. typhi , P. aeruginosa | |
| 5.6 mg/ml | S. aureus | ||||
| Achyranthes Aspera
[Amaranthaceae] |
Elenj/Qaytelenj
(Leaf) |
MEOH | 15.6 mg/ml | S.aureus, P. aeuruginosa, E. coli, P. vulgaris | [16] |
| 7.81 mg/ml | S. pyogens | ||||
| Bruceaanti dysenterica
[Simaroubaceae] |
Waginos/YedegaAbalo
(Root) |
MEOH | 15.6 mg/ml | S. aureus, E. coli | |
| 7.81 mg/ml | S. pyogens | ||||
| 62.5 mg/ml | P. aeuruginosa | ||||
| Phytolaca Dodecandra
[Phytolaccaceae] |
Indodi
(Root) |
MEOH | 1.96 mg/ml | S. pyogens | |
| 15.6 mg/ml | P. aeuruginosa | ||||
| Solanum incanum
[Solanaceae] |
Inbuay
(Leaf) |
MEOH | 31.3 mg/ml | S. aureus | |
| 62.5 mg/ml | S. pyogens | ||||
| 125 mg/ml | P. aeuruginosa | ||||
| Rumex nepalensis [Polygonaceae] | Tult
(Leaf) |
Ac | 3.13 mg/ml | E. coli, S. typhi | [17]
|
| 6.26 mg/ml | S. aureus | ||||
| Phytolac adodecandra [Phytolaccaceae] | Indodi
(Fruits) |
Ac | 12.5 mg/ml | E. coli, S. typhi | |
| 25.1 mg/ml | S. aureus , P. aeruginosa | ||||
| Grewia ferruginea [Tiliaceae] | Lenkoata (Bark) | Ac | 3.13 mg/ml | E. coli | |
| 25.1 mg/ml | S. aureus, P. aeruginosa | ||||
| 12.5 mg/ml | S. typhi | ||||
| Aloe vera [Xanthorrhaeceae] | ERET
(Aerial part) |
MEOH | .098 mg/ml | S. typhi, S. agalactia | [18]
|
| .02 mg/ml | E. coli | ||||
| .42 mg/ml | S. aureus | ||||
| .10 mg/ml | S. agalactia | ||||
| Zingiber officinale [Zingiberaceae] | Jinjible (Rhizome) | MEOH | .093 mg/ml | S. aureus | |
| .05 mg/ml | S. typhi | ||||
| .16 mg/ml | E. coli | ||||
| .19 mg/ml | S. agalactia | ||||
| Allium ursinium
[Amaryllidaceae] |
Yejibshinkurt
(Bulb) |
MEOH | 250 mg/ml | M. tuberculosis | [19] |
| Pterolobium stellatum
[Fabaceae] |
Kentefa (Leaf) | MEOH | 250 mg/ml | M. tuberculosis | |
| Dodonaea anguistifolia [Sapindaceae] | Kitkita (Leaf) | MEOH | 12.5 mg/ml | M. tuberculosis | |
| Calpurnia aurea [Fabaceae] | Digita (Leaf) | MEOH | .063 mg/ml | S. aureus, P. aeruginosa, E. coli. | [20] |
| Croton macrostachyus [Euphorbiaceae] | Bissana (Leaf) | MEOH CHCl3 | 7.5 mg/ml | E. coli | [21] |
| 3.75 mg/ml | S. aureus | ||||
| Lippia adoensis
[Verbenaceae] |
Kesse / Koseret (Leaf) | MEOH CHCl3 | 2.5 mg/ml | S. aueres, P. aeroginosa, S. typhi | [22]
|
| 10 mg/ml | E. coli | ||||
| Acokanthera schimperi
[Apocynaceae] |
Merztekil
(Leaf) |
MEOH | 7.81 mg/ml | S. pyogens | [16] |
| 15.6 mg/ml | E. coli, P. aeuruginosa P. vulgaris, S. aureus | ||||
| Brucea Antidysenterica
[Simaroubaceae] |
Waginos/YedegaAbalo
(Root) |
MEOH | 15.6 mg/ml | S. aureus | |
| 7.8 mg/ml | S. pyogens | ||||
| 62.5 mg/ml | P. aeuruginosa | ||||
| Phytolacca Dodecandra
[Phytolaccaceae] |
Endodi
(Root) |
MEOH | 1.95 mg/ml | S. pyogens | |
| 15.6 mg/ml | P. aeuruginosa | ||||
| Solanum incanum
[Solanaceae] |
Imbuay
(Leaf) |
MEOH | 31.3 mg/ml | S. aureus | |
| 62.5 mg/ml | S. pyogens | ||||
| 125 mg/ml | P. aeuruginosa | ||||
| Verbena officinalis
[Verbenaceae] |
Atuch
(Leaf) |
MEOH | 31.3 mg/ml | S. aureus | [22] |
| 7.81 mg/ml | S. pneumoniae | ||||
| 250 mg/ml | E. coli | ||||
| Hypericum revolutum
[Hypericaceae] |
Amija
(Leaf) |
MEOH | 125mg/ml | Shigella | [23] |
| 250 mg/ml | P. aeuroginosa | ||||
| Peterollobium stellatum
|
Kentefa
(Roots) |
MEOH | 250 mg/ml | S. typhi, S. aureus | |
| 125 mg/ml | E. coli | ||||
| Thymus serrulatus [Lamiaceae] | Tosign
(Leaf ) |
ETOH | 4 mg/ml | B. cereus, S. aureus | [24] |
| 2 mg/ml | E. coli | ||||
| Terminalia brownii [Combretaceae] | Weba
(Leaf) |
ETOH | 4 mg/ml | S. typhi | |
| Oliniaro chetiana
[Penaeaceae] |
Tife
(Leaf) |
MEOH | 5 mg/ml | S. aureus | [25] |
| 2.5 mg/ml | P. aeruginosa | ||||
| 10 mg/ml | E. coli, C. albicans | ||||
| Calpurinia aurea [Fabaceae] | Digita
(Leaf) |
MEOH | 25 mg/ml | S. aureus | [26] |
| 100 mg/ml | S. dysgalactiae, D. congolensis | ||||
| Croton macrostachyus
[Euphorbiaceae] |
Bissana
(Leaf) |
MEOH | 12.5 mg/ml | S. aureus | |
| 25 mg/ml | S. dysgalactiae, D. congolensis | ||||
| Nicotiana tabacum [Solanaceae] | Zeka-keba
(Leaf) |
MEOH | 25 mg/ml | S. aureus, S. dysgalactiae | |
| 50 mg/ml | S. agalactia, D. congolensis | ||||
| Achyranthes aspera
[Asparagaceae] |
Leaf
(Mechalo) |
MEOH | 6.25 mg/ml | E. coli, S. hycus, S. aureus | |
| 50 mg/ml | K. pneumonae, S. intermedi | ||||
| Salvia schimperi [Lamiaceae] | Leaf
(Yahiya Joro) |
MEOH | 31.3mg/ml | S. typhi, S.aureus | [38] |
| 62.5mg/ml | S. paratyphi, Shigella | ||||
| 125 mg/ml | E. coli, P. aeuroginosa | ||||
| Foenculum vulgare
[Apiaceae] |
Leaf
(Insilal) |
Aqueous extract | .01mg/l | P. gallinarum, S. agalactae | [39] |
| 0.1 mg/ml | M. haemolyticum | ||||
| Lyonitesocymifolia [Lamiaceae] | Aerial part
(Raskimir) |
MEOH | 0.1 mg/ml | P. gallinarum | |
| 1 mg/ml | M. haemolyticum, S. typhi | ||||
| Jasminum abysinicum
[Oleaceae] |
Leaf
(Tembelel) |
Aqueous extract | 0.1 mg/ml | P. gallinarum, S. gallinarum, M. haemolyticum, S. agalactae | |
| 0.01 mg/ml | S. typhimurium | ||||
| Myrsine Africana
[Myrsinaceae] |
Leaf
(Kechemo) |
MEOH | 0.1 mg/ml | P. gallinarum, S. agalactae | |
| 0.01 mg/ml | S. aureus | ||||
| Moring astenopetala [Moringaceae] | Shiferaw
(Seeds) |
MEOH | 1.25 mg/ml | S. aureus | [40] |
| 10 mg/ml | E. coli | ||||
| 5 mg/m | S. boydii | ||||
| Oliniaro chetiana
[Oliniaceae] |
TIFE
(Leaf) |
MEOH | 2.5 g/ml | P. aeuroginosac, T. mentagrophytes | [26]
|
| 5 g/ml | S. aureus | ||||
| 10 g/ml | E. coli, Candida albica | ||||
| Combretum molle
[Combretaceae] |
Yekolaavalo
(Stem bark) |
MEOH | 0.1 mgml | E. coli, V. cholerae | [41] |
| 0.2 mg/ml | S. typhi, B. pumilus | ||||
| 0.05 mg/ml | Shigella dysentery | ||||
| 0.8 mg/ml | C.albicans | ||||
| 1.5 mg/ml | P. notatum, P. citrinum | ||||
| Albizia gummifera [Fabaceae] | Seed | Butanol | .125 mg/ml | N. gonorrhea | [42] |
| Achyranthes aspera
[Asparagaceae] |
Leaf
(Mechalo) |
MEOH | 12.3 mg/ml | E. coli, P. aeruginosa, S. aureus | [43] |
| 6.26 mg/ml | S. boydii, S. typhi | ||||
| Aloe elegans
[Aloeacea] |
Leaf | ETOH | 1.56 mg/ml | E. coli, S. aureus | [44] |
| 6.25 mg/ml | P. aeruginosa, C. albicans | ||||
| 12.5 mg/ml | K. pneumonia, S. typhi | ||||
| Lepidium sativum [Brassicaceae] | Seed
(Fetto) |
ETOH | 6.5 mg/ml | E. coli | [45] |
| 12.5 mg/ml | P. aeruginosa, | ||||
| Ocimum lamiifolium
[lamiaceae] |
Leaf
(Damakesse) |
Aqueous extract | 10 mg/ml | E. coli, P. aeruginosa, S. aureus, S. boydii | [46] |
| Taverniera abyssinica [Fabaceae] | Root (Dingetegna) | MEOH | 12.5 mg/ml | S. aureus, E. faecalis, A. flavus | [47] |
| 6.5 mg/ml | C. albicans | ||||
| Rosmarinus officinalis
[Lamiaceae] |
Leaf
(Sigametibesha) |
MEOH | 15.8 mg/ml | S. epidermidis | [48] |
| 31.3 mg/ml | S. pyogenes, E. coli, S. typhi,
S. aureus |
||||
| Eucalyptus globules
[Myrtaceae] |
Leaf
(Nechbahirzaf) |
MEOH | 15.8 mg/m | S. epidermidis, S. aureus, | |
| 20.6 mg/ml | S. pyogene, Shigella, E.coli | ||||
| Vernonia Auriculifera
[Compositae] |
Leaf
(Grawa) |
CHCl3
MEOH |
200 mg/ml | S. aureus |
[49] |
| Verbena officinalis
[Verbenaceae] |
Leaf
(Atuch) |
MEOH | 7.8 mg/ml | S. boydii | [24] |
| 15.8 mg/ml | S. aureus | ||||
| 3.9 mg/ml | S. pneumonia | ||||
| 62.5 mg/ml | MRSA | ||||
| Punica Granatum
[Punicaceae] |
Leaf
(Yeromanzaf) |
MEOH | 6.25 mg/ml | S. aureus, S. pneumonia, Shigella | [50] |
| 12.5 mg/ml | MRSA, E. coli, C. albican | ||||
| Plectranthus caninus
[Lamiaceae] |
Aerial part
(Endifdif) |
Essential oil | 0.03 mg/ml | E. coli, S. typhi, Shigella, S. aureus, V. cholera, B. pumilus | [51] |
| Aloe sinana
[Xanthorrhaeceae] |
Latex
(Rate ) |
MEOH | 5 μg/ml | Shigella boydii | [52] |
| 10 μg/ml | E. coli, V. cholera, S. typhi | ||||
| 25 μg/ml | S. aureus, S. enterica | ||||
| 0.8 mg/ml | A. niger, C. albica | ||||
| 1.5 mg/ml | P. funiculosum, P. notatum |
Abbreviation: Methanol: MEOH, ethanol: ETOH, CHCl3: Chloroform, PE: Petroleum ether, Ac: Acetone Methicillin resistant Staphylococcus aureus, MRSA
TABLE 3: MEDICINAL PLANT EXTRACTS WITH THEIR CONCENTRATION ACTIVE AGAINST DIFFERENT MICRO ORGANISM
| Botanical name
[Family] |
local name
(Parts used) |
Extract | Extract concentration | Activity against microorganism | Ref |
| Clematis simensis
[Ranunculacae] |
Azohareg
(Leaf) |
MEOH | 5 mg/ml | S. aureus, E. coli, S. typhi, P. aeruginosa | [28] |
| PE | 5 mg/ml | E. coli, S. typhi | |||
| Ruta graveolens
[rutaceae] |
Tena Adam
(Leaf) |
CHCl3 MEOH | 5 mg/ml | S. aureus, B. subtilis,
E. coli, P. aeruginosa |
[29] |
| Syzygeum guineense [Myrtaceae] | Dokma (Leaf) | MEOH | .25 mg/ml | N. gonorrhea, E.coli | [30]
|
| Laggerato mentosa
[Compositae] |
Keskeso
(Areal part) |
MEOH | 2 mg/ml | N. gonorrhea, S. pyogenous, S. pneumonia | |
| Artemisia abyssinica
[Compositae] |
Chikugn
(Areal) |
ETOH/
PE |
2 mg/ml | B. cereus, N. gonorrhea,
S. aureus |
|
| Dovyalis abyssinica
[Flacourtiaceae] |
Koshim
(Leaf) |
MEOH | 2 mg/ml | B. cereus, N. gonorrhea,
S. aureus, Shigella |
|
| Warburgia ugandensis [Canaleaceae] | Bifti
(Leaf/stem bark) |
MEOH | 2 mg/ml | B. cereus, N. gonorrhea,
S. aureus |
|
| Syzygeum guineense
[Myrtaceae] |
Dokma
(Leaf) |
MEOH | 1 mg/ml | N. gonorrhea, S. aureus S. pyogene S. pneumonia | |
| Bersama abyssinica
[Melianthaceae] |
Azamir
(Root bark) |
MEOH | 1 mg/ml | B. cereus, Shigella | |
| Ferula communis
[Umbelliferae] |
Doge
(Root) |
MEOH | 1 mg/ml | N. gonorrhea, S. aureus S. pyogene, S. pneumonia | |
| Gardenia lutea
[Rubiaceae] |
Gambilo
(Stem bark) |
MEOH | 2 mg/ml | B. cereus, N. gonorrhea,
S. aureus, Shigella |
|
| Myricasalci folia
[Myricaceae] |
Shinet
( Leaf) |
MEOH | 1 mg/ml | B. cereus, N. gonorhea
S. aureus, Shigella |
|
| Discopodiu peninervum
[Solanaceae] |
Ameraro
(Leaf) |
MEOH | 2 mg/ml | N. gonorrhea, S. aureus S. pyogene, S. pneumonia | |
| Olea europea
[Oleaceae] |
Woyera
(Stem bark) |
MEOH | 1 mg/ml | N. gonorrhoea, S. aureus S. pyogene, S. pneumonia | |
| Trichila emetica
[Melianthaceae] |
Roka (Fruit) | MEOH | 2 mg/ml | B. cereus, N. gonorrhea,
S. aureus, S. typhi |
|
| Combertum molle
[Combretaceae] |
Yekolaabola
(Leaf) |
ETOH | 1 mg/ml | B. cereus N. gonorrhea,
S. aureus, S. typhi |
|
| Maesa lanceolata
[Myrsinaceae] |
Kelewo (Fruits) | MEOH | 250 mg/ml | S. aureus, E. coli | [31]
|
| Cissus quadrangularis
[Vitaceae] |
Yezhonanjent (Aerial part) | MEOH | 250 mg/ml | S. aureus | |
| Dodonae angustofolia
[Sapindaceae] |
Kitkita
(Leaf) |
MEOH | 250 mg/ml | S. aureus, E. coli | |
| Artemisia afra
[Asteraceae] |
Ariti
(Leaf) |
MEOH PE | 100mg/ml | S. aureus , E. coli, B. subtilis, shigella | [32]
|
| Nicotia natabacum
[Solanaceae] |
Zeka-keba
(Leaf) |
MEOH CHCl3 | 50 mg/ml | E. coli, S. aureus,
P. aeruginosa |
[20]
|
| Croton macrostachyus
[Euphorbiaceae] |
Bissana
(Leaf) |
MEOH CHCl3 | 50 mg/ml | S. aureus E. coli,
P. aeruginosa |
|
| Calpurnia aurea
[Fabaceae] |
Digita
(Leaf) |
MEOH CHCl3 | 50 mg/ml | S. aureus, E. coli,
P. aeruginosa |
|
| Withania somnifera
[Solanaceae] |
Giziewa
(Root) |
MEOH CHCl3 | 50 mg/ml | S.aureus, E. coli, P. aeruginosa | |
| Datura stramonium
[Solanaceae] |
Astenagir
(Leaf) |
ETOH & Ac | 20 mg/ml
|
S. aureus, B. subtilis, S. typi, E. coli, F. solani, F. oxysporum, A. niger | [33] |
| Commicarpus pedenculosus
[Nyctaginaceae] |
Tihuantila
(Leaf) |
MEOH | 2.5 mg/ml | S. aureus | [34] |
| Rosmarinus officinalis
[Lamiaceae] |
YetibsKitel (Leaf) | Oil extract | 10 μl | S. aureus, E. coli,
F. oxysporum |
[35] |
| Eculaptus Globulus
[Myrtaceae] |
nechbahirzaf
(leaf) |
Oil extract | 20 μl
|
S. typhi, E. coli, B. subtilis S. aureus | [36] |
| Hagenia abyssinica
[Leucosidea] |
Kosso
(Flower, Fruit) |
MEOH
ETOH |
50 mg/ml | S. aureus, S. typhi | [37] |
| Combertum molle
[Combretaceae] |
Yekolaavalo
(Bark) |
An | 50 mg/ml | E. coli, Shigella,
C. albican |
[38] |
| Acokanthera schimperi
[Apocynaceae] |
Mrenz
(Leaf) |
MEOH | 25 mg/ml | S. aureus, P. aeruginosa,
T. mentagrophytes |
[26] |
| Kalanchoe petitiana
[Crassulaceae] |
Endohahila
(Leaf) |
MEOH | 25 mg/ml | S. aureus, P. aeruginosa,
E. coli |
|
| Malva parviflora
[Malvaceae] |
Lit
(Root) |
MEOH | 25 mg/ml | T. mentagrophytes,
S. aureus |
|
| Verbascumsinaiticum
[Scrophulariaceae] |
Ketetina
(Leaf) |
MEOH | 25 mg/ml | S. aureus, P. aeruginosa |
Abbreviation: Methanol: MEOH, ethanol: ETOH, CHCl3: Chloroform, PE: Petroleum ether, Ac: Acetone Methicillin-resistant Staphylococcus aureus: MRSA
Anti-microbial Studies of Medicinal Plant Extracts in Ethiopia: Several in-vitro antimicrobial studies on crude extracts of medicinal plant species have been done in Ethiopia with promising results against pathogenic micro-organisms. Approximately 91 medicinal plants in Ethiopia, distributed in 49 families, showed important anti-microbial activity in Table 1 and 2. From these medicinal plant families, the most common representatives are Lamiaceae, Solanaceae, Fabaceae, Compositae, and Myrtaceae, which contain nine, six, four, three, and three plant species respectively. About 109 extracts belonging to these medicinal plants have been evaluated for their antimicrobial effect against 27 species of bacteria and 11 species of fungi. The findings showed that methanol extracts of Guizoti aschimperi 13, Combretum molle 14, Foenculum vulgare 39, Myrsine Africana 40 and Aloe sinana 52, showed the highest growth inhibition against the microorganism evaluated with the anti-microbial activity ranged between 5 μg/ml and 250 mg/ml.
Phytochemical Compounds from Medicinal Plants with Anti-microbial Actions: Phytochemical compounds are bioactive substances found in medicinal plants. Phytochemical screening is a simple and cheap procedure that gives a rapid response to the different types of phytochemicals in a mixture and is a vital tool in identifying bioactive compounds of medicinal and industrial value. The antimicrobial effect of these medicinal plants is attributed to the presence of several classes of bioactive compounds such as quinines, phytoalexins, phenols, alkaloids, flavonoids, terpenoids, tannins, glucosinolates and pathogenesis-related proteins 49, 50.
Alkaloids are heterocyclic nitrogen bioactive compounds synthesized from amino acid building blocks that exert their antimicrobial effect due to their ability to intercalate with DNA thereby interfering with cell division, and also by disrupting cell membranes leading to increase membrane permeability in the bacteria. Morphine, quinine, caffeine, strychnine, narcotine, and emetin are well-known alkaloids that have medicinal value. Besides, berberine, piperine, palmatine, and tetrahydropalmatine are examples of alkaloids with antimicrobial, anthelmintic and anti-diarrhoeal activity 63. Phenols are bioactive compounds that have one or more unsaturated rings with one or more hydroxyl groups, responsible for the color of fruits of plants and as well as defense against pathogenic microorganisms. Phenol acid exerts its antimicrobial activity by diffusion across the membrane, leading to acidification of the cytoplasm and also cause a reduction in adherence of organisms to the cell lining. Catechol, epicatechin and cinnamic acid are examples of phenol with antimicrobial activity 64.
Flavonoids are another group of phenolic compounds that contain more than one benzene ring in its structure, provide colors attractive to plant pollinators, and are well known for their antioxidant or free radical scavenging activities. Flavonoids have antimicrobial, antiviral, anti-allergic, and anti-inflammatory properties. Their antimicrobial activity is through inhibition of bacterial virulence factors such as toxins, quorum-sensing signal receptors and enzymes, destabilization, and permeabilization of the cytoplasmic membrane, inhibition of extracellular microbial enzymes and deprivation of the substrates required for microbial growth such as iron and zinc. Quercetin, rutincatechin, chrysin, kaempferol, flavones, and calchones are some of the other important flavonoids with anti-microbial activity 64, 65.
Anthraquinones are aromatic compounds with a 9, 10-dioxoanthracene. The antimicrobial activity of Anthraquinones occurs through different mechanisms including destabilization of the cell wall and alterations of metabolic pathways. For example, emodin disrupts cell wall and membrane, intercalates into DNA base pairs and inhibits the activity important enzymes, which generally affect transcription, repress expression and cause cell death. Aloe-emodin, hypericin, 1, 8-dihydroxyanthraquinone, chrysophanol, emodin, physcion, and rhein are some of the Anthraquinones with antimicrobial activity 63, 66. Terpenoids are bioactive compounds that confer characteristic smell on plants. They are effective against bacteria, fungi, viruses, and protozoa. Terpenoids exert their antimicrobial activity by disturbing the cell membrane, disrupting the proton motive force and coagulation of cell contents. Typical examples include menthol, citral, camphor, and salvinorin 64, 67.
The essential oil contains about 500 different compounds like terpenoids, aliphatic and aromatic compounds such as aldehydes and phenols. Essential oils have a hydrophobic property that disturbs cell membrane, electron flow, and coagulation of cell contents, which are the possible mechanisms of their antimicrobial activity. Carvacrol, eugenol, and thymol are some examples of essential oil with anti-microbial activity 68. Tannin is a polyphenolic bioactive compound with the astringent effect that helps to protect against a variety of illnesses by enhancing the immune system through activation of phagocytic cells.
Tannin also exerts its effect by inhibiting microbial essential enzymes, deprivation of the substrates needed for microbial growth, and inhibition of oxidative phosphorylation, which affects microbial metabolism. Condensed tannins bind to the cell wall of bacteria thereby inducing bacterial stasis and protease activity.
Ellagitannin is a typical tannin with antimicrobial activity 69. Saponins are bioactive compounds derived from triterpenoid glycosides with ‘soap like’ property in water. It is a poisonous compound that causes hemolysis of red blood cells. Saponins exert antimicrobial activity by altering the permeability of cell walls to elicit changes in cell morphology leading to cell lysis, as well as immunostimulant activity 70. Glucosinolates are bioactive compounds that consist of sulfur and nitrogen. Glucosinolates such as glucoiberverine, sinigrin, glucoiberin, glucoerucin, and isothiocyanates have significant antifungal, anticancer antimicrobial, antioxidant and anti-inflammatory activity 70, 71. Anti-microbial peptides are used as host defenses against different micro-organisms. Anti-microbial peptides are more potent than conventional antibiotics because they have different targets and mechanisms of actions.
They inhibit the growth of bacteria by interacting with cellular membranes. The cationic amino acids bind to negatively charged compounds such as teichoic acid, phospholipids, and lipo-polysaccharide. This leads to membrane destabilization and leakage of cytoplasmic contents, loss of membrane potential, the entry of peptides and the occlusion of anionic cell components or the activation of autolytic enzymes.
Thionins, hevein-like peptides, defensins, lipid transfer proteins, α-hairpinin families, snakins, knottin-type peptides, and puroindolines are examples of antimicrobial peptide families of plant origin that are classified based on their Cys motifs, sequence similarity and distinctive disulfide bond patterns 63, 71.
CONCLUSION: Medicinal plants have provided important sources of antimicrobial drugs. This review reveals that in Ethiopia, medicinal plants from over 90 species extracted with different solvents showed promising anti-microbial activity. The traditional uses of medicinal plants for the management of microbial infections in Ethiopia have been supported by in-vitro and in-vivo studies. Based on the anti-microbial assay from this review, Staphylococcus aureus was found to be more susceptible to the plant extracts than Escherichia coli or Pseudomonas aeruginosa.
ACKNOWLEDGEMENT: The author acknowledges the School of Pharmacy, University of Gondar, for providing necessary resources.
CONFLICT OF INTEREST: No conflict of interest is declared.
REFERENCES:
- Ventola CL: The anti-biotic resistance crisis: Part 1: causes and threats. Pharm Therapeutic 2015; 40(4): 277.
- Rios JL and Recio MC: Medicinal plants and anti-microbial activity. J of Ethno 2005; 100(1): 80-4.
- Briskin DP: Medicinal plants and phytomedicines. Linking plant biochemistry and physiology to human health. Plant Physiology 2000; 124(2): 507-14.
- Azmir J, Zaidul IS and Rahman MM: Techniques for extraction of bioactive compounds from plant materials: a review. J of Food Engineering 2013; 117(4): 426-36.
- Suffness MA and Douros JO: Drugs of plant origin. Methods in Cancer Research 1979; 16(1): 73-126.
- Bahar M, Deng Y, Fletcher JN and Kinghorn AD: Plant-derived natural products in drug discovery and development: an overview. Selected Topics in the Chemistry of Natural Products 2007; 31(1): 11-16.
- Teklehaymanot T and Giday M: Ethnobotanical study of medicinal plants used by people in Zegie Peninsula, North western Ethiopia. Journal of Ethnobiology and Ethnomedicine 2007; 3(1): 12.
- Abdullahi AA: Trends and challenges of traditional medicine in Africa. African J of Trad Complementary and Alternative Medicines 2011; 8(5): 13-18.
- Farnsworth NR and Soejarto DD: Global importance of medicinal plants. The Con of Medi Pla 1991; 7(1): 25-51.
- Yineger H and Yewhalaw D: Traditional medicinal plant knowledge and use by local healers in Sekoru District, Jimma Zone, South western Ethiopia. Journal of Ethnobiology and Ethnomedicine 2007; 3(1): 24.
- Fahey JW: Moringa oleifera: a review of the medical evidence for its nutritional, therapeutic and prophylactic properties. Part 1. Trees for Life Journal 2005; 1(5): 1-5.
- Bacha K, Tariku Y and Gebreyesus G: Anti-microbial and anti-quorum sensing activities of selected medicinal plants of Ethiopia: Implication for development of potent anti-microbial agents. BMC Microbio 2016; 16(1): 139.
- Teka A, Rondevaldova J and Asfaw Z: In-vitro anti-microbial activity of plants used in traditional medicine in Gurage and Silti Zones, south central Ethiopia. BMC Compl and Alternative Medicine 2015; 15(1): 286.
- Molla Y, Nedi T and Tadesse G: Evaluation of the in-vitro anti-bacterial activity of the solvent fractions of the leaves of Rhamnus prinoides L. Herit (Rhamnaceae) against pathogenic bacteria. BMC Complementary and Alternative Medicine 2016; 16(1): 287.
- Begashawu T, Tariku Y and Bacha K: Anti-bacterial activity of selected medicinal plants used in South-western Ethiopia. African Journal of Microbiology Research 2016; 10(46): 1961-72.
- Taye B, Giday M, Animut A and Seid J: Anti-bacterial activities of selected medicinal plants in traditional treatment of human wounds in Ethiopia. Asian Pacific Journal of Tropical Biomedicine 2011; 1(5): 370-75.
- Tura GT, Eshete WB and Tucho GT: Anti-bacterial efficacy of local plants and their contribution to public health in rural Ethiopia. Anti-microbial Resistance & Infection Control 2017; 6(1): 76.
- Redda YT, Kebede E and Cruz GG: Potential anti-bacterial activity of crude extracts from aloe vera, Zingiber officinale and vinca major medicinal plants. Int J 2014; 5(3): 202-07.
- Balcha E, Mengiste B and Gebrelibanos M: Evaluation of in-vitro anti-mycobacterial activity of selected medicinal plants in Mekelle, Ethiopia. World Appl Sci J 2014; 31 (6): 1217-20.
- Romha G, Admasu B and Hiwot GT: Anti-bacterial activities of five medicinal plants in Ethiopia against some human and animal pathogens. Evidence-Based Complementary and Alternative Medi 2018; 31(6): 17-20.
- Aylate AL, Agize MA and Ekero DE: In-vitro and in-vivo anti-bacterial activities of Croton macrostachyus methanol extract against E. coli and S. aureus A Adv Anim Vet Sci 2017; 171: 162-71.
- Wasihun Y, Adraro T and Ali S: Evaluation of anti-bacterial activity and phytochemical constituents of leaf extract of Lippia adoensis. APJEE 2014; 1(1): 45-53.
- Sahile S, Habtamu M and Chandrodyam S: Anti-bacterial activity of Verbena officinalis (Linn.) Crude Extract against Some Human Pathogens. Int J Pure App Biosci 2015; 3(6): 34-41.
- Andualem G, Umar S and Getnet F: Anti-microbial and phytochemical screening of methanol extracts of three medicinal plants in Ethiopia. Advan Biol Res 2014; 8: 101-06.
- Mechesso AF, Asrade B, Hailu H and Toma A: In-vitro anti-microbial activity of three medicinal plants of Ethiopia against some selected bacterial isolates. Afr Journal of Microbiol Res 2016; 10(42): 1779-83.
- Kalayou S, Haileselassie M and Gebre-egziabher G: In-vitro anti-microbial activity screening of some ethnoveterinary medicinal plants traditionally used against mastitis, wound and gastrointestinal tract complication in Tigray Region, Ethiopia. Asian Pac J Trop Biomed 2012; 2(7): 516-22.
- Teshome S and Teshale C: Preliminary phytochemical screening and evaluation of the anti-bacterial activity of the gradient extracts of leaves of Clematis simensis. Int J Biotechnol Res 2013; 1(8): 116-19.
- Amabye TG: Phytochemical screening and evaluation of anti-bacterial activity of Ruta graveolens L. a medicinal plant grown around Mekelle, Tigray, Ethiopia. Natural Products Chemistry and Research 2015; 4(2): 87-91.
- Geyid A, Abebe D and Debella A: Screening of some medicinal plants of Ethiopia for their anti-microbial properties and chemical profiles. J Ethno 2005; 97(3): 421-27.
- Mengiste B, Hagos Y, Moges F, Tassew H, Tadesse G and Teklu A: In-vitro anti-bacterial screening of extracts from selected Ethiopian medicinal plants. Momona Ethiopian Journal of Science 2014; 6(1): 102-10.
- Keshebo DL, Washe AP and Alemu F: Determination of anti-microbial and anti-oxidant activities of extracts from selected medicinal plants. American Scientific Research Journal for Engineering, Technology and Sciences (ASRJETS) 2016; 16(1): 212-22.
- Girmay S: Preliminary phytochemical screening and in-vitro anti-microbial activity of Datura stramonium leaves extracts collected from Eastern Ethiopia. Int Res J Biol Sci 2015; 4(1): 55-59.
- Tadele T, Henok W and Fekadu R: A study on in-vitro anti-microbial effects of some selected plants on Staphylococcus aureus isolated from bovine clinical mastitis. Int J of Veterinary Medicine 2010; 8(1): 54-60.
- Asressu KH and Tesema TK: Chemical and Anti-microbial investigations on essential oil of Rosmarinus officinalis leaves grown in Ethiopia and comparison with other countries. J Appl Pharm 2014; 6(1): 132-42.
- Amabye TG, Bezabh AF and Mekonen F: Phytochemical and anti-microbial potentials leaves extract of Eucalyptus globulus oil from Maichew Tigray Ethiopia. Int J of Compl and Alternative Medicine 2016; 2(3): 56-59.
- Wolde T, Behailu BTH and Tiruha K: Phytochemical analysis and anti-microbial activity of Hagenia abyssinica. Ind J Pharm Pharmacol 2016; 3(3): 127-134.
- Asres K, Mazumder Ab and Bucar F: Anti-bacterial and anti-fungal activities of extracts of Combretum molle. Ethiop Med J 2006; 44(3): 269.
- Umer S, Andualem G and Getnet F: Anti-diarrheal and anti-bacterial activities of hydroalcoholic extracts of Salvia schimperi benth from Ethiopia. Int J Pharmacogn 2015; 2: 290-95.
- Yitberek H, Tedesse E and Alegne W: In-vitro anti-microbial activity of selected Ethiopian medicinal plants against some important bacteria of veterinary importance. Afr J Microbiol Res 2010; 4: 1230-34.
- Chekesa B and Mekonnen Y: Anti-bacterial activity of Moringa stenopetala against some human pathogenic bacterial strains. Science, Technology and Arts Research Journal 2015; 4 (2): 190-198.
- Tefera M, Geyid A and Debella A: In-vitro anti-Neisseria gonorrhoeae activity of Albizia gummifera and Croton macrostachyus revista CENIC. Ciencias Biologicas 2010; 41(1): 312-17.
- Habtamu A and Mekonnen Y: Evaluation of the anti-bacterial activities of leaf extracts of Achyranthus aspera. AfrJ Bacteriol Res 2017; 9(2): 9-14.
- Habtemariam M and Medhanie G: Screening of biologically active constituents from leaves of Aloe elegans and their anti-microbial activities against clinical pathogens. Afr J Microbiol Res 2017; 11(8): 366-71.
- Besufekad Y, Beri S, Adugnaw T and Beyene K: Anti-bacterial activity of Ethiopian Lepidium sativum L. against pathogenic bacteria. J Med Plants Res 2018; 12(6): 64-68.
- Damtie D and Mekonnen Y: In-vitro evaluation of the anti-bacterial activity of methanol, aqueous and N- hexane extract of Ocimum lamiifolium from Ethiopia 2015; 9(4): 91-9.
- Buli GA, Gure A and Dessalegn E: Anti-microbial activity of Taverniera abyssinica A. rich against human pathogenic bacteria and fungi. Afr J Microbiol Res 2015; 9(50): 2385-90.
- Mekonnen A, Yitayew B, Tesema A and Taddese S: In-vitro anti-microbial activity of essential oil of Thymus schimperi, Matricaria chamomilla, Eucalyptus globulus and Rosmarinus officinalis. Int J Micro 2016; 3(1): 12-17
- Albejo B, Endale M, Kibret B and Anza M: Phytochemical investigation and anti-microbial activity of leaves extract of Vernonia auriculifera Hiern. J Pharm Pharmacogn Res 2015; 3(6): 3-9
- Alemu F, Tilahun A and Elias E: In-vitro Anti-microbial activity screening of Punica granatum extracts against human pathogens 2017; 7(1): 2-5.
- Tadesse S, Mazumder A and Bucar F: Chemical composition and biological activities of the essential oil of P. caninus Roth. Pharmacogn J 2011; 3 26): 1-7.
- Minale G, Bisrat D, Asres K and Mazumder A: In-vitro anti-microbial activities of anthrones from the leaf latex of Aloe sinana Reynolds. International Journal of Green Pharmacy (IJGP) 2014; 8(1): 1-9.
- Cushnie TT, Cushnie B and Lamb AJ: Alkaloids: an overview of their anti-bacterial, anti-biotic-enhancing and anti-virulence activities. Int J Anti-microb Agents 2014; 44(5): 377-86.
- Levey M: Ancient chemical technology in a Sumerian pharmacological tablet. Journal of Chemical Education 1955; 32(1): 11-16.
- Nair R, Sellaturay S and Sriprasad S: The history of ginseng in the management of erectile dysfunction in ancient China (3500-2600 BCE). Indian Journal of Urology: IJU: Journal of the Urological Society of India 2012; 28(1): 15-19.
- Sarker SD, Latif Z and Gray AI: Natural product isolation. in natural products isolation. Hum press 2006; 1(1): 1-25.
- Shoeb M: Anti-cancer agents from medicinal plants. Bangladesh Journal of Pharmacology 2006; 1(2): 35-41.
- Khan H: Medicinal plants in light of history: recognized therapeutic modality. J of Evidence-Based Complementary & Alternative Medicine 2014; 19(3): 216-19.
- Abu-Rabia A: Herbs as a food and medicine source in Palestine. Asian Pacific Journal of Cancer Prevention 2005; 6(3): 404.
- Van AMA, Eggelte TA and van Boxtel CJ: Artemisinin drugs in the treatment of malaria: from medicinal herb to registered medication. Trends in Pharmacological Sciences 1999; 20(5): 199-05.
- Balouiri M, Sadiki M and Ibnsouda SK: Methods for in-vitro evaluating anti-microbial activity: a review. Journal of Pharmaceutical Analysis 2016; 6(2): 71-9.
- Rios JL, Recio MC and Villar A: Screening methods for natural products with anti-microbial activity: a review of the literature. Journal of Ethno 1988; 23(2-3): 127-49.
- Valgas C, Souza SM, Smânia EF and Smânia Jr A: Screening methods to determine anti-bacterial activity of natural products. Brazilian Journal of Microbiology 2007; 38(2): 369-80.
- Omojate GC, Enwa FO, Jewo AO and Eze C: Mechanisms of anti-microbial actions of phytochemicals against enteric pathogens: a review. J Pharm Chem Biol Sci 2014; 2(2): 77-85.
- Cushnie TT and Lamb A: Anti-microbial activity of flavonoids. Int J Antimicrob Agents 2005; 26(5): 343-356.
- Kurek AN, Grudniak AM, Kraczkiewicz-Dowjat A and Wolska KI: New anti-bacterial therapeutics and strategies. Pol J Microbiol 2011; 60(1): 3-12.
- Sakkas H and Papadopoulou C: Anti-microbial activity of basil, oregano and thyme essential oils. J Microbiol Biotechnol 2017; 27(3): 429-38.
- Chouhan S, Sharma K and Guleria S: Anti-microbial activity of some essential oils-present status and future perspectives. Medicines 2017; 4(3): 58.
- Wafa N, Sofiane G and Mouhamed K: The anti-oxidant and anti-microbial activities of flavonoids and tannins extracted from Phlomis bovei De Noé. Eur J Exper Biol 2016; 6(3): 55-61.
- Guil-Guerrero JL, Ramos L, Moreno C and Zúñiga-Paredes JC: Anti-microbial activity of plant-food by-products: a review focusing on the tropics. Livestock Science 2016; 189: 32-49.
- Monte J, Abreu AC, Borges A, Simões LC and Simões M: Anti-microbial activity of selected phytochemicals against Escherichia coli and Staphylococcus aureus and their biofilms. Pathogens 2014; 3(2): 473-98.
- Tam JP, Wang S, Wong KH and Tan WL: Anti-microbial peptides from plants. Pharmaceuticals 2015; 8(4): 711-757.
How to cite this article:
Belete TM: Review on anti-microbial properties of medicinal plants in Ethiopia. Int J Pharmacognosy 2019; 6(9): 287-99. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(9).287-99.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
287-299
852
1560
English
IJP
T. M. Belete
Department of Pharmacology, College of Medicine and Health Sciences, University of Gondar, Gondar, Ethiopia.
mutafere@yahoo.com
28 August 2019
23 September 2019
28 September 2019
10.13040/IJPSR.0975-8232.IJP.6(9).287-99
30 September 2019