PRILIMINARY PHYTOCHEMICAL SCREENING AND IN-VITRO ANTICONVULSANT ACTIVITY OF METHANOLIC EXTRACT OF LEAVES OF CINNAMOMUM TAMALA (BUCH.-HAM) T. NEES & EBERM (FAMILY- LAURACEAE)
HTML Full TextPRELIMINARY PHYTOCHEMICAL SCREENING AND IN-VITRO ANTICONVULSANT ACTIVITY OF METHANOLIC EXTRACT OF LEAVES OF CINNAMOMUM TAMALA (BUCH.-HAM) T. NEES & EBERM (FAMILY- LAURACEAE)
Rajdeep Phukan* and Dibyendu Shil
Girijananda Chowdhury Institute of Pharmaceutical Science, Azara, Guwahati - 781017, Assam, India.
ABSTRACT: Objective: To perform the phytochemical screening and anticonvulsant activity of methanolic extract of leaves of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm. Method: The physicochemical parameter and in-vitro anticonvulsant study were done. The anticonvulsant activity of methanolic extract of leaves of Cinnamomum tamala (100 and 400 mg/kg, p.o) in Wistar rats was assessed by using the Maximal Electric Shock test. Result: The present study shows different physicochemical analysis which reveals the presence of various metabolites. The methanolic extract of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm significantly reduced the duration of seizure induced by maximal electric shock. The methanolic extract of leaves of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm in doses of 100 and 400mg/kg conferred protection (39.65% and 45.97% respectively) on a rat. Conclusion: The methanolic extract of leaves of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm possess anticonvulsant activity since it reduced the duration of seizures produced by maximal electric shock.
| Keywords: |
Anticonvulsant activity, Cinnamomum tamala, Seizures, Maximal Electric Shock
INTRODUCTION: An epilepsy seizure is a transient paroxysm of uncontrolled discharges of neurons causing an event that is discernible by the person experiencing the seizure. The tendency of recurrent attack is called epilepsy. Epileptic seizures are the symptom of many different diseases, and the term epilepsy is loosely applied to several conditions that have in common a tendency to have recurrent epileptic attacks. A patient with epilepsy will show recurrent epileptic seizures that occur unexpectedly and stop spontaneously 1.
Epilepsy is a central nervous system disorder which is characterized by recurrent seizures unprovoked by an acute systemic or neurologic slur. Epileptogenesis is the sequence of events that turns a normal neuronal network into a hyperexcitable network. Identifying the difference between seizures and epilepsy is essential. Epilepsy may require chronic treatment (with antiepileptic medication and, in some cases, surgery) whereas therapy for an isolated seizure is directed toward the underlying cause and may not require antiepileptic drugs (AEDs) 2.
The traditional system is believed to be an important source of chemical substances with potential therapeutic effects. Herbs may have antiepileptic effects in several ways. Some herbs may increase brain levels, which quiets nerve activity.
Simultaneously, phytochemicals identified from traditional medicinal plants are presenting the development of new types of medicines. This has enhanced the global effort to harness and harvest those medicinal plants that allow a significant amount of potential phytochemicals is showing multiple beneficial effects in convulsion 3, 4.
According to TOI, there are near about 10% -12% total population of India are suffering from epilepsy (around 14 people per 1000 population). In India, many plants have been used from years, for the prevention and treatment of diseases. Indian ayurvedic system is one of the noteworthy systems of traditional medicine practice that uses mainly certain plants for the treatments of ailments in both man and other animals.
Although the popularity of herbal medicine recorded a sharp decline after the introduction of allopathic chemical drugs, herbal medicines are gaining growing interest because of their cost-effective, ecofriendly attributes, and true relief from disease condition. The harmful side effects and high cost of the other forms of treatment and their non-availability to the poor populations, who live in remote areas, are also the reasons for the demand for herbal medicine 5.
Cinnamomum tamala (Buch-Ham) Nees & Eberm an evergreen monoecious species tree of the “Lauraceae” family is a promising medicinal tree species. It is commonly known as the Tejpat or Indian bay leaf in India. Its leaves were used in rheumatism, cardiac disorders, colic, diarrhea, nausea, and vomiting. The aim of the present study was, therefore, to evaluate the anticonvulsant potential of an extract of leaves of Cinnamomum tamala (Buch-Ham) Nees & Eberm in experimental animal models, to provide a pharmacological justification for the traditional use of the plant in the management of epilepsy in India 6, 7, 8.
MATERIALS AND METHODS:
Collection of Plant Materials: The fresh leaves of Cinnamomum tamala (Buch.-Ham.) T. Nees & Eberm was collected from Jorhat, Assam and it was authenticated by P.P. Baruah, Head, Department of Botany, Gauhati University, Guwahati, Assam. The voucher specimen has been stored in the Herbarium of the Department of Botany, Gauhati University with voucher no Acc. No.18231 dated 25-04-2017 for future reference.
Chemicals and Reagents: Petroleum Ether, Chloroform, Methanol, Carboxymethyl cellulose, hydrochloric acid, Dragondorff reagent, Mayer’s reagent, Wagner’s reagent, Benedict’s reagent, sulphuric acid, lead acetate, Molisch’s reagent, Fehling solution A & B, sodium citrate, copper sulphate, ferric chloride, sodium hydroxide, glacial acetic acid, benzene, chloroform, ammonia, nitric acid, potassium nitrite, gelatine. All the chemicals and solvents used were of standard analytical grades.
Preparation of the Plant Extract: The fresh leaves of the plant were collected, washed, shade dried and then kept at tray drier at 37 ºC for 48 h. The dried leaves were ground to a crushed powder. The crush powdered material was defatted by using Petroleum Ether through hot percolation method and then extracted with Chloroform and followed by methanol in the order of their increasing polarity by cold maceration process until it got to be distinctly dreary as indicated by the standard techniques. The extracts were then concentrated and kept inside of vacuum desiccator for further investigation. The extract acquired was weighed, and the rate yield was figured as far as the dried weight of the plant material utilizing the equation:
% Yield = (Dry weight of the extract/Dry weight of leaf sample) × 100.
The extracts were stored in a cool, dark place in further desiccator use.
Phytochemical Screening: The plant extract was investigated for the nearness of alkaloids, saponin, flavonoids, phenol, carbohydrates, proteins and amino acids, cardiac glycosides, steroids, anthraquinone and terpenoids using the standard strategies. The concentrated extracts were subjected to a chemical test as per the methods mentioned below for the identification of the various phytoconstituents
1. Detection of Alkaloid: Solvent-free extract, 50 mg is stirred with few ml of dilute hydrochloric acid & filtered. The filtrate is tested carefully with various alkaloidal reagents as follows:
- Mayer’s test: To a few ml of filtrate, a drop or two of Mayer’s reagent are added by the side of the test tube. A white or creamy ppt indicates test as positive.
- Wagner’s Test: To a few ml of filtrate, few drops of Wagner’s reagent are added by the side of the test tube. A reddish-brown ppt indicates test as positive.
- Hager’s Test: To a few ml of filtrate, 1 or 2 ml of Hager’s reagent is added by the side of the test tube. A prominent yellow ppt indicates test as positive.
- Dragendorff’s test: To a few ml of filtrate, 1 or 2 ml of Dragendorff’s reagent is added by the side of the test tube. A prominent yellow ppt indicates test as positive.
2. Detection of Carbohydrates: The extract (50mg) is dissolved in 5 ml of water & filtered. The filtrate is subjected to the following test.
- Molish’s Test: To 2 ml of filtrate, 2 drops of alcoholic soln of alpha-naphthol are added, the mixture is shaken well & 1ml of conc. H2SO4 is added slowly along the side of the test tube & allowed to stand. A violet ring indicates the presence of carbohydrate.
- Fehling’s Test: 1 ml of filtrate is boiled on a water bath with 1ml each of Fehling solution A & Fehling solution B; a red ppt indicates the presence of sugar.
- Benedict’s Test: To 0.5 ml of filtrate, 1ml of Benedict’s reagent is added. The mixture is heated on a boiling water bath for 2 min. A characteristic colored ppt indicates the presence of sugar.
- Barfoed’s Test: To 1 ml of filtrate, 1 ml of Barfoed’s reagent is added & heated on a water bath for 2 min. Red ppt. Indicates the presence of sugar.
3. Detection of Saponins:
- Foam Test: The extract (50 mg) is diluted with distilled water & made up to 20 ml. The suspension is shaken for 15 min. A layer of 2 cm of foam indicates the presence of saponins.
4. Detection of Phenolic compounds:
- Lead Acetate Test: The extract (50 mg) is dissolved in distilled water & to this; 3 ml of 10% lead acetate solution is added. A bulky white ppt. indicates the presence of Phenolic compounds.
- Ferric Chloride Test: The extract (50 mg) is dissolved in 5 ml of distilled water. To this few drops of natural 5%, ferric chloride solution is added. The blue color appears if the hydrolyzable tannins present and dark green color indicate the presence of Phenolic compound.
5. Detection of Glycoside: 50 gm of the extract is hydrolyzed with concentrated hydrochloric acid for 2 h on a water bath, filtered & the hydrolysate is subjected to the following test:
6. Detection of Anthraquinone glycoside:
- Borntrager’s Test: To 2ml of filtered hydrolysate, 3ml of CHCl3 is added & shaken, CHCl3 layer is separated & 10% NH3 solution is added to it; pink color indicates the presence of glycosides.
7. Detection of Cardiac glycoside:
- Legal’s Test: 50 mg of the extract is dissolved in pyridine, sodium nitroprusside solution is added & made alkaline using 10% NaOH & presence of glycoside is indicated by pink color.
- Keller-Killiani Test: To an extract of the drug in glacial acetic acid, few drops of ferric chloride & conc. Sulfuric acid is added. A reddish brown color is formed at the junction of two layers & the upper layer turns bluish green.
8. Detection of Flavonoid:
- Shinoda Test: The extract (50mg) is dissolved in 5ml of alcohol & few fragments of magnesium ribbon & conc. HCl acid (dropwise) is added. If any pink to crimson color develops, presence of flavanol glycosides is inferred.
- Alkaline Reagent Test: An aqueous solution of the extract is treated with 10% NH4OH solution. Yellow fluorescence indicates the presence of flavonoids.
9. Detection of Proteins & Amino acids: The extract (100 mg) is dissolved in 10 ml of distilled water & filter through Whatman filter paper no-1 & the filtrate is subjected to tests for proteins & amino acids.
- Millon’s Test: To 2ml of filtrate, few drops of Millon’s reagent are added. A white ppt. Indicates the presence of proteins.
- Biuret Test: An aliquot of filtrate is treated with one drop of 2% copper sulfate solution. To this 1 ml of ethanol (95%) is added, followed by an excess of potassium hydroxide palate. The pink color in the ethanolic layer indicates the presence of proteins.
- Ninhydrin test: Two drops of ninhydrin solution (10 mg of ninhydrin in 200 ml of acetone) are added to 2 ml of the aqueous filtrate. A characteristic purple color indicates.
10. Detection of Tannin:
- Gelatin Test: The extract (50mg) is dissolved in 5 ml of distilled water & 1% gelatin containing 2ml of 10% sodium chloride solution is added. White ppt. indicates the presence of tannin compounds.
11. Detection of Phytosterols:
- Salkowski Test:0 ml of concentrated sulphuric acid were added to 10 mg of extract and dissolved in 1.0 ml of chloroform. A reddish blue color exhibited by the chloroform layer and green fluorescence by the acid layer suggested the presence of steroids.
Experimental Animals: Wister albino rats of either sex weighing 150-200 gm were used for animal study. The animals were grouped in polypropylene cages and maintained under standard laboratory conditions (temperature 25 ± 2 °C) and relative humidity (50 ± 5%) with dark and light cycle (12/12 h). All the experimental protocols are reviewed and accepted by the Institutional Animal Ethical Committee (IAEC) before the initiation of the experiment. [GIPS/IAEC/B.Ph/2017/04]
Acute Toxicity Study: The acute toxicity study was executed as described in the OECD guideline 425. The methanolic extract of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm leaves were administered at of different doses of 2000 mg/kg and 5000 mg/kg bw p.o to overnight fasted experimental mice as suggested in OECD Guidelines 425.
The animal behavioral changes and mortality, abnormalities were observed next 24 h and then recorded up to 14 days.
Selection of Dose: After performing the acute toxicity studies it was found that there was no mortality at the dose of 2000 mg/kg body weight as well as 5000 mg /kg body weight of leaf extract. So the drug was found as safe. Therefore dose optimization was done and 100mg/kg and 400 mg/kg, p.o. were selected for the experimental study 11.
Assessment of Anticonvulsant Activity:
Maximal Electric Shock (MES) Induced Seizures: The animals will be divided into 4 groups with each group consisting of 3 animals. Group A received normal saline 10ml/kg served as the control; Group B received phenytoin sodium (25mg/kg, i.p.) as standard in MES method.
Group C, D will be administered the test drug (methanolic extract of Cinnamomum tamala, 100 and 400mg/kg, respectively).
In MES model, convulsion will produce in all groups by giving a maximal electric shock of 150 mA for 0.2 sec after 1 h of giving test extract orally and standard drug i.p., the tonic-clonic seizure will produce after giving an electric shock. Time duration (in a sec) of each phase, i.e., tonus, clonus, stupor will be noted and compare with control.
Statistical Analysis: All the values are expressed as mean ± SEM. Statistical differences between means were determined by one way ANOVA followed by Dunnett’s post hoc test. p<0.05 was considered as significant.
RESULTS AND DISCUSSION:
Phytochemical Analysis: Different phytochemical investigations with the different solvents have demonstrated the nearness of alkaloids, saponin, flavonoids, carbohydrate, glycosides, steroids, proteins and amino acids and tannins Table 1.
Acute Toxicity Study: There were no mortality and signs of toxicity observed in the animal at a dose of 5000 mg/kg of methanolic extract of Cinnamomum tamala leaf after 14 days.
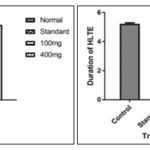
Anticonvulsant Activity: Maximal electroshock produced hind limb tonic extension seizures (HLTE) in all the animals. The vehicle-treated rats showed tonic hind limb extension for the duration of 5.22 ± 0.07 sec. The doses of 100 and 400 mg/kg, respectively, protected 39.65% and 45.97% of rats and significantly reduced the duration of the seizures. However, phenytoin completely abolished the MES-induced tonic seizures in all the animals Table 2.
TABLE 1: PHYTOCHEMICAL SCREENING OF DIFFERENT SOLVENTS EXTRACTS OF LEAVES OF CINNAMOMUM TAMALA (BUCH.-HAM) T. NEES & EBERM
| Chemical Constituents | Test | Pet. Ether | Chloroform | Methanol |
| Alkaloid | ||||
| Mayer’s test | - | - | + | |
| Wagner’s test | - | - | + | |
| Hager’s test | - | + | + | |
| Dragondroff’s test | + | + | + | |
| Carbohydrate | ||||
| Molish’s test | - | + | + | |
| Fehling’s test | - | + | + | |
| Benedit’s test | - | + | + | |
| Barfoed’s test | - | + | + | |
| Saponins | ||||
| Foam’s test | + | + | + | |
| Phenolic Compounds | ||||
| Lead acetate test | - | - | + | |
| Ferric Chloride | - | + | + | |
| Anthraquinone Glycoside | ||||
| Borntrager’s test | + | + | + | |
| Cardiac Glycoside | ||||
| Legel’s test | - | - | - | |
| Killer-killiani test | - | + | + | |
| Flavanoid | ||||
| Shinoda test | + | + | + | |
| Alkaline reagent test | + | + | + | |
| Protein & Amino Acid | ||||
| Millon’s test | - | - | + | |
| Biuret test | - | - | - | |
| Ninhydrin test | + | - | + | |
| Tannin | ||||
| Gelatin test | + | + | + |
+: Present; - : Absent (Based On Colour Reaction).
TABLE 2: ASSESSMENT OF ANTICONVULSANT ACTIVITY OF METHANOLIC EXTRACT OF CINNAMOMUM TAMALA (BUCH.-HAM) T.NEES & EBERM
| Treatment | Dose | Onset of time
(Sec) |
Duration of HLTE (Sec) | % of inhibition of Convulsion |
| Normal (Normal saline) | 10 ml/kg | 1.01 ± 0.03 | 5.22 ± 0.07 | 00 |
| Standard (Phenytoin Sodium) | 25 mg/kg | 0 | 0 | 100 |
| Methanolic extract of C. tamala | 100 mg/ kg | 6.67 ± 0.28* | 3.15 ± 0.017* | 39.65% |
| Methanolic extract of C. tamala | 400 mg/ kg | 8.77 ± 0.54* | 2.08 ± 0.113* | 45.97% |
* Results are expressed as Mean ± SEM; (n=3). Significance at P<0.05*, as compared to control, Dunnett‘s test
FIG. 1: MAXIMAL ELECTROSHOCK PRODUCED HIND LIMB TONIC EXTENSION SEIZURES (HLTE)
DISCUSSION: Now a day there is a renewed interest in drugs of natural origin simply because they are considered as green medicine and green medicine always supposed to be safe. In this study, the effort has been made to establish the identity, purity, quality and the therapeutic value of Cinnamomum tamala (Buch.-Ham.) T. Nees & Eberm which has been used as a traditional drug from ancient time to cure many diseases.
The Pharmacognostical study was done to authenticate the crude drug through morphological and microscopical screening, find out the adulteration through different physiological methods. The powder drugs were subjected to chemical analysis to establish its purity and quality.
Furthermore, the preliminary phytochemical screening of methanolic extract of the leaf had shown the presence of alkaloids, carbohydrates, flavonoids, tannins, proteins, amino acids, reducing sugar and phenols.
The anticonvulsant study has shown that Cinnamomum tamala (Buch.-Ham.) T. Nees & Eberm significantly increases the onset time and decreases the duration of seizures by electroconvulsive shock. In MES‐induced convulsion, animals represent the grand mal type of epilepsy. It has often been suggested stated that antiepileptic drugs that block MES‐induced tonic extension phase act by blocking seizure spread. Moreover, MES‐induced tonic extension phase can be prevented either by drugs that inhibit voltage-dependent Na+ channels, such as phenytoin, valproate, felbamate and lamotrigine or by drugs that block glutaminergic excitation mediated by the N-methyl‐ D‐Aspartate (NMDA) receptor such as felbamate. The methanolic extract of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm showed anticonvulsant activity against MES‐induced convulsion; it was abolished tonic extension phase due to it might be either inhibit voltage‐dependent Na+ channels or act as an NMDA antagonist.
The methanolic extract of Cinnamomum tamala leaves reduced the duration of seizures may be blocking sodium channels which are in an activated state and delayed the recovery of these channels from inactivation. It decreases the number of channels which are available for the generation of action potential and inhibits excitatory of these voltage-gated sodium channels.
CONCLUSION: “Nature itself is the Best Physician”- Hippocrates. The plant has long been a very vital source of the drug, and many plants have been screened on the off chance that they contain mixes with helpful movement. In this manner, it is essential to assess the anticonvulsant activity of leaves of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm. In this study, the anticonvulsant activity of the plant of Cinnamomum tamala was evaluated by using the maximal electric shock method. The animals have chosen to be studied Wister albino rats. From the results, further investigation is warranted to isolate and identify the specific molecules which are responsible for the anticonvulsant activity.
ACKNOWLEDGEMENT: The authors acknowledged the Head of Department, Guwahati University for authenticating the plant and Principal and Department of Pharmacognosy and Pharmacology, Girijananda Chowdhury Institute of Pharmaceutical Science, Azara, Guwahati, Assam for conveying this exploration work.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Tripathi KD: Essentials of Medical Pharmacology. Jaypee Brothers Medical publisher, seven editions 2014.
- Barar FSK: Essentials of Pharmacotherapeutics. S Chand and Company Ltd, 2005.
- Walker R: Clinical Pharmacy and Therapeutics. Churchill Livingstone, 2012.
- Sharma JD, Dandiya PC, Baxter RM and Kandel SI: Pharmadynamical effects of asarone and asarone. London, 1961
- Mukherjee PK, Kumar V, Mal M and Peter J: Scientific validation of ayurvedic tradition from natural resources. Journal of Pharmaceutical Bioscience 2007; 45: 651-666.
- Saluja AK and Santani DD: Pharmacological screening of an ethanolic extract of defatted seeds of Annona squamosal. International Journal of Pharmacognosy 1994; 32: 154-162.
- Hosseinzadeh H and Khosraran V: Anticonvulsant effects of aqueous and ethanolic extracts of Crocus sativus stigmas in mice. Archives of Iranian Medicine 2002; 5(1): 44-47.
- Bharal N, Sahaya K, Jain S, Mediratta PK and Sharma KK: Curcumin has anticonvulsant activity on increasing current electroshock seizures in mice. Journal of Phytochemical Research 2008; 22(12): 1660-1664.
- Pal D, Sahoo M and Mishra AK: Analgesic and anticonvulsant effects of saponin in mice. Herbal Drug Research 2005; 13(2): 91-97.
- Ambawade SD, Kasture VS and Kasture SB: Anticonvulsant activity of Glycyrrhiza glabra. International Journal Pharmacognosy 2002; 34(1): 251-55.
- Prema V: Expert consultant. CPCSEA. OECD guide line No. 42 2000.
- Jaggi RK, Madaan R and Singh B: Anticonvulsant potential of holy basil, Ocimum sanctum L, and its culture. Indian Journal of Experimental Biology 2003; 41(11): 1329-33.
How to cite this article:
Phukan R and Shil D: Priliminary phytochemical screening and in-vitro anticonvulsant activity of methanolic extract of leaves of Cinnamomum tamala (Buch.-Ham) T. Nees & Eberm (family- Lauraceae). Int J Pharmacognosy 2017; 4(10): 345-51. doi link: http://dx. doi.org/10.13040/IJPSR.0975-8232.IJP.4(10).345-51.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
345-351
727
1490
English
IJP
R. Phukan * and D. Shil
Girijananda Chowdhury Institute of Pharmaceutical Science, Azara, Guwahati, Assam, India.
rajdeepgips671@gmail.com
06 June 2017
22 August 2017
17 September 2017
10.13040/IJPSR.0975-8232.IJP.4(10).345-51
01 October 2017