PREPARATION AND EVALUATION OF ANTI-ACNE PHARMACEUTICAL GEL CONTAINING HENNA AND CHAMOMILE EXTRACTS
HTML Full TextPREPARATION AND EVALUATION OF ANTI-ACNE PHARMACEUTICAL GEL CONTAINING HENNA AND CHAMOMILE EXTRACTS
Mahsa Bagheria 1, Yalda Shokoohiniab 2, Zahra Pourmanouchehrib 2, Salar Khaledianc 3, Fereshteh Jalilianb 2, Shahla Mirzaieb 2 and Leila Behboodb * 2
School of Pharmacy 1, Pharmaceutical Sciences Research Center, Health Institute 2, Nano Drug Delivery Research Center 3, Kermanshah University of Medical Sciences, Kermanshah, Iran.
ABSTRACT: Acne is a skin-related condition entailing many psychological complications. Although several medications have been developed, natural remedies attracted more attention because of the less side effects and multifunctional. The objective of this study was to prepare and evaluate the anti-acne effects of a pharmaceutical gel containing herbal extracts, including Lawsonia inermis and Matricaria chamomilla. The total phenolics of extracts were determined with the Folin-Ciocalteu method. Using Hydroxypropyl methylcellulose (HPMC), carboxymethyl cellulose (CMC) and propylene glycol (PEG), the pharmaceutical gel was formulated and the physical properties of the formulation were specified at 37 ± 2 °C. The release of the active ingredients from the optimum formulation was investigated using Franz cell device. The results showed that the optimum formulation was stable at least for 3 months. The total phenol content of the aqueous extract of Henna leaves, hydroalcoholic extract of chamomile flowers, and the optimum formulation was 57.8 mg/g ex, 181.08 mg/g and 202.75 mg/g, respectively. Nearly 80 % of the phenolic compounds in the optimum formulation were released over 4 h. The phenolic compounds have inhibitory effects on the growth of S. aureus and P. aeruginosa. It was concluded that the formulation exhibits excellent stability, viscosity, homogeneity, greater extrudability, and enhanced antibacterial activity, which can be employed as a local pharmaceutical gel in acne treatment.
| Keywords: |
Lawsonia inermis, Henna, pharmaceutical gel, Anti-acne, Matricaria chamomilla
INTRODUCTION: Acne is the most common inflammatory dermatosis that occurs in adolescence and young adults, which involves sebaceous glands 1. Many factors contribute to the development of acne, including excessive sebum production, hormonal imbalance, inflammation, dead cells 2 and external bacterial infection 3, 4, although other factors such as diet, mental stress, and sunlight can also play major roles 5.
In principle, acne is characterized by extreme bacterial colonization such as propionibacterium acnes, Staphylococcus epidermidis and Staphylo-coccus aurous besides follicular hyper-keratinization 6, 7.
The ultimate purpose of the acne treatment is to reduce the numbers of inflamed and non-inflamed scars with the least side effects 8. Besides, prevent the development of scars and undesirable psychological effects from improving the ability of the individual to establish social communication in all stages of the personal life 9. In this regard, antibiotics play important roles 10. The common antibiotics widely using for the treatment of acne are erythromycin, clindamycin, and tetracycline, which available as a gel, liquid, lotion, or ointment. However, the problem caused by the widespread use of these antibiotics is drug resistance 11, which, in addition to the complications of the treatment failure, imposes a financial burden 12. In this regard, Different strategies have been improvised to reduce the shortcomings of antibiotics and enhance their therapeutic efficiency. For example, a lot of attention has been paid to topical treatment of acne, especially in patients with mild to moderate papulopustular acne 7comprisesbenzoyl peroxide, topical retinoid; azelaic acid, clindamycin, and erythromycin but their adverse effects like burning, scaling, photosensitivity, and flare-up or even less-therapeutic effect limited their widespread use. Importantly, the long-term use of oral or topical antibiotics could facilitate development of passage antibiotic resistance to other organisms for instance Staphylococcus, Streptococcus, or Chlamydia 13.
Medicinal plants have made significant strides during the past decade through the development of herbal compounds in the pharmaceutical field and natural health care 14, 15, along with extending the potential number of viable solutions to tackle antibiotic resistance 16. They have a long history of use, and their extracted bioactive compounds have been shown to possess antimicrobial properties, which can use as the best alternative treatments for acne diseases 17, 18. These plants are a reliable source for the preparation of new pharmaceutical dosage forms 16. Matricaria chamomilla belongs to the family of Compositae, and its flowers have anti-inflammatory and antimicrobial properties 19. Chamomile extract contains several bioactive compounds such as terpenoids, monoterpenoids, sesquiterpenoids and phenolics 20. This plant has been used in herbal medicine as an anti-inflammatory and antispasmodic substance. It is also employed in the treatment of antibacterial and anti-fungal diseases 21-23. Lawsoniainermis (also known as henna), is yet another medicinal plant which belongs to the family of Lythraceae 21. Henna leaves contain 1.3-22% Lawson or hydroxy naphthoquinone, which is the active ingredient and the color compound 23, 24. The antimicrobial and fungicidal effects of henna have long been known and reported in Myriad studies 25-27.
The purpose of this study was to investigate the synergism anti-acne effects of hydroalcoholic and aqueous extract of chamomile and henna in a topical pharmaceutical dosage form in order to slow the release of secondary metabolites to the successful treatment of acne.
MATERIALS AND METHODS:
Materials: Mueller Hinton agar, Folin-Ciocalteu reagent, gallic acid, Hydroxypropyl Methyl-cellulose (HPMC), Carboxymethylcellulose (CMC), propylene glycol (PG), and sodium carbonate were purchased from Sigma–Aldrich, USA. Two pathogenic strains of bacteria, namely Staphylococcus aureus (ATCC 25923) and P. aeruginosa (ATCC 27853)were supplied from the microbial control laboratory, Faculty of Pharmacy, Kermanshah University of Medical Sciences, Kermanshah, Iran.
Procurement of Plant Material: The samples of Matricaria chamomillaand Lawsonia inermis were collected from Kermanshah and Kerman province, Iran, respectively. It was identified by Dr. Mirtajeddini, Bahonar University, Kerman, and a voucher specimen for Matricariachamomilla (No 075 101 003) and Law soniainermis (No 167 003 001) was deposited at the Herbarium of the School of Pharmacy and Pharmaceutical Sciences, Kermanshah University of Medical Sciences, Kermanshah, Iran.
Preparation of the Plant Extract: Primarily, leaves of henna and aerial parts of Matricaria chamomilla including flowers, leaves, and the end of the stem, were washed to remove dirt via deionized water and then air-dried and ground using a mechanical grinder. The powder was then placed in a dark space at room temperature. Next, 50 g of henna leaves powder was mixed with 500 mL distilled water in an Erlenmeyer flask under stirring at room temperature for 24 h. In the case of chamomile extraction, 50:50 ratios of distilled water and ethanol was added about one-tenth of the weight of the powdered henna plant on the basis of its adhering property for 72 h. The macerated mixture was filtered through Whatman grade No. 42 filter paper to get the appropriate aqueous or hydroalcoholic extract. Finally, the extracts were concentrated, lyophilized, and stored at 4 °C for further studies.
Preparation of Gel Formulation: Different formulations containing henna and chamomile extract with total concentrations of 5% (w/v) were also prepared. Gels were formulated using hydroxypropyl Methylcellulose (HPMC), carbo-xymethyl cellulose (CMC), and propylene glycol (PG) as excipients. The amount of preservative was considered constant. The gelling agent (HPMC or CMC) was mixed with PG and dissolved in an adequate quantity of phosphate buffer at pH 5.8 (equal to skin’s pH) by stirring, after which, the preservative was added to the prepared gel. Table 1 presents the ingredients used in different formulations. In order to use henna and chamomile extracts in the gel formulation, lyophilized extracts were dissolved in phosphate buffer and added to the gel with a final concentration of 5% w/v in each formulation.
TABLE 1: AMOUNTS (% w/v) OF GEL EXCIPIENTS IN DIFFERENT FORMULATIONS
| Role in Formulation | Gelling Agent | Humectant | Preservative | Active Ingredient | ||||||
| Formulation number | CMC | HPMC | PG | Benzalkonium chloride | Lyophilized henna Ex | Lyophilized Chamomile Ex | ||||
| F1 | 2.5 | - | 10 | 0.1 | 5 | 5 | ||||
| F2 | 3 | - | 10 | 0.1 | 5 | 5 | ||||
| F3 | 3.5 | - | 10 | 0.1 | 5 | 5 | ||||
| F4 | 4 | - | 10 | 0.1 | 5 | 5 | ||||
| F5 | 4.5 | - | 10 | 0.1 | 5 | 5 | ||||
| F6 | 3.5 | - | 7.5 | 0.1 | 5 | 5 | ||||
| F7 | 4.5 | - | 7.5 | 0.1 | 5 | 5 | ||||
| F8 | - | 3.5 | 7.5 | 0.1 | 5 | 5 | ||||
| F9 | - | 4.5 | 7.5 | 0.1 | 5 | 5 | ||||
HPMC: Hydroxypropyl methylcellulose, CMC: Carboxymethylcellulose, PG: Propylene glycol
Physical Evaluation of the Gel Formulations: Optimum formulation was chosen according to a comparative evaluation of the physical properties of the prepared gels. These parameters were tested in all formulations:
- pH was measured using a digital pH meter (827PH Lab, Metrohm, Switzerland).
- Color was tested via a white surface as the gel background.
- Homogeneity was checked through observing the presence of any particle in the formulations.
- Extrudability was determined by means of Aiyalu et al., method, where 20 g of gel in a collapsible tube was pressed from the crimped end with prevention any rollback. The percentage of the extruded gel was then calculated 28.
Viscosity was evaluated using the viscometer (DV-111ULTRA, Brookfield, USA).
Stability Studies: The stability studies were carried out in different temperature conditions (25 and 40 °C) for 3 months. All the evaluation parameters, namely pH, viscosity, and appearance, were studied at time intervals of 30th, 60th, and 90th days.
Total Phenolic Assay: The total phenolic compounds (TPC) of henna and chamomile extracts were determined at the concentration of 5% (w/v) using Folin-Ciocalteu method 29. Briefly, 0.2 mL of henna or chamomile extracts was added to 1 mL of freshly prepared Folin-Ciocalteu reagent, to which 0.8ml of sodium carbonate (7.5 % (w/v) of Na2CO3) was added after 10 min. After 2 h incubation at room temperature, the absorbance of the reaction mixture was recorded by UV Spectrophotometer at 760 nm. Gallic acid (GA) was used as standard, and TPC was explained asmg GAE/g extracts equivalents. The calibration equation for GA at different concentrations is as follows (1):
Y = 0.0037 x + .0081….1
R2 = 0.9987
The Release of Phenolic Compounds: The extract release from optimum formulation was determined using a Franz cell device at 37 °C in the phosphate buffer medium at pH=5.8. The amount of phenolic compounds was determined using the Folin-Ciocalteu method, as already mentioned. The release of the phenolic compound was measured across the dialysis membrane (12 kd) using Franz diffusion cell, with a diffusional area of 2 cm2 and receptor volume of 50 mL.
The membrane was soaked in the receptor compartment, and 5 g of the gel was placed on the membrane surface in the donor compartment. The receptor compartment of the cell was filled with 50 mL of phosphate buffer pH 5.8 and 37o C.
One mL aliquots we recollected from the receptor side at intervals of 0, 15, 30, 60, 90, 120, 180, and 240 min and replaced by the same volume of fresh buffer in the receptor to maintain the constant volume. The concentration of phenolic compounds in the samples was determined through the use of a UV spectrophotometer and GA calibration curve. All experiments were done in triplicate.
Determination of Antimicrobial Activity of Gel: The antibacterial activity of optimum formulation, 5% (w/v) henna, and chamomile extracts were tested by disc diffusion method against the two pathogenic strains of bacteria S. aureus and P. aeruginosa. In this test, clindamycin disc and preservative (0.1%) were used as a positive control. The bacterial suspension (5 × 104 CFU/mL) was swabbed onto sterile Mueller Hinton Agar plates using a sterile cotton swab. Disks soaked in 5% henna or chamomile extract, optimal gel formulation, and 0.1% preservative with clindamycin disc was placed in the environment of Muller-Hinton agar. The plates were incubated at 37 °C for 24 h, after which time, the inhibition zone of bacteria was measured.
The minimal inhibitory concentrations (MICs) of the henna and chamomile extracts were determined using the standard broth dilution method. Live cells of experimented pathogenic bacterial strains at final concentrations of 5 × 104 CFU/mL were inoculated into 48-well plates followed by 100 µL of henna and chamomile extract. After incubation for 24 h at 37 °C, their concentrations were recorded. The pure medium and the medium containing bacteria were respectively used as the negative and positive controls. The MIC was calculated based on the lowest concentration of extract or gel formulations inhibiting the bacterial growth.
RESULTS AND DISCUSSION: Hydrogel masks are 3D networks of polymers with cooling and soothing effects in acne treatment for sensitive skins, which can adsorb water several times the gel weight 30. For reinforcement of prepared hydrogel, CMC was essential. The extract of henna and chamomile was used as an anti-acne ingredient facial gel. The stability studies of the different parameters of hydrogels are exhibited in Table 2.
Among the prepared formulations, formulation number 6 was selected as the optimum formulation for further calculation of drug release due to the better appearance and physical properties. Physical stability parameters of optimum formulation such as appearance, pH N, and viscosity were investigated at 37 ± 2 °C within 3 months. According to the results obtained in this study, after three months, no significant change in pH, viscosity, and appearance was observed in an optimal gel formulation. The pH of hydrogel was measured to be in the range of 5.6-5.9, which was in compliance with human skin pH (4.0 - 6.0).
TABLE 2: PHYSICOCHEMICAL PROPERTIES OF PREPARED GEL FORMULATIONS
| pH | Viscosity (Cp) | Color | Homogeneity | Extrudability | |
| F1 | 5.8 | 716 | brown | +++ | +++ |
| F2 | 5.8 | 847 | brown | +++ | +++ |
| F3 | 5.9 | 1650 | brown | +++ | +++ |
| F4 | 5.8 | 2400 | brown | ++ | + |
| F5 | 5.8 | 2740 | brown | + | + |
| F6 | 5.8 | 2700 | brown | +++ | +++ |
| F7 | 5.6 | 2823 | brown | + | +++ |
| F8 | 5.8 | 2940 | brown | +++ | + |
| F9 | 5.7 | 3261 | brown | ++ | + |
+: good, ++: very good, +++: excellent
Determination of Total Phenolic Contents: Total phenolic compounds in plants normally have antioxidant activity. Hence, they are an important factor in health promotion because of their antioxidant and antimicrobial activity.
In fact, their beneficial free radical scavenging came from the hydroxyl group of phenolic compounds, which acts as a hydrogen donor 31. These compounds were studied in the aqueous extract of henna leaves, hydroalcoholic extract of chamomile flowers, and optimum formulation using Folin-Ciocalteu method. The results are shown in Table 3. The total phenolic contents in various compounds such as flavonoids, coumarins, and gallic acid (GA) derivatives, obtained from henna and chamomile extracts is documented by many publications, although just a few studies have investigated the amounts of the phenolic compounds in a mixture of these two plants.
Our observations confirmed the results obtained by Saoudi Mongi et al. 32 It could be concluded that the high quantity of phenolic compounds in chamomile flower extract is due to the use of hydro alcoholic solvent as a higher polar solvent than less polar solvents such as alcohol, which is more efficient in extracting the phenolic compounds 33.
TABLE 3: TOTAL PHENOLIC CONTENT OF THE AQUEOUS EXTRACT OF HENNA LEAVES, HYDROALCOHOLIC EXTRACT OF CHAMOMILE FLOWERS AND OPTIMUM FORMULATION (MEAN ±SD)
| Sample | Henna Leaves Extract (mg/g) | Chamomile Flowers Extract (mg/g) | Optimum Formulation (mg/g) |
| Total phenol content | 57.8 ± 1.2 | 181.08 ± 2.57 | 202.75 ± 3.78 |
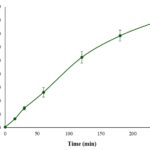
In-vitro Phenolic Compounds Release: Phenolic compounds were considered as the effective ingredient index in the optimum formulation. As shown in Table 4 and Fig. 1, nearly 80% of phenolic compounds in the optimal formulation were released over a period of 4 h due to the slow and regular erosion of the polymeric matrix in the gel and the presence of PG, which can play a penetration enhancer role and facilitate drug release from the 3D matrix of the gel.
In order to investigate the effects of polymer on total phenol content, the base formulation without henna and chamomile extract was checked out similar to its release process.
The results showed that the basic formulation did not interfere with the observed total phenol content.
FIG. 1: THE RELEASE PROFILE OF THE PHENOLIC COMPOUNDS IN THE OPTIMUM FORMULATION
FIG. 2: RELEASE KINETIC MODEL FOR OPTIMIZED FORMULATION; (A) ZERO-ORDER KINETIC, (B) FIRST-ORDER KINETIC AND (C) HIGUCHI KINETIC
TABLE 4: CORRELATION COEFFICIENT OF DIFFERENT KINETIC MODELS FOR PHENOLIC COMPOUNDS RELEASE
| Zero order | Higuchi | First-order |
| Y = 0.3407x + 4.0311 R2 = 0.9782 | Y = 5.5799x-9.9828 R2 = 0.9587 | Y = 0.0029x+2.0176 R2 = 0.9973 |
Kinetics and Mechanism of Drug Release: Regarding to the in-vitro release profile, data was subjected to different release kinetic models such as zero-order Eq (2), first-order Eq (3) and Higuchi Eq (4) 34, 35: where Q is the phenolic compounds (active ingredients) released at time t, Q0 is the percent of active ingredients remaining to be released, and k0, k1 and kH are the coefficients of the equations. The experiments to determine the kinetics of release were carried out in triplicate. Fig. 2 the analyzed release data using mathematical models for zero-order kinetic, first-order kinetic and Higuchi kinetic. The best fit model was determined by highest R2 value, as shown in Table 5. The highest regression value was calculated for first order and zero-order models, indicating the drug release with diffusion-controlled 36.
Q=K0t....2
Ln (100-Q) = Ln (Q0) - k1t...3
Q = KHt 1/2...4
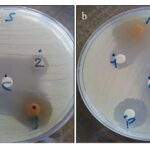
Antibacterial Activity: Antibacterial activity of the formulation plays a crucial role in skin acne treatment. Thus, the antibacterial properties of the prepared henna aqueous extract, chamo-milehydroalcoholic extract and optimum formulation against P. aeruginosa and S. aureus were evaluated in this part. Table 6 summarizes the results of inhibition zones of phenolic compounds in extracts and optimum formulation against two pathogenic bacteria under disk diffusion. Preservative (0.1%) and clindamycin disk were used as controls. The antibacterial activity of extracts is also presented in Fig. 3. The MIC values of S. aureus and P. aeruginosa were 50 µg/ml (chamomile extract), 5 µg/ml (henna extract), and 1 µg/ml (optimum formulation). According to obtained results, the phenolic compounds had higher effects ongram-negative bacteria P. aeruginosa in comparison to gram-positive bacteria S. aureus. Interestingly, fewer antibiotics are effective against P. aeruginosa 37. This is may be attributed to the thicker cell wall of gram-positive bacterium, causing more resistance to be attacked by active polyphenol groups than P. aeruginosa 38. The antibacterial activities of the henna and chamomile leaf extracts have been reported against both gram-negative and gram-positive bacteria 26, 27, 39-44. It is worth mentioning that various experimental conditions comprise extract concentration, type of the solvent, and environmental conditions of plant growth that could affect the antimicrobial activities of extracts 45-47. The antimicrobial activity of formulation containing chamomile and henna extract is to be caused by synergies and multiple mechanisms. The probable antibacterial mechanism of chamomile is based on the abundant amount of active constituents such as α- bisabolol and chamazulene, which gavedual bactericidal and bacteriostatic antimicrobial action. From the above data, it is assumed that sesquiterpenoid compounds of chamomile with the ability to interrupts cell wall permeability barrier and inhibition of cell membrane enzymes could induce antibacterial effects 48. Similar to chamomile extract, henna is a rich source of polyphenol compounds such as Lawson, Gallic acid, tannic acid, mucilage, and mannitol 49 which the free hydroxyl group of these compounds with the capability to combine with the bacterial cell wall carbohydrates and proteins and attached to sites of the enzyme rendering them inactive 50-52. Although, the exact antibacterial mechanism of henna and chamomile bioactive compounds are required tounderst and relied on the promising result, the combination of these extracts shows a synergistic antimicrobial effect.
TABLE 5: ANTIBACTERIAL ACTIVITY OF HENNA, CHAMOMILE EXTRACT AND OPTIMUM FORMULATION (N=3)
| Test samples | Inhibition Zone Diameter (mm) , Mean ±SD | |
| S. aureus | P. aeruginosa | |
| Henna extract (5%) | 11.6 ± 0.057 | 16.3 ± 0.11 |
| Chamomile extracts (5%) | 14.3 ± 0.057 | 22.6 ± 0.057 |
| Optimum formulation | 25.1 ± 0.15 | 36 ± 0.36 |
| Preservative 0.1% | 17.3 ± 0.15 | 19.6 ± 0.057 |
| Clindamycin disk | 35.3 ± 0.057 | 0 |
FIG. 3: ANTIBACTERIAL EFFECTS OF PHENOLIC COMPOUNDS AGAINST (A) S. AUREUS (B) P. AERUGINOSA 1: HENNA EXTRACT DISK, 2: CHAMOMILE EXTRACT DISK, G: OPTIMUM FORMULATION DISK, P: PRESERVATIVE, C: CLINDAMYCIN DISK
CONCLUSION: The chamomile and henna extract were found to have good potency against acne-inducing bacteria. Phenolic compounds were assumed as effective ingredients in both herbal extracts. The gel formulation developed from gelling agent, and these extracts also showed good physical properties and acceptable kinetics in the release of the phenolic compound. It is concluded that the aqueous extract of Lawsonia inrmis and the hydroalcoholic extract of Matricaria chamomilla act as potential reducing compounds against S. aureus and P. aeruginosa, and the formulation exhibits excellent stability, viscosity, homogeneity, greater extrudability, and enhanced antibacterial activity, which can be employed as aqueous-based gel formulation in anti-acne topical dosage forms.
ACKNOWLEDGEMENT: The authors gratefully acknowledge the Research Council of Kermanshah University of Medical Sciences (Grant Number: 96205) for financial support. This work was performed in partial fulfillment of the requirements for Pharm. D of Mahsa Bagheri, in the school of Pharmacy, Kermanshah University of Medical Sciences, Kermanshah, Iran.
CONFLICTS OF INTEREST: The authors declare no conflicts of interest, financial or otherwise.
REFERENCES:
- Budiman A, Aulifa DL, Kusuma ASW, Kurniawan IS and Sulastri A: Peel-off gel formulation from black mulberries (Morus nigra) extract as anti-acne mask. National Journal of Physiology, Pharmacy and Pharmacology 2017; 7(9): 987-94.
- Pendyala V, Suryadevara V, Tokala D and Nelluri N: Formulation and evaluation of a polyherbal ointment for the treatment of acne. Asian Journal of Pharmacy and Pharmacology 2019; 5(1): 143-48.
- Kurokawa I, Danby FW, Ju Q, Wang X, Xiang LF, Xia L, Chen W, Nagy I, Picardo M and Suh DH: New developments in our understanding of acne pathogenesis and treatment. Experimental dermatology 2009; 18(10): 821-32.
- Williams HC, Dellavalle RP and Garner S: Acne vulgaris. The Lancet 2012; 379(9813): 361-72.
- Simpson NB and Cunliffe WJ: Disorders of the sebaceous glands. Rook's Txtbook of Dermatology 2004; 2121-96.
- Karlapudi AP, Kodali VP, Kota KP, Shaik SS, Kumar NS and Dirisala VR: Deciphering the effect of novel bacterial exopolysaccharide-based nanoparticle cream against Propionibacterium acnes. Biotech 2016; 6(1): 35.
- Mazzarello V, Donadu M, Ferrari M, Piga G, Usai D, Zanetti S and Sotgiu M: Treatment of acne with a combination of propolis, tea tree oil, and Aloe vera compared to erythromycin cream: two double-blind investigations. Clinical Pharmacology Advances and Applications 2018; 10: 175.
- Leyden JJ, McGinley KJ, Mills OH and Kligman AM: Propioni bacterium levels in patients with and without Acne vulgaris. Journal of Investigative Dermatology 1975; 65(4): 382-84.
- Mallon E, Newton J, Klassen A, Stewart-Brown SL, Ryan T and Finlay A: The quality of life in acne: a comparison with general medical conditions using generic questionnaires. The British Journal of Dermatology 1999; 140(4): 672-76.
- Cunliffe WJ, Meynadier J, Alirezai M, George SA, Coutts I, Roseeuw DI, Hachem JP, Briantais P, Sidou F and Soto P: Is combined oral and topical therapy better than oral therapy alone in patients with moderate to moderately severe Acne vulgaris. a comparison of the efficacy and safety of lymecycline plus adapalene gel 0.1%, versus lymecycline plus gel vehicle. Journal of the American Academy of Dermatology 2003; 49(3): 218-26.
- Humphrey S: Antibiotic resistance in acne treatment. Skin Therapy Lett 2012; 17(9): 1-3.
- Patel M, Bowe WP, Heughebaert C and Shalita AR: The development of antimicrobial resistance due to the antibiotic treatment of Acne vulgaris: a review. Journal of Drugs in Dermatology: JDD 2010; 9(6): 655-64.
- Toyoda M and Morohashi M: An overview of topical antibiotics for acne treatment. Dermatology 1998; 196(1): 130-34.
- Moradi S, Taran M, Mohajeri P, Sadrjavadi K, Sarrami F, Karton A and Shahlaei M: Study of dual encapsulation possibility of hydrophobic and hydrophilic drugs into a nanocarrier based on bio-polymer coated graphene oxide using density functional theory, molecular dynamics simulation and experimental methods. Journal of Molecular Liquids 2018; 262: 204-17.
- Moradi S, Sadrjavadi K, Farhadian N, Hosseinzadeh L and Shahlaei M: Easy synthesis, characterization and cell cytotoxicity of green nano carbon dots using hydrothermal carbonization of Gum Tragacanth and chitosan bio-polymers for bioimaging. Journal of Molecular Liquids 2018; 259: 284-90.
- Krishnaiah D, Sarbatly R and Nithyanandam R: A review of the antioxidant potential of medicinal plant species. Food and Bioproducts Processing 2011; 89(3): 217-33.
- Chomnawang MT, Surassmo S, Nukoolkarn VS and Gritsanapan W: Antimicrobial effects of Thai medicinal plants against acne-inducing bacteria. Journal of Ethnopharmacology 2005; 101(1-3): 330-33.
- Kumar G, Jayaveera K, Kumar C, Sanjay UP, Swamy B and Kumar D: Antimicrobial effects of Indian medicinal plants against acne-inducing bacteria. Tropical Journal of Pharmaceutical Research 2007; 6(2): 717-23.
- Singh O, Khanam Z, Misra N and Srivastava MK: Chamomile (Matricaria chamomilla L.): an overview. Pharmacognosy Reviews 2011; 5(9): 82.
- Nováková L, Vildová A, Mateus JP, Gonçalves T and Solich P: Development and application of UHPLC–MS/MS method for the determination of phenolic compounds in Chamomile flowers and Chamomile tea extracts. Talanta 2010; 82(4): 1271-80.
- Makhija IK, Dhananjaya D, Kumar VS, Devkar R, Khamar D, Manglani N and Chandrakar S: Lawsonia inermis-from traditional use to scientific assessment. African J of Pharmace Sciences and Pharmacy 2011; 2(1).
- Chaudhary G, Goyal S and Poonia P: Lawsonia inermis Linnaeus: a phytopharmacological review. Int J Pharm Sci Drug Res 2010; 2(2): 91-98.
- Mahkam M, Nabati M and Kafshboran RH: Isolation, identification and characterization of lawsone from henna leaves powder with Soxhlet technique. Iranian Chemical Communication 2014; 2: 34-38.
- El‐Shaer NS, Badr JM, Aboul‐Ela MA and Gohar YM: Determination of lawsone in henna powders by high performance thin layer chromatography. Journal of Separation Science 2007; 30(18): 3311-15.
- Muhammad H and Muhammad S: The use of Lawsonia inermis Linn. (Henna) in the management of burn wound infections. African Journal of Biotechnology 2005; 4(9).
- Habbal O, Hasson S, El-Hag A, Al-Mahrooqi Z, Al-Hashmi N, Al-Bimani Z, Al-Balushi M and Al-Jabri A: Antibacterial activity of Lawsonia inermis Linn (Henna) against Pseudomonas aeruginosa. Asian Pacific Journal of Tropical Biomedicine 2011; 1(3): 173-76.
- Saadabi MA: Evaluation of Lawsonia inermis Linn. (Sudanese henna) leaf extracts as an antimicrobial agent. Research journal of biological sciences 2007; 2(4): 419-23.
- Aiyalu R, Govindarjan A and Ramasamy A: Formulation and evaluation of topical herbal gel for the treatment of arthritis in animal model. Brazilian Journal of Pharmaceutical Sciences 2016; 52(3): 493-07.
- Singleton VL, Orthofer R and Lamuela-Raventós RM: Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods in enzymology Elsevier; 1999; 299: 152-78.
- Nilforoushzadeh MA, Amirkhani MA, Zarrintaj P, Salehi Moghaddam A, Mehrabi T, Alavi S and Sisakht M: Skin care and rejuvenation by cosmeceutical facial mask. Journal of Cosmetic Dermatology 2018; 17(5): 693-02.
- Nakamura K, Ishiyama K, Sheng H, Ikai H, Kanno T and Niwano Y: Bactericidal activity and mechanism of photoirradiated polyphenols against Gram-positive and-negative bacteria. Journal of Agricultural and Food Chemistry 2015; 63(35): 7707-13.
- Hsouna AB, Mongi S, Culioli G, Blache Y, Ghlissi Z, Chaabane R, El Feki A, Jaoua S and Trigui M: Protective effects of ethyl acetate fraction of Lawsonia inermis fruits extract against carbon tetrachloride-induced oxidative damage in rat liver. Toxicology and Industrial Health 2016; 32(4): 694-06.
- Hernández-Hernández E, Ponce-Alquicira E, Jaramillo-Flores ME and Legarreta IG: Antioxidant effect rosemary (Rosmarinus officinalis L.) and oregano (Origanum vulgare L.) extracts on TBARS and colour of model raw pork batters. Meat Science 2009; 81(2): 410-17.
- Kalam MA, Humayun M, Parvez N, Yadav S, Garg A, Amin S, Sultana Y and Ali A: Release kinetics of modified pharmaceutical dosage forms: a review. Cont J Pharm Sci 2007; 1: 30-35.
- Dash S, Murthy PN, Nath L and Chowdhury P: Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm 2010; 67(3): 217-23.
- Siepmann J and Peppas N: Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Advanced Drug Delivery Reviews 2012; 64: 163-74.
- Page MG and Heim J: Prospects for the next anti-Pseudomonas drug. Current Opinion in Pharmacology 2009; 9(5): 558-65.
- Chahardoli A, Karimi N, Sadeghi F and Fattahi A: Green approach for synthesis of gold nanoparticles from Nigella arvensis leaf extract and evaluation of their antibacterial, antioxidant, cytotoxicity and catalytic activities. Artificial Cells, Nanomedicine and Biotechnology 2018; 46(3): 579-88.
- Roby MHH, Sarhan MA, Selim KA-H and Khalel KI: Antioxidant and antimicrobial activities of essential oil and extracts of fennel (Foeniculum vulgare L.) and chamomile (Matricaria chamomilla L.). Industrial Crops and Products 2013; 44: 437-45.
- Kazemi M: Chemical composition and antimicrobial activity of essential oil of Matricaria chamomilla. Bulletin of Environment, Pharmacology and Life Sciences 2014; 3(2): 148-53.
- Mekonnen A, Yitayew B, Tesema A and Taddese S: In-vitro antimicrobial activity of essential oil of Thymus schimperi, Matricaria chamomilla, Eucalyptus globulus, and Rosmarinus officinalis. International Journal of Microbiology 2016.
- Habbal OA, Al-Jabri AA, El-Hag AH, Al-Mahrooqi ZH and Al-Hashmi NA: In-vitro antimicrobial activity of Lawsonia inermis Linn (henna). A pilot study on the Omani henna. Saudi medical Journal 2005; 26(1): 69-72.
- Al-Rubiay KK, Jaber NN, Al-Mhaawe B and Alrubaiy LK: Antimicrobial efficacy of henna extracts. Oman Medical Journal 2008; 23(4): 253.
- Arun P, Purushotham K, Jayarani J and Vasantha K: In-vitro antibacterial activity and flavonoid contents of Lawsonia inermis (Henna). International Journal of Pharm Tech Research 2010; 2(2): 1178-81.
- Jeyaseelan EC, Jenothiny S, Pathmanathan M and Jeyadevan J: Antibacterial activity of sequentially extracted organic solvent extracts of fruits, flowers and leaves of Lawsonia inermis L. from Jaffna. Asian Pacific Journal of Tropical Biomedicine 2012; 2(10): 798-02.
- Dai J and Mumper RJ: Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 2010; 15(10): 7313-52.
- Cowan MM: Plant products as antimicrobial agents. Clinical microbiology reviews 1999; 12(4): 564-82.
- Alkuraishy HM, Al-Gareeb AI, Albuhadilly AK and Alwindy S: In-vitro assessment of the antibacterial activity of Matricaria chamomile alcoholic extract against pathogenic bacterial strains. Microbiology Research J Inte 2015: 55-61.
- Semwal RB, Semwal DK, Combrinck S, Cartwright-Jones C and Viljoen A: Lawsonia inermis L. (Henna): ethnobotanical, phytochemical and pharmacological aspects. J of Ethnopharmacology 2014; 155(1): 80-103.
- Rahmoun NM, Boucherit-Atmani Z, Benabdallah M, Boucherit K, Villemin D and Choukchou-Braham N: Antimicrobial activities of the henna extract and some synthetic naphthoquinones derivatives 2013.
- Karpe ST, Kulkarni SR, Shaikh SA and Manikrao AM: bactericidal ad bactriostatic activity of isolated aphthoqui oe fractio of lawso ia i ermis ad sy thetic lawso eo staphylococcus epidermidis.
- Abulyazid I, Mahdy EM and Ahmed RM: Biochemical study for the effect of Henna (Lawsonia inermis) on Escherichia coli. Arabian J of Chemist 2013; 6(3): 265-27.
How to cite this article:
Bagheria M, Shokoohiniab Y, Pourmanouchehrib Z, Khaledianc S, Jalilianb F, Mirzaieb S and Behboodb L: Preparation and evaluation of anti-acne pharmaceutical gel containing Henna and Chamomile extracts. Int J Pharmacognosy 2020; 7(8): 208-16. doi link: http://dx.doi. org/10.13040/IJPSR.0975-8232.IJP.7(8).208-16.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
208-216
896
980
English
IJP
M. Bagheria, Y. Shokoohiniab, Z. Pourmanouchehrib, S. Khaledianc, F. Jalilianb, S. Mirzaieb and L. Behboodb *
Pharmaceutical Sciences Research Center, Health Institute, Kermanshah University of Medical Sciences, Kermanshah, Iran.
leila_behbood@yahoo.com
21 April 2020
24 July 2020
28 July 2020
10.13040/IJPSR.0975-8232.IJP.7(8).208-16
01 August 2020