POTENTIAL ANTIOXIDANT ACTIVITIES OF SPONDIAS PINNATA (FAMILY: ANACARDIACEAE) BARK EXTRACT IN RATS WITH STREPTOZOTOCIN INDUCED DIABETES MELLITUS
HTML Full TextPOTENTIAL ANTIOXIDANT ACTIVITIES OF SPONDIAS PINNATA (FAMILY: ANACARDIACEAE) BARK EXTRACT IN RATS WITH STREPTOZOTOCIN INDUCED DIABETES MELLITUS
A. P. Attanayake * 1, K. A. P. W. Jayatilaka 1, C. Pathirana 1 and L. K. B. Mudduwa 2
Department of Biochemistry 1, Department of Pathology 2, Faculty of Medicine, University of Ruhuna, Matara, Sri Lanka.
ABSTRACT: The present study was to investigate the antioxidant activity of aqueous bark extract of Spondias pinnata (Linn. f.) Kurz (Family: Anacardiaceae) in streptozotocin-induced diabetic rats. The antioxidant activity was evaluated by the activities of liver enzymes, antioxidant enzymes, and the extent of lipid peroxidation (LPO) in the liver of streptozotocin (STZ) ‑diabetic rats. S. pinnata extract decreased the LPO by 17%, activities of alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase decreased by 20%, 17% and 36% respectively (P<0.05). The liver reduced glutathione, activities of glutathione reductase, glutathione peroxidase, and glutathione S-transferase of plant extract treated diabetic rats were increased by 43%, 44%, 69% and 52% respectively (P<0.05). The light microscopic appearance of the liver tissue in S. pinnata treated rats has corroborated the biochemical results. The present investigation revealed that the bark extract of S. pinnata exerts significant in-vivo antioxidant activities in diabetic rats.
| Keywords: |
Antioxidant activities, Oxidative stress, Spondias pinnata, Streptozotocin-induced diabetic rats
INTRODUCTION: Diabetes mellitus is a complex heterogeneous disorder affecting millions of people worldwide. The pathogenesis involves the development of insulin resistance accompanied by defective insulin secretion from pancreatic β cells resulting in hyperglycaemia 1. It has been proved experimentally and clinically that the oxidative stress is involved in the development and progression of diabetes mellitus 2. This is due to the deleterious invasion of free radicals or impaired antioxidant defense mechanisms 3. Increased lipid peroxidation and insulin resistance are linked with abnormal production of free radicals and the simultaneous decline of antioxidant defense mechanisms 4.
Current treatments provide a satisfactory glycaemic control; however, do little in preventing complications associated with diabetes mellitus. Besides, most of the prescribed hypoglycaemic drugs and insulin are associated with side effects; there is an increasing demand for natural products for the treatment of diabetes mellitus and its associated complications 5. There is an increasingly powerful case for revisiting natural products for drug discovery. Historically, natural products from plants and animals were the source of virtually all medicinal preparations, and more recently, natural products have continued to provide leads for compounds that have effective against different chronic complications.
S. pinnata (Linn.f.) Kurz (Family: Anacardiaceae) is a deciduous tree distributed in Sri Lanka, India, and other South-East Asian countries. The bark of the tree is used for treating dysentery, muscular rheumatism and diabetes mellitus in traditional Ayurvedic medicine 6. The preliminary investigations by our group confirmed that the aqueous bark extract of S. pinnata at a dose of 1.00 g/kg was found to be the optimum effective anti-hyperglycemic dose in healthy (normoglycaemic) and diabetic rats 7, 8. Furthermore, the anti-hyperglycaemic, anti-hyperlipidaemic and β cell regenerative effects of S. pinnata (Linn.f.) Kurz. bark extract in streptozotocin-induced diabetic rats was reported previously by our group (9). The isolation of 24-methylene cycloartanone, stigma-4en-3one, lignoceric acid, β-sitosterol and its β-D-glucoside from S. pinnata have also described previously 10. The bark extract of S. pinnata was found to be toxicologically safe as a potent anti-hyperglycaemic agent in healthy Wistar rats 11. However, the effect of the S. pinnata bark extract at the optimum effective therapeutic dose on oxidative stress in diabetic rats was not previously reported. Therefore, the present study is aimed at investigating the potency of the S. pinnata bark extract to attenuate the oxidative stress in streptozotocin-induced diabetic rats.
MATERIALS AND METHODS:
Chemicals: All chemicals were of analytical grade and used without any purification.
Plant Material: Stem bark parts of S. pinnata were collected during May-June 2013 from the Southern region of Sri Lanka. Botanical identity was determined by the descriptions given by Jayaweera 6 and confirmed by comparing authentic samples at the National Herbarium, Royal Botanical Gardens, Peradeniya, Sri Lanka. A voucher specimen was preserved at the Department of Biochemistry, Faculty of Medicine, University of Ruhuna, Sri Lanka (Attanayake/2011/ 01).
Preparation of the Aqueous Plant Extract: The bark parts of S. pinnata were cut into small pieces, dried at 40 °C until a constant weight was reached and coarsely ground. Powdered plant material (50g) was dissolved in 400 ml of distilled water and refluxed for 4h. The mixture was strained, and the final volume was adjusted to 50 ml. A single dose of 1.00 g/kg (the optimum effective anti-hyperglycaemic dose) was administered orally to streptozotocin-induced diabetic rats.
Animals: Healthy adult male rats of Wistar strain (200 ± 25 g, body weight) were purchased from the Medical Research Institute (MRI), Sri Lanka and used to carry out the experiments. They were housed in standard environmental conditions at the animal house of Faculty of Medicine, the University of Ruhuna, Sri Lanka (Tem 25 ± 2 °C, relative humidity 55-65% and 12 ± 1 h light/ dark cycle). Rats were fed with a standard diet (MRI rat formulae, Sri Lanka) with free access to water before and during the experiment. The rats were randomized into various groups and allowed to acclimatize for seven days under standard environmental conditions before commencing the experiments. The animals described as fasting were deprived of food and water for 12 h ad libitum.
All protocols used in this study were approved by the Ethics Committee of Faculty of Medicine, University of Ruhuna, Sri Lanka guided by the Council for International Organization of Medical Sciences (CIOMS) international guiding principles of biomedical research involving animals.
Development of Diabetes Mellitus in Wistar Rats: Streptozotocin dissolved in citrate buffer (0.1M, pH 4.4) at a dose of 65 mg/kg was administered intraperitoneally to rats fasted for 12 h. After that, rats have maintained on 5% D-glucose solution for the next 24 h. Rats were allowed to stabilize for three days, and on the 4th day, blood samples were drawn from the tail vein to determine the blood glucose concentration to confirm the development of diabetes mellitus. Rats with fasting blood glucose concentration of 12.0mmol/L or above were considered as hyperglycaemic and used for the experiments 12.
Experimental Design: Rats were randomly allotted to four groups of six animals per group. The group 1 and group 2 served as healthy and diabetic untreated (control) groups respectively. Group 3 and group 4 diabetic rats received the aqueous bark extract of S. pinnata at the optimum effective dose (1.00 g/kg) and glibenclamide (0.50 mg/kg) daily for 30 days respectively. At the end of the study (on the 30th day), blood was collected for the estimation of glycated hemoglobin (HbA1c) and serum activities of liver enzymes. The liver tissues of rats were excised for the estimation of the concentration of reduced glutathione, activities of antioxidant enzymes, and the extent of lipid peroxidation.
Assessment of Biochemical Parameters: The ion exchange resin method as described by Abraham et al. was followed for the estimation of glycated hemoglobin using spectrophotometric enzyme assay kit (Stanbio, USA) 13. Fasting serum activities of liver enzymes; alanine amino-transferase (ALT), aspartate aminotransferase (AST) and alkaline phosphatase (ALP) were estimated using spectrophotometric enzyme assay kits 14, 15.
The estimation of reduced glutathione (GSH), activities of glutathione reductase (GR, EC 1.6.4.2), glutathione peroxidase (GPx, EC 1.11.1.9) and glutathione S-transferase (GST, EC2.5.1.18) in the liver homogenates were done using reported protocols 16, 17, 18. Further, the extent of lipid peroxidation and total protein were estimated in liver homogenates by the formation of malondialdehyde (MDA) using thiobarbituric acid and Lowry methods respectively 19, 20.
Assessment of Liver Histopathology: The liver tissues of test rats were fixed in 10% formalin. Tissues were processed routinely and embedded in paraffin wax. Sections were stained with hematoxylin and eosin.
Statistical Analysis: The replicates of each sample were used for statistical analysis, and the values were expressed as the mean ± standard deviation in the in-vitro study. The data were analyzed using analysis of variance (ANOVA) and the mean values for each group were compared by Dunnett’s multiple comparison tests in the in-vivo study. The level of significance was set at P<0.05.
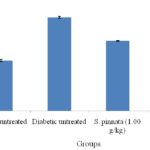
RESULTS: As shown in Fig. 1, the percentage of HbA1C in S. pinnata extract treated rats were significantly lower than that in the diabetic control group (P<0.05). Table 1 and 2 shows the effect of the S. pinnata bark extract on liver enzymes and hepatic oxidative stress markers in healthy control and STZ-diabetic experimental rats. There was an elevation in the activities of ALT, AST, ALP (by 195%, 104%, 170%) and the concentration of MDA (by 199%), while the concentration of GSH, GR, GPx and GST (by 42%, 41%, 56%, 57% respectively) were decreased in STZ-diabetic control when compared with corresponding healthy control rats. Treatment of diabetic rats with the extract and glibenclamide decreased the activities of ALT (by 20%, 16%), AST (by 17%, 17%) and ALP (by 36%, 5%) compared to the corresponding diabetic control group (P<0.05).
FIG. 1: EFFECT OF S. PINNATA (1.00 g/kg) ON GLYCOSYLATED HAEMOGLOBIN (%) IN STREPTOZOTOCIN INDUCED DIABETIC RATS AFTER 30 DAYS OF TREATMENT
TABLE 1: EFFECT OF PLANT EXTRACTS ON SERUM HEPATIC ENZYMES IN STREPTOZOTOCIN INDUCED DIABETIC RATS AFTER 30 DAYS OF TREATMENT
| Group | ALT(U/L) | AST(U/L) | ALP(U/L) |
| Healthy untreated | 12.40±0.12 | 44.21±1.75 | 61.48±1.00 |
| Diabetic untreated | 36.64± 2.24 | 90.08 ± 0.07 | 165.69 ± 2.52 |
| S. pinnata (1.00 g/kg) | 29.13±1.10* | 74.34±1.90* | 106.11± 0.07* |
| Glibenclamide (0.50 mg/kg) | 30.67± 1.03* | 74.98 ± 3.00* | 156.65 ± 2.30* |
The values are expressed as mean ± SEM (n=6/group). *Statistically significant from streptozotocin-induced diabetic control rats at P<0.05 (ANOVA followed by Dunnett’s test). ALT: Alanine aminotransferase, AST: Aspartate aminotransferase, ALP: Alkaline phosphatase.
TABLE 2: EFFECT OF PLANT EXTRACTS ON HEPATIC ANTIOXIDATIVE STRESS MARKERS IN STREPTOZOTOCIN INDUCED DIABETIC RATS AFTER 30 DAYS OF TREATMENT
| Group | MDA (nmol/
mg protein) |
GSH
(µg/g liver) |
GR (nmol/ min/
mg of protein) |
GPx (nmol/ min/
mg of protein) |
GST (nmol/ min/
mg of protein) |
| Healthy untreated | 12.15±0.30 | 719.80±7.45 | 9.12±1.06 | 10.58±0.98 | 10.92±1.47 |
| Diabetic untreated | 36.38±1.01 | 419.65±8.77 | 5.33±0.54 | 4.61±1.03 | 4.75±0.15 |
| S. pinnata (1.00 g/kg) | 30.23±1.32* | 600.15±12.78* | 7.70±0.36* | 7.79±0.34* | 7.23±0.89* |
| Glibenclamide (0.50 mg/kg) | 22.09±0.25* | 564.71±9.86* | 6.39±0.14* | 6.38±0.18* | 5.34±0.20* |
The values are expressed as mean ± SEM (n=6/group). *Statistically significant from streptozotocin-induced diabetic control rats at P<0.05 (ANOVA followed by Dunnett’s test). MDA: Malondialdehyde, GSH: Reduced glutathione, GR: Glutathione reductase, GPx: Glutathione peroxidase, GST: Glutathione S-transferase.
Also, the concentration of MDA was reduced by 17% in S. pinnata extract treated diabetic rats (P <0.05). The administration of the S. pinnata extract restored the There was an increase in the activities of GR, GPx, and GST in glibenclamide treated diabetic rats (by 20%, 38%, 12%) however it was less effective than the attainment of the above biochemical parameters by S. pinnata extract.
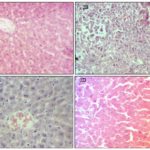
FIG. 2: PHOTOMICROGRAPHS OF H&E SECTIONS OF THE PANCREAS OF ALLOXAN INDUCED DIABETIC RATS AFTER 30 DAYS OF PLANT TREATMENT (X400). A: Islets of Langerhans are normal with a normal islet cell population, B: Few preserved islet cells, fibrosis, and infiltration by inflammatory cells s as a result of islet cell destruction by streptozotocin, C: Restoration of pancreatic islet cells and with no evidence of inflammation, D: Pancreatic islet cells are restored with evidence of islet cell hyperplasia.
As shown in Fig. 2, liver histology was normal in untreated healthy rats. In contrast, STZ-diabetic control diabetic rats showed very early microvesicular fatty change in centrilobular areas of the liver, mild congestion, moderate lymphocytic infiltrate mostly around portal tract, increased fibrosis with parenchymal infiltrates and focal necrosis. The light microscopic appearance of the liver tissue in S. pinnata treated rats are in line with biochemical results, with a reduction in micro-vesicular fatty change, mild lymphocytic infiltrates and no necrosis. Further, reduction in micro-vesicular fatty change, moderate lymphocytic infiltrates were also observed in glibenclamide treated diabetic rats.
DISCUSSION: Oxidative stress is likely to be involved in the progression of diabetic complications 21. High levels of free radicals, due to increasing production and insufficiency of the antioxidant defense system, may lead to disruption of cellular function, oxidative damage to membranes, and enhance their susceptibility to lipid peroxidation 22. In recent years, medicinal plant extracts with strong in-vivo antioxidative property have been the center of focus. It is believed that these plant extracts can prevent or protect tissues by direct antioxidative and anti-hyperglycaemic effects 23. The protection of hepatocyte cells from the effects of oxidative stress by treatment with vitamin E was evidenced in a recent study. Also, it has been shown that dietary supplementation with natural antioxidants such as vitamins C and E, melatonin and flavonoids attenuates the oxidative stress and diabetic state induced by STZ 24, 25. The present study evaluates the in-vivo antioxidant activity of S. pinnata extract in diabetic rats
Increased concentration of malonaldehyde, an end-product of lipid peroxidation, were found in the liver homogenates of streptozotocin-induced diabetic rats 26, 27. In this study, the increase confirms this finding, which indicates an overall increase in oxidative stress in diabetic rats. Treatment with S. pinnata bark extract at a dose of 1.00 g/kg suggests an amelioration of oxidative stress. Oxidative stress is the result of a redox imbalance between the generation of ROS and the compensatory response from the endogenous antioxidant network. The activities of glutathione reductase, glutathione peroxidase and glutathione S-transferase in diabetic rats showed a reduction in the concentrations of these enzymes which is similarly mentioned by other authors 28, 29. Hyperglycaemia results in the increased enzymatic conversion of glucose to the polyalcohol sorbitol with concomitant decreases in NADPH and glutathione 29, 30. The resulting loss of antioxidant reducing equivalents as glutathione results in enhanced sensitivity to oxidative stress associated with intracellular ROS.
In the present study, the concentration of glutathione in the liver homogenates of S. pinnata extract treated diabetic rats is increased. This increased GSH may be one of the factors responsible for the inhibition of lipid peroxidation in rats treated with S. pinnata extract.
The AST and ALT are found in large quantities in the liver where they play an important role in the metabolism of amino acids. However, as a result of cellular damage caused by reactive oxygen species or toxicity to the liver, these enzymes may leak from the hepatocytes into the circulation where their levels become elevated. Therefore, the elevated levels of AST and ALT are indicators of functional disturbance of liver cell membranes and cellular infiltrations. Also, ALP is membrane-bound, and its alteration is likely to affect the membrane permeability and produce derangements in the transport of metabolites. A significant increase in serum activities of AST, ALT, and ALP in STZ-diabetic rats is also consistent with published data 31. However, the values were significantly decreased in plant extract-treated diabetic rats. Accordingly, results revealed the extract of S. pinnata accelerated the regeneration in hepatocytes, thus decreased the leakage of ALT, AST, and ALP into systemic circulation.
Further, the liver histopathology matches with biochemical results. Even though there was a pronounced antihyperglycemic effect in diabetic rats treated with glibenclamide, we report that the extract of S. pinnata appeared to be more effective than glibenclamide in ameliorating the oxidative stress in diabetic rats. The anti-hyperglycemic action of glibenclamide is mediated through stimulating insulin secretion via β-cells in the pancreatic tissue 32, and the results suggest that there is no direct effect on oxidative stress in diabetic rats. This may further strengthen the direct in-vivo antioxidant activity of the plant extract.
CONCLUSION: The results revealed that the aqueous bark extract of S. pinnata possessed antioxidant activities in STZ-diabetic rats.
ACKNOWLEDGEMENT: The financial assistance given by the University Grants Commission in Sri Lanka is greatly appreciated (UGC/ICD/CRF 2009/2/5). The authors wish to thank Dr. D.A.B.N. Gunarathne of the Department of Crop Science, Faculty of Agriculture, University of Ruhuna, Sri Lanka for the guidance given in statistical data analysis, Mrs. B.M.S. Malkanthie, Mr. G.H.J.M. Priyashantha and Mr. D.G.P. Pathmabandu Faculty of Medicine, University of Ruhuna, Sri Lanka for technical assistance.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Henriksen EJ, Diamond-Stanic MK, Marchionne EM and Henriksen EJ: Oxidative stress and the etiology of insulin resistance and type 2 diabetes. Free Radical Biology and Medicine 2010; 51: 993-9.
- Feng B, Ruiz MA and Chakrabarti S: Oxidative-stress-induced epigenetic changes in chronic diabetic complications. Canadian Journal of Physiology and Pharmacology 2013; 91(3): 213-20.
- Tupe RS, Diwan AG, Mittal VD, Narayanam PS and Mahajan KB: Association of plasma proteins at multiple stages of glycation and antioxidant status with erythrocyte oxidative stress in patients with type 2 diabetes. British Journal of Biomedical Sciences 2014; 71(3): 93-9.
- Murdolo G, Piroddi M, Luchetti F, Tortoioli C, Canonico B, Zerbinati C, Galli F and Iuliano L: Oxidative stress and lipid peroxidation by-products at the crossroad between adipose organ dysregulation and obesity-linked insulin resistance. Biochimie 2013; 95(3): 585-94.
- Singh R, Kaur N, Kishore L and Gupta GK: Management of diabetic complications: A chemical constituents based approach. Journal of Ethnopharmacology 2013; 150(1): 51-70
- Jayaweera DMA: Medicinal Plants (indigenous and exotic) used in Ceylon. Sri Lanka, Colombo, Edition 2nd, 1982.
- Attanayake AP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Study of anti-hyperglycaemic activity of some Sri Lankan medicinal plants in alloxan-induced diabetic rats. Ancient Science of life 2013a; 32(4): 193-8.
- Attanayake AP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Acute hypoglycaemic and anti-hyperglycaemic effects of ten Sri Lankan medicinal plant extracts in healthy and streptozotocin-induced diabetic rats. International Journal of Diabetes in Developing Countries 2014a; doi.10.1007/s13410-014-0217-8.
- Attanayake AP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Antihyperglycaemic, antihyperlipidaemic and β cell regenerative effects of Spondias pinnata (Linn. f.) Kurz. bark extract on streptozotocin-induced diabetic rats. European Journal of Integrative Medicine 2014b; 6(5): 588-596.
- Tandon S and Rastogi RP: Studies on the chemical constituents of Spondias pinnata. Planta Medica 1976; 29(2): 190-2.
- Attanayake AP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Toxicological investigation of Spondias pinnata (Linn. F.) Kurz. (Family: Anacardiaceae) bark extract in Wistar rats. International Journal of Green Pharmacy 2015; 9(1): 26-31.
- Vasconcelos CF, Maranhao HM, Batista TM, Carneiro EM, Ferreira F and Costa J: Hypoglycaemic activity and molecular mechanisms of Caesalpinia ferrea Martiusbark extract on streptozotocin-induced diabetes in Wistar rats. Journal of Ethnopharmacology 2011; 137(3): 1533-41.
- Abraham EC, Huff TA, Cope ND, Wilson JB Jr, Bransome ED Jr and Huisman TH: Determination of the glycosylated hemoglobin (HbA1) with a new micro column procedure. Suitability of the technique for assessing the clinical management of diabetes mellitus. Diabetes 1978; 27: 931-7.
- Bergmeyer HU, Scheibe P and Wahlefeld AW: Optimization of methods for aspartate aminotransferase and alanine aminotransferase. Clinical Chemistry 1978; 24: 58-73.
- Bowers GN Jr and Mc Comb RB: A continuous spectrophotometric method for measuring the activity of serum alkaline phosphatase. Clinical Chemistry 1966; 12: 70-89.
- Sedlak J and Lindsay RH: Estimation of total, protein-bound and non-protein sulfhydryl groups in tissue with Ellman's reagent. Analytical Biochemistry 1968; 25: 192-205.
- Jodynis-Liebert J, Murias M and Bloszyk E: Effect of sesquiterpene lactones on antioxidant enzymes and some drug metabolizing enzymes in rat liver and kidney. Planta Medica 2000; 66: 199-205.
- Habig WH, Pabst MJ and Jakoby WB: Glutathione S-transferase: The first enzymatic step in mercapturic acid formation. J of Biological Chemistry 1974; 249: 7130-9.
- Ohkawa H, Ohishi N and Yagi K: Assay for lipid peroxides in animal tissues by the thiobarbituric acid reaction. Analytical Biochemistry 1979; 95: 351-8.
- Lowry OH, Rosebrough NJ, Farr AL and Randall RJ: Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry 1951; 193: 265-75.
- Kajimoto and Kaneto H: Role of oxidative stress in pancreatic β-cell dysfunction. Annals of the New York Academy of Sciences, 2004.
- Baynes JW: Role of oxidative stress in the development of complications in diabetes. Diabetes 1991; 40(4): 405-12.
- Rivera-Ramírez F, Escalona-Cardoso GN, Garduño-Siciliano L, Galaviz-Hernández C, and Paniagua-Castro N: Anti-obesity and hypoglycaemic effects of aqueous extract of Ibervillea sonorae in mice fed a high-fat diet with fructose. Journal of Biomedicine and Biotechnology 2011; doi.org/10.1155/2011/968984.
- Coskun O, Kanter M, Korkmaz A and Oter S: Quercetin, a flavonoid antioxidant, prevents & protects streptozotocin-induced oxidative stress and β-cell damage in rat pancreas. Pharmacological Research 2005; 51(2): 117-23.
- Maciel MAM, Pinto AC, Arruda AC, Pamplona SGSR, Vanderlinde FA and Lapa AJ: Ethnopharmacology, phyto-chemistry and pharmacology: A successful combination in the study of Croton cajucara. Jour of Ethnopharmacology 2000; 70(1): 41-55.
- Dias AS, Porawski M, Alonso M, Marroni NPS, Collado and González-Gallego J: Quercetin decreases oxidative stress, NF-κB activation, and iNOS overexpression in the liver of streptozotocin-induced diabetic rats. Journal of Nutrition 2005; 135(10): 2299-304.
- Di Naso FC, de Mello RN, Bona S, Dias AS, Porawski M and Ferraz ABF: Effect of Agaricus blazei Murill on the pulmonary tissue of animals with streptozotocin-induced diabetes. Experimental Diabetes Research 2010; org/10.1155/2010/543926
- Sanders RA, Rauscher FM and Watkins JB: Effects of quercetin on antioxidant defense in streptozotocin-induced diabetic rats. Journal of Biochemical and Molecular Toxicology 2001; 15(3): 143-9.
- Vijayakumar M, Govindarajan R, Rao GMM, Rao ChV, Shirwaikar A, Mehrotra S and Pushpangadan P: Action of Hygrophila auriculata against streptozotocin-induced oxidative stress. Journal of Ethnopharmacology 2006; 104(3): 356-61.
- Stephen Irudayaraj S, Sunil C, Duraipandiyan V and Ignacimuthu S: Antidiabetic and antioxidant activities of Toddalia asiatica (L.) Lam. leaves in streptozotocin-induced diabetic rats. Journal of Ethnopharmacology 2012; 143: 515-23.
- Jarald E, Joshi SB and Jain DC: Biochemical study on the hypoglycemic effects of extract and fraction of Acasia catechu wild in alloxan-induced diabetic rats. International Journal of Diabetes and Metabolism 2009; 17: 63-9.
- Rajasekaran S, Sivagnanam K and Subramanian S: Modulatory effects of Aloe vera leaf gel extract on oxidative stress in rats treated with streptozotocin. Journal of Pharmacy and Pharmacology 2005; 57: 241-6.
How to cite this article:
Attanayake AP, Jayatilaka KAPW, Pathirana C and Mudduwa LKB: Potential antioxidant activities of Spondias pinnata (Family: Anacardiaceae) bark extract in rats with streptozotocin induced diabetes mellitus. Int J Pharmacognosy 2015; 2(4): 166-72. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(4).166-72.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
166-72
587
2077
English
IJP
A. P. Attanayake *, K. A. P. W. Jayatilaka, C. Pathirana and L. K. B. Mudduwa
Department of Biochemistry, University of Ruhuna, Matara, Sri Lanka
anoja715@yahoo.com
15 January 2015
19 March 2015
28 March 2015
10.13040/IJPSR.0975-8232.IJP.2(4).166-172
01 April 2015