PHYTOCHEMICAL STUDIES AND EVALUATION OF THE ANTIOXIDANT AND ANTIDIABETIC ACTIVITIES OF TERMINALIA CATAPPA L. (COMBRETACEAE) AND SENNA OCCIDENTALIS (L.) LINK (FABACEAE) FROM IVORY COAST
HTML Full TextPHYTOCHEMICAL STUDIES AND EVALUATION OF THE ANTIOXIDANT AND ANTIDIABETIC ACTIVITIES OF TERMINALIA CATAPPA L. (COMBRETACEAE) AND SENNA OCCIDENTALIS (L.) LINK (FABACEAE) FROM IVORY COAST
Koidio Bindé Boris *, Kadja Amani, Mamyrbekova-Békro Janat Akhanovna and Bekro Yves-Alain
Laboratoire de Chimie Bio-Organique et de Substances Naturelles (LCBOSN), UFRSFA, Université Nangui ABROGOUA, 02 BP 801 Abidjan 02, Ivory Coast.
ABSTRACT: Diabetes mellitus is a major public health challenge worldwide, particularly in sub-Saharan Africa where conventional therapeutic resources remain limited. In Ivory coast, the use of medicinal plants in the management of diabetes remains widespread despite advances in modern medicine. This study aims to evaluate the antidiabetic potential of two Ivorian medicinal plants, Terminalia catappa L. (Combretaceae) and Senna occidentalis (L.) Link (Fabaceae), through phytochemical analyses, antioxidant assays, and in-vivo pharmacological tests. Phytochemical screening revealed the presence of flavonoids, coumarins, sterols, terpenes, and tannins in all tested extracts. Extracts derived from T. catappa leaves stood out for their particularly high levels of total polyphenols and condensed tannins. Assessment of antioxidant activity revealed a significantly high free radical scavenging capacity for the ethanolic and hydroethanolic extracts of T. catappa (extracts Ca3 and Ca4), with respective CR₅₀ values of 0.0316 and 0.0318 mg/ml. In-vivo tests demonstrated that extracts from both plants significantly (p < 0.05) reduce glucose-overload-induced postprandial hyperglycemia, with efficacy comparable to that of low-dose glibenclamide. These results provide a solid scientific basis for the traditional use of Terminalia catappa and Senna occidentalis in diabetes management in Ivory Coast. The high content of phenolic compounds and marked antioxidant activity suggest that these properties may contribute to the observed antihyperglycemic effects. Further mechanistic studies are needed to elucidate the molecular targets involved.
Keywords: Terminalia catappa, Senna occidentalis, DPPH antioxidant activity, Antihyperglycemic effect, Phytochemistry, Ivorian medicinal plants
INTRODUCTION: Diabetes mellitus, particularly the more common type 2, is now one of the major global public health challenges. Considered one of the scourges of the third millennium, it affects both industrialized countries and low- and middle-income countries, with a steadily rising prevalence 42. This alarming increase is accompanied by high morbidity and mortality, linked in particular to associated metabolic and cardiovascular complications.
In Ivory coast, the situation is particularly concerning: approximately 78.7% of type 2 diabetes cases remain undiagnosed, contributing to an estimated 5,382 deaths per year among people aged 20 to 79 (IDF, 2025). This high proportion of undiagnosed cases constitutes a major obstacle to early and effective management of the disease.
Although conventional treatments (oral antidiabetics and insulin therapy) have proven effective, their use remains limited by several constraints, including high cost, associated side effects, and sometimes limited accessibility in resource-constrained settings 34. In this context, the search for accessible, safe, and effective therapeutic alternatives is a necessity. Medicinal plants represent an important source of bioactive compounds that can be utilized in the management of diabetes. The traditional Ivorian pharmacopoeia constitutes a rich reservoir of plant species used empirically for the treatment of diabetes and its complications 1. From this perspective, it seems appropriate to undertake an in-depth scientific investigation aimed at validating the traditional uses of these species and characterizing their biological properties. Thus, the general objective of this study is to evaluate the phytochemical potential as well as the antioxidant and antidiabetic activities of Terminalia catappa L. (Combretaceae) and Senna occidentalis (L.) Link (Fabaceae).
MATERIALS AND METHODS:
Plant Material: The plant matrices used in this study consist of leaves from Terminalia catappa L. (Combretaceae) and Senna occidentalis (L.) Link (Fabaceae), formerly known by the synonym Cassia occidentalis L. The samples were collected in the Abidjan district (Ivory Coast) during February 2020, during the dry season. The botanical identification of the two species was carried out at the National Floristic Center (NFC) of Félix Houphouët-Boigny University (UFHB) in Abidjan, using existing herbarium specimens: No. UCJ003136 for Terminalia catappa and No. UCJ009170 for Senna occidentalis. After harvesting, the leaves were carefully cleaned with distilled water to remove impurities, then dried away from direct light under air conditioning at 16 °C for 7 days. This low-temperature drying method preserves the integrity of heat-sensitive secondary metabolites. The dried leaves were then ground into a fine powder using an electric grinder (Moulinex, model LM 2201). The resulting powders were stored in airtight bags, protected from light and moisture, until use.
Animal Materials: Male Wistar rats (Rattus norvegicus, Wistar strain) weighing between 80 and 150 g and aged 6 to 12 weeks were used for this study. The animals were obtained from the vivarium of Félix-Houphouët-Boigny University located at the École Normale Supérieure d’Abidjan and housed in polypropylene cages in a climate-controlled animal facility (temperature: 22 ± 2 °C, humidity: 55 ± 5%), maintained on a 12-hour light/dark cycle. The animals had ad libitum access to drinking water and a standard diet. A 7-day acclimatization period was observed prior to the start of the experiments. All experimental procedures were conducted in accordance with international guidelines for the ethical use of laboratory animals (Guide for the Care and Use of Laboratory Animals, NRC, 2011) and were approved by the Committee of the African Pharmacopoeia and Natural Substances Research Center (PPASN) at Nangui Abrogoua University.
Methods:
Preparation of Plant Extracts:
Obtaining Aqueous Extracts by Decoction: Aqueous extraction was performed by decoction. Thirty grams (30 g) of plant powder were placed in a 500 mL Erlenmeyer flask fitted with a reflux condenser, to which 300 mL of distilled water were added, resulting in a drug-to-solvent ratio of 1:10 (w/v). The mixture was brought to a boil (100 °C) under stirring for 30 minutes. After cooling to room temperature, the decoction was filtered through a Büchner funnel fitted with Whatman No. 1 filter paper. The recovered filtrate was concentrated under reduced pressure using a rotary evaporator (water bath temperature: 45 °C), then dried in an oven at 45 °C for 16 hours until dry extracts of constant mass were obtained. The dry aqueous extracts thus obtained were designated Ca1 (Terminalia catappa) and Se1 (Senna occidentalis). The extraction yields were calculated using the following formula:
R (%) = (mass of dry extract / mass of plant powder) × 100
The extracts were stored in hermetically sealed glass vials, protected from light and moisture, until use.
Preparation of Aqueous, Hydroethanolic, and Ethanolic Extracts by Maceration: Maceration extraction was performed using three solvents of increasing polarity: distilled water, 80% (v/v) ethanol, and absolute ethanol (99.9%). For each solvent, 30 g of plant powder was placed in contact with 300 mL of solvent in a hermetically sealed glass flask, resulting in a drug-to-solvent ratio of 1:10 (w/v). Maceration was conducted at room temperature (25 ± 2 °C) under continuous magnetic stirring for 24 hours. This procedure was repeated three consecutive times (exhaustive maceration over 3 × 24 h), with the solvent replaced at each cycle to optimize extraction yield and ensure complete exhaustion of the plant material.
The resulting macerates were filtered through a Büchner funnel, and the filtrates were combined. The combined solutions were concentrated under reduced pressure using a rotary evaporator (water bath temperature: 40 °C for the ethanolic extracts, 45 °C for the aqueous extracts), then dried in an oven at 45 °C for 24 hours until dry extracts of constant mass were obtained. Extraction yields were calculated using the formula described above (section 2.3.1.1.).
The dry extracts thus obtained were coded as follows:
- Ca2 and Se2: aqueous extracts obtained by maceration of catappa and S. occidentalis, respectively;
- Ca3 and Se3: 80% hydroethanolic extracts of catappa and S. occidentalis, respectively;
- Ca4 and Se4: ethanolic extracts of catappa and S. occidentalis, respectively.
All extracts were stored in tightly sealed amber glass vials, protected from light and moisture at 4 °C until use.
Obtaining Selective Fractions by Liquid-Liquid Fractionation: To obtain fractions enriched in families of specialized metabolites with distinct polarities, each crude extract was subjected to liquid-liquid fractionation using a gradient of increasing polarity. One gram (1 g) of each crude extract was dissolved in 25 mL of distilled water, then transferred to a separatory funnel. The resulting aqueous solution was successively eluted with four organic solvents of increasing polarity: hexane (nonpolar solvent), chloroform (low-polarity solvent), ethyl acetate (medium-polarity solvent), and n-butanol (polar solvent), using three successive extractions of 15 mL for each solvent (3 × 15 mL), for a total volume of 45 mL per solvent. After each extraction, the organic phases were separated from the residual aqueous phase, then combined and washed with a saturated sodium chloride (NaCl) solution to remove residual water. The organic phases were then dehydrated over anhydrous sodium sulfate (Na₂SO₄), filtered, and evaporated to dryness under reduced pressure using a rotary evaporator. The resulting dry residues were weighed and stored at 4 °C in amber glass vials until use. The fractionation yields were calculated using the formula described above.
Phytochemical Screening by Thin-Layer Chromatography (TLC): Phytochemical screening of the crude extracts and fractions obtained from the liquid-liquid fractionation of Terminalia catappa and Senna occidentalis was performed by thin-layer chromatography (TLC), according to the methodologies described by 14, 37 adapted to laboratory conditions. Chromatographic separations were performed on 10 × 20 cm aluminum-backed silica gel 60 F₂₅₄ plates (Merck, Darmstadt, Germany).
Aliquots of extracts were dissolved in the appropriate solvent, then spotted using a capillary, 1 cm from the bottom of the plate and 1 cm from the side edges. Spotting was performed under a stream of hot air to ensure rapid evaporation of the carrier solvent. The chromatographic plates were developed in chromatographic chambers saturated with solvent vapor, using developing agents (solvent systems) specific to each family of specialized metabolites being sought. After migration, the plates were air-dried and then visualized under a UV lamp at 254 nm (extinguished fluorescence) and 366 nm (induced fluorescence), prior to the application of specific detection reagents.
Phytochemical Quantification:
Assay of Phenolic Compounds: Phenolic compound contents were quantified by spectrophotometry. Total phenols were determined according to Singleton et al. (1999) 47 and Flavie et al. (2024) 22, while total flavonoids were measured using the method of Hariri et al. (1991) 27 as adapted by N’guessan et al. (2011) 38. Finally, hydrolyzable tannins were assessed according to Dif et al. (2015) 19 and Flavie et al. (2024) 22, and condensed tannins according to the protocols of Broadhurst and Jones (1978) 13 and Heilmer et al. (2006) 28.
Total Sugar Determination: The total sugar content was determined using the sulfuric acid-phenol method, according to the protocol of Dubois et al. (1956) 21. Briefly, a 0.1 mL volume of ethanolic extract (1 mg/mL) was made up to 1 mL with distilled water, then mixed with 1 mL of phenol (5%) and 2 mL of concentrated sulfuric acid. After stirring and incubation at room temperature for 30 min, the absorbance was measured at 490 nm against a blank. Concentrations were determined from a glucose calibration curve (1 mg/mL) and expressed in milligrams of glucose equivalent per gram of dry matter (mg GE/g) according to formula (1):
Q (mg/g) = Vf × q / Vi × m (1)
Q: total sugar content; Vf: final volume after extraction; m: mass of powder sampled for extraction in g; q: amount of material calculated from the calibration curve in mg; Vi: initial volume of plant extract
Determination of Reducing Sugars: The reducing sugar content was quantified using the 3,5-dinitrosalicylic acid (DNS) method, adapted from Bernfeld (1955) 9. A volume of 0.1 mL of crude extract was mixed with 0.9 mL of distilled water and 0.5 mL of DNS reagent (prepared from 2 g of DNS, 60 g of sodium potassium tartrate, and 3.2 g of NaOH, all adjusted to 200 mL).
The reaction mixture was heated in a boiling water bath (100°C) for 5 min, then cooled to room temperature for 10 min. After adding 3.5 mL of distilled water, the absorbance was measured at 540 nm. Concentrations were determined using a glucose calibration curve (1 mg/mL), and the reducing sugar content was calculated according to equation (2):
Q (mg/g) = Vf × q / Vi × m (2)
Q: total sugar content; Vf: final volume after extraction; m: mass of powder sampled for extraction in g; q: amount of material calculated from the calibration curve in mg; Vi: initial volume of plant extract
Evaluation of Antioxidant Activity by DPPH: The antioxidant activity of the extracts was evaluated using the DPPH (2,2-diphenyl-1-picrylhydrazyl) free radical scavenging assay, according to the method of Blois (1958) 10 adapted by Tanoh et al. (2019) 57. Antioxidant efficacy was expressed as the median effective concentration (EC₅₀), corresponding to the extract concentration required to reduce the initial DPPH coloration by 50%. This value was determined graphically by linear regression from the curve of scavenging activity versus concentration 57.
Evaluation of Hypoglycemic Activity: Fifty-five (55) male Wistar rats, aged 6 to 12 weeks and weighing between 80 and 150 g, were randomly divided into 11 groups of 5 animals. After a 12-hour water fast, baseline blood glucose (t0) was measured using a glucose meter (One Call Extra®, USA).
The rats were then administered the following substances orally: distilled water (10 mL/kg, healthy control), glibenclamide (10 mg/kg, positive control), anhydrous glucose (4 g/kg, negative control/vehicle), or plant extracts at doses of 250 and 500 mg/kg. Blood glucose levels were monitored every 60 minutes over a 4-hour period (t60, t120, t (180), and t240), according to the protocols described by Gisèle et al. (2017) 25 and Nko’o et al. (2024) 39.
Evaluation of Antihyperglycemic Activity: An oral glucose tolerance test (OGTT) was performed on fifty-five (55) male Wistar rats (80–150 g; 6–12 weeks), divided into 11 groups of 5 animals. After a 12-hour fast, hyperglycemia was induced by oral administration of an anhydrous glucose solution (4 g/kg). The various treatments were administered 60 minutes after the glucose load to evaluate their ability to inhibit the glycemic response.
The experimental groups included: a healthy control (distilled water, 10 mL/kg), a negative control (glucose alone, 4 g/kg), a reference or positive control (glibenclamide, 10 mg/kg), and the groups treated with the extracts (250 and 500 mg/kg). Basal blood glucose (t0) was measured immediately before administration, and glucose levels were then monitored every 60 min for 4 h (t60 , t120 , t(180), and t240) using a glucose meter (One Call Extra®, USA), according to the protocols of Gisèle et al. (2017) 25 and Nko’o et al. (2024) 39.
Statistical Analyses: Data are presented as means ± standard error of the mean (SEM). Analyses were performed using one-way ANOVA followed by Tukey’s post-hoc test to compare the treated groups with the negative control. GraphPad Prism software (version 9.0) was used, and differences were considered significant at p < 0.05.
RESULTS AND DISCUSSION:
Extraction Yields: The extraction yields presented in Table 1 and 2 show significant variability depending on the plant organ and the polarity of the extraction solvent used.
TABLE 1: YIELDS OF CRUDE EXTRACTS
| Yield (%) | ||||||||
| Crude extract | Ca1 | Ca2 | Ca3 | Ca4 | Se1 | Se2 | Se3 | Se4 |
| Powder mass (g) | 30 | 30 | 30 | 30 | 30 | 30 | 30 | 30 |
| Mass of crude extract (g) | 6.23 | 7.24 | 5.67 | 3.4 | 5.85 | 7.17 | 4.15 | 2.3 |
| Yield (%) | 20.77 | 24.13 | 18.90 | 11.33 | 19.50 | 23.90 | 13.83 | 7.67 |
Ca1: aqueous decoction, Ca2: aqueous maceration, Ca3: hydroethanolic maceration (80%), Ca4: ethanolic maceration of Terminalia catappa; Se1: aqueous decoction, Se2: aqueous maceration, Se3: hydroethanolic maceration (80%), Se4: ethanolic maceration of Senna occidentalis.
TABLE 2: YIELDS OF LIQUID-LIQUID FRACTIONATION (IN %)
| Yield (%) | ||||||||
| Selective fraction | Ca1 | Ca2 | Ca3 | Ca4 | Se1 | Se2 | Se3 | Se4 |
| Hexane | ~0.4 | ~0 | ~0.4 | ~1 | ~0.5 | 1.1 | ~0.7 | ~1 |
| Chloroform | ~1.3 | 3.5 | 2.7 | ~0.2 | 0.6 | 0.7 | 0.9 | ~0.7 |
| Ethyl acetate | ~0.9 | 3.6 | 2 | 5.6 | 1.6 | 1.6 | 3.6 | 0.4 |
| n-Butanol | 10.6 | 15 | 2.9 | 8 | 13 | 7.1 | 14.2 | 7.9 |
Ca1: aqueous decoction, Ca2: aqueous maceration, Ca3: hydroethanolic maceration (80%), Ca4: alcoholic maceration of Terminalia catappa; Se1: aqueous decoction, Se2: aqueous maceration, Se3: 80% hydroalcoholic maceration, Se4: alcoholic maceration of Senna occidentalis
Analysis of the yields Table 1 and 2 highlights the decisive influence of the nature of the plant part and the solvent system on extraction efficiency, suggesting a differential distribution of specialized metabolites within the plant.
Crude Extracts: Extraction yields for crude extracts vary significantly depending on the type of solvent used. Aqueous extracts obtained by maceration show the highest yields, with values of 24.13% for T. catappa (Ca2) and 23.90% for S. occidentalis (Se2). In contrast, ethanolic extracts show the lowest yields, at 11.33% for Ca4 and 7.67% for Se4. This inverse relationship between solvent polarity and extraction yield is explained by the hydrophilic nature of the parietal constituents of plant cells, which promotes the solubilization and transfer of polar compounds to the aqueous phase 15. Comparable results have been reported in the literature for other tropical medicinal plants, where aqueous extractions consistently outperform organic solvents in terms of crude yield 14, 15.
Selective Fractions: Regarding the fractions obtained from liquid-liquid fractionation Table 2, the hexane and chloroform fractions exhibit yields below 1.3%, reflecting a low content of nonpolar compounds such as waxes, essential oils, and chlorophylls. This profile is characteristic of polyphenol-dominant plants, whose major specialized metabolites are polar in nature 15. In contrast, the butanol fractions exhibit the highest yields among the selective fractions, which is consistent with n-butanol’s strong affinity for flavonoid glycosides and glycosylated phenolic compounds 33. These observations suggest that the two species studied exhibit a predominantly polyphenolic metabolic profile, which justifies their traditional use in the management of diabetes and directs further phytochemical investigations toward the polar fractions.
Phytochemical Profile of the Selective Fractions: The different classes of phytochemicals identified by thin-layer chromatography (TLC) are presented in Table 3 through 6. Qualitative analysis reveals the presence of several major chemical groups, including sterols, terpenes, coumarins, flavonoids, alkaloids, and tannins, consistent with the phytochemical profiles generally reported for bioactive plant extracts.
The Liebermann–Bürchard reagent is classically used for the detection of sterols and triterpenes. In this study, it revealed sterols as yellow spots and terpenes as orange to violet stains under UV irradiation at 365 nm, which is consistent with observations described in the literature 4, 46. Godin’s reagent also confirmed the presence of sterols, with blue coloration under UV light (365 nm) and violet to brown hues in visible light, indicating reactions specific to steroid rings. Coumarins were detected using a 5% KOH solution, which induces characteristic fluorescence. The observed spots appear yellow in visible light and exhibit green to blue fluorescence under UV light at 365 nm, consistent with the well-documented photophysical properties of these compounds 11, 57. Flavonoids were identified using Neu’s reagent, which forms fluorescent complexes with these compounds. Flavonoids appear yellow in visible light and exhibit blue to red fluorescence under UV light (365 nm), depending on their structure and degree of substitution 18, 43.
Alkaloids were detected using Dragendorff’s reagent, producing orange spots under visible light, which is a widely used standard method for screening these nitrogen-containing compounds 29. Finally, tannins were detected using ferric chloride (2% FeCl₃), yielding grayish to dark stains under visible light, indicative of the formation of phenol–iron complexes, characteristic of high-molecular-weight phenolic compounds 52, 54, 55. Overall, these results confirm the phytochemical richness of the extracts studied and highlight the value of CCM as a rapid, reliable, and inexpensive technique for the preliminary screening of secondary metabolites.
TABLE 3: TLC PROFILE OF HEXANE EXTRACTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Extracts | Rf, Colors, possible phytochemicals |
| Ca1I | 0.86, ob -vic , teb /stc |
| Ca2I | 0.86, ob, teb, 0.95, vic, st; 0.50, jb, st; 0.13, mc, st |
| Ca3I | 0.98, ob - vic , te/st, 0.74, Vic, 0.49, Jpb, 0.30, Jb, 0.26, vic, st |
| Ca4I | 0.49, jpb , st; 0.98, vic , st |
| Se1I | 0.98, ob - vic ,te/st |
| Se2I | 0.93, ob , te; 0.90, Vic , st |
| Se3I | 0.95, ob- Vic , te/st ; 0.15, Bb , te; 0.46, Jb , st; 0.11,Vic , st |
| Se4I | 0.73, jb, st; 0.64, jb, st; 0.53, jb, st; 0.48, vic, st; 0.48, bb, te; 0.20, ob , te; 0.15, bpb , te; 0.11, ob -bc , te/st |
b: Liebermann-Bürchard; c: Godin; J: yellow; V: green; B: blue; G: gray; O: orange; M: brown; Vi: violet; R: red; st/sterol; te/terpene
TABLE 4: TLC PROFILE OF CHLOROFORM EXTRACTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Extract | Rf , Colors, possible phytochemicals |
| Ca1II | 0.48, ba , yellow; 0.56, ba , yellow; 0.53, Jb, st |
| Ca2II | 0.64, vib– bd, te/fl; 0.59, ja, cou; 0.56, ja, cou; 0.49, ba, cou; 0.46, ba– vib, cou/te |
| Ca3II | 0.86, rd, fl; 0.79, vib, bd, ja, fl/te/cou; 0.75, jb, st; 0.64, b²d - vib, fl/te; 0.59, ja, cou; 0.48, ba, cou; 0.46, vib - ba, te/cou; 0.43, ja, cou |
| Ca4II | 0.64, vib - bd, te/fl |
| Se1II | 0.56, ba, cou; 0.50, ba- bd- Jb, cou/fl/st; 0.44, jd- bb, fl/te; 0.40, ge- ja, ta/cou; 0.34, bd, fl; 0.24, ba -bb -bd, cou/te/st; 0.08, ja - jb -jd, cou/st/fl |
| Se2II | 0.94, jb, st; 0.71, jb, st; 0.56, ja, cou; 0.48, ba, cou; 0.48, bb, te; 0.44, jb- jd, st/fl; 0.41, ja - ge, cou/ta; 0.38, ja, cou; 0.34, jd , fl; 0.25, jd , fl; 0.23, va , cou; 0.08, jd -ja -jb , fl/cou/st |
| Se3II | 0.88, ja, neck; 0.55, ba, neck; 0.48, ba, neck; 0.45, ja-bb-jd-ge, neck/te/fl/ta; 0.43, jb, st; 0.38, ja -jd , cou/fl; 0.36, va, cou; 0.33, bd, fl ; 0.30, va, cou; 0.25, bb -bd, te/fl; 0.20, va, neck; 0.08, jb, st; 0.06, ja- jd, neck/fl |
| Se4II | 0.08,; 0.25, bd; 0.91, rd , fl; 0.86, rd, fl; 0.80, jb, st; 0.58, ba, cou; 0.48, ba, cou; 0.44, ja - jb -jd -ge, cou/st/fl/ta; 0.40, bd ,fl; 0.38, ja , cou; 0.34, bd , fl; 0.30, ja , cou; 0.25, bb, te; 0.23, va, cou; 0.20, ja, cou; 0.08, jb -jd, st/fl |
a: KOH; b: Liebermann-Bürchard; c: Godin; d: Neu; e: FeCl₃; f: Dragendorff; J: yellow; V: green; B: blue; G: gray; O: orange; M: brown; Vi: violet; Br: brown; R: red; st/sterol; te/terpene; cou/coumarin; fl/flavonoid; ta/tannin
TABLE 5: TLC PROFILE OF ETHYL ACETATE EXTRACTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Extract | Rf, Colors, potential phytochemicals |
| Ca1III | 0.93, ja, stem; 0.68, va, stem; 0.64, vd, flower; 0.60, bd, flower; 0.55, ba -vd, stem/flower; 0.49, ge, stem; 0.35, ba - bd, stem/flower; 0.25, ja -ge, stem/stem; 0.08, jd, flower; 0.04, od - ge , flower/stem |
| Ca2III | 0.69, ba, cou; 0.64, vd, fl; 0.59, bd, fl; 0.55, ba - vd, cou/fl; 0.36, ba, cou; 0.30, bd, fl; 0.24, ja - ge, cou/ta; 0.08, jd , fl ; 0.04, od -ge , fl/ta |
| Ca3III | 0.90, ba - bd, neck/flute; 0.83, ja, neck; 0.80, ja, neck; 0.75, ba, neck; 0.71, ja, neck; 0.68, ja, cou; 0.64, jd, fl; 0.53, ja- vd, cou/fl; 0.41, ja, cou |
| Ca4III | 0.04, od; 0.09, jd; ; ; 0.63, vd, fl; 0.55, ba - bd, cou/fl; 0.41, ja, cou; 0.35, ba, cou; 0.28, bd, fl; 0.15, ja, cou; 0.04, ge, ta |
| Se1III | 0.93, rd, fl; 0.74, jd, fl; 0.68, ja, cou; 0.65, va- jd- ge, cou/fl/ta; 0.60, va, cou; 0.56, va -ge , cou/ta; 0.53, vd, fl; 0.50, va, cou ; 0.30 ja, cou; 0.19, ja -jd , cou/fl; 0.13, ja - jd, cou/fl; 0.04, ja -jd, cou/fl |
| Se2III | 0.93, rd, fl; 0.75, jd, fl; 0.68, ja -of, cou/al; 0.65, jd -ge, fl/ta; 0.61, va -of, cou/al; 0.56, ge, ta; 0.30, ja, cou; 0.18, jd, fl ;0.15, ja, cou; 0.11, jd, fl ; 0.05, ja - jd, cou/fl |
| Se3III | 0.96, rd, fl; 0.94, ge, ta; 0.83, rd, fl; 0.81, jd, fl; 0.75, rd-ge, fl/ta; 0.66, jd, fl; 0.61, va -jd -of, cou/fl/al; 0.58, ja , cou; 0.53, jd, fl; 0.49, ja, cou; 0.28, ja -jd, cou/fl; 0.19, jd, fl ; 0.14, ja, cou; 0.11, jd , fl ; 0.05, ja -jd , cou/fl; 0.03, ja, cou |
| Se4III | 0.65, va - jd, neck/fl; 0.61, va - rd - of, neck/fl/al; 0.58, ja, neck; 0.54, va - jd, neck/fl; 0.50, va, cou; 0.43, ja, cou; 0.34, ja, cou; 0.26, ja, cou; 0.18, jd, fl; 0.15, ja, cou; 0.04, ja -jd, cou/fl |
a: KOH; b: Liebermann-Bürchard; c: Godin; d: Neu; e: FeCl₃; f: Dragendorff; J: yellow; V: green; B: blue; G: gray; O: orange; M: brown; Vi: violet; Br: brown; R: red; cou/coumarin; fl/flavonoid; ta/tannin; al/alkaloid
TABLE 6: TLC PROFILE OF N-BUTANOL EXTRACTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Extract | Rf , Colors, possible phytochemicals |
| Ca1IV | 0.19, jd, fl;; 0.13, jd, fl ; 0.09, jd, fl; 0.04, va -jd, cou/fl; 0.03, jd, fl; |
| Ca2IV | 0.40, bd, fl; 0.20, jd -ge, fl/ta; 0.16, ge, ta; 0.14, ja -jd, cou/fl; 0.11, ge, ta; 0.04, va - jd, cou/fl |
| Ca3IV | 0.66, jd - ja, fl/cou; 0.39, jd, fl; 0.31, ja, cou; 0.21, jd, fl; 0.19, ge, ta; 0.15, jd -ge, fl/ta; 0.13, ja -jd, cou/fl; 0.09, jd -ge , fl/ta; 0.04, ge , ta |
| Ca4IV | 0.25, bd, fl; 0.21, jd, fl; 0.14, jd, fl; 0.13, ja -jd, cou/fl; 0.11, ja, cou; 0.09, jd, fl; 0.08, jd, fl ; 0.04, va -ge, cou/fl |
| Se1IV | 0.64,; 0.75, jd, fl; 0.73, jd, fl; 0.65, va - jd , cou/fl; 0.58, jd, fl; 0.54, va, cou; 0.40, va, cou; 0.38, jd, fl; 0.29, jd, fl; 0.23, ja, cou; 0.18, jd, cou; 0.13, ja - jd, cou/fl ; 0.11, ja, cou 0.05, ja - jd, cou/fl |
| Se2IV | 0.74, jd -ja, cou/fl; 0.65, jd, fl; 0.04, ja, cou |
| Se3IV | 0.98, rd, fl;; 0.96, jd, fl; 0.83, rd, fl; 0.76, jd, fl; 0.64, ja- jd, cou/fl; 0.53, ja, cou; 0.28, ja, cou; 0.13, ja, cou; 0.11, jd, fl; 0.05, jd, fl; 0.04, ja -jd, cou/fl |
| Se4IV | 0.64, jd - ja , fl/cou; 0.11, jd - ja, fl/cou; 0.05, jd - ja; fl/cou |
a: KOH; b: Liebermann-Bürchard; c: Godin; d: Neu; e: FeCl3; f: Dragendorff; J: yellow; V: green; B: blue; G: gray; O: orange; M: brown; Vi: violet; Br: brown; R: red; cou/coumarin; fl/flavonoid; ta/tannin
Phytoconstituent Content: Table 7 presents the total phenolic compound contents of the various crude extracts. The Ca2and Ca3extracts of Terminalia catappa stand out for having the highest concentrations, reaching 67.53 ± 2.09 mg EAG/g and 55.38 ± 0.395 mg EAG/g, respectively, compared to the Se3 and Se1 extracts from Senna occidentalis (formerly Cassia occidentalis), which contain 39.69 ± 0.298 mg EAG/g and 19.60 ± 0.447 mg EAG/g, respectively. These results confirm the high polyphenol content of T. catappa, previously reported in the literature, with levels reaching up to 95 mg EAG/g in certain parts of the fruit, particularly the epicarp 23, 24. Furthermore, studies using technical assisted extraction techniques, such as ultrasonication, have shown even higher yields of phenolic compounds in the leaves and fruits of this species 2, 3, 35, 36. The high total phenolic content observed in T. catappa extracts is of significant pharmacological interest, particularly in the context of type 2 diabetes. Indeed, phenolic compounds are recognized for their ability to neutralize reactive oxygen species (ROS) and mitigate oxidative stress, a key factor in the pathophysiology of diabetes and its complications 50, 51, 52, 54.
Thus, aqueous and hydroethanolic (80%) macerates could be promising extracts for the development of phytotherapeutic approaches to treat diabetes. Regarding flavonoids, the contents range from 2.76 ± 0.370 to 6.54 ± 0.277 mg EQ/g, with higher concentrations observed in S. occidentalis, particularly in the hydroethanolic extract (80%). These results are consistent with chromatographic observations (TLC), where Neu’s reagent revealed several fluorescent spots characteristic of flavonoids, particularly in the chloroform and ethyl acetate fractions. These compounds are well documented for their ability to inhibit key enzymes of carbohydrate metabolism, such as α-glucosidase and α-amylase, thereby contributing to the regulation of postprandial blood glucose levels 18, 31, 32, 61, 62. The hydrolyzable tannin contents range from 0.7346 ± 0.126 to 7.773 ± 0.144%, while those of condensed tannins range from 41,167 ± 231 to 119,000 ± 346 µg ECT/mg. The Se4 extract of S. occidentalis has the highest content of hydrolyzable tannins, whereas the Ca2 extract of T. catappa has the highest concentration of condensed tannins. These results differ from those reported by Benchaachoua et al. (2018) 7, 8, who demonstrated greater efficacy of the decoction for the extraction of condensed tannins, suggesting that extraction parameters (solvent, temperature, duration) strongly influence the recovery of these metabolites. Tannins, particularly condensed forms, are known for their antioxidant properties and their ability to modulate carbohydrate absorption, thereby reinforcing their potential in diabetes management 53, 55, 56. Total sugar contents Table 8 range from 0.976 ± 0.43 mg/g to 13.894 ± 1.357 mg/g across all extracts. Ethanol macerates (absolute and 80%) generally exhibit the highest concentrations, while the aqueous decoction of S1 of S. occidentalis shows the lowest content. This decrease could be explained by partial thermal degradation of simple sugars during the decoction process, a phenomenon already described in plant matrices subjected to high temperatures. As for reducing sugars, their levels remain lower than those of total sugars in all extracted, with values ranging from 0.036 ± 0.00 mg/g to 0.0748 ± 0.020 mg/g. These low concentrations suggest a predominance of complex carbohydrates, such as polysaccharides and glycosides, over free monosaccharides. From a nutritional and pharmacological standpoint, the low content of reducing sugars, particularly in the Se4and Se3extracts of S. occidentalis, is an advantage for potential use in diabetes management by limiting the intake of rapidly assimilable sugars.
TABLE 7: PHYTOCONSTITUENT CONTENTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Total Phenols (µg EAG/g) | Total Flavonoids
(in %) |
Hydrolyzable tannins (in %) | Condensed tannins (µg ECT/mg) | |
| Ca1 | 46,471.3 ± 199.1 | 3.58 ± 0.306 | 2.557 ± 0.154 | 94,167 ± 288.7 |
| Ca2 | 67,534.4 ± 2,090 | 4.85 ± 0.530 | 4.426±0.146 | 119,000±346 |
| Ca3 | 55,379.3 ± 395 | 3.71±0.496 | 6.289±0.116 | 55,833±252 |
| Ca4 | 40,609.2 ± 647 | 3.09 ± 0.052 | 4.214±0.176 | 70,000±278 |
| Se1 | 19,603.4 ± 447.9 | 2.96±0.034 | 0.9006±0.152 | 51,667±317 |
| Se2 | 21,040.2 ± 199.1 | 2.76 ± 0.370 | 0.7346±0.126 | 43,667 ± 231 |
| Se3 | 39,689.6 ± 298.6 | 6.54 ± 0.277 | 2.975±0.165 | 51,833 ± 480 |
| Se4 | 22,534.5 ± 149.3 | 3.69 ± 0.152 | 7.773±0.144 | 41,167±231 |
Ca1: aqueous decoction, Ca2: aqueous maceration, Ca3: 80% hydroethanolic maceration, Ca4: ethanolic maceration of Terminalia catappa; Se1: aqueous decoction, Se2: aqueous maceration, Se3: 80% hydroethanolic maceration, Se4: ethanolic maceration of Senna occidentalis.
TABLE 8: TOTAL AND REDUCING SUGAR CONTENTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Ca1 | Ca2 | Ca3 | Ca4 | Se1 | Se2 | Se3 | Se4 | |
| Total sugars (mg/g) | 2.8810
±0.104 |
1.0891
±0.116 |
13.1804
±1.925 |
13.8937
±1.357 |
0.9760
±0.435 |
8.9354
±1.531 |
7.7871
±0.766 |
5.7777
±0.122 |
| Reducing sugars (mg /g) | 0.0808
±0.014 |
0.0531
±0.009 |
0.1341
±0.042 |
0.0748
±0.020 |
0.0816
±0.028 |
0.0418
±0.009 |
0.0966
±0.022 |
0.0362
±0.001 |
Ca1: aqueous decoction, Ca2: aqueous maceration, Ca3: 80% hydroethanolic maceration, Ca4: ethanolic maceration of Terminalia catappa; Se1: aqueous decoction, Se2: aqueous maceration, Se3: 80% hydroethanolic maceration, Se4: ethanolic maceration of Senna occidentalis.
DPPH Antioxidant Profile: The CR₅₀ values of the various extracts Table 9, determined using the DPPH radical scavenging assay, provide a reliable quantitative indicator of their antioxidant capacity. The CR₅₀, defined as the concentration required to inhibit 50% of free radical, is inversely proportional to antioxidant activity 12, 57. In the present study, the CR₅₀ values ranged from 0.0316 ± 0.002 mg/mL to 0.9828 ± 0.058 mg/mL, highlighting significant variability in antioxidant activity depending on the extracts and solvents used. The hydroethanolic (Ca3; CR₅₀ = 0.0316 ± 0.002 mg/mL) and ethanolic (Ca4; CR₅₀ = 0.0318 ± 0.004 mg/mL) macerates of Terminalia catappa leaves exhibit the highest activities. This efficacy can be attributed to the strong ability of polar solvents to extract phenolic compounds, particularly flavonoids and tannins, which are known for their capacity to neutralize free radicals via electron or hydrogen atom transfer mechanisms 16, 45. However, these extracts remain less active than vitamin C (IC₅₀ = 0.0070 ± 0.001 mg/mL), used as a standard. This difference is explained by the pure and highly reactive nature of ascorbic acid, unlike plant extracts, which are complex mixtures of compounds that may exhibit synergistic or antagonistic effects. In comparison, extracts from Senna occidentalis leaves exhibit significantly higher CR₅₀ values, indicating lower antioxidant activity. This observation could be related to a lower content of total phenolic compounds or to a different composition of secondary metabolites. Indeed, several studies have established a positive correlation between total polyphenol content and antioxidant activity measured by the DPPH, ABTS, or FRAP methods 44, 58. Furthermore, the results obtained confirm the decisive influence of the extraction solvent on the yield of bioactive compounds. Hydroalcoholic solvents are often described as optimal for the extraction of natural antioxidants, due to their intermediate polarity, which allows for the solubilization of a wide range of molecules 20. Thus, Terminalia catappa extracts, particularly the hydroethanolic and ethanolic macerates (Ca3 and Ca4), exhibit significant antioxidant potential and could serve as promising natural sources for pharmacological, food, or cosmetic applications. Nevertheless, further investigations, including the quantification of total polyphenols, the identification of active compounds using chromatographic techniques (HPLC, LC-MS), and evaluation via other antioxidant assays (ABTS, FRAP), would be necessary for a better understanding of the mechanisms involved and the optimal utilization of these extracts.
TABLE 9: EC₅₀ (MG/ML) OF CRUDE EXTRACTS OF TERMINALIA CATAPPA AND SENNA OCCIDENTALIS
| Se1 | Se2 | Se3 | Se4 | Ca1 | Ca2 | Ca3 | Ca4 | VIT C | |
| CR50 (mg/mL) | 0.9828
±0.058 |
0.4716
±0.051 |
0.1553
±0.023 |
0.2144
±0.038 |
0.2578
±0.032 |
0.0857
±0.012 |
0.0316
±0.002 |
0.0318
±0.004 |
0.0070
±0.001 |
Ca1: aqueous decoction, Ca2: aqueous maceration, Ca3: 80% hydroethanolic maceration, Ca4: ethanolic maceration of Terminalia catappa; Se1: aqueous decoction, Se2: aqueous maceration, Se3: 80% hydroethanolic maceration, Se4: ethanolic maceration of Senna occidentalis
Effect of Terminalia catappa and Senna occidentalis extracts on Blood Glucose Levels in Rats:
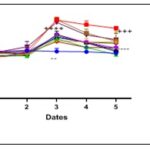
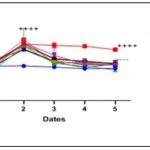
Effect of Terminalia catappa and Senna occidentalis extracts on Blood Glucose Levels in Pretreated Rats: The effects of Terminalia catappa and Senna occidentalis extracts in pretreated rats are shown in Fig. 4 and 5. One hour after administration of the various crude extracts, no significant change in blood glucose levels was observed compared to healthy controls (78.2 ± 0.46 mg/dL for Terminalia catappa and 80.0 ± 2.6 mg/dL for Senna occidentalis). These results indicate that the tested extracts do not induce basal hypoglycemia, suggesting an absence of direct hypoglycemic effect under normoglycemic conditions. Following oral administration of glucose (4 g/kg), a significant increase in blood glucose levels (p < 0.0001) was observed in all groups, confirming the effective induction of transient hyperglycemia. This model replicates the postprandial response and is widely used to evaluate the anti-hyperglycemic potential of plant extracts 25, 39. At a dose of 250 mg/kg body weight, the blood glucose levels measured at t60 for the extracts of Terminalia catappa and Senna occidentalis are comparable to those observed in animals treated with glibenclamide (104–109 mg/dL), suggesting a similar ability to modulate the initial hyperglycemic peak at low doses. In contrast, at a dose of 500 mg/kg, notable differences emerge among the extracts. For Terminalia catappa, the Ca3 hydroethanolic extract stands out with a blood glucose level of 93.6 ± 1.2 mg/dL at t60, lower than that of the other extracts and comparable to, or even slightly better than, that of glibenclamide. The other extracts show higher values (120 ± 4.2 to 130 ± 3.2 mg/dL), indicating lower efficacy in limiting the glycemic peak. Conversely, in Senna occidentalis, the 500 mg/kg extracts show reduced efficacy, with high blood glucose levels for Se2 (138.4 ± 1.7 mg/dL) and Se3 (133.0 ± 4.5 mg/dL), close to those of the positive control (140 mg/dL), indicating a low capacity to contain the initial rise in blood glucose. Starting at t120, a gradual and significant decrease in blood glucose is observed in the groups treated with both plant species. This kinetics is consistent with data in the literature attributing to polyphenolic compounds a delayed action involving, in particular, the inhibition of digestive enzymes (α-amylase, α-glucosidase), the improvement of peripheral glucose uptake, and the modulation of insulin sensitivity 26. In Terminalia catappa, at t240, the majority of extracts administered at 250 mg/kg, with the exception of Ca2, induce a marked reduction in hyperglycemia. Similarly, at 500 mg/kg, the Ca3and Ca4extracts yielded final blood glucose levels of 86 ± 2 mg/dL, close to normoglycemic values, indicating high antihyperglycemic activity. In contrast, the Ca2 extracts at 250 mg/kg, as well as Ca1 and Ca2 at 500 mg/kg, showed significantly higher blood glucose levels (100 ± 3.2 mg/dL), suggesting more limited efficacy. Glibenclamide also induces a significant decrease in blood glucose levels, with a final value of 80.4 ± 5 mg/dL. Regarding Senna occidentalis, the reduction in hyperglycemia becomes noticeable starting at t180, unlike the positive control, in which blood glucose levels remain high throughout the experiment. At t240, the extracts administered at 250 mg/kg showed mean blood glucose levels of 85.4 ± 1 mg/dL, close to those obtained with glibenclamide (76.8 ± 0.4 mg/dL) and to normoglycemic values. In contrast, the 500 mg/kg extracts showed higher final blood glucose levels (96.8 ± 0 to 100 ± 2 mg/dL), suggesting a lack of a favorable dose-dependent effect and, potentially, saturation or an antagonistic interaction between bioactive compounds at high doses. Overall, these results indicate that Terminalia catappa exhibits more pronounced antihyperglycemic activity than Senna occidentalis, particularly for the hydroethanolic and ethanolic extracts. This activity may be linked to a higher content of bioactive phenolic compounds capable of intervening at various levels of carbohydrate metabolism. However, further analyses, including the quantification of total polyphenols, the identification of active molecules, and the study of mechanisms of action, are necessary to confirm these hypotheses and better assess the therapeutic potential of these species.
FIG. 1: EFFECT OF TERMINALIA CATAPPA ON BLOOD GLUCOSE LEVELS IN PRETREATED RATS
FIG. 2: EFFECT OF SENNA OCCIDENTALIS ON BLOOD GLUCOSE LEVELS IN PRETREATED RATS
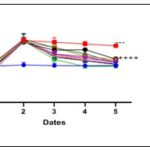
Effect of Terminalia catappa and Senna occidentalis on Blood Glucose Levels in Post-Treated Rats: The effects of Terminalia catappa and Senna occidentalis extracts on blood glucose levels in rats under post-treatment conditions are presented in Fig. 6 and 7. One hour after oral administration of glucose, significant hyperglycemia (p < 0.0001) was observed in all groups.
FIG. 3: EFFECT OF TERMINALIA CATAPPA ON BLOOD GLUCOSE LEVELS IN RATS AFTER TREATMENT
FIG. 4: EFFECT OF SENNA OCCIDENTALIS ON BLOOD GLUCOSE LEVELS IN POST-TREATED RATS
In rats treated with Terminalia catappa extracts, the mean blood glucose level reached 135 ± 4 mg/dL at t60. Similarly, in those receiving Senna occidentalis extracts, blood glucose levels rose from 81.8 ± 1 mg/dL to 138.2 ± 5.6 mg/dL during the first hour, representing an increase of approximately 65%. These results confirm the reproducibility and robustness of the model of hyperglycemia induced by oral glucose overload. The positive control group, consisting of untreated hyperglycemic rats, maintained elevated blood glucose levels throughout the experiment, with final blood glucose levels of 120.6 ± 3 mg/dL for Terminalia catappa and 126 ± 7 mg/dL for Senna occidentalis, corresponding to limited spontaneous decreases of 10% and 8%, respectively.
This demonstrates the stability and persistence of the induced hyperglycemic state. As early as t120, i.e., one hour after administration of the extracts, a significant reduction in blood glucose (p < 0.0001) is observed in all treated groups. In Terminalia catappa, the mean blood glucose level decreased to approximately 100±3.2 mg/dL. In Senna occidentalis, blood glucose values at t120 ranged from 104±2 to 122±3.7 mg/dL, indicating a significant antihyperglycemic response, albeit slightly more gradual. At the end of the experiment (t240), all extracts from both species induced a significant reduction in hyperglycemia compared to the positive control.
The extracts of Terminalia catappa showed a marked decrease in blood glucose levels, consistent with the results obtained in the pretreatment. For Senna occidentalis, final blood glucose levels ranged from 85 ± 1.7 mg/dL to 95 mg/dL, values close to normoglycemia. Glibenclamide, used as a reference, caused a rapid and pronounced reduction in blood glucose levels as early as t120, with a final value of 96.2±6 mg/dL, confirming its efficacy in this experimental model. Overall, these results demonstrate that extracts of Terminalia catappa and Senna occidentalis possess significant antihyperglycemic activity under postprandial conditions. The absence of a hypoglycemic effect under basal conditions, combined with a marked reduction in induced hyperglycemia, suggests a regulatory rather than a sudden corrective effect on blood glucose levels. Mechanistically, these effects could be attributed to the presence of bioactive secondary metabolites such as polyphenols, flavonoids, tannins, alkaloids, and coumarins. These compounds are known to act at various levels of carbohydrate metabolism: stimulation of insulin secretion by pancreatic β-cells, improvement of insulin sensitivity, inhibition of digestive enzymes (α-amylase and α-glucosidase), reducing intestinal glucose absorption, and modulating hepatic gluconeogenesis 26, 64. Flavonoids, in particular, play a key role in the enzymatic regulation of carbohydrate metabolism, while tannins may promote peripheral glucose uptake and limit protein glycation 6, 17.
Furthermore, the observed effect of glibenclamide, which stimulates insulin secretion via pancreatic islet β-cells 49, suggests that certain compounds present in the extracts may act through partially similar mechanisms, albeit with lesser potency. These results are consistent with several previous studies reporting the antihyperglycemic properties of plants from the African pharmacopoeia, notably Terminalia catappa and Senna occidentalis 48. However, the observed activity could also result from synergistic effects between different bioactive compounds present in the extracts. In conclusion, the extracts studied show promising potential in regulating postprandial hyperglycemia. Nevertheless, further studies are needed to identify the active molecules, clarify their mechanisms of action, and evaluate their long-term efficacy and safety, particularly through pharmacological and clinical approaches.
CONCLUSION: This study highlights the phytochemical, antioxidant, and antihyperglycemic potential of the leaves of Terminalia catappa (Combretaceae) and Senna occidentalis (Fabaceae). Thin-layer chromatography analysis revealed the presence of major secondary metabolites (sterols, terpenes, flavonoids, coumarins, saponins, and tannins), confirming the bioactive richness of these two species. Quantitative analyses showed that the solvent and extraction method had a decisive influence on the levels of phenolic compounds. The aqueous macerates of Terminalia catappa (Ca2) are distinguished by high levels of total phenols and condensed tannins, while Senna occidentalis exhibits higher concentrations of flavonoids. Assessment of antioxidant activity using the DPPH assay reveals a clear superiority of Terminalia catappa extracts, particularly the hydroethanolic (Ca3) and ethanolic macerates (Ca4), with respective CR₅₀ values of 0.0316 and 0.0318 mg/mL, lower than those of Senna occidentalis (0.1553 to 0.9828 mg/mL). Pharmacologically, both species demonstrated significant antihyperglycemic activity (p < 0.0001) in the pretreatment and post-treatment models in rats subjected to hyperglycemia induced by oral glucose overload (4 g/kg). In the pretreatment model, extracts administered at 250 mg/kg, particularly Ca3 from Terminalia catappa (93.6±1.2 mg/dL at t₆₀), showed the greatest ability to limit the glycemic peak, with performance comparable to that of glibenclamide. In the post-treatment model, all extracts induced a significant decrease in blood glucose levels starting from the second hour, reaching values close to normoglycemia (85–95 mg/dL). Overall, the in-vivo results confirm that these two plant species exert a regulatory effect on postprandial hyperglycemia without inducing basal hypoglycemia, which constitutes a potential therapeutic advantage. These effects could be attributed to the high content of phenolic compounds and other specialized metabolites, which are likely to act via multiple mechanisms involving the modulation of insulin secretion, the improvement of peripheral glucose sensitivity, and the inhibition of intestinal absorption. These results provide scientific validation of the traditional use of Terminalia catappa and Senna occidentalis and suggest their potential as sources of bioactive compounds for the development of antidiabetic herbal medicines. However, further studies are needed to identify the active molecules, elucidate their precise mechanisms of action, and evaluate their safety and long-term efficacy in preclinical and clinical models.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Aké AL: Some medicinal properties of Cassia occidentalis L. in Lower Ivory coast. Boihalia 1983; 14(3–4): 617–620.
- Annegowda HV, Nee CW, Mordi MN, Ramanathan S & Mansor SM: Evaluation of phenolic content and antioxidant properties of hydrolyzed extracts of Terminalia catappa L. leaf. Food Chemistry 2010; 122(4): 1038–1043. https://doi.org/10.1016/j.foodchem.2010.03.078
- Annegowda HV, Ween NC, Mordi MN, Ramanathan S, Mansor SM: Evaluation of phenolic content and antioxidant properties of hydrolyzed extracts of Terminalia catappa L. leaf. Asian Journal of Plant Science 2010; 9(8): 479–485.
- Banu KS & Cathrine L: General techniques involved in phytochemical analysis. International Journal of Advanced Research in Chemical Science 2015; 2(4): 25–32.
- Békro YA, Mamyrbekova-Békro JA, Boua BB, Tra Bi FH. & Éhilé E: Ethnobotanical study and phytochemical screening of Caesalpinia benthamiana (Baill.) Herend & Zarucchi (Caesalpiniaceae). Sciences & Nature 2007; 4(2): 217–225.
- Benhamida R, Bourouh M and Maizi S: Antidiabetic effect of a from the Guelma region. Master’s thesis, University of 8 May 1945, Guelma 2023; 83.
- Benchaachoua A, Djeridane A, Yousfi M & Nadjemi B: Influence of extraction solvent on phenolic content and antioxidant capacity of tannins from medicinal plants. Industrial Crops and Products 2018; 115: 248–255. https://doi.org/10.1016/j.indcrop.2018.02.014
- Benchaachoua A, Bessam HM, Saidi I and Bel-abbes S: Effects of different extraction methods and solvents on the phenolic composition and antioxidant activity of Silybum marianum leaf extracts. International Journal of Medical Science and Clinical Invention 2018; 5(3): 3641–3647.
- Bernfeld P: α- and β-amylase. Methods in Enzymology, ed. Colowich and N.O. Kaplan, 1949–154, Academic Press 1955.
- Blois MS: Antioxidant determinations by the use of a stable free radical. Nature 1958; 181: 1199-1200.
- Borges F, Roleira F, Milhazes N, Santana L & Uriarte E: Simple coumarins and analogues in medicinal chemistry: occurrence, synthesis and biological activity. Current Medicinal Chemistry 2021; 28(7): 1345–1379.
- Brand-Williams W, Cuvelier ME & Berset C: Use of a free radical method to evaluate antioxidant activity. LWT - Food Science and Technology 1995.
- Broadhurst RB, Jones WT: Analysis of condensed tannins using acidified vanillin. Journal of the Science of Food and Agriculture 1978; 29(9): 788–794.
- Brou KG, Mamyrbekova-Békro JA, Dogbo DO, Gogbeu SJ and Békro YA: On the Qualitative Phytochemical Composition of Crude Hydromethanolic Extracts of the Leaves of 6 Varieties of Manihot esculenta Crantz from Ivory Coast. European Journal of Scientific Research 2010; 45(2): 200-211.
- Bruneton J: Pharmacognosy, Phytochemistry, Medicinal Plants. 5th ed., Lavoisier Tec. & Doc., Paris (France) 2016; 1487.
- Dai J & Mumper RJ: Plant phenolics: extraction, analysis, and their antioxidant and anticancer properties. Molecules 2010.
- De Boeck M, Jose G, Derrai K and Wayne S: Polyphenol and Glucose Homeostasis in Humans. J Acad Nutr Dietetics 2012; 112(6): 808-815.
- Dias MC, Pinto DCGA & Silva AMS: Plant flavonoids: chemical characteristics and biological activity. Molecules 2021; 26(17): 5377. https://doi.org/10.3390/molecules26175377
- Dif MM, Benchiha H, Mehdadi Z, Benali-Toumi F, Benyahia M and Bouterfas K: Quantitative study of polyphenols in the different organs of the species Papaver rhoeas L. Phytotherapy 2015; 13(5): 324-319.
- Do QD, Angkawijaya AE, Tran-Nguyen PL, Huynh LH, Soetaredjo FE, Ismadji S & Ju YH: Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. Journal of Food and Drug Analysis 2014; 22(3): 296–302. https://doi.org/10.1016/j.jfda.2013.11.001
- Dubois M, Gilles A, Hamilthon JJ, Rebers PA and Smith F: Colorimetric method for determination of sugars and related substances. Analytical Chemistry 1956; 28: 350–356.
- Flavie ASS, Mida KGR, Diane BBF and Mamyrbékova-Békro JA: Chemical Composition and Antioxidant Activity of Dialium guineense Fruit (Fabaceae), Ivory coast. International Journal of Biochemistry Research & Review 2024; 33(6): 604–6015.
- Fousseyni T, Diallo A, Sanogo R & Maïga A: Phytochemical profile and antioxidant activity of Terminalia catappa leaf extracts. Journal of Herbal Medicine 2021; 28: 100446. https://doi.org/10.1016/j.hermed.2021.100446
- Fousseyni D, Savio S, Issiaka T and Moussa K: Ethnobotanical and phytochemical studies of ornamental species used in Mali: the case of Terminalia catappa (L.) in the Bamako district. International Journal of Applied Research 2021; 7(6): 01-07.
- Gisèle EL, Didier DS, Vanessa BN, Jean-Pierre N and Pouka K: Evaluation of Hypoglycemic and Anti-Hyperglycemic Activity of the Aqueous Extract of the Roots of Jatropha curcas L. (Euphorbiaceae). Saudi Journal of Medical and Pharmaceutical Sciences 2017; 4929: 1397–413.
- Hanhineva K, Tunoner R, Bon PI, Pekkinen J, Kokhannina H and Poutanen K: Impact of dietary polyphenols on carbohydrate metabolism. J Mol Sci 2010; 11: 1365–1402.
- Hariri EB, Sallé G and Andary C: Involvement of flavonoids in the resistance of two poplar cultivars to mistletoe (Viscum album L.). Protoplasma 1991; 162(1): 20–26.
- Heilmer D, Vigndini P, Dini MG, Vincieri FF and Romani A: Antiradical activity and polyphenol composition of local edible Brassicaceae varieties. Food Chemistry 2006; 99(3): 464-469.
- Heinrich M, Mah J & Amirkia V: Alkaloids used as medicines: structural phytochemistry meets biodiversity an update and forward look. Molecules 2020; 25(5): 1254.
- International Diabetes Federation (IDF), Diabetes Atlas. Intern Diabetes Federation, 11th Edition 2025; 74–75.
- Iheagwam FN, Israel EN, Kayode KO, De Campos OC, Ogunlana OO & Chinedu SN: GC-MS Analysis and Inhibitory Evaluation of Terminalia catappa Leaf Extracts on Major Enzymes Linked to Diabetes. Evidence-based complementary and alternative medicine: eCAM 2019; 6316231. https://doi.org/10.1155/2019/6316231
- Iheagwam FN, Ogunlana OO, Ogunlana OE, Isewon I & Oyelade J: Potential antidiabetic activity of plant polyphenols: in-silico and in-vitro evaluation of selected compounds. Biomedicine & Pharmacotherapy 2019; 114: 108728. https://doi.org/10.1016/j.biopha.2019.108728
- Kadja AB, Mamyrbékova-Békro JA, Benie A, Boua BB, N’gaman KC and Békro YA: Erythrophleum africanum Afzel (Caesalpiniaceae), an African toothpick: Phytochemical screening, total flavonoid content, and antioxidant activity, Journal of Medicinal Plants Research 2011; 5(27): 6273–6277.
- Konkon NG, Ouattara D, Kpan WB and Kouakou TH: Medicinal plants used for the treatment of diabetes by traditional practitioners in the Abidjan district, Ivory coast. Journal of Medicinal Plants Studies 2017; 5(2): 39–48.
- Marques MR, Paz DD, Batista LPR, Barbosa CO, Mota AMA and Moreira-Araújo RSR: An in-vitro analysis of the total phenolic content, antioxidant power, physical, physicochemical, and chemical composition of Terminalia catappa Linn fruits. Food Sci Tech 2012; 32(1): 209-213.
- Marques TR, Corrêa AD, Alves APC, Simão AA & Pinheiro ACM: Chemical constituents and biological activities of extracts obtained by ultrasound-assisted extraction of phenolic compounds from plant matrices. Food Science and Technology 2012; 32(3): 512–518. https://doi.org/10.1590/S0101-20612012005000072
- N’Gaman KCC: Phytochemical study and effect of extracts of Gmelina arborea Roxb. (Verbenaceae) from Ivory coast on the osmotic stability of erythrocytes. (Doctoral dissertation). Nangui Abrogoua University, Abidjan, Ivory coast 2013; 152.
- N’Guessan K, Fofie NBY and Zirihi GN: Effect of aqueous extract of Terminalia catappa leaves on the glycemia of rabbits. Journal of Applied Pharmaceutical Science 2011; (08): 59-64.
- Nko’o NDL, Nko’o MH, Ngoule CC, Nyangono NM, Mbole MJ, Soppo LC, Benga MC, Foumane MS and Nnanga N: Evaluation of the hypoglycemic and anti-hyperglycemic activity of the aqueous extract of Phyllostachys aurea (Poaceae). Health Research in Africa 2024; 2(6): 103-108.
- National Research Council. (2011). Guide for the Care and Use of Laboratory Animals (8th ed.). The National Academies Press. https://doi.org/10.17226/12910
- OECD (2001). OECD Test Guideline 423, adopted on December 17, 2001.
- WHO World Diabetes Report 2016; 11–17
- Panche AN, Diwan AD & Chandra SR: Flavonoids: an overview. Journal of Nutritional Science 2016; 5: 47.
- Prior RL, Wu X & Schaich K: Standardized methods for the determination of antioxidant capacity. Journal of Agricultural and Food Chemistry 2005.
- Rice-Evans CA, Miller NJ & Paganga G: Antioxidant properties of phenolic compounds. Trends in Plant Science 1997.
- Sasidharan S, Chen Y, Saravanan D, Sundram KM & Yoga Latha L: Extraction, isolation, and characterization of bioactive compounds from plants. African Journal of Traditional, Complementary, and Alternative Medicines 2011; 8(1): 1–10.
- Singleton VL, Ortofer R and Lamuela-Raventos RM: Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Packer L (ed) Methods in Enzymology, Orlando: Academic Press 1999; 299: 152–178.
- Shaik AN, Ramesh BK, Swapna S, Thandaiah KT, Malaka VJK and Chippada AR: Antidiabetic and antihyperlipidemic activity of Piper longum root aqueous extract in STZ-induced diabetic rats. BMC Complementary Medicine and Therapies 2013; 13: 37. 9.
- Rydberg T, Jönsson A, Roder M & Melander A: Hypoglycemic activity of glyburide (glibenclamide) metabolites in humans. Diabetes Care 1994; 17(9): 1026–1030
- Shahidi F & Danielski G: Phenolic compounds and their role in oxidative stress modulation. Trends in Food Science & Technology 2024.
- Shahidi F & Danielski R: Review on the role of polyphenols in preventing and treating type 2 diabetes: evidence from in-vitro and in-vivo studies. Nutrients, 2024; 16(18): 3159. https://doi.org/10.3390/nu16183159
- Sharifi-Rad J, Behalf V, Mnayer D, Kregiel D, Ezzat S, M, Muhsinah AB & Martorell M: Plants of the genus Ziziphus: classification, biosynthesis of secondary metabolites and biological activities — a review. Molecules 2022; 27(1): 1–45. https://doi.org/10.3390/molecules27010001
- Shahidi F & Yeo J: Bioactivities of phenolics. Journal of Food Bioactives 2018.
- Sharifi-Rad J, Cirone Silva NC, Jantwal A, Bhatt ID, Bhatt DL, Sharopov F & Calina D: Biological activities of phenolic compounds present in diet and their role in oxidative stress and inflammation. Molecules 2022; 27(11): 3442. https://doi.org/10.3390/molecules27113442
- Sieniawska E: Activities of tannins—from in-vitro studies to clinical trials. Natural Product Communications 2015; 10(11): 1877–1884.
- Sieniawska E: Activities of tannins — from in-vitro studies to clinical trials. Natural Product Communications, 2015; 10(9): 1608–1610. https://doi.org/10.1177/1934578X1501000936
- Tanoh SK, N'Gaman-Kouassi C, Boa D, Mamyrbekova-Békro JA, Békro YA: Antioxidant activity of crude hydroethanolic and hydroacetonic extracts from the organs of four medicinal plants from Côte d'Ivoire. Nature & Technology 2019; (21): 28–34.
- Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L & Byrne DH: Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. Journal of Food Composition and Analysis 2006; 19(6–7): 669–675.
- Venugopala KN, Rashmi V & Odhav B: Review on natural coumarin lead compounds for their pharmacological activity. Bio Med Research International 2013; 1–14.
- Verma L, Khatri A, Kaushik B, Patil UK and Pawar RS: Antidiabetic activity of Cassia occidentalis (Linn) in normal and alloxan-induced diabetic rats. Indian Journal of Pharmacology 2010; 42(4): 224–228.
- Wende-Konté HCN, Yssouf K, Ousmane I, Téeda HG, Issa T: Inhibition of α-glucosidase activity by polyphenol compounds from C. occidentalis: Phytochemical screening and antidiabetic studies. Asian Journal of Research in Chemistry 2023; 16(4): 257-4. doi: 10.52711/0974-4150.2023.00043
- Wende-Konté F, Ouédraogo N, Compaoré M, Coulibaly AY, Kiendrebeogo M & Nacoulma OG: Antidiabetic activity of African medicinal plants: a review of preclinical and clinical studies. Journal of Ethnopharmacology 2023; 301: 115771. https://doi.org/10.1016/j.jep.2022.115771
- Yassa HD & Tohamy AF: Extract of Moringa oleifera leaves ameliorates streptozotocin-induced diabetes mellitus in adult rats. Acta Histochemica 2014; 116(5): 844–854. https://doi.org/10.1016/j.acthis.2014.02.002
- Zougrou NE: Evaluation of the toxicological and pharmacological effects of an aqueous extract of Cnestis ferruginea Vahl ex DC. (Connaraceae) on the reproductive system of male and female rats. (Doctoral dissertation), Félix Houphouet-Boigny University, Ivory Coast 2017; 267.
How to cite this article:
Boris KB, Amani K, Akhanovna MJ and Yves-Alain B: Phytochemical studies and evaluation of the antioxidant and antidiabetic activities of Terminalia catappa L. (Combretaceae) and Senna occidentalis (L.) link (Fabaceae) from ivory coast. Int J Pharmacognosy 2026; 13(5): 445-58. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(5).445-58.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
8
445-458
2314 KB
6
English
IJP
Koidio Bindé Boris *, Kadja Amani, Mamyrbekova-Békro Janat Akhanovna and Bekro Yves-Alain
Laboratoire de Chimie Bio-Organique et de Substances Naturelles (LCBOSN), UFRSFA, Université Nangui ABROGOUA, 02 BP 801 Abidjan 02, Ivory Coast.
dr-boris@hotmail.fr
18 April 2026
06 May 2026
12 May 2026
10.13040/IJPSR.0975-8232.IJP.13(5).445-58
15 May 2026