PHYTOCHEMICAL SCREENING AND FREE RADICAL SCAVENGING ACTIVITY OF HYDROETHANOLIC LEAF EXTRACT OF SENNA SIEBERIANA DC (CAESALPINIACEAE) AND ITS FRACTIONS
HTML Full TextPHYTOCHEMICAL SCREENING AND FREE RADICAL SCAVENGING ACTIVITY OF HYDROETHANOLIC LEAF EXTRACT OF SENNA SIEBERIANA DC (CAESALPINIACEAE) AND ITS FRACTIONS
Alioune Dior Fall * 1, Serigne Ibra Mbacké Dieng 1, Awa Ndiaye Sy 2, Abdou Sarr 1 and Mbaye Dieng 1
Laboratory of Pharmacognosy and Botany 1, Faculty of Medicine, Pharmacy and Odontology, Cheikh Anta Diop University, BP5005 Dakar Fann, Dakar, Senegal.
Laboratory of Pharmacology and Pharmacodynamy 2, Faculty of Medicine, Pharmacy and Odontology. Cheikh Anta Diop University, Dakar, Senegal.
ABSTRACT: The aim of this study was to identify the phytochemical groups of hydroethanolic leaf extract of Senna sieberiana DC and its fractions and to investigate their free radical scavenging activity. S. sieberiana leaves were extracted with a hydroethanolic solvent. From the hydroethanolic extract 3 fractions were obtained after a liquid/liquid fractionation (dichloromethane, ethyl acetate, and water). Phytochemical screening of the leaf extract and its fractions was done using standard reactions. Free radical scavenging activity was assessed using DPPH and ABTS assays. Tannins, flavonoids, anthracene derivatives, sterols, and triterpenoids were the main phytochemical constituents of the leaf extract and fractions. The hydro-ethanolic leaf extract of Senna sieberiana, its dichloromethane, ethyl acetate, and aqueous fractions and ascorbic acid had respective IC50 values of 191.6 ± 3.82; 495.73 ± 8.96; 165.8 ± 4.85; 50.40 ± 2.65; 19.53 ± 0.13 µg/ml in ABTS assay. In DPPH assay, the IC50 values were 44.8 ± 1.22; 218.93 ± 9.01; 32.13 ± 1.8; 26. 4 ± 0.11; 4.66 ± 0.07 µg/ml respectively for the leaf extract, dichloromethane, ethyl acetate, and aqueous fractions and ascorbic acid. The hydroethanolic leaf extract of S. sieberiana had shown free radical scavenging activity. The aqueous fraction was more active among plant tested samples.
| Keywords: |
Senna sieberiana, Leaf extract, Fractions, Phytochemical, Scavenging activity
INTRODUCTION: Senna sieberiana DC (synonym: Cassia sieberiana DC) is a plant widely spread in West Africa. Its leaves are used in senegalese traditional medicine as detoxing, antipyretic, diuretic and against anemia 1. Leaf extract is also used to treat various diseases such as stomach ache, ulcer, and diarrhea.
Leaf extract of Senna sieberiana has been reported for their antimicrobial 2, antitrypanosomal 3, antiviral 4 and anthelmintic 5 properties. Oxidative stress due to overproduction of free radicals has been involved in neurodegenerative disease (Alzheimer's and Parkinson's diseases), diabetes, cardiovascular disease, atherosclerosis, rheumatoid arthritis 6, 7. What makes us assess the phytochemical composition and the free radical scavenging activity of hydroethanolic leaf extract of S. sieberiana and its fractions.
MATERIALS AND METHODS:
Plant Collection: Leaves of Senna sieberiana DC were collected at the Botanical Garden of the Faculty of Medicine, Pharmacy and Odontology (Cheikh Anta DIOP University, Dakar / Senegal). The plant was identified and authenticated by Dr. W. Diatta (Herbarium of the Botanical Garden of the Faculty of Medicine, Pharmacy and Odontology of Dakar) where a voucher specimen was kept. Plant leaves were air-dried at room temperature. Dried leaves were ground to a fine greenish powder.
Extraction and Fractionation: An amount of 125 g of powdered leaves of S. sieberiana was decocted twice for 30 min using one liter of ethanol/water (80:20, v/v) and filtered through Whatman no. 1 filter paper. The solvent was removed under reduced pressure using a rotary evaporator in order to get the hydroethanolic leaf extract. For liquid/liquid fractionation, 2.5 g of dried leaf extract was dissolved in a mixture (distilled water/ dichloromethane; 1:1). After decantation in a separatory funnel, the aqueous solution obtained was extracted again twice with dichloromethane. The dichloromethane solutions were combined and evaporated to give the dichloromethane fraction. The aqueous solution was again subjected to liquid-liquid extraction with ethyl acetate under the same conditions as above. The ethyl acetate and aqueous solutions obtained were evaporated separately and lead to the corresponding fractions.
Phytochemical Screening: To test for the presence of phytoconstituents groups, standard phyto-chemical analyses were carried out on the hydroethanolic leaf extract and its fractions. Chemical tests were carried out on these samples using standard procedures for the detection of condensed and hydrolysable tannins (Stiasny test followed by ferric chloride test), flavonoids (Shibata’s test), anthracene derivatives (Borntraeger test), cardiac glycosides (Baljet, Kedde and Raymond-Marthoud reagents tests), steroids and triterpenoids (Liebermann-Buchard test), carotenoids (antimony chloride/chloroform test), alkaloïds (Bouchardat, Valser-Mayer and Dragendorff’s reagents tests), in order to identify the presence of phytochemical constituents 8.
Free Radical Scavenging Activity:
ABTS Assay: Reduction of free radical ABTS (2, 2-azinobis-3ethylbenzothiazoline-6-sulfonic acid) was investigated using the described method 9. Two stock solutions of 7.4 mm ABTS and 2.6 mm potassium persulfate were prepared and mixed in equal volumes before allowing them to react for 12 h at room temperature in darkness. This mixture was diluted by adding ethanol, to obtain an absorbance of 0.7 at 734 nm. Samples (2 ml) were mixed with 2 ml of ABTS solution, and the mixture was left at room temperature for 2 h in darkness. The absorbance of each sample was measured at 734 nm after 30 min using a spectrophotometer. Experiments were done in triplicate and the ABTS free radical scavenging effect was expressed as IC50 (concentration of sample required to scavenge 50% of free radicals).
DPPH Assay: The determination of the DPPH free radical scavenging activity of samples was done using the described method 10. An ethanol solution of DPPH was prepared by dissolving 4 mg in 100 ml of ethanol. An aliquot of each sample (0.8 ml) at appropriate concentration was added to 3 ml of ethanol solution of DPPH. The hydroethanolic leaf extract of Senna sieberiana, its fractions (dichloromethane, ethyl acetate, and water) and ascorbic acid were tested at different concentrations. The absorbance of each sample was measured at 517 nm after 30 min using a spectrophotometer. Each experiment was done in triplicate and the absorbance of the initial ethanol DPPH solution did not change after 30 min. The DPPH free radical scavenging effect was expressed as IC50.
Statistical Analyses: Data were expressed as mean ± SEM. Analyses of variance (ANOVA) were done for the comparison of results using Fischer's test. Statistical significance was set at p˂0.05.
RESULTS:
Extraction and Fractionation: From 125 gm of dried powdered leaves, 21.87 gm of dried leaf extract were obtained corresponding to a yield of 17.5%. The dichloromethane, ethyl acetate and water fractions represented respectively 12.74; 31.79 and 54.46% of the dried hydroethanolic leaf extract.
Phytochemical Screening: Phytochemical screening revealed that anthracene derivatives, hydrolyzable and condensed tannins, flavonoids, sterols, and triterpenoids were identified in the hydroethanolic leaf extract of the plant. Negative reactions were obtained for the presence of carotenoids, alkaloids, and cardiac glycosides. The dichlormethane fraction contained anthracenic derivatives, sterols, triterpenoids, and condensed tannins. Anthracenic derivatives, condensed and hydrolysable tannins, flavonoids were found in the ethyl acetate fraction while in the aqueous one flavonoids, hydrolyzable and condensed tannins were identified Table 1.
TABLE 1: PHYTOCHEMICAL GROUPS IDENTIFIED IN LEAF EXTRACT OF S. SIEBERIANA AND ITS FRACTIONS
| Phytochemical groups | HE | DF | EAF | AF |
| Alkaloids | - | - | - | - |
| Anthracenic derivatives | + | + | + | - |
| Cardiac glycosides | - | - | - | - |
| Carotenoids | - | - | - | - |
| Flavonoids | + | - | + | + |
| Hydrolyzable tannins | + | - | + | + |
| Condensed tannins | + | + | + | + |
| Sterols and triterpenoids | + | + | - | - |
HE: hydro-ethanolic extract, DF: dichloromethane fraction EAF: ethyl acetate fraction AF: aqueous fraction
Free Radical Scavenging Activity:
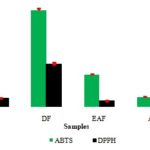
ABTS assay: The hydroethanolic leaf extract of Senna sieberiana had an IC50 value (191.6 ± 3.82µg/ml) higher than those of ethyl acetate and aqueous fractions (respective IC50: 165.8 ± 4.85 µg/ml and 50.40 ± 2.65 µg/ml/ml) (p < 0.05). The dichloromethane fraction had exhibited the highest IC50 value (495.73 ± 8.96 µg/ml). Ascorbic acid had shown the lowest IC50 value (19.53 ± 0.13 µg/ml) (p < 0.05) Fig. 1.
FIG. 1: IC50 VALUES OF DIFFERENT SAMPLES IN DPPH AND ABTS ASSAYS. HE: Hydroethanolic extract DF: Dichloromethane fraction EAF: Ethyl acetate fraction AF: aqueous fraction AA: ascorbic acid
DPPH Assay: The aqueous fraction had shown the lowest IC50 value (26.4 ± 0.11 µg/ml) among plant samples, followed by the ethyl acetate fraction (IC50: 32.13 ± 1.8 µg/ml) and the hydroethanolic leaf extract (IC50: 44.8 ± 1.22 µg/ml) (p<0.05). Dichloromethane fraction (IC50: 218.93 ± 9.01 µg/ml) had revealed the highest IC50 value among plant samples. For ascorbic acid, an IC50 value of 4.66 ± 0.07 µg/ml was obtained (p < 0.05) Fig. 1.
DISCUSSION: Leaves of S. sieberiana contained mainly phenolic compounds such as flavonoids, tannins and anthracenic derivatives 11. The polyphenol extraction is a crucial step for the valorization of these active principles recognized for their scavenging effect 12. The appropriate choice of solvents preserving the biological properties of these phenolic constituents is very important 13. As part of our study, we first carried out a decoction using a mixture of two polar solvents such as ethanol and water. The solvents had ability to extract polar compounds represented in S. sieberiana leaves.
However, ethanol is also able to extract nonpolar constituents such as sterols, triterpenoids and aglycones represented in the leaves of S. sieberiana. The yield of this extraction was 17.5%. From leaves of Senna alata, it has been recorded after hydroethanolic maceration an extraction yield of 12.5% 14. The difference between these values may be due to the process.
Subsequently, the liquid-liquid fractionation allowed us to successively obtain three fractions: a dichloromethane fraction containing non-polar compounds; an ethyl acetate fraction with compounds of intermediate polarity, and an aqueous fraction containing the most polar compounds. The lowest yield relative to the hydroethanolic extract was that of the dichloromethane fraction followed by those of the ethyl acetate and aqueous fractions. These results suggest that the bioactive constituents most represented in the leaves of S. sieberiana were mainly polar compounds.
The aqueous and ethyl acetate fractions had shown the better capacity to scavenge free radicals, both in ABTS and DPPH assays, than the hydro-ethanolic leaf extract and its dichloromethane fraction. Among plant samples, the aqueous fraction was seen to be the more active while the dichloro-methane fraction exhibited the lowest ability to scavenge the free radicals. What makes us suggest that polar phytoconstituents, identified in ethyl acetate and aqueous fractions had a better ability to scavenge the free radicals. The phytochemical screening had revealed the presence of flavonoids and hydrolysable tannins in ethyl acetate and aqueous fractions and not in the non-polar fraction (Dichloromethane fraction). These phenolic compounds such as flavonoids and tannins contained in these fractions could be responsible for the free radical scavenging activity. Indeed polyphenolic compounds are known for their scavenging ability 15, 16. It has been established that the antioxidant efficiency of a proton-bound A radical (AH) increases if the binding force A-H is low and the resulting radical A is as stable as possible.
This is the case for phenolic compounds such as flavonoids which are among the best electron or proton donors 17. Besides in the dichloromethane fraction had been detected condensed tannins which were also found in the ethyl acetate and aqueous fractions. The low ability of the dichloromethane solvent to extract polar phytoconstituents such as polyphenols would explain its low free radical scavenging activity in DPPH and ABTS assays.
CONCLUSION: The hydroethanolic leaf extract of Senna sieberiana which contained tannins, flavonoids, anthracenic derivatives, sterols, and triterpenoids had exhibited free radical scavenging activity. The ethyl acetate and water fractions were seen to be more active than the leaf extract and the dichloromethane fraction. Aqueous fraction had shown better ability to scavenge free radicals among plant tested samples.
AUTHOR CONTRIBUTIONS: This work was carried out in collaboration between all authors. Authors ADF and SIMD planned all experiments. Authors AS and MD supported the extraction, fractionation and phytochemical screening. Authors ADF, ANS, and SIMD carried out the free radical scavenging study and provided the statistical analyses of data. Author ADF wrote the first draft of the manuscript. All authors read and approved the final manuscript.
ACKNOWLEDGEMENT: Nil
FUNDING: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CONFLICT OF INTEREST: Authors have declared that no competing interests exist.
REFERENCES:
- Pousset JL: Plantes médicinales Africaines, Utilisation pratiques. Ed. ACCT Ellipses, Paris 1989: 156.
- Asase A, Kokubun T, Grayer RJ, Kite G, Simmonds MSJ, Oteng‐Yeboah AA and Odamtten GT: Chemical constituents and antimicrobial activity of medicinal plants from Ghana: Cassia sieberiana, Haematostaphis barteri, Mitragyna inermis and Pseudocedrela kotschyi. Phytotherapy Research 2008; 22(8): 1013-16.
- Hoet S, Opperdoes S, Brun R, Adjakidjé V and Quentin-Leclerq J: In-vitro anti-plasmodial activity of ethno-pharmacolocally selected Beninese plants. Journal of Ethnopharmacology 2004; 91(1): 37-42.
- Silva O, Barbosa S, Diniz A, Valdeira ML and Gomes E: Plant extracts antiviral activity against herpes simplex virus 1 and African swine fever virus. International Journal of Pharmacognosy 2008, 35(1): 12-16.
- Waterman C, Smith RA, Pontiggia L and Der Marderosian A: Anthelmintic screening of Sub-Saharan African plants used in traditional medicine. J of Ethnopharmacology 2010, 127(3): 755-59.
- Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M and Telser J: Free radicals and antioxidants in normal physiological functions and human disease. The Int Journal of Biochemistry and Cell Biology 2007; 39: 44-84.
- Chen X, Guo C and Kong J: Oxidative stress in neurodegenerative diseases. Neural Regeneration Research 2012; 7(5): 376-85.
- Joslyn MA: Methods in food analysis. physical, chemical and instrumental methods of analysis. Academic Press, London and New York 1970.
- Dudonne S, Vitrac X, Coutière P, Woillez M and Merillon JM: Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J of Agriculture and Food Chemistry 2009; 57(5): 1768-74.
- Molyneux P: The use of the stable free radical diphenylpicryl-hydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J Sci Technol 2004; 26: 211-19.
- Awomukwu DA, Nyananyo BL, Ikpeama AI and Adieze CU: Comparative chemical constituents of some Cassia species and their pharmacognistic importance in South Eastern Nigeria. Science J of Chemistry 2015, 3(3): 40-49.
- Bonnaillie C, Salacs M, Vassiliova E and Saykova I: Etude de l’extraction de composés phénoliques à partir de pellicules d’arachide (Arachis hypogaea) Revue de Génie Industriel 2012; 7: 35-45.
- Babbar N, Oberoi HS, Sandhu SK and Bhargav VK: Influence of different solvents in extraction of phenolic compounds from vegetable residues and their evaluation as natural sources of antioxidants. J Food Sci Technol 2014; 51(10): 2568-75.
- Pieme CA, Penlap VN, Nkegoum B, Taziebou PCL, Tekwu EM, Etoa FX and Ngongang J: Evaluation of acute and sub acute toxicities of aqueous ethanolic extract of leaves of Senna alata (L.) roxb (Caesalpiniaceae). African Journal of Biotechnology 2006; 5(3): 283-89.
- 15Álvarez R, Araya H, Navarro-Lisboa R and Lopez de Dicastillo C: Evaluation of polyphenol content and antioxidant capacity of fruits and vegetables using a modified enzymatic extraction. 2016; 54(4): 462-67.
- Tresserra-Rimbau A, Lamuela-Raventos RM and Moreno JJ: Polyphenols, food and pharma current knowledge and directions for future research. Biochem Pharmacol 2018; 156: 186-95.
- Leopoldini M, Marino T, Russo N and Toscano M: Antioxidant properties of phenolic compounds: h-atom versus electron transfer mechanism. J Phys Chem A 2004; 108 (22): 4916-22.
How to cite this article:
Fall AD, Mbacké Dieng SI, Ndiaye AS, Sarr A and Dieng M: Phytochemical screening and free radical scavenging activity of hydroethanolic leaf extract of Senna sieberiana DC (Caesalpiniaceae) and its fractions. Int J Pharmacognosy 2019; 6(8): 268-72. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(8).268-72.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
268-272
561
1139
English
IJP
A. D. Fall *, S. I. M. Dieng, A. N. Sy, A. Sarr and M. Dieng
Laboratory of Pharmacognosy and Botany, Faculty of Medicine, Pharmacy and Odontology, Cheikh Anta Diop University, Dakar Fann, Dakar, Senegal.
elieufall@yahoo.fr
03 August 2019
23 August 2019
27 August 2019
10.13040/IJPSR.0975-8232.IJP.6(8).268-72
31 August 2019