PHYTOCHEMICAL SCREENING AND ANTIOXIDANT ACTIVITY OF JUSTICIA TRANQUEBARIENSIS AND BAUHINIA RACEMOSA
HTML Full TextPHYTOCHEMICAL SCREENING AND ANTIOXIDANT ACTIVITY OF JUSTICIA TRANQUEBARIENSIS AND BAUHINIA RACEMOSA
R. Krishnamoorthi
PG and Research Department of Zoology, Presidency College (Autonomous), Chennai - 600005, Tamil Nadu, India.
ABSTRACT: Introduction: The phytochemicals in medicinal plants are defined as bioactive non-nutrient plant compounds in fruits, vegetables, and grains that have been linked to reducing the risk of major chronic diseases. The medicinal plant is promising to have such have effective medicinal property due to the presence of phytochemical compounds like alkaloids, tannins, flavonoids, and phenolic compounds. The present study aimed to find the phytochemicals present in hexane and ethyl acetate extracts of Justicia tranqubarieansis and Bauhinia racemosa and also find out the antioxidant property of these plant extracts. Methods: Chemical methods identified phytochemicals present in the hexane and ethyl acetate extract of Justicia tranqubarieansis and Bauhinia racemosa. The antioxidant property was done by DPPH free radical scavenging method. Results: In Justicia tranqubarieansis, hexane extract showed positive for flavonoids, alkaloids, and coumarins. Ethyl acetate extract showed positive for flavonoids, alkaloids, cardiac glycosides, terpenoids, coumarins, and steroids. The hexane extract of Bauhinia racemosa showed positive for flavonoids, alkaloids, quinines, cardiac glycosides, terpenoids, and coumarins. Ethyl acetate extract showed positive for flavonoids, alkaloids, cardiac glycosides, terpenoids, coumarins, and steroids. Ethyl acetate showed better extraction of phyto-chemicals than hexane. Hexane and ethyl acetate extracts of Justicia tranqubarieansis exhibited 66% and 90% respectively of free radical scavenging activity at a concentration of 150 µg. Extracts of Bauhinia racemosa showed less antioxidant activity. Conclusion: This study suggests that the Justicia tranqubarieansis extracts have good antioxidant property, which might help prevent of slowing the progress of diseases involved as a result of oxidative stress.
| Keywords: |
Alkaloids, Flavonoids, Justicia tranqubarieansis, Antioxidant activity, Ethyl acetate
INTRODUCTION: Medicinal plants, as a source of remedies, are widely used as alternative therapeutic tools for the prevention or treatment of many diseases.
The recent studies have investigated that the antioxidant effect of plant products is mainly attributed to phenolic compounds such as flavonoids, phenolic acids, tannins, etc. 1, 2
Accumulation of free radicals can cause pathological conditions such as ischemia, asthma, arthritis, inflammation, neuro-degeneration, Parkinson’s diseases, mongolism, the aging process and perhaps dementia. Natural antioxidants have become the target of a great number of research studies in finding the sources of potentially safe, effective and cheap antioxidants 3. Herbal drugs containing free radical scavengers are known for their therapeutic activity 4. In the present study, the phytochemical screening and antioxidant activities of two medicinally important herbs Justicia tranquebariensis and Bauhinia racemosa were taken to identify its phytoconstituents and radical scavenging capacity.
Justicia tranquebariensis (Acanthaceae) is a small shrub, which is widely distributed in the southern parts of India. Some species of the genus Justicia have been used in the traditional system of medicine for the treatment of fever, 5 pain 6, inflammation 7, diabetes 8, diarrhea 9 and liver diseases 10. The juice of small of small and somewhat fleshy leaves of this species of Justicia is considered by the natives of India as cooling and aperients, and is prescribed for the children in smallpox, in the doses of a table-spoonful or two, twice daily; bruised leaves are also applied to blows and other external injuries 11. As far as our literature survey could ascertain, no information was available on the protective and curative effect of Justicia tranquebariensis leaf extract. Therefore this study aimed to investigate the protective and curative effect of Justicia tranquebariensis from India.
Bauhinia racemosa Lam. (Caesalpiniaceae) is a small bushy tree with drooping branches. The leaves are green and broader than long. The flowers are white or pale yellow, terminal or leaf-opposed racemes 12, 13, 14. A new tetracycliclupeol, betulin, β-sitosterol, and tetracyclic 2, 2-dimethylchroman have been isolated from the roots 15, 16. The seed contains flavonoids, crude protein, and lipid 17, 18. A methanolic extract of the stem and bark are used as an anti-inflammatory, analgesic and antipyretic 19. A methanolic extract of the flower buds is used in the treatment of peptic ulcer 20. The whole plant is used as veterinary medicine in central India.
MATERIALS AND METHODS:
Chemicals and Reagents: DPPH (2, 2-diphenyl-1-picrylhydrazyl) and all other chemicals and reagents used were of the highest analytical grade commercially available 21.
Collection and Identification of Plant Materials: Fresh leaves of Justicia tranquebariensis and Bauhinia racemosa were collected from Chinnapaliyapattu, Tiruvannamalai, Tamil Nadu, India and Presidency College, Chennai, Tamil Nadu, India. The Taxonomist identified the plants at the Institute (NIPRD).
Phytochemical Tests: Preliminary phytochemical tests were carried out for aqueous extract of A. deliciosa to identify different phytoconstituents 21.
Test for Carbohydrates: To 2 ml of plant extract, 1 ml of Molisch’s reagent and few drops of concentrated sulphuric acid were added. Presence of purple or reddish color indicates the presence of carbohydrates 22.
Test for Tannins: To 1 ml of plant extract, 2 ml of 5% ferric chloride was added. Formation of dark blue or greenish black indicates the presence of tannins 23.
Test for Saponins: To 2 ml of plant extract, 2 ml of distilled water was added and shaken in a graduated cylinder for 15 min lengthwise. Formation of a 1 cm layer of foam indicates the presence of saponins 24.
Test for Flavonoids: To 2 ml of plant extract, 1 ml of 2N sodium hydroxide was added. Presence of yellow color indicates the presence of flavonoids 25.
Test for Alkaloids: To 2 ml of plant extract, 2 ml of concentrated hydrochloric acid was added. Then few drops of Mayer’s reagent were added. Presence of green color or white precipitate indicates the presence of alkaloids 26.
Test for Quinines: To 1 ml of extract, 1 ml of concentrated sulphuric acid was added. Formation of red color indicates the presence of quinones 27.
Test for Glycosides: To 2 ml of plant extract, 3 ml of chloroform and 10% ammonia solution was added. Formation of pink color indicates the presence of glycosides 28.
Test for Cardiac Glycosides: To 0.5 ml of extract, 2 ml of glacial acetic acid and few drops of 5% ferric chloride were added. This was under layered with 1 ml of concentrated sulphuric acid. Formation of the brown ring at the interface indicates the presence of cardiac glycosides 25.
Test for Terpenoids: To 0.5 ml of extract, 2 ml of chloroform was added and concentrated sulphuric acid was added carefully. Formation of red-brown color at the interface indicates the presence of terpenoids 25.
Test for Phenols: To 1 ml of the extract, 2 ml of distilled water followed by a few drops of 10% ferric chloride was added. Formation of blue or green color indicates the presence of phenols 27.
Test for Coumarins: To 1 ml of extract, 1 ml of 10% NaOH was added. Formation of yellow color indicates the presence of coumarins 27.
Phlobatannins: To 1 ml of plant extract a few drops of 2% HCl was added the appearance of red color precipitate indicates the presence of phlobatannins 25.
Steroids and Phytosteroids: To 1 ml of plant extract equal volume of chloroform is added and subjected with few drops of the concentrated sulphuric acid appearance of brown ring indicates the presence of steroids and appearance of bluish-brown ring indicates the presence of phytosteroids29.
Anthraquinones: To 1 ml of plant extract, a few drops of 10% ammonia solution were added, appearance pink color precipitate indicates the presence of anthraquinones 25.
Antioxidant Activity: The ability of the extracts to annihilate the DPPH radical (1, 1-diphenyl-2-picrylhydrazyl) was investigated by the method described by (Blois 1958). A stock solution of leaf extracts was prepared for the concentration of 1mg/ml. 100µg of each extract was added, at an equal volume, to a methanolic solution of DPPH (0.1mM). The reaction mixture is incubated for 30min at room temperature; the absorbance was recorded at 517 nm. The experiment was repeated for three times. BHT was used as standard controls. The annihilation activity of free radicals was calculated in % inhibition according to the following formula:
% of Inhibition = (A of control – A of Test)/A of control × 100
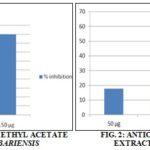
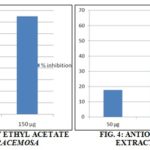
RESULTS: In Justicia tranqubarieansis, hexane extract showed positive for flavonoids, alkaloids, and coumarins. Ethyl acetate extract showed positive for flavonoids, alkaloids, cardiac glycosides, terpenoids, coumarins, and steroids. The hexane extract of Bauhinia racemosa showed positive for flavonoids, alkaloids, quinines, cardiac glycosides, terpenoids, and coumarins. Ethyl acetate extract showed positive for flavonoids, alkaloids, cardiac glycosides, terpenoids, coumarins, and steroids. Ethyl acetate showed better extraction of phytochemicals than Hexane. Hexane and ethyl acetate extract of Justicia tranqubarieansis exhibited 66% and 90% respectively of free radical scavenging activity at a concentration of 150 µg. Extract of Bauhinia racemosa showed less antioxidant activity Fig. 1, 2, 3, & 4 and Table 1 & 2.
TABLE 1: PHYTOCHEMICAL SCREENING OF JUSTICIA TRANQUEBARIENSIS EXTRACTS
| Justicia tranquebariensis | ||
| Phytochemical
test |
Inference | |
| Hexane | Ethyl acetate | |
| Carbohydrates | - | - |
| Tannins test | - | - |
| Saponin test | - | - |
| Flavonoid test | + | + |
| Alkaloid test | + | + |
| Quinones | - | - |
| Glycosides test | - | - |
| Cardiac glycosides test | - | + |
| Terpenoids test | - | + |
| Triterpenoids | - | - |
| Phenols | - | - |
| Coumarins | + | + |
| Proteins | - | - |
| Steroids and Phytosteroids | - | + |
| Phlobatannins | - | - |
| Anthraquinones | - | - |
TABLE 2: PHYTOCHEMICAL SCREENING OF BAUHINIA RACEMOSA EXTRACTS
| Bauhinia racemosa | |||
| Phytochemical
test |
Inference | ||
| Hexane | Ethyl acetate | ||
| Carbohydrates | - | - | |
| Tannins test | - | - | |
| Saponin test | - | - | |
| Flavonoid test | + | + | |
| Alkaloid test | + | + | |
| Quinones | + | - | |
| Glycosides test | - | - | |
| Cardiac glycosides test | + | + | |
| Terpenoids test | + | + | |
| Triterpenoids | - | - | |
| Phenols | - | - | |
| Coumarins | + | + | |
| Proteins | - | - | |
| Steroids and Phytosteroids | - | + | |
| Phlobatannins | - | - | |
| Anthraquinones | - | - | |
Concentration µg/ml, X-axis – concentration of the sample, Y-axis – the percentage of inhibition
Concentration µg/ml, X-axis – concentration of the sample, Y-axis – the percentage of inhibition
DISCUSSION: The importance of medicinal values of plants has been accepted in the past few decades. Plants produce a very diverse group of secondary metabolites with antioxidant potential. Antioxidant blocks the action of the free radicals which have been implicated in the pathogenesis of many diseases and the aging process. The free radicals are governing the important biological processes which are necessary for the body. The free radicals are necessary but at the same time harmful for the body. Hence it has some mechanisms are minimized the free radical-induced damage. The enzymes involved in repairs, the damage caused by free radicals are superoxide dismutase, catalase, glutathione, peroxidase, and glutathione reductase 30, 31, 32. In our study, hexane and methanol extract of Bauhinia racemosa showed positive for the presence of flavonoid, alkaloid, quinones, cardiac glycosides, terpenoids, coumarins steroids. Manohas et al., 33 reported that methanol and aqueous extracts showed positive for carbohydrates, glycosides, alkaloids, phytosterol, saponins, flavonoids, gums and mucilage, tannins, fixed oil, and fats.
Akilandeswari et al., 34 reported that the leaf of Justicia tranquebariensis showed, positive for phytosteroids, flavonoids, glycosides, and absence of triterpenoids, alkaloids, saponins, tannins. From our study, hexane and ethyl acetate extracts of Justicia tranquebariensis exhibited positive for the presence of flavonoids, alkaloids, cardiac glycosides, terpenoids, coumarins, and steroids. Balamurugan et al., 35 reported that ethanol extract of the aerial portions of Justicia tranquebariensis showed free radical scavenging activity. In this study, hexane and ethyl acetate extracts of Justicia tranquebariensis exhibited 66% to 90% free radical scavenging activity at a concentration of 150 µg.
CONCLUSION: Phytochemical screening of hexane and ethyl acetate extracts of Justicia tranquebariensis and Bauhinia racemosa had revealed the presence of flavonoids, alkaloids, cardiac glycosides, terpenoids, coumarins, steroids. From this study, it has been concluded that Justicia tranquebariensis extracts have shown significant antioxidant activity than Bauhinia racemosa. This plant might help prevent or slowing the process of diseases involved as a result of oxidative stress.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Nagavani V and Rao TR: Evaluation of antioxidant potential and qualitative analysis of major polyphenols by RP-HPLC in Nymphaea nouchali Burm flowers. International Journal of Pharmacy and Pharmaceutical Sciences. 2010; 2(S-4): 98-104.
- Cartea ME, Francisco M, Lema M, Soengas P and Velasco P: Resistance of cabbage (Brassica oleracea capitata Group) crops to Mamestra brassicae. Journal of Economic Entomology 2010; 103(5): 1866- 1874.
- Mundhe KS, Kale AA, Gaikwad SA, Deshpande NR and Kashalkar RV: Evaluation of phenol, flavonoid contents and antioxidant activity of Polyalthia longifolia. J Chem Pharm Res 2011; 3(1): 764-769.
- Hakiman M and Maziah M: Non-enzymatic and enzymatic antioxidant activities in aqueous extract of different Ficus deltoidea Journal of Medicinal Plants Research 2009; 3(3): 120-131.
- Chen CC, Hsin WC, Ko FN, Huang YL, Ou JC and Teng CM: J Nat Prod 1996; 59: 1149.
- Bhattarai NK: Fitoterapia 1993; 64: 483.
- Panthong A and Kanjanapothi D: J Ethnopharmacol 1986; 18: 213.
- Mahabir D: Am J Pub Health. 1997; 1: 174.
- Heinrich M, Kuhnt M, Wright CW, Rimplex H, Phillipson JD, Schandelmaier A and Warhurst DC: J Ethnopharmacol 1992; 36: 81.
- Yangfg LL, Yen KY, Kiso Y and Kikino H: J Ethnopharmacol 1987; 19: 103.
- Nadkarni KM: Indian meteria medica. Bombay Popular Prakashan; Vol. 1, 715.
- Nadkarni KM: Indian materia medica. Bombay: Bombay Popular Prakashan; Edition 3rd, Vol. 1, 1982: 183.
- Kirtikar KR and Basu BD: Indian Medicinal Plants. Dehradun: Bishen Singh Mahendra Pal Singh; Edition 2nd, Vol. 3, 1991: 894-5.
- Anonymous: The Wealth of India. New Delhi: Council of Scientific Research and Industrial Research 1991: 54-5.
- Jain R, Alam S and Saxena UA: A new tetracyclic phenol and other constituents from the roots of Bauhinia racemosa. Indian J Chem 2002; 41: 1321-2.
- Prabhakar P, Gandhidasan R, Raman PV, Krishnasamy NR and Nanduri S: A tetracyclic 2,2-dimethyl chroman from the roots of Bauhinia racemosa. Phytochemistry 1994; 36: 817-8.
- Mohan VR and Janardhanan K: Chemical and nutritional evaluation of two germplasms of the tribal pulse, Bauhinia racemosa. Plant Foods Hum Nutr 1994; 96: 367-74.
- El-Hossary GA, Selim MA, Sayed AE and Khaleel AE: Study of the flavonoids content of Bassia muricata and Bauhinia racemosa. Cairo University: Bulletin of the Faculty of Pharmacy; 2000: 56.
- Gupta M, Mazumdar UK, Kumar RS, Gomathi P, Rajishwar Y and Kakoti BB: Anti-inflammatory, analgesic and antipyretic effects of methanol extract from Bauhinia racemosa stem bark in animal models. J Ethnopharmacol, 2005; 98: 267-73.
- Akhtar AH and Ahmad KV: Anti-ulcerogenic evaluation of the methanolic extracts of some indigenous medicinal plants of Pakistan in aspirin-ulcerated rats. Journal Ethnopharmacol 1995; 46: 1-6.
- Aruoma OI: Methodological considerations for characterizing potential antioxidant actions of bioactive components in plant foods. Mutation Res 2003; 523 (524): 9-20.
- Dasgupta N and De B: Antioxidant activity of Piper betle Leaf extract in vitro. Food Chem. 2004; 88: 219-224.
- Coruh N, Celep AGS and Ozgokce F: Antioxidant properties of Prangos ferulacea (L) Lindl, Chaerophyllum macropodum And Heracleum persicum Desf. from Apiaceae family used as food in Eastern Anatolia and their inhibitory effects on glutathione-S-transferase. Food Chem 2007; 100: 1237-1242.
- Blois MS: Antioxidant determinations by the use of a stable free radical. Nature 1958; 29: 1199-1200.
- Sofowora A: Medicinal Plants and Traditional Medicinal in Africa. Sunshine House, Ibadan, Nigeria: Spectrum Books Ltd; Screening Plants for Bioactive Agents; Edition 2nd, 1993: 134–156.
- Harborne JB: Phytochemical Methods: A guide to modern techniques of plant analysis. Chapman and Hall, New York, Edition 3rd, 1973: 279.
- Smolenski SJ, Silinis H and Farnswoth NR: Alkaloids screening. V. Lloydia, 1974; 37: 506-536.
- Kapoor LD, Singh A, Kapoor SL and Shrivastava SN: Survey of Indian medicinal plants for saponins, alkaloids and flavonoids. Lloydia 1969; 32: 297-302.
- Jana S and Shekhawat GS: Phytochemical analysis and antibacterial screening of in-vivo and in-vitro extracts of Indian medicinal herbs: Anethum graveolens. Research Journal of medicinal plants 2010; 4(4): 206-212.
- Ayoola GA, Coker HAB, Adesegun SA, Adepoju-Bello AA, Obaweya K, Ezennia EC and Atangbayila TO: Phytochemical screening and antioxidant activities of some selected medicinal plants used for malaria therapy in Southwestern Nigeria. Tropical Journal of Pharmaceutical Research 2008; 7(3): 1019-1024.
- Sureshkumar CA, Varadharajan R, Muthumani P, Meera R, Devi P, and Kameswari B: Pharmacognostic and preliminary phytochemical investigations on the stem of Saccharum spontaneum. J Pharm Sci & Res 2009; 1(3): 129-136.
- Manasboxi, Rajesh Y, Rajakumar V, Praveen B and Mangamma K: Extraction, phytochemical screening and in-vitro evaluation of anti-oxidant properties of Commicarpus chinesis (aqueous leaf extract), International
- Journal of Pharma and Bio Sciences 2010; 1(4).
- Kolawole OM, Oguntoye SO, Agbede O and Olayemi AB: Studies on the efficacy of Bridelia ferruginea bark extract in reducing the coliform load and BOD of domestic waste water. Ethnobotanical Leaflets 2006; 10: 228-238.
- Akilandeswari S, Manimaran S, Valarmathi R, Kumar SK and Loganathan SV: Phytochemical observation on a leaf of Justicia tranquebariensisF. Aceiont Science of Life 2001; 20(3), 60-61.
- Manohar P, Rajesham VV, Kiran M, Kumar S and Kumara P: Pharmacognostical, phytochemical and antimicrobial activity of Bauhinia racemosa International Journal of Pharmaceutical Biology 2011; 1(1): 10-14.
How to cite this article:
Krishnamoorthi R: Phytochemical screening and antioxidant activity of Justicia tranquebariensis and Bauhinia racemosa. Int J Pharmacognosy 2015; 2(7): 362-67. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(7).362-67.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.