PHYTOCHEMICAL PROFILE, ACUTE TOXICITY, AND ANTINOCICEPTIVE POTENTIAL OF METHANOLIC EXTRACT OF CITRUS AURANTIFOLIA PEELS
HTML Full TextPHYTOCHEMICAL PROFILE, ACUTE TOXICITY, AND ANTINOCICEPTIVE POTENTIAL OF METHANOLIC EXTRACT OF CITRUS AURANTIFOLIA PEELS
Elijah Oladapo Oyinloye *, Abdullahi Akanji Murtala, Farouk Adedeji Oladoja, Rasidat Olufunke Tijani, Mu’d-Uwaiz Oluwatomisin Abdullah, Esther Oluwabunmi Adeniyi and Emmanuel Pelumi Sodamola
Department of Pharmacology and Toxicology, Faculty of Pharmacy, Olabisi Onabanjo University, Sagamu Campus, Ogun State, Nigeria.
ABSTRACT: People worldwide have long sought effective ways to alleviate pain, a major symptom of many clinical conditions. To achieve better pain control, there is a continuous need to discover new and effective therapeutic agents. Lime peels (Citrus aurantifolia) have been traditionally used to manage various ailments, including pain and infections, and have also been reported to protect several vital organs. Preliminary phytochemical screening and oral acute toxicity studies were conducted on Citrus aurantifolia peels extract (CAPE). No mortality was recorded during the acute toxicity study; therefore, the LD₅₀ of CAPE was estimated to be greater than 2000 mg/kg. In the acetic acid–induced writhing test, CAPE produced significant (p < 0.001) and non–dose-dependent inhibition of pain, with percentage reductions of 56.9%, 66.5%, and 64.9% at doses of 100, 200, and 400 mg/kg, respectively. In the formalin-induced pain model, CAPE at 200 and 400 mg/kg significantly (p < 0.001) reduced paw-licking time in both the first and second phases. In addition, CAPE significantly increased latency time (area under the curve) and maximal effect compared with the control group in the hot plate test. These findings suggest that Citrus aurantifolia peels extract may serve as a promising phytotherapeutic agent for the management of nociceptive pain.
Keywords: Phytochemical, Citrus aurantifolia, Indomethacin, Antinociceptive
INTRODUCTION: Almost all clinical illnesses involve pain, which is often the primary symptom that leads patients to seek medical attention. For individuals of all ages, it poses a significant clinical and financial obstacle. According to Jibira et al.1, the total estimated cost of pain may reach 3.0% of global, demonstrating the substantial economic burden of this condition. Globally, the annual expense of treating pain exceeds that of cardiovascular disease or cancer 2.
According to Henschke et al. 3, 20% of adults in developing nations suffer from pain as a result of insufficient management or a lack of efficient treatment plans. When tissue is damaged by mechanical, thermal, or chemical stimulation, pro-nociceptive chemicals are released, activating nociceptors at peripheral nerve terminals 1.
According to Kuschner et al. 4, non-steroidal anti-inflammatory drugs (NSAIDs), such as aspirin, diclofenac, and indomethacin, are frequently used to treat pain. However, because of gastrointestinal problems, for example, peptic ulcers, bleeding, perforation, blockages, and hepatotoxicity, their clinical usefulness is restricted. These drugs function by blocking the production of inflammatory mediators such as prostaglandins (PGs) and cyclooxygenase (COX) 5. According to Sloot et al.6, prolonged use of these painkillers is associated with adverse side effects or an increased risk of substance misuse, and many of them are not very effective in treating chronic pain. The aforementioned restrictions encourage researchers to look for new natural compounds derived from various plant sources for use as medications. Citrus aurantifolia (Christm.) Swingle is a polyembryonic herb that is cultivated in South Florida, India, Mexico, Africa, the West Indies, Guiyang, Nanning in southwestern China and other temperate, subtropical, and tropical climates 7.
This plant is a member of the kingdom: Plantae; Order: Sapindales; Family: Rutaceae; Phylum: Magnoliophyta; Class: Magnoliopsida; Citrus aurantifolia (C. aurantifolia) is a species in the genus Citrus. According to the USDA 8, C. aurantifolia is also known as lime (Nigeria), key lime (Mexican lime), and sour lime (China). Citrus aurantifolia (Christm.) Swingle (lime) peels promotes and alleviates organ dysfunction in various organs of the body. Lime has been used for years in traditional medicine to treat discomfort resulting from pain, primarily in the abdomen, peripheral and respiratory system 9. According to earlier studies, the presence of fatty substances such as pentadecanoic acid and 9-octadecenoic acid in Citrus aurantifolia peels has been ascribed to their antinociceptive properties 10. The inhibition of prostaglandin E2, nitric oxide, and tumor necrosis factor-α production has been attributed to the antinociceptive properties of pentadecanoic acid and 9-octadecenoic acid 11.
The phytochemical analysis, pharmacological characteristics of Citrus aurantifolia peels have not been thoroughly studied, despite this plant being widely used in traditional Chinese medicine to treat various illnesses. Hence, the antinociceptive potential of Citrus aurantifolia peels extract (CAPE), are reported herein antinociceptive effects of Citrus aurantifolia peels extract (CAPE), are reported herein.
MATERIALS AND METHODS: Three experimental models (n = 25) were randomly selected from 75 albino mice, and each model was then divided into five groups (n = 5). Group I received 10 mL/kg of distilled water (vehicle) {control} in the three models, whereas groups II–IV received 100, 200, and 400 mg/kg of CAPE, respectively. Group V was given 10 mg/kg of Indomethacin, {standard drug}. The doses of CAPE were chosen based on our previous investigation 14.
Chemicals: The following substances were used: glacial acetic acid, formaldehyde, and methanol. Indomethacin was a product of Sigma Aldrich, USA. Analytical-grade reagents and solvents were employed.
Plant Collection and Identification: Swingle fruits of Citrus aurantifolia (Chrism.) were gathered in Ibadan, Nigeria, which is southwest of Oyo State. A taxonomist from the University of Ibadan's Department of Botany in Ibadan, Oyo State, Nigeria, named D.P.O. Esimekhuai identified the plant. The plant was sampled and given the same Herbarium's voucher number, UIH-22957.
Preparation and Extraction of Citrus aurantifolia Peels: For some days, the entire peels of Citrus aurantifolia were gathered and left to air dry at room temperature (27 to 30 °C) until they reached a consistent weight. After that, they were processed with a mechanical grinder into a coarse powder. A solution of 80% methanol (1.0 L) and 20% water (0.2 L) was used to extract 200 grams of peel for 72 hours. The peel was then filtered and concentrated in a water bath that was set at 60°C. The raw methanol extract was stored at -20°C until it was time for analysis.
Animals and Ethical Statement: Swiss mice, weighing 18–22 g and of both sexes, were obtained from an animal house at the Institute for Advanced Medical Research and Training (IAMRAT), College of Medicine, University of Ibadan. The animals were kept in cages with normal humidity and lighting conditions, fed pelletised feed, and allowed unrestricted access to water. The Institutional Animal Ethics Committees' (IAEC) rules were followed in every instance. The WMA Statement on the Use of Animals in Biomedical Research, the EU rules (Directive 2010/63/EU) for experimental design and analysis in pharmaceutical care, and/or the recommendations of an internationally recognised authority were all adhered to in this work.
The use of animals in the study was approved ethically by the University of Lagos College of Medicine's Animal Care and Use Research Ethics Committee (CMUL/ACUREC). The study was authorised under permission number CMUL/ACUREC/06/22/1116.
Phytochemicals Screening: The methanol extract of Citrus aurantifolia peels was screened for phytochemicals both qualitatively and quantitatively by Trease and Evans 12 and De Silva et al.13, as previously reported by Oyinloye et al. 14.
Oral acute toxicity and lethality (LD50) study: The mice were randomly allotted to four groups of five mice. The individual groups received different concentrations of CAPE (100, 500, and 2000 mg/kg) orally. The control group was administered orally with 0.1 mL of distilled water. All the groups received treatment once at the beginning of the experiment. For 14 days following the first four hours of dosage administration, the mice were observed daily for any changes in their gross morphology, physiology, and behaviour, as well as for any signs of mortality 15.
Evaluation of Antinociceptive Effect of CAPE in Acetic Acid-Induced Nociception: Acetic acid was used to induce writhing in mice by modifying the method of Bastos et al. 16, as reported by Nwaneloet al. 17. The mice, weighing 18–22 g, were divided into five groups (n = 5), and each group spent 30 minutes acclimating within a Plexiglas observation chamber. Depending on when each treatment was scheduled, the mice were pre-administered with graded doses of CAPE (100, 200, and 400 mg/kg), indomethacin (10 mg/kg), and vehicle (10 mL/kg), all administered orally. To induce the distinctive writhing, mice were given 10 mL/kg of 0.6% acetic acid intraperitoneally one hour after treatment, which induced the characteristic writhing. Following an injection of acetic acid, the latency time to the first contraction of the abdominal muscles (writhe) was measured. Five to twenty minutes following the acetic acid injection, the number of writhes was also recorded. The results of the treatment groups were compared to those of the vehicle-treated controls. With only slight modifications,
The percentage of the number of writhes is calculated by = Vehicle peak- Test compound / Vehicle peak x 100
Evaluation of Antinociceptive Activity of CAPE in Formalin-Induced Nociception: The protocol described by Nwanelo et al.17 was followed when performing the formalin test. After being divided into five groups (n = 5), the 18-22 g mice were given a 30-minute acclimatisation period in a plexiglass observation chamber. The sub-plantar area of the left hind paw received an injection of 1% formalin (20µL). The injection of formalin caused paw licking, biting, and grooming. The length of time spent licking the injected paw was recorded and considered an indication of discomfort. An hour before the mice were injected with formalin, they were pretreated with CAPE (100, 200, and 400 mg/kg), indomethacin (10 mg/kg), and a control group (10 mL/kg). The duration of paw licking was measured at 0 to 5 min (first/neurogenic phase) and 15 to 30 min (second/inflammatory phase) after formalin administration 18.
Evaluation of Antinociceptive Effect of CAPE in Thermal-Induced Nociception (Hot Plate Method): Hot plate-based thermally generated nociceptive behaviour in mice utilising a modified Ezeja et al. 19 techniques. The 18-22 g mice were split up into five groups (n = 5), and each group spent 30 minutes acclimating inside a plexiglass observation chamber. Depending on the duration allotted for each treatment, Pretreatment of the mice included indomethacin (10 mg/kg p.o.), CAPE (100, 200, and 400 mg/kg p.o.), and control (10 mL/kg p.o.). The baseline latency was set. A UgoBasile hot/cold plate (35100, Italy) was used to maintain each animal at 55±10 °C, and the animals' reaction times to the heat stimulation were recorded. The nociceptive response latency (as measured by paw licking or jumping) was assessed prior to treatment and at 60, 90, 120, and 150 minutes post-treatment. The post-treatment cut-off period was set at 20 seconds, and only animals that displayed initial nociceptive reactions within 2 seconds were chosen. Calculations were made to determine the area under the curve (AUC) and mean percentage maximum probable effect (% MPE) as follows:
Area under the curve (AUC) = Time spent before jumping for the extract and negative control (vehicle)
% MPE = post-drug latency – Pre-drug latency / Cut-off time – Post drug latency ×100
Statistical Analysis: The results are presented as the Mean ± SEM of five mice. Data were analysed using 1-way ANOVA analysis of variance (ANOVA) followed by Dunnett’s/Turkey’s post hoc tests using Graph Prism 6 Software (Graph Pad Software, Inc., CA, USA). The results were considered significant at p<0.05.
RESULTS: The phytochemical (qualitative and quantitative) analysis results showed the presence of the following constituents in the following order: alkaloids (1.6% w/w), terpenoids (1.15% w/w), tannins (0.94% w/w), phenols (0.79% w/w), flavonoids (0.72% w/w), and saponins (0.65% w/w).
Oral Acute Toxicity: Mice given oral doses of methanolic extract from Citrus aurantifolia peels at 100 mg/kg, 500 mg/kg, and 2000 mg/kg did not die or show any changes in behavior, Additionally, the animals' cutaneous, digestive, neurological, and sensory systems remained unaltered. This proved that the extracts' LD50 was estimated to be above 2000 mg/kg, indicating that they were safe to take orally and that the study doses were not toxic.
Antinociceptive Effect of CAPE in Acetic Acid–Induced Writhing Test: The number of writhes decreased significantly (p<0.001; p<0.0001) in CAPE treated groups, which was not differ statistically among the CAPE treated groups as compared to control (vehicle).
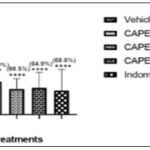
Furthermore, CAPE at 100, 200, and 400 mg/kg exhibited writhing percentage inhibitions of 56.9%, 66.5%, and 64.6%, respectively, relative to control. The CAPE (100, 200, and 400 mg/kg) increased the onset of writhes caused by acetic acid, which was not significant Fig. 1 and 2.
FIG. 1: ANTI-NOCICEPTIVE EFFECTS IN ACETIC ACID –INDUCED WRITHING. NO OF WRITHES. Data represent Mean ± SEM of five mice, values in parenthesis are percentages of inhibition. ***p<0.001; ****p<0.0001 by 1-way ANOVA followed by Newman-Keuls Multiple comparison post hoc test compared to vehicle group.
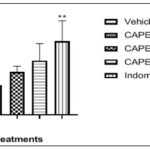
FIG. 2: ANTI-NOCICEPTIVE EFFECTS IN ACETIC ACID –INDUCED WRITHING. ONSET OF WRITHES (MIN). Data represent Mean ± SEM of five mice, **p<0.01, by 1-way ANOVA followed by Newman-Keuls Multiple comparison post hoc test compared to vehicle group.
Antinociceptive Effect of CAPE in Formalin Test: In the first phase of the test, mice administered with CAPE (100, 200 and 400 mg/kg) independently and significantly decreased the time spent in paw licking as compared to the control (vehicle). Meanwhile, in the second phase a significant (p<0.01, p<0.001) decrease in the time spent in paw licking were observed at 200 and 400 mg/kg relative to control. The decrease in the time spent in paw licking by CAPE 400 mg/kg is comparable with indomethacin (standard drug) Fig. 3.
FIG. 3: ANTI-NOCICEPTIVE EFFECTS OF CAPE IN MICE IN THE FIRST AND SECOND PHASE OF THE FORMALIN-INDUCED NOCICEPTION TEST. Data represent Mean ± SEM of five mice. **p<0.01, ***p<0.001, by 1-way ANOVA followed by Newman-Keuls Multiple comparison post hoc test compared to vehicle group.
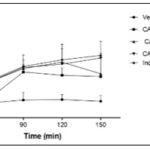
Antinociceptive Effect of CAPE in Hot Plate Test: Results of CAPE 100, 200, and 400 mg/kg in the hotplate test are presented in Fig. 4 and Fig. 5. The extract was found to exhibit an increase in percentage maximum possible effect (% MPE) relative to control. The extract dependently and significantly (*p<0.05; p<0.01) increase the area under the curve (AUC) {time spent before jumping} in comparison with the control.
FIG. 4: ANTI-NOCICEPTIVE EFFECTS OF CAPE IN THERMALLY-INDUCED NOCICEPTION ON HOTPLATE, MAXIMAL POSSIBLE EFFECT. Data represent Mean ± SEM of five mice
FIG. 5: ANTI-NOCICEPTIVE EFFECTS OF CAPE IN THERMALLY-INDUCED NOCICEPTION ON HOT PLATE, AREA UNDER THE CURVE. Data represent Mean ± SEM of five mice. *p<0.05; **p<0.01, by 1-way ANOVA followed by Newman-Keuls Multiple comparison post hoc test compared to vehicle group.
DISCUSSION: Alkaloids, terpenoids, tannins, phenols, flavonoids, and saponins were detected in the methanol extract of Citrus aurantifolia peels after preliminary phytochemical screening. This supports a previous study by Oyinloye et al. 14. According to Jibira et al, 1 plant components such as flavonoids, saponins, and tannins have influenced drug discovery and development studies due to their inherent pharmacological potential. Research on Citrus aurantifolia peels extract has shown that it contains a wide range of bioactive compounds, contributing to its diverse medicinal potential. Meanwhile, Citrus aurantifolia peels has been found to have antinociceptive properties. Several induction models, including acetic acid, formalin induction, and thermally induced nociceptive, were used to examine the antinociceptive activity. The mice did not exhibit any behavioral abnormalities, sickness, or mortality up to a dose of 2000 mg/kg, indicating that CAPE is safe and non-toxic at this dose. Because substances with an oral LD50 value of greater than 1000 mg/kg are believed to have a low toxicity profile, the extract can thus be regarded as being extremely non-toxic in mice 1.
The acetic acid-induced abdominal writhing test is a highly sensitive and practical test that offers an excellent analgesic profile for both centrally and peripherally acting drugs 1. Nociception is initiated by inducing inflammation in the peritoneum, which lowers the pH of the system and releases endogenous pro-inflammatory substances. Citrus aurantifolia peels extract at 100, 200, and 400 mg/kg (56.9%, 66.5%, and 64.9%, respectively) produced a significant (P < 0.001) non-dose-dependent inhibition of writhes compared to the control. However, at 100, 200, and 400 mg/kg, CAPE showed a delay in the onset of writhing after acetic acid administration compared to the control.
This indicates that the CAPE exhibits antinociceptive activity and also inhibits the release of inflammatory mediators or cytokines. It is thought that acetic acid works indirectly by causing the release of prostaglandins and other mediators into the peritoneum. These mediators then trigger nociceptive neurons that are responsive to analgesic anti-inflammatory medications 20. CAPE's analgesic action may be attributed to its ability to inhibit pain mediators, which excite pain nerve endings similar to that of indomethacin and NSAIDs 21. In drug discovery, the formalin-induced nociception is a well-established in-vivo model of acute pain that has been used to assess the analgesic potential of compounds 1. Two different nociceptive response phases define the formalin test. The ability to distinguish between non-inflammatory (first) and inflammatory (second) phase of pain is one advantage of the formalin paradigm. A few minutes make up the first phase, which represents the neurogenic aspect of nociception. The release of chemical mediators such as bradykinins, histamine, interleukins, and tumor necrosis factors is linked to the second phase, which follows the silent period of 15 minutes 22. Moreover, the majority of non-steroidal anti-inflammatory drugs (NSAIDs) target prostaglandins at the local level, which contributes to the advancement of nociception 23. Our results in the present study showed that our extract demonstrated a significant (p<0.01; P<0.001) antinociceptive effect in the first and second phases of the formalin test in mice, except CAPE 100 mg/kg in the second phase.
The antinociceptive effect of indomethacin in the first phase is in agreement with earlier research by Jurna and Brune 24, who reported the central activity of indomethacin, ibuprofen and diclofenac responsible for their analgesic effect. The hot-plate method is a widely used test that is thought to be selective for medicines that act centrally. It is well-established that mice's nociceptive response to heat stimuli can be used to identify opiate analgesics (centrally acting agents) and other spinal-derived analgesic medications 25. In the hot plate test, CAPE extract and indomethacin (10 mg/kg) demonstrated a larger percentage maximum possible effect (% MPE) as the dose increased relative to the control group (distilled water).
The graded dosages of CAPE (100, 200, and 400 mg/kg) exhibited a dose-dependent and significant increase in the area under the curve (AUC) {time spent prior to jumping}. It is known that an agent must be working centrally if it prolongs the hot plate latency in mice by inducing thermally generated discomfort 26. The effect shown by our extract in the hot plate test, which is a test used to evaluate the analgesic activity mediated by central mechanisms, confirmed that the analgesic activity of CAPE could be mediated by both central and peripheral mechanisms, as described by Elisabetsky et al. 27 and Pal et al 28. Our study demonstrated that indomethacin, a non-steroidal anti-inflammatory drug, exhibited an antinociceptive effect in the hot plate test, a method used to assess the antinociceptive activity mediated by central mechanisms. However, the central effect of indomethacin in the present study is in agreement with Hu et al 29, who asserted that indomethacin, a non-steroidal anti-inflammatory drug (NSAID), exhibits both the central and peripheral mechanisms of pain.
Overall, Citrus aurantifolia peels extract demonstrated significant antinociceptive activity across multiple pain models, indicating both peripheral and central analgesic effects. These activities are likely mediated by its bioactive phytoconstituents.
CONCLUSION: The Citrus aurantifolia peels extract (CAPE) is relatively non-toxic in mice, with an LD50 above 2000 mg/kg, and exerts both central and peripheral analgesic effects. However, the results obtained provide the pharmacological basis for the ethnomedicinal use of Citrus aurantifolia peels extract in managing pain-related disorders. Furthermore, future research should focus on isolating the active constituents to investigate the processes underlying the central and peripheral antinociceptive effects.
Animal Ethics Approval: The Institutional Animal Ethics Committees' (IAEC) rules were followed in every instance. The WMA Statement on the use of Animals in Biomedical Research, the EU rules (Directive 2010/63/EU) for experimental design and analysis in pharmaceutical care, and/or the recommendations of an internationally recognised authority were all adhered to in this work. The use of animals in the study was approved ethically by the University of Lagos College of Medicine's Animal Care and Use Research Ethics Committee (CMUL/ACUREC). The study was authorised under permission number CMUL/ACUREC/06/22/1116.
ACKNOWLEDGEMENTS: The authors would like to thank all the Technical Staff of Department of Pharmacology and Toxicology, Olabisi Onabanjo University, Sagamu Campus Ogun State, Nigeria, for their assistance during bench work.
Ethical Approval: An ethical clearance request was submitted to the University of Lagos, (Lagos State, Nigeria), College of Medicine's Animal Care and Use Research Ethics Committee (CMUL/ACUREC), and was granted permission number CMUL/ACUREC/06/22/1116.
CONFLICTS OF INTERESTS: The authors have no relevant conflicts of interest to disclose.
REFERENCES:
- Jibira Y, Boakye-Gyasi E, Abotsi WKM, Amponsah IK, Adongo DW and Woode E: Hydroethanolic stem bark extract of Burkea africana attenuates vincristine-induced peripheral neuropathy in rats. Advances in Pharmacological and Pharmaceutical Sciences 2020; 2020: 1–10.
- Bond M and Breivik H: Why pain control matters in a world full of killer diseases. In: Wittink HM and Carr DB (eds): Pain Management: Evidence, Outcomes, and Quality of Life. Elsevier, London 2008; 407–411.
- Henschke N, Kamper SJ and Maher CG: The epidemiology and economic consequences of pain. Mayo Clinic Proceedings 2015; 90(1): 139–147.
- Kushner P, McCarberg BH and Grange L: The use of non-steroidal anti-inflammatory drugs (NSAIDs) in COVID-19. NPJ Primary Care Respiratory Medicine 2022; 32: 1–10.
- Monteiro C, Silvestre S, Duarte AP and Alves G: Safety of non-steroidal anti-inflammatory drugs in the elderly. International Journal of Environmental Research and Public Health 2022; 19: 3541.
- Sloot S, Boland J and Snowden JA: Side effects of analgesia may significantly reduce quality of life in symptomatic multiple myeloma. Supportive Care in Cancer 2015; 23(3): 671–678.
- Patil JR, Murthy KN, Jayaprakasha GK, Chetti MB and Patil BS: Bioactive compounds from Mexican lime juice induce apoptosis in human pancreatic cells. Journal of Agricultural and Food Chemistry 2009; 57(22): 10933–10942.
- USDA Agricultural Research Service: National Nutrient Database for Standard Reference, Release 26. USDA, Washington DC 2013.
- Igimi H, Nishimura M, Kodama R and Ide H: Absorption, distribution and excretion of d-limonene in rats. Xenobiotica 1974; 4(2): 77–84.
- Api AM, Belsito D and Biserta S: RIFM fragrance ingredient safety assessment of nonanoic acid. Food and Chemical Toxicology 2020; 144: 111683.
- Xie C, Wang S, Cao M, Xiong W and Wu L: (E)-9-octadecenoic acid ethyl ester ameliorates inflammatory responses in macrophages. Evidence-Based Complementary and Alternative Medicine 2022; 2022: 6731360.
- Trease GE and Evans WC: Textbook of Pharmacognosy. 15th ed. Saunders, London 2002; 393.
- De Silva GO, Abeysundara AT and Aponso MM: Extraction methods and phytochemical screening techniques. American Journal of Essential Oils and Natural Products 2017; 5(2): 29–32.
- Oyinloye EO, Murtala AA and Oladoja FA: Citrus aurantifolia peel extract attenuates doxorubicin-induced nephrotoxicity. Pharmacological Research – Modern Chinese Medicine 2024; 11: 100412.
- Adeyemi OO, Akindele AJ and Ogunleye EA: Antidiarrhoeal effect of Sansevieria liberica root extract. Journal of Ethnopharmacology 2009; 123(3): 459–463.
- Bastos GNT, Santos ARS and Ferreira VMM: Antinociceptive effect of Physalis angulata root extract in mice. Journal of Ethnopharmacology 2006; 103(2): 241–245.
- Nwanelo V, Agwu L and Ugwuoke S: Antinociceptive effect of Chasmantheradependens root extract. Letters in Applied Bioscience 2023; 12: 176.
- Pereira CKS, Oliveira AMF and Penha ARS: Antinociceptive effect of Herissantia crispa extract. Journal of Medicinal Plants Research 2012; 6(40): 5813–5818.
- Ezeja MI, Ezeigbo II, Madubuike KG and Ekpe IJ: Analgesic activity of Delonix regia leaf extract. Nigerian Veterinary Journal 2012; 33(2): 515–522.
- Hasan SMR, Hossain MM and Akter R: Analgesic activity of Commelina benghalensis aerial parts. International Journal of Pharmacology 2010; 6(1): 63–67.
- Valerio DA, Cunha TM and Arakawa NS: Anti-inflammatory and analgesic effects of budlein A in mice. European Journal of Pharmacology 2007; 562(1–2): 155–163.
- Hama A and Menzaghi F: Nicotinic receptor antagonists enhance formalin-induced nociception. Brain Research 2001; 888(1): 102–106.
- Ferreira SH: Peripheral analgesic sites of action of anti-inflammatory drugs. International Journal of Clinical Practice Supplement 2002; 128: 2–10.
- Jurna I and Brune K: Central effects of NSAIDs on thalamic neurons. Pain 1990; 41(1): 71–80.
- Alhaider AA, Lei SZ and Wilcox GI: Spinal 5-HT mediated antinociception and possible GABA release. Journal of Neuroscience 1991; 11(7): 1881–1888.
- Ibironke GF and Ajiboye KI: Anti-inflammatory and analgesic properties of Chenopodium ambrosioides. International Journal of Pharmacology 2007; 3(2): 111–115.
- Elisabetsky E, Amador TA, Albuquerque RR, Nunes DS and Carvalho ACT: Analgesic activity of Psychotria colorata alkaloids. Journal of Ethnopharmacology 1995; 48(2): 77–83.
- Pal S, Sen T and Chaudhuri AK: Neuropsychopharmacological profile of Bryophyllum pinnatum leaf extract. Journal of Pharmacy and Pharmacology 1999; 51(3): 313–318.
- Hu X, Tang H, Li QX and Huang Y: Central mechanism of indomethacin analgesia. European Journal of Pharmacology 1994; 263(1–2): 53–57.
How to cite this article:
Oyinloye EO, Murtala AA, Oladoja FA, Tijani RO, Abdullah MO, Adeniyi EO and Sodamola EP: Phytochemical profile, acute toxicity, and antinociceptive potential of methanolic extract of Citrus aurantifolia peels. Int J Pharmacognosy 2026; 13(2): 80-87. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(2).80-87.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
80-87
582 KB
109
English
IJP
Elijah Oladapo Oyinloye *, Abdullahi Akanji Murtala, Farouk Adedeji Oladoja, Rasidat Olufunke Tijani, Mu’d-Uwaiz Oluwatomisin Abdullah, Esther Oluwabunmi Adeniyi and Emmanuel Pelumi Sodamola
Department of Pharmacology and Toxicology, Faculty of Pharmacy, Olabisi Onabanjo University, Sagamu Campus, Ogun State, Nigeria.
oyinloye.oladapo@oouagoiwoye.edu.ng
27 January 2026
23 February 2026
26 February 2026
10.13040/IJPSR.0975-8232.IJP.13(2).80-87
28 February 2026