PHYTOCHEMICAL ANALYSIS AND IN-VIVO SCREENING OF PSEUDODRYNARIA CORONANS (WALL. EX. METT.) CHING ON ITS ANALGESIC AND ANTI-INFLAMMATORY PROPERTIES
HTML Full TextPHYTOCHEMICAL ANALYSIS AND IN-VIVO SCREENING OF PSEUDODRYNARIA CORONANS (WALL. EX. METT.) CHING ON ITS ANALGESIC AND ANTI-INFLAMMATORY PROPERTIES
Kirangngaulule Elu 1, Probin Kr. Roy 1, Phaibiang Lapasam 1, Charles Lalnunfela 2, H. Lalhlenmawia 1, Yasangam Umbon 1 and T. C. Lalhriatpuii * 1
Department of Pharmacy 1, Regional Institute of Paramedical & Nursing Sciences (RIPANS), Aizawl -796017, Mizoram, India.
Deparment of Zoology 2, Mizoram University, Tanhril - 796004, Mizoram, India.
ABSTRACT: The methanolic rhizome extract of Pseudodrynaria coronans (MEPC) was assessed for its preliminary phytochemical constituents, anti-nociceptive and anti-inflammatory activity. Acute toxicity of the extracts was tested following the OECD – 425 guidelines and after giving MEPC at the dose of 2000 mg/kgb.w indicating that the LD50 is higher than the given dose. Two doses 200 and 400 mg/kg bw. were selected for all the experiments. Hot plate and Tail immersion methods showed that the extract was statistically significant (p<0.05 and p<0.01). Different concentrations of the MEPC showed significant inhibition of protein denaturation and membrane stabilization. Also, the MEPC exhibited significant inhibition of carrageenan-induced paw oedema (p<0.01) and cotton pellet-induced granuloma (p<0.05 & p<0.01) inflammatory responses in the rats.
| Keywords: |
Anti-nociceptive, Anti-inflammatory, Pseudodrynaria coronans, NSAIDS, Acute Toxicity, MEPC
INTRODUCTION: The advancement in pharmaceutical research makes natural products, mainly medicinal plants, a new hope for drug development. The pure compound(s) or standardized extracts derived from a wide pool of fauna become the greatest assets for researchers in their pursuit of combating different ailments 1. Methods were developed to isolate the bioactive compounds from the highly acclaimed remedial plants instead of using the whole plant, thereby using them for developing drugs with higher efficacy and lower side effects 2.
Pain may be defined as the uncomfortable sensation produced by nerve impulses produced by the nervous stimuli in the brain, the drugs used for terminating the pain or alter pain sensitivity are named ‘Analgesic’.
Opiates and non-steroidal anti-inflammatory drugs are our only defense and remedy in our fight against pain, unfortunately leaving behind and adverse effects that are detrimental to our physiological system. Because of these side effects and the low potency of current analgesia-inducing drugs, there is an urgent call for the search for an alternative. Medicinal plants with their diverse phytochemical compounds may be the answer for the long-sought alternatives from which novel analgesic could be discovered 3. Man is equipped with a sturdy physiological system in which inflammation is one of the defense mechanisms in which pain is the primary associate followed by alteration in a membrane, vascular permeability, and increase denaturation of protein. Inflammation is classified into two categories, namely acute and chronic inflammation which is triggered by the release of kinins, prostaglandins, and histamine the inflammatory mediators. Steroidal and Non-steroidal anti-inflammatory drugs (NSAIDs) are clinically available treatments in the market, but due to their side effects, searching for phytochemicals alternative is the need of the hour 4.
Pseudodrynaria coranans (Wall. Ex. Mett.) Ching is locally known as “Awmvel” among the Mizo. This epiphytic basket fern belongs to the Polypodiaceae family having a white rhizome that resembles a paw encircling the host plant. The plant is native to tropical Africa, South Asia, East Asia, Southeast Asia, Australia, and Oceania in which the Chinese use the rhizome for the treatment of traumatic injury, rheumatic arthritis, tinnitus, and toothache 5. In Mizoram, the crushed rhizomes are applied externally for infection against herpes zoster and taken orally for an irregular heartbeat as traditional medicine. Unfortunately, this medicinally potential plant has not been studied intensively; therefore investigation of phytochemical constituents, acute toxicity, anti-nociceptive and anti-inflammatory studies have been carried out to study the true nature of this plant and to justify its traditional uses.
MATERIALS AND METHODS:
Collection and Identification of the Plant Material: The rhizomes of Pseudodrynaria coronans were collected from Aizawl, Mizoram (India) in the month of September. The specimens were identified and authenticated by Botanical Survey of India (BSI), Kolkata (Reference No: BSI/ERC/TECH/2018-19/688) and herbarium was preserved in RIPANS library for future reference.
Preparation of Plant Extract: The rhizomes of the plants were washed, cut into pieces, dried in shade and powdered using mechanical grinder. The powdered material was then subjected to successive extraction using Soxhlet apparatus. The powdered sample was subjected to extraction with petroleum ether for the removal of fats, which was followed by the extraction with chloroform and methanol for 72 h each. Then the extracts were obtained by using rotary vacuum evaporator and stored in the refrigerator at 4 °C for further analysis.
Phytochemical Screening: The preliminary phyto-chemical screening of the methanolic extracts was carried out as per the standard protocol. In the phytochemical screening, methanolic extract of Pseudodrynaria coronans was screened for the presence of phytochemical constituents like alkaloids, carbohydrates, glycosides, tannins, phenols, steroids, flavonoids, proteins and amino acids, fats, and fixed oil.
Animals Care and Maintenance: Six to eight weeks old Wistar rats of both sexes were maintained in an institution animal house in RIPANS in simulated atmospheric conditions (temperature: 25±2 °C; relative humidity: 60-70%; 12 h of light-dark alternative cycles). Animals were acclimatized for 7 days in a sterile polypropylene cage using sawdust as bedding. Experimentation and methodology were approved by the Institutional Animal Ethics Committee of Regional Institute of Paramedical & Nursing Sciences (RIPANS), bearing Approval no. IAEC/RIPANS/ 34, 35 & 36.
Diet: All the animals were given water ad libitum and fed with a standard commercial rodent diet.
Acute Oral Toxicity Test: An acute oral toxicity test was performed as per the Organization for Economic Co-operation and Development guidelines 425 (OECD-425). A total of 10 animals (5 males and 5 nulliparous females) were selected and divided into two groups randomly. Group-I received methanol extract of Pseudodrynaria coronans (MEPC), and group - II received normal saline.
Administration of Dose: Animals were fasted overnight before oral administration. The weight of animals was measured, and dosage calculation was done accordingly. MEPC was administered at the dose of 2000 mg/kg body weights of the rat orally by feeding gavage. During the study, changes in body weight, a sign of toxicity, food and water intake were observed.
In-vivo Analgesic Activity:
Hot Plate Method: Analgesic activity of MEPC using hot plate method was determined by the method described by Hijazi et al. with slight modification 6. Animals were divided into 4 groups consisting of 5 animals each (both sexes). The first group (control group) was given normal saline, and group- II (Standard group) was given Morphine sulfate at the dose of 5 mg/kg b.w intraperitoneally. Group – II and group – IV were treatment groups receiving MEPC at the dose of 200 and 400 mg/kg b.w, respectively. The animals were placed on a hot plate (55 ± 1 ºC), the reaction time before the treatment was taken, and four more readings were taken after drug administration at 30, 60, 90, and 120 min. The cut-off time of 15 seconds was maintained to avoid paw damage.
Tail Immersion Method: The tail Immersion method was performed as described by Malairajan et al. with slight modification (Malairajan et al., 2011). The water bath was maintained at a temperature of 55± 1 ºC, and the rat’s tail about 3 cm was emersed in the water bath; the tail withdrawal time was noted as reaction time or tail-flick latency. Animals were divided into four groups (namely control, standard, 200 mg/kg b.w, and 400 mg/kg b.w) of 5 animals each. To avoid the injury of the tissues of the tail, the maximum cut-off time for immersion was taken as 15 sec.
The initial reading was taken before administration of the test and standard drugs and then at 30, 60, 90,120, and 180 min after the administration. The reaction time was measured and calculated.
In-vitro Anti-Inflammatory Activity:
Anti-Denaturation Activity: Anti-denaturation assay was carried out using a method developed by William et al. with slight modification 7. 0.2% BSA was prepared in Tris buffer saline, and the pH was adjusted using acetic acid to 6.8. Extract stock solution of 1000 µg/ml was prepared in methanol, and 4 different concentrations of 200, 400, 600, 800, and 1000 µg/ml were prepared from this stock solution. From these working concentrations, 50 µl each was taken in an Eppendorf tube, and 5 ml of 0.2% w/v BSA was added. The standard consisted of 10 µg/ml of Aspirin in methanol with 5 ml 0.2% w/v BSA solution. They were heated at 72 °C for 5 minutes and then cooled for 10 min. The absorbance of these solutions was determined by using UV/Vis Double beam spectrophotometer (Elico SL-196) at a wavelength of 660 nm. The % inhibition of precipitation (denaturation of the protein) was determined using the following formula:
% inhibition of denaturation = (Absorbance of control - Absorbance of extract) × 100 / Absorbance of control
Inhibition of Albumin Denaturation: Albumin denaturation was determined following Chandra et al. method with slight modification 8. The reaction mixture was made up of 0.2 ml of eggs albumin (Fresh hen egg), 2.8 ml of phosphate-buffered saline (PBS, pH 6.4), and 2 ml of varying concentrations (200, 400, 600, 800 & 1000 µg/ml) of MEPC. 2 ml of Aspirin was used as a reference drug instead of MEPC and double-distilled H2O served as control.
The mixtures were incubated at 37 °C for about 15 min and then heated at 70 °C for 5 min. After cooling, their absorbance was measured at 660 nm, and the percentage inhibition of protein denaturation was calculated by using the formula;
% Inhibition of denaturation = (Absorbance of control – Absorbance of extracts) × 100 / Absorbance of control
HRBC Membrane Stabilization Method: Blood samples were collected from healthy individuals who had not taken any NSAIDS drugs two weeks before the experiment. It was then mixed with an equal volume of sterilized Alsevers solution and centrifuged at 3000 rpm. The packed cells were washed with is saline solution, and a 10% v/v solution was prepared with normal saline and kept at 4 °C undisturbed before use.
Different concentrations of MEPC extract (200, 400, 600, 800, and 1000 µg/ml), Diclofenac sodium (standard), and control were separately mixed with 1 ml of phosphate buffer, 2 ml of hyposaline, and 0.5 ml of 10% HRBC suspension. All the mixtures were incubated at 37 °C for 30 min and centrifuged at 3000 rpm. The supernatant liquid was decanted, and the hemoglobin content was estimated spectro-photometrically at 560 nm. The percentage of HRBC membrane stabilization was calculated by using the formula 9:
Percentage stabilization = Absorbance of control – Absorbance of extract × 100 / Absorbance of control
In-vivo Anti-Inflammatory Activity:
Carrageenan-Induced Rat Paw Edema (Acute Model of Inflammation): Animals were divided into four groups, namely Control, Standard drug (Indomethacin 10 mg/kg, p.o.), MEPC 200 mg/kg b.w, and MEPC 400 mg/kg b.w consisting of four rats each in a group. Treatments were given 1 h prior to the administration of carrageenan. Paw edema was induced by sub plantar injection of 0.1 ml 0f 1%w/v carrageenan suspended in 0.9% normal saline into the right hind paw of each group of rats 10. Paw edema was measured by taking the paw diameter using Vernier calliper at (0, 1, 2, 3, 4, 5, 6, and 24 h 11. The increase in paw thickness, as well as the percentage inhibition, was calculated by the following equations:
Increase in paw thickness in control
(Pc) = Pt – Po
Increase in paw thickness in treated
(PT) = Pt – Po
Percentage inhibition = [(Pc – PT)/Pc] × 100
Where, Pt is the paw thickness at time t, Po is initial paw thickness carrageenan-Induced Paw edema in rats.
Cotton Pellet- Induced Granuloma in Rats (Chronic Model of Inflammation): Chronic inflammation was assessed through cotton pellet induced granuloma model according to D’Arcy et al. method with slight modification. Animals were divided into four groups consisting of four animals each in a group and were anesthetized, and sterile cotton pellets (10±1 mg) were implanted subcutaneously along the axillae of the rats. Control vehicle (0.5% CMC), MEPC (200 and 400 mg/kg p.o.), and standard drug Diclofenac sodium (10 mg/kg p.o.) were administered successively for 7 days starting from the day of cotton pellet implantation. The cotton pellets were surgically removed on the 8th day, and the weight of the wet pellets were measured immediately. After that, the wet pellets were dried in an oven at 60 ºC for 24 h. An increase in the wet and dry weight of the pellets was recorded as a measure of granuloma formation, and also their percentage inhibition was calculated by using the formula.
% inhibition = Mean granuloma weight of control (wet/dried) - Mean granuloma weight of test (wet/dried) × 100 / mean weight of control
Statistical Analysis: For statistical analysis, GraphPad Prism 8.0.2 software was used. All the data were statistically analyzed using one-way ANOVA followed by Dunnet’s multiple comparison test. P values <0.05 were considered statistically significant.
RESULTS:
Phytochemical Analysis: The phytochemical investigation from the methanolic rhizomes extract of Pseudodrynaria coronans revealed the presence of steroids, flavonoids, tannin, saponin, triterpenoids, amino acids, and carbohydrates.
TABLE 1: PHYTOCHEMICAL ANALYSIS OF METHANOL EXTRACT OF PSEUDODRYNARIA CORONANS
| Phytochemical test | Different extract | ||
| Petroleum ether | Chloroform | Methanol | |
| Alkaloids | - | + | - |
| Steroids | - | + | + |
| Flavonoid | - | - | + |
| Reducing sugar | - | + | |
| Tannins | - | - | + |
| Saponins | - | - | + |
| Triterpenoids | - | + | + |
| Amino acid | - | - | + |
| Fats and Fixed oils | + | - | - |
| Carbohydrates | - | + | + |
| Glycosides | - | - | - |
Acute Toxicity Studies: The acute oral toxicity study from the rhizomes of methanolic extract of Pseudodrynaria coronans showed no toxicity or mortality within 24 h, and there were no signs of behavioral changes within 14 days after administering 2000 mg/kg dose of the extract (Table 2, 3 & 4). Therefore 200 and 400 mg/kg, b.w were selected as doses for the study of analgesic activity.
In-vivo Analgesic Activity:
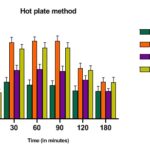
Hot Plate Method: The results of the hot plate method revealed that oral administration of the MEPC at the doses of 200 and 400 mg/kg, b.w., produced significant increases in the latency time, which can be compared to that of the standard drug (morphine, 5 mg/kg., b.w., p.o) as shown in Table 5. No significant difference in the thermal stimulus in rats treated with the vehicle (control) was observed throughout the different time points of the experiment. Administration of Morphine significantly increases the response time of the rats (8.34 seconds 90 min) and its analgesic effect decreased with time but remained significant even after 1 h and 30 min for both the extracts. In comparison, 400 mg/kg is more effective as compared to the 200 mg/kg extract, and the results were found to be highly statistically significant (P<0.05, P<0.01, respectively).
FIG. 1: ANALGESIC ACTIVITY OF MEPC AND MORPHINE ON HOT PLATE METHOD. THE ANIMALS WERE TREATED DAILY WITH THE EXTRACTS (P.O) OR MORPHINE (I.P). Each point represents the mean ± S.E.M. of change of paw size. Both extracts and morphine were effective compared to control (normal saline), by one-way ANOVA, Dunnet’s multiple comparison tests
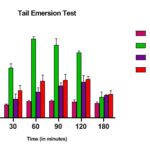
The results of the Tail immersion method presented in Table 6 showed that oral administration of the methanolic extracts at the doses of 200 and 400 mg/kg, b.w., produced significant increases in the reaction time in comparison to that of the standard drug (morphine, 5 mg/kg, b.w., p.o).
When treated with the vehicle (control), no significant difference in the thermal stimulus in rats was observed throughout the experiment. The analgesic effect of Morphine decreased with time but remained significant even after an hour.
All doses of the extracts showed significant increases in the latency time of rats when compared to the control group. The analgesic activity in the standard and extracts treated in animals is noted from 30 min onwards till 120 min in which response peaked at 90 min for Morphine and both the extracts.
FIG. 2: ANALGESIC ACTIVITY OFMEPC AND MORPHINE ON TAIL EMERSION METHOD. THE ANIMALS WERE TREATED DAILY WITH THE EXTRACTS (P.O) OR MORPHINE (I.P). Each point represents the mean ± S.E.M. of change of paw size. Both extracts and morphine were effective compared to control (normal saline), by one-way ANOVA, Dunnet’s multiple comparison tests.
In-vitro Anti-Inflammatory Activity:
Anti-denaturation Activity: The methanolic rhizomes extract of Pseudodrynaria coronans has the ability to protect Bovine Serum Albumin (BSA) against heat denaturation, as shown in Table 7. The methanolic extract has shown 60.36% and 82.02% anti-denaturation activity on BSA at a concentration of 800 µg/ml and 1000 µg/ml, respectively.
This result indicates that the anti-denaturation action of MEPC increased with the increased concentration. The standard Aspirin had shown 87.0% of anti-denaturation effect at 1000 µg/ml.
TABLE 2: EFFECT OF MEPC AND ASPIRIN ON ITS ANTI-DENATURATION ACTIVITY BY INHIBITING THE HEAT DENATURATION OF BOVINE SERUM ALBUMIN (BSA)
| Concentration (µ/ml) | Aspirin | MEPC | ||
| Mean ± SEM
(Absorbance 660nm) |
%Inhibition | Mean ± SEM
(Absorbance 660 nm) |
% Inhibition | |
| 200 | 0.14±0.01** | 35.2 | 0.16±0.01* | 23.96 |
| 400 | 0.19±0.01** | 49.76 | 0.14±0.01** | 35.02 |
| 600 | 0.07±0.02** | 66.82 | 0.12±0.01** | 44.23 |
| 800 | 0.05±0.01** | 76.49 | 0.08±0.02** | 60.36 |
| 1000 | 0.02±0.01** | 87.09 | 0.03±0.01** | 82.02 |
*All the results presented here are Mean ± SEM, compared with the standard by one-way ANOVA, Dunnet’s multiple comparison tests and the statistical significance taken at the level of P<0.05 as significant and p<0.01 as very significant
Inhibition of Albumin Denaturation: In this assay, the denaturation of egg albumin is induced by treatment of heat, and the denatured protein expresses antigen associated with type III hypersensitive reaction 12. From the results, concentration-dependent inhibition of protein albumin denaturation by the extracts and the standard drug Aspirin throughout the concentration range (200, 400, 600, 800, 1000 µg/ml) were highly prominent 8. Maximum inhibition of 75.59% for MEPC and 83.82% for Aspirin standard drug was observed at the concentration of 1000 µg/ml.
TABLE 3: EFFECT OF MEPC AND ASPIRIN ON ITS ANTI-DENATURATION ACTIVITY BY INHIBITING THE DENATURATION OF EGG ALBUMIN
| Concentration (µ/ml) | Aspirin | MEPC | ||
| Mean ±SEM
(Absorbance 660 nm) |
%Inhibition | Mean ± SEM
(Absorbance 660 nm) |
% Inhibition | |
| 200 | 0.31±0.02** | 43.01 | 0.42±0.02** | 21.5 |
| 400 | 0.22±0.02** | 59.55 | 0.33±0.02** | 38.23 |
| 600 | 0.14±0.01** | 73.89 | 0.22±0.01** | 58.63 |
| 800 | 0.12±0.01** | 76.28 | 0.14±0.01** | 73.34 |
| 1000 | 0.08±0.02** | 83.82 | 0.11±0.01** | 75.59 |
*All the results presented here are Mean±SEM, compared with the standard by one-way ANOVA, Dunnet’s multiple comparison tests and the statistical significance taken at the level of p<0.01 as very significant
HRBC Membrane Stabilization Method: As shown in Table 9, MEPC has shown significant anti-inflammatory activity when compared to the standard diclofenac sodium. The percentage of stabilization of MEPC and standard diclofenac at a concentration of 1000 µg/ml was found to be 83.35% and 90.62%, respectively. The activity of the extracts increased with the increase in the concentration, which indicates the activity of the extract was concentration-dependent.
TABLE 4: EFFECT OF MEPC AND ASPIRIN ON ITS ANTI-DENATURATION ACTIVITY BY HRBS MEMBRANE STABILIZATION
| Concentration (µ/ml) | Diclofenac | MEPC | ||
| Mean ± SEM
(Absorbance 560 nm) |
%Inhibition | Mean ± SEM
(Absorbance 560 nm) |
%Inhibition | |
| 200 | 0.11±0.028* | 42.7 | 0.15±0.01ns | 20.31 |
| 400 | 0.07±0.010** | 60.93 | 0.10±0.02* | 45.31 |
| 600 | 0.04±0.01** | 78.12 | 0.08±0.02** | 58.33 |
| 800 | 0.03±0.01** | 80.12 | 0.05±0.01** | 71.35 |
| 1000 | 0.01±0.01** | 90.62 | 0.03±0.01** | 83.85 |
*All the results presented here are Mean±SEM, compared with the standard by one-way ANOVA, Dunnet’s multiple comparison tests and the statistical significance taken at the level of *p<0.05 as significant and **p<0.01 as very significant.
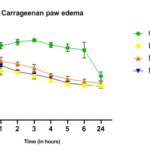
FIG. 3: EFFECT OF MEPC AND INDOMETHACIN ON CARRAGEENAN INDUCED PAW EDEMA. THE INFLAMMATION WAS PRODUCED BY SUBAPONEUROTIC. THE ANIMALS WERETREATED DAILY WITH THE EXTRACTS (P.O) OR INDOMETHACIN (I.P). Each point represents the mean ± S.E.M. of change of paw size. Both extracts and Indomethacin were effective compared to control (normal saline), by one-way ANOVA, Dunnet’s multiple comparison tests
In-vivo Anti-inflammatory Activity:
Carrageenan-Induced Rat Paw Edema (Acute Model of Inflammation): The results of the present finding shown in Table 10 revealed that the MEPC (200 & 400 mg/kg) significantly inhibited the carrageenan-induced edema, which is comparable with the standard drug Indomethacin (10 mg/kg). The anti-inflammatory activity of the extract was found to be dose-dependent.
Cotton Pellet- Induced Granuloma in Rats (Chronic Model of Inflammation): Based on the results shown in Table 11, the MEPC at the dose of 200 mg/kg and 400 mg/kg revealed a significant anti-inflammatory activity by the reduction of wet and dry weight granuloma when compared with the standard drug Indomethacin (10 mg/kg).
TABLE 5: EFFECT OF MEPC AND INDOMETHACIN ON ITS ANTI-INFLAMMATORY ACTIVITY AGAINST COTTON PELLET-INDUCED GRANULOMA
| Groups | Treatment | Doses (mg/Kg) | Wet weight of cotton pellets (Mean ± SEM) | Dry weight of cotton pellets
(Mean ± SEM) |
% Inhibition |
| 1 | Control
(1%carrageenan + Normal saline) |
-------- |
63.97±1.97 |
25.75±0.85 |
------- |
| 2 | MEPC | 200 | 56.1±1.08* | 19.75±0.75** | 23.3 |
| 3 | MEPC | 400 | 43.5±1.84** | 14.75±0.47** | 42.75 |
| 4 | Indomethacin | 10 | 42.75±1.79** | 13.75±1.03** | 46.6 |
DISCUSSION: In the present study, the methanol extract of the plant Pseudodrynaria coronans (MEPC)is analyzed for its analgesic and anti-inflammatory activities. Hot plate method and tail emersion methods were employed for the analgesic activity of MEPC. Fig. 1 revealed that oral administration of the MEPC at the doses of 200 and 400 mg/kg, b.w., produced significant increases in the latency time which can be compared to that of the standard drug (morphine, 5 mg/kg., b.w., p.o). Hot plate method produces two measurable behavioral components in response to thermal pain (i.e., paw licking and jumping in rats which are considered supraspinally integrated) concerning their reaction times. Administration of Morphine significantly increases the response time of the rats to reach 8.34 secs (at 90 min). Its analgesic effect decreased with time but remained significant even after 1 h, 30 min for both the extracts. In comparison, 400mg/kg is more effective as compared to the 200 mg/kg extract, and the results were found to be highly statistically significant (P<0.05, P<0.01, respectively).
Similarly, the results of the Tail immersion method presented in Fig. 2 showed that oral administration of the methanolic extracts at the doses of 200 and 400 mg/kg, b.w., produced significant increases in the reaction time which can be compared to that of the standard drug (morphine 5 mg/kg, b.w., p.o). Analgesic drugs which are centrally acting elevate the pain threshold of animals towards heat and pressure. Morphine administration significantly increased the response time of the animal to reach 6.994 seconds (at 60 min). Its analgesic effect decreased with time but remained significant even after an hour. All doses of the extracts showed significant increases in the latency time of rats when compared to the control group.
Protein denaturation is a well-documented cause of inflammation and arthritic diseases. In certain arthritic diseases, the production of auto-antigens may be due to the denaturation of proteins (Sangita, 2012). The methanolic extract has shown 60.36% and 82.02% anti-denaturation activity on BSA at a concentration of 800 µg/ml and 1000 µg/ml respectively. This result indicates that the anti-denaturation action of MEPC increased with the increased concentration. The standard Aspirin had shown 87.0% of anti-denaturation effect at 1000 µg/ml.
The denaturation of egg albumin is induced by treatment of heat, and the denatured protein expresses antigen associated with type III hypersensitive reaction 12. The conventional NSAIDs like phenylbutazone and Indomethacin were already proved that they not only act in the inhibition of endogenous prostaglandins production by blocking COX enzyme but also by prevention of denaturation of proteins 12.
Maximum inhibition of 75.59% for MEPC and 83.82% for Aspirin standard drug was observed at the concentration of 1000 µg/ml. Since the erythrocyte membrane is similar to the lysosomal membrane and its stabilization suggests that the extract may also stabilize the lysosomal membrane, the HRBC method has been employed for the evaluation of in-vitro anti-inflammatory activity 13. The percentage of stabilization of MEPC and standard diclofenac at a concentration of 1000 µg/ml was found to be 83.35% and 90.62%, respectively.
The results of the present finding shown in Fig. 3 revealed that the MEPC (200 & 400 mg/kg) significantly inhibited the carrageenan-induced edema, which is comparable with the standard drug Indomethacin (10 mg/kg). The anti-inflammatory activity of the extract was found to be dose-dependent. Through this result, it indicates that the extract was likely to act by hindering the release and/or action of prostaglandin 14 and 15.
Cotton pellet-induced granuloma represents a chronic inflammation model that is used extensively to assess the proliferative components of the inflammation by the development of granuloma in rodents 28 13. Based on the results of this study shown in Table 5, the MEPC at the dose of 200 mg/kg and 400 mg/kg revealed a significant anti-inflammatory activity by the reduction of wet and dry weight granuloma, which is found to be comparable with the standard drug Indomethacin (10 mg/kg).
It was reported that from the phytochemical screening, flavonoids, tannins, and saponins possessed analgesic and anti-inflammatory activities. Therefore, these phytochemical constituents found to be present in the methanolic extract of Pseudodrynaria coronans may be responsible for the observed analgesic effects and anti-inflammatory activity 1.
Based on the present investigation, it can be concluded that the methanolic rhizomes extract of Pseudodrynaria coronans possessed significant anti-nociceptive and anti-inflammatory activity, which may be due to the presence of various phytochemicals like flavonoids, triterpenoids, tannins, saponin, amino acids, carbohydrates, and steroids. These studies also support the traditional use of this plant in various ailments and also proved to be non-toxic through the findings of the Acute oral toxicity test. Further analysis is however required to isolate the phytochemical constituents responsible for the observed effects, which may be important for the discovery of lead compounds that can be used as an alternative for the new phase treatment of pain and inflammation.
ACKNOWLEDGEMENT: The authors are grateful to the Director, RIPANS, for providing all the support and help needed for the completion of this experiment.
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Atiku I, Sule MI, Pateh UU, Musa AM, Ya’u J, Sani YM, Hanwa UA, Abdullahi SM, Adamu SA, Lawal EA and Abdulrahman H: Phytochemical, anti-inflammatory and analgesic studies of the crude ethanolic leaf extract of Globimetularbrounii Van Tieghem (Family: Loranthaceae). Natural Products Chemistry & Research 2015; 54: 275-87.
- Sasidharan S, Chen Y, Saravanan D, Sundram KM and Globimetularbrounii Latha LY: Extraction, isolation and characterization of bioactive compounds from plants’ extracts. African Journal of Traditional, Complementary and Alternative Medicines 2011; 8(1): 1-10.
- Malairajan P, Gopalakrishnan G, Narasimhan S and Veni KJ: Analgesic activity of some Indian medicinal plants. Journal of Ethnopharmacology 2006; 106(3): 425-8.
- Prabha PM, Kamalakkannan V, Kumaran KS and Sambathkumar R: Antioxidant and hepatoprotective activities of ethanolic root extract of Bauhinia variegata Journal of Pharmacognosy and Phytochemistry 2014; 3(3): 92-8.
- Tai Z, Zhang F, Cai L, Shi J, Cao Q and Ding Z: Flavonol glycosides of Pseudodrynaria coronans and their antioxidant activity. Chemistry of Natural Compounds. 2012; 48(2): 221-4.
- Hijazi MA, El-Mallah A, Aboul-Ela M and Ellakany A: Evaluation of analgesic activity of Papaver libanoticum extract in mice: involvement of opioids receptors. Evidence-Based Complementary and Alternative Medicine 2017; 2017.
- Williams LA, O'Connar A, Latore L, Dennis O, Ringer S, Whittaker JA, Conrad J, Vogler B, Rosner H and Kraus W: The in-vitro anti-denaturation effects induced by natural products and non-steroidal compounds in heat treated (immunogenic) bovine serum albumin is proposed as a screening assay for the detection of anti-inflammatory compounds, without the use of animals, in the early stages of the drug discovery process. West Indian Medical Journal 2008; 57(4): 327-31.
- Chandra S, Dey P and Bhattacharya S: Preliminary in-vitro assessment of anti-inflammatory property of Mikania scandens flower extract. J Adv Pharm Edu Res 2012; 2(1): 25-31.
- Saleem TM, Azeem AK, Dilip C, Sankar C and Prasanth NV and Duraisami R: Anti–inflammatory activity of the leaf extacts of Gendarussa vulgaris Asian Pacific Journal of Tropical Biomedicine 2011; 1(2): 147-9.
- Winter CA, Risley EA and Nuss GW: Carrageenin-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Proceedings of the Society for Experimental Biology and Medicine 1962; 111(3): 544-7.
- Bhatt KR, Mehta RK and Shrivastava PN: A simple method for recording anti-inflammatory effects on rat paw oedema. Indian Journal of Physiology and Pharmacology. 1977; 21(4): 399-400.
- Ahmad F, Khan RA and Rasheed S: Study of analgesic and anti-inflammatory activity from plant extracts of Lactucascariola and Artemisia absinthium. Journal of Islamic Academy of Sciences 1992; 5(2): 111-4.
- Ullah HA, Zaman S, Juhara F, Akter L, Tareq SM, Masum EH and Bhattacharjee R: Evaluation of antinociceptive, in-vivo & in-vitro anti-inflammatory activity of ethanolic extract of Curcuma zedoaria BMC Comple and Alternative Medicine 2014; 14(1): 1-2.
- Shenoy S, Shwetha K, Prabhu K, Maradi R, Bairy KL and Shanbhag T: Evaluation of anti-inflammatory activity of Tephrosia purpurea in rats. Asian Pacific Journal of Tropical Medicine 2010; 3(3): 193-5.
- Guo D, Xu L, Cao X, Guo Y, Ye Y, Chan CO and Chen S: Anti-inflammatory activities and mechanisms of action of the petroleum ether fraction of Rosa multiflora hips. Journal of Ethnopharmacology 2011; 138(3): 717-22.
- Rock EM, Limebeer CL and Parker LA: Effect of cannabidiolic acid and∆ 9-tetrahydrocannabinol on carrageenan-induced hyperalgesia and edema in a rodent model of inflammatory pain. Psychopharmacology 2018; 235(11): 3259-71.
- Mondal A, Maity TK and Bishayee A: Analgesic and Anti-Inflammatory Activities of Quercetin-3-methoxy-4′-glucosyl-7-glucoside Isolated from Indian Medicinal Plant Melothria heterophylla. Medicines. 2019; 6(2): 59.
- Mishra D, Ghosh GO, Kumar PS and Panda PK: An experimental study of analgesic activity of selective COX-2 inhibitor with conventional NSAIDs. Asian Journal of Pharmaceutical and Clinical Research 2011; 4(1): 78-81.
- Leelaprakash G and Dass SM: In-vitro anti-inflammatory activity of methanol extract of Enicostemma axillare. International Journal of Drug Development and Research. 2011; 3(3): 189-96.
- CK K, Purohit AP and Gokhale SB: Pharmacognosy, 11th edition, Nirali Prakashan 1999; 78-83.
- Thanzami K, Pachuau VL, Lalremruati C and Kakoti BB: Phytochemical screening, anti-oxidant and anti-inflammatory activities of ethyl acetate extract of Combretum punctatum squamosum. Indian Journal of Natural Products and Resources (IJNPR)[Formerly Natural Product Radiance (NPR)] 2020; 11(1): 38-45.
- D'arcy PF, Howard EM, Muggleton PW and Townsend SB: The anti‐inflammatory action of griseofulvin in experimental animals. Journal of Pharmacy and Pharmacology 1960; 12(1): 659-65.
- Patil KR and Patil CR: Anti-inflammatory activity of bartogenic acid containing fraction of fruits of Barringtonia racemosa in acute and chronic animal models of inflammation. Journal of Traditional and Complementary Medicine 2017; 7(1): 86-93.
- Subha D and Geetha N: Evaluation of acute toxicity of the methanolic extract of Tanacetum parthenium in albino wistar rats. J Sci Innov Res 2017; 6(3): 113-5.
- WOOLFE G and MacDonald AD: The evaluation of the analgesic action of pethidine hydrochloride (Demerol). Journal of Pharmacology and Experimental Therapeutics. 1944; 80(3): 300-7.
- Toma W, Gracioso JD, Hiruma-Lima CA, Andrade FD, Vilegas W and Brito AS: Evaluation of the analgesic and antiedematogenic activities of Quassia amara bark extract. Journal of Ethnopharmacology 2003; 85(1): 19-23.
How to cite this article:
Elu K, Roy PK, Lapasam P, Lalnunfela C, Lalhlenmawia H, Umbon Y and Lalhriatpuii TC: Phytochemical analysis and in-vivo screening of Pseudodrynaria coronans (wall. ex. mett.) ching on its analgesic and anti-inflammatory properties. Int J Pharmacognosy 2021; 8(5): 196-04. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(5).196-04.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
196-204
761
1305
English
IJP
K. Elu, P. K. Roy, P. Lapasam, C. Lalnunfela, H. Lalhlenmawia, Y. Umbon and T. C. Lalhriatpuii *
Department of Pharmacy, Regional Institute of Paramedical & Nursing Sciences, Zemabawk, Aizawl, Mizoram, India.
tclalhriatpuii@gmail.com
20 April 2021
24 May 2021
26 May 2021
10.13040/IJPSR.0975-8232.IJP.8(5).196-04
31 May 2021