PHYTOCHEMICAL ANALYSIS AND BIOLOGICAL ACTIVITIES OF STEM BARK EXTRACT OF JATROPHA CURCAS LINN.
HTML Full TextPHYTOCHEMICAL ANALYSIS AND BIOLOGICAL ACTIVITIES OF STEM BARK EXTRACT OF JATROPHA CURCAS LINN.
Naqab Khan * 1, Abdul Haleem Shah 2, Ejaz Ahmad Khan 3, Muhammad Sadiq 3, Samiullah Khan 1 and Natasha Baloch 4
Gomal Center of Biochemistry and Biotechnology 1, Department of Biological Sciences 2, Faculty of Agriculture 3, Gomal University, Dera Ismail Khan, Khyber Pakhtunkhwa, Pakistan.
Department of Basic Medical Sciences 4, Khyber Medical University, Peshawar, KPK, Pakistan.
ABSTRACT: Objective: The study was designed to explore the antibacterial, antifungal and antioxidant potential of Jatropha curcas Linn. (J. curcas) stem bark extract. Materials and Methods: The chemotherapeutic action was measured in the crude extract using agar well diffusion method. The antibacterial and antifungal activities of different extracts were tested against two gram-positive and nine gram negative human pathogenic bacteria using agar well diffusion method. Area of the reticence of crude extracts was matched with that of various antibiotics like ampicillin, streptomycin for antibacterial property and fluconazole for antifungal activity. The antioxidant and free radical scavenging activities of different extracts of the Jatropha curcas were also investigated against 2,2 Diphenyl –1- picrylhydrazyl (DPPH), 2,2, Azion-bis- (3- ethylbenzothia-zoline-96- sulphonic acid) (ABTS), superoxide anion (O-2) and nitric oxide (NO) using spectroscopic absorption methods. The data were statistically analyzed by ANOVA, arranged in completely randomized design (CRD) in the factorial arrangement having four replications. Results: The average inhibition zone for methanol extract (25.31mm) was establish to be extra valuable than the average inhibition zone of ethanol extract (19.72 mm).The average least inhibitory quantity for the methanol and ethanolic extracts were 4.31 and 5.09 mgml-1 respectively. The average minimum bactericidal concentration for the methanol and ethanol extracts were 8.27 and 9.81 mgml-1 respectively. The average inhibition zone for methanol extract (16.60 mm) was observed to be most active followed by the average inhibition zone of ethanol extract (15.15 mm). Among the tested free radical activities, the maximum percent DPPH scavenging action was shown by the methanolic extract of J. curcas stem bark extract (90.5%). Quantitative estimation of secondary metabolites showed that tannins were most abundant (24.1 ± 0.1) followed by steroids (19.7 ± 0.1). Conclusion: It is concluded that the significant inhibition of the bacterial growth was observed against the tested pathogens. The phytochemical analysis of the plant showed the presence of phenolic compounds, which may have contributed to the antioxidant activity of J. curcas stem bark extract. Therefore, such plants can be used for isolation of biologically active natural products that may lead to the improvement of new pharmacological research accomplishment.
| Keywords: |
Jatropha curcas, Crude extract, Antibacterial activity, Antifungal activity, Antioxidant activity, Phytochemical analysis
INTRODUCTION: Jatropha curcas (J. curcas) is an ornamental plant belongs to family Euphorbiaceae, is a drought-resistant perennial, growing well in marginal areas as well as in poor soil.
It is easily established, grows comparatively quickly and survives in varied habitat. Jatropha the miracle plant provides seeds, for 45 years which are oily contain 37% oil. In crude from the oil can be combusted as a fuel. It burns with clear smoke, free flame, tested accurately as fuel for running a diesel engine. The residue of the plant acts as an excellent chemical fertilizer; the oil can also repel and kill the insects. It is successfully cultivated and grown in many parts of the Khyber Pakhtunkhwa like Mardan, Kohot and Dera Ismail Khan, Pakistan.
Plants provide a huge range of biologically active compounds; therefore plants are the main source of various medicines. A lot of drugs nowadays are extractable directly from natural sources or by some little modification in the process of extraction 1. The use of various plant crude extracts and phytochemical compounds, both with understood antibacterial activities, can be of significant importance in diseases management. The majority of the community in many countries uses these old-style drugs which are directly derived from herbal resources 2. About 75% of the peoples from developed countries use traditional medicine, which has bio-active molecule extracted from herbal plants. In the previous few years, a lot of work has been performing in various countries to demonstrate such potency. A large number of plants have been studied due to their antibiotics activities, which are mainly produced during subordinate plant breakdown. That way, such types of plants should be exposed to much better understand their activities 3. Antimicrobial compounds are found in various parts of the medicinal plant like in whole plants, stems, leaves, barks, roots, flowers and fruits 4.
According to one of the reports of world health organization, among natural sources such medicinal shrubberies should be studied to obtain complete pharmacological knowledge about their activities, method of isolation, method of preparation, uses, pharmacokinetics, pharmacodynamics and bioavailability date, etc. 5 Practice of herbal medicine treatment were used thousands of year ago especially in Africa, and herbal medicine were considered as the basic treatment for their primary health care disorders nowadays also the herbal medicine treatment is continued in African countries and about 75% of the people depends upon these plants extract for their primary health problems. 6 In tropical countries among the health problems about 50% of the death occurs due to chronic infections 7. Preferably, antimicrobial agents disrupt microbial processes or structures that differ from those of the host. They may damage pathogens by hampering cell wall synthesis, inhibiting microbial protein and nucleic acid synthesis, disrupting microbial membrane structure and function, or blocking metabolic pathways through inhibition of key enzymes. Numerous works have established the chemotherapeutic potency of various Jatropha species, though there is inadequate evidence concerning the antibacterial properties of J. curcas Linn. So, on restricted figures are existing on the therapeutic activities of J. curcas stem bark while frequently on the leaf latex of the plant.
Antioxidants are those agents which can protect the human body from free radicals and reactive oxygen species (ROS) effect 8. Antioxidants are those plants secondary metabolites which slow down the progress of many life-threatening diseases as well as lipid peroxidation. Now- days the most abundantly use synthetic antioxidant agents are butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), tert-butylhydroquinone and propyl gallate, but due to having the toxic and carcinogenic effect the use of these antioxidants are restricted nowadays 9. Methanolic extracts of J. curcas stem bark possess various secondary metabolites which act as an antimicrobial and antioxidants, the polyphenolic substance especially the flavonoids play a vital role as an antioxidant agent, scavenging free radical, inhibition of lipid peroxidation 10. Hence, having a role in protecting food, cells, and organs from oxidative damage 11.
In the current study, various extracts of stem bark of J. curcas were used as an antimicrobial agent against different pathogenic microorganisms. Along with the broad spectrum antibacterial, anti-fungal, and antioxidant activities, the plant is also used for the various diseases like piles, tumor, paralysis, snakebite, and dropsy, etc. Keeping in view all the facts mentioned above, the present research study has been designed to assess the different biological activities and phytochemical analysis of crude methanolic, ethanolic and aqueous extract of crushed stem bark of J. curcas Linn.
MATERIALS AND METHODS:
Collection of Plant Material: The fresh plant material of the main stem bark of J. curcas was obtained from Jatropha Orchard laid out in Agronomic Research area by Department of Agronomy Faculty of Agriculture Gomal University Dera Ismail Khan in the month January and was identified by taxonomist of Pharmacognosy Department, Faculty of Pharmacy, Gomal University, Dera Ismail Khan.
Preparation of Extract: The selected plant was thoroughly washed and cleaned with distilled water and then was dried to constant weight for 30 days under shade to prevent decomposition of chemical ingredients of the plant. The dried plant material was ground into a fine powder by the electrical grinder. Accurately 300 g of the selected plant material was dissolved in ethanol and methanol separately with 1:16 respectively. Another 300 g of the selected plant’s fine powder was dissolved in water in the same ratio for 6 days with spontaneous shaking in shaking incubator. 12 The organic solvents used were of analytical grade. The dissolved extracts were first filtered from Muslin cloth and then were sieved through Whitman's No. 1 filter paper. After the process of filtration, the extracts were subjected for drying, by rotary evaporator. The evaporation process was performed at 40 0C to avoid decomposition of chemical ingredients present in the crude extract. The water extract was freeze-dried to get dry powder form.
Bacterial Strains: Pathogenic strains of two gram-positive bacteria, nine gram-negative bacteria, and ten fungi were obtained from American Type Culture Collection (ATCC) Sigma chemical Lahore, and preserved at the Department of Biological sciences Gomal University DI Khan KPK Pakistan. The fresh bacterial broth cultures were prepared before the screening procedure. gram-positive bacteria include Staphylococcus epidermidis ATCC 25021 and Staphylococcus aureus 25323. gram negative bacteria are Klebsiella pneumonia ATCC 25945, Salmonella typhi ATCC 25820, Escherichia coli ATCC 25922, Aeromonos ATCC 12045, Vibrio cholerae ATCC 12546, Pseudomonas aeruginosa 25356, Shigella ATCC 12022, Corynae diptherie ATCC 25465 and Streptococus penemoniae 12075. Fungal strains included Candida albicans ATCC 90028, Candida glaberata ATCC 6258, Fusarium solani ATCC 750, Microsporium canis ATCC 22019, Aspergillus flavus ATCC 21007, Penicillium notatum ATCC 851, Aspergillus niger ATCC 6200, Trychophytons longifolia ATCC 90118, Conidobokus coronatus ATCC 22001, and Rhodotorula mucilagnosa ATCC 91005.
Antibacterial Susceptibility Tests:
Bacterial Zone of Inhibition (mm): The selected microbes were grown in nutrient broth media for 18 h. The fresh broth was stocked in shaking incubator. The turbidity of the broth was standardized to 0.5 McFarland standards (106 cfu ml-1). The nutrient agar media was prepared in the flask. The media was poured into sterile Petri dishes in the aseptic condition in a laminar flow hood. The fresh broth of the respective bacteria was applied by the streaking method 13.
The Petri dishes were kept in a laboratory oven at 37 °C for 24 h. After 24 h, the Petri dishes were examined for bacterial growth. After formation of colonies, the powdered crude extracts were reconstituted in dimethyl sulphoxide (DMSO) which is a neutral solvent, wells were formed with a sterilized 8 mm borer, and the bores were occupied with 100 µl of crude extract with the help of micropipette at 10 mgml-1 were introduced into the wells. The plates were allowed to stand on the laboratory bench for 60 min to allow the adequate diffusion of the extract into the media. After 60 min the plates were kept in an oven at 37 °C for 24 h for the incubation process. After incubation, the plates were observed and measured with the help of venire caliper in mm. The effectiveness of the extracts on bacterial species was compared with Ampicillin and Streptomycin at a quantity of 10 mgml-1.
Determination of Minimum Inhibitory Concentration (MIC) (mg ml-1): The assessment of the lowest inhibitory amount of the various crude extract of Jatropha stem bark was determined by the protocol of 14 serial dilutions of the extract were formulated in (DMSO). From these serial dilutions of different concentration, 2 ml of sample was supplemented to 18 ml of autoclaved liquid nutrient agar media for each bacterial strains at 40 °C. Then these Petri dishes having media as well as crude extracts were placed in a laminar flow to dryness. After 30 min of drying the process of streaking was started, the 24 h old bacterial broth was applied to the Petri dishes by streaking method. The uniform Lon was prepared in Petri dishes with respective bacterial strains; they were allowed for incubation at 37% for 24 h for bacterial strains. After the incubation period, the Petri dishes were observed for the occurrence or absence of bacterial growth. The MIC was considered as the lowermost quantity that retarded the growth of the test micro-organisms.
Determination of Minimum Bactericidal Concentration (MBC) (mg ml-1): The lowest bactericidal amount of the crude extract of stem bark extract of Jatropha was calculated by process 15, with little amendments. Those Petri dishes having no visible growth were taken from the MIC test and were subculture again on newly prepared nutria agar plates for bacterial strains, and then were incubated in an oven at 37 % for 48 h for bacteria strains. The MBC was considered as the quantity of the crude solution that did not show any growth of the test micro-organism on a new set of Petri dishes.
Fungal Zone of Inhibition (mm): The ATCC strains were first grown on a special type of nutrient media called. Sabouraud dextrose agar (SDA) media. After growing the fungi, the fungal spores were formed. The spores were obtained by pouring a mixture of distilled water and glycerol on the surface of the Petri dishes having spores; the spores were removed by a sterile glass rod. The spores were standardized to OD 600 nm of 0.1 before using. The standardized fungal spores suspension was used as a fungal broth and was used for the determination of zone of inhibition.
The fungal suspension was uniformly spread on the Petri dishes having media; wells were formed with a 6 mm cork borer and allowed for incubation, were kept in an oven at a temperature of 25 °C for 72 h. After 72 h the zone of inhibition was measured with the help of venire caliper and compared with a control that was fluconazole.
Anti-Oxidant Activity:
Diphenyl-1 Picrylhydrazyl (DPPH) Radical Scavenging Assay: The method 16 was adopted for the investigation of scavenging capability of DPPH free radicals in the various extract of J. curcas. First of all the solution of 0.125mM DPPH in methanol was prepared, and from this solution, 1.0 ml was vigorously mixed with 1 mg of the extract in methanol having 0.2-1.0mg/ml was completely vortex and then was kept in the dark for 60 min. After that the mixture was subjected for absorbance by double beam spectrophotometer, the spectro-photometer was rennet at 517 mm wavelength. The BHT was compared as a standard. The scavenging capacity of the plant extract was determined by the equation.
DPPH scavenging activity (%) = [(Abs of control - Abs of sample)] / (Abs of control)] × 100
Whereas, Abs of control is the absorbance of DPPH + methanol. Abs sample is the absorbance of DPPH radical + sample extract or standard.
2, 2-Azino- bis - (3 – ethylbenzothiazoline - 6 sulphonic acids) (ABTC) Radical Scavenging Assay: The ABTS free radical scavenging activity was determined according to 17 to by mixing 7mM of ABTS with 2.4 mM of potassium persulphate solution in equal volume. In this way, these two stock solution were mixed and allowed to react and kept for 12 h in the dark room. Then this solution was diluted by mixing 1ml of ABTS solution along with 60 ml of methanol and then the absorbance was measured at 734 nm. After that 1 ml of the plant extracts were mixed with 1ml of ABTS solution and the absorbance was measured at the same wavelength by using double beam spectro-photometer, the ABTS scavenging ability of the plant extract was compared with that of standard BHT and determined by the following equation.
ABTS scavenging activity % = [(Abs of control – Abs of sample)/Abs of control)] × 100
Whereas, Abs of control was the absorbance of ABTS radical + methanol and Abs sample is the absorbance of ABTS radical + sample extract or BHT.
Superoxide Anion Scavenging Capacity: The antioxidant capacity of the methanolic, ethanolic and aqueous extracts of Jatrupha curcas were studied by the method Liyana-Pathiranan 18 using superoxide anion as free radicals. In this method one milliliter of nitroblue tetrazolium (NBT) solution (156 mM) in 100 mM phosphate buffer, pH 7.4), 1 ml NADH solution (46 mM in 100 mM phosphate buffer (pH 7.4) and 100 µl of the plant extract sample solution in distilled water and BHT were also mixed separately at various concentration 0.2 -1.0 mg/ml.
To this solution 100 µL of the phenazine methosulphate (PMS) solution was also added, the reaction mixture was incubated at 25 °C for 10 min, and then the absorbance was measured at 560 nm. The percentage inhibition of superoxide anion was determined by using the following formula.
% Inhibition superoxide anion scavenging ability = [(Ao-A1) / Ao] × 100
Whereas, Ao is the absorbance of control and A is the absorbance of extract or standard.
Nitric Oxide Scavenging Assay: The activity of J. curcas as an antioxidant was investigated by the method. 19 In this method nitric oxide was used as free radicals, 2 ml of sodium nitroprusside was prepared in 0.5 ml phosphate buffer saline (pH 7.4) and was mixed with 0.5 ml of plant extracts, BHT at a concentration of 0.2-1 mg/ml. This mixture was incubated at 25 °C for 120 min. A sample of 0.5 ml of this solution was added to 0.5 ml of Griess reagents [(1 ml of sulfanilic acid reagent (0. 33% prepared in 20% glacial acetic acid at room temp for 5 min with 1 ml of napthylenediamine chloride ( 0.1% w/v)]. The absorbance of the mixture was measured at 540 nm by double beam spectrophotometer- with the help of the following formula.
% inhibition of Nitric Oxide Radical Scavenging [Ao –A1] / Ao × 100
Whereas, Ao = absorbance of control and A1 = absorbance of extract or standard.
Qualitative Phytochemical Analysis of Jatropha Extract: The following standard procedures were adopted for qualitative analysis of stem bark extract of Jatropha curcas 12.
Testing for Saponins: In a clean autoclaved test tube 10ml of sterile water was taken. Then 0.5 g of the methanolic extract was supplemented to the test tube containing distilled water. The test tube was stirred vigorously; foaming was formed in the test tube which was the indication of the presence of saponins in the methanolic extract of the J. curcas.
Testing for Tannins and Phenolic: A clean, the sterilized test tube was taken, 0.5 g of methanolic extract was supplemented to the test tube, 12 ml of sterile water was also supplemented to the test tube.
It was stirred and then filtered. The filtrate was transferred to another test tube. Some droplets of 6% FeCl3 was supplemented to the filtrate, blue-black or blue-green coloration or aggregation indicated the occurrence of phenolic and tannins in the methanolic extract of J. curcas.
Testing for Steroids: A cleaned and dried test tube was taken, 0.5 g of the methanolic extract was supplemented along with 4 drops of acetic anhydride and a droplet of strong sulphuric acid (H2SO4) was also supplemented. The combination was allowed to stand for 1 h and neutralized with sodium hydroxide (NaOH). Along with the tallying of a blue-green color indicated the presence of steroid in the methanolic extract.
Testing for Glycosides: A cleaned and dried test tube was taken, 0.5 g of methanolic extract was supplemented to the test tube along with 3 ml of chloroform. The extract softened in chloroform. Tetraoxosalphate VI acid was carefully supplemented to the test tube. A reddish brown color at the border showed the existence of a steroidal molecule, which is a glycine portion of the cardiac glycosides.
Test for the Phenolic Compounds:
Flavonoids: In a cleaned and dried test tube 5 ml of methanolic extract was supplemented along with a concentrated sulphuric acid (H2SO4) (1 ml) and 0.5 g of Magnesium (Mg). A pink or red coloration that disappears on standing for 3 mints showed the presence of flavonoids.
Test for Alkaloids: In a 100 ml cleaned flask 20 ml methanolic solution was supplemented, the solvent was evaporated by heating the beaker. The dried residue obtained was dissolved in 5 ml of HCl (2N) and filtered. A few drops of Meyers reagent was supplemented, the presence of precipitate indicated the alkaloids.
Test for Phlobatannins: In a cleaned test tube 3 ml of methanolic extract was supplemented along with 3 ml of 2% hydrochloric acid and the combination was allowed to bioleach. The appearance of a red aggregation was taken as evidence for the occurrence of phlobatannins in the methanolic extract.
Tests for Anthraquinones: For testing anthraquinones, a specific test is used called borntragers test. Three ml of methanolic extract was shaken and filtered, and 6 ml of 12% ammonia extract was also supplemented to the filtrate. The combination was allowed to stand, and the presence of a pink, color in the ammonical segment showed the existence of anthraquinones.
Test for Terpenoids: In a cleaned test tube 3 ml of the methanolic extract was dissolved in 3 ml of chloroform and vaporized to dryness. Three ml of concentrated H2SO4 was than supplemented and heated for about 3 minutes; grayish color showed the existence of terpenoids.
Quantitative Phytochemical Analysis of Jatropha Extract: The quantitative photochemical analysis was performed at Nuclear Institute of Agriculture and Biology (NIAB) by using High-performance Liquid Chromatography (HPLC) and spectrophotometry by little modification of the method. 12
RESULTS:
Antibacterial Susceptibility Tests:
Zone of Inhibition in Bacteria (mm): Zone of inhibition of different bacteria as affected by stem bark extracts of J. curcas are presented in Table 1. It is indicated that methanolic, ethanolic and aqueous extracts of J. curcas affected the zone of inhibition of the test bacteria significantly and differently. The antimicrobial properties of the methanol and ethanol extract compared positively with that of broad-spectrum antibiotics, i.e., Streptomycin and Ampicillin. They seemed to be broad-ranging as its potency were autonomous on gram-positive as well as on gram-negative pathogenic bacteria species. The inhibition zone for bacterial test species ranged from 23.50 mm to 29.50 mm, being minimum (23.50) for Corynae diphtheria and maximum (29.50 mm) for Escherichia coli. The average inhibition zone for methanol extract (25.31 mm) was established to be extra valuable than the average inhibition zone of ethanol extract (19.72 mm). The mean aqueous extract showed low antibacterial activity with inhibition zone 7.77 mm ranging between 1-11 mm. The interaction between different extracts and test bacteria species ranged between 1-37.50 mm. The maximum zone of inhibition (37.50 mm) was recorded for Escherichia coli with methanol extract and the minimum (1.00 mm) for Klebsiella pneumonia with aqueous extract.
TABLE 1: ZONE OF INHIBITION (MM) OF DIFFERENT BACTERIA AS AFFECTED BY STEM BARK EXTRACT OF J. CURCAS
| Test
Bacteria |
Methanol (10 mgml-1) | Eethanol (10 mgml-1) | Aqueous
(10 mgml-1) |
ST
(10 mgml-1) |
AMP
(10 mgml-1) |
Mean |
| Klebsiella pnemoniae | 26.25 MN | 21.25 P-R | 1.00 Z | 42.25 B-F | 39.75F-I | 26.10 C |
| Salmonella typhi | 26.0 NO | 15.75 U | 7.75 W-Y | 42.50 B-E | 36.0 JK | 25.60 CD |
| Escherichia coli | 37.50 IJ | 17.75 S-U | 9.25 V-X | 43.75 A-C | 39.25 HI | 29.50 A |
| Aeromonos | 28.50 MN | 19.50 R-T | 8.75 V-Y | 42.0 C-G | 37.75 IJ | 27.30 B |
| Staphylococcus aureus | 26.75 MN | 19.75 Q-S | 8.25 V-Y | 45.75A | 40.00 E-I | 28.10 B |
| Vibrio cholerae | 21.50 P-R | 20.25 Q-S | 8.50 V-Y | 41.0 D-H | 35.50 JK | 25.35 CD |
| Pseudomonas aeruginosa | 28.75 M | 16.75 U | 6.25 Y | 39.50 G-I | 34.50 K | 25.15 CD |
| Shigella | 22.25 PQ | 26.50 MN | 11.0 V | 44.75 AB | 34.50 K | 28.00 B |
| Staphylococcus epidermidis | 23.0 P | 23.50 OP | 6.75 XY | 42.75 B-D | 35.50 JK | 26.00 C |
| Corynae diptherie | 17.0 TU | 16.25U | 8.25 W-Y | 41.5 C-H | 34.00 KL | 23.50 E |
| Streptococus penemoniae | 21.0 P-R | 19.75 Q-S | 9.75 VW | 42.0 C-G | 31.50 L | 24.80 D |
| Mean | 25.318 C | 19.727 D | 7.773 E | 42.523 A | 36.205 B | |
| LSD values | Extracts | 0.7591 | Test bacteria | 1.1259 | Interaction | 2.5176 |
Means followed by different alphabets are significant at 1% level of probability.
Zone of inhibition of Jatropha curcas stem bark extract against the selected human pathogenic gram positive and gram negative bacteria in methanolic, ethanolic and aqueous extracts. Positive control was two broad-spectrum antibiotics, ampicillin and streptomycin while negative control was DMSO, which was a neutral solvent. The maximum inhibitory effect was shown by methanolic extract against Escherichia coli.
Determination of Minimum Inhibitory Concentration (MIC mg ml-1): The minimum inhibitory concentration of different extracts of J. curcas stem bark is given in Table 2 which shows that methanolic, and ethanolic extract significantly affected the growth of various test bacteria, and the test bacteria also showed a significant variation against these extracts. The least inhibitory amount of various extracts of J. curcas for different bacteria species ranged between 1.91 and 5.00 mgml-1. The average least inhibitory quantity for the methanol and the ethanolic suspension was 4.31, and 5.09 mgml-1 respectively. Hence, it was observed that the methanolic suspension was more potent than the other extracts. The interaction between methanol extract and test pathogen extended between 2.50 and 6.00 mgml-1, while between ethanol and test bacteria was recorded from 3.00 mgml-1to 7.00 mgml-1. Both the extracts exhibited excellently and statistically the same antimicrobial activities (MIC) against E coli, and Pseudomonas aeroginosa.
TABLE 2: MINIMUM INHIBITORY CONCENTRATION (MIC mgml-1) REGIMES OF STEM BARK EXTRACTS OF J. CURCAS
| Test
Bacteria |
Methanol
(mgml-1) |
Ethanol
(mgml-1) |
Aqueous
(mgml-1) |
ST
(mgml-1) |
Mean |
| Klebsiella pnemoniae | 4.00 e | 5.00 cd | ND | 1.00 ij | 3.33 cd |
| Salmonella typhi | 5.50 bc | 5.50 bc | ND | 1.50hi | 4.16 b |
| Escherichia coli | 2.50 fg | 3.00 f | ND | 0.25 j | 1.91 f |
| Aeromonos | 4.00e | 5.00 cd | ND | 1.00 ij | 3.33 cd |
| Staphylococcus aureus | 4.50 be | 5.00 cd | ND | 1.50 hi | 3.66 bcd |
| Vibrio cholerae | 5.00 cd | 5.50 bc | ND | 0.50 j | 3.66 bcd |
| Pseudomonas aeruginosa | 3.00 f | 4.00 e | ND | 1.00 ij | 2.66 e |
| Shigella | 5.00 cd | 6.00 b | ND | 0.50 j | 3.83 bc |
| Staphylococcus epidermidis | 4.00 e | 5.00 cd | ND | 0.65 ij | 3.21 de |
| Corynae diptherie | 6.00 b | 7.00 a | ND | 2.00 gh | 5.00 a |
| Streptococus penemoniae | 4.00 e | 5.00 cd | ND | 1.50 hi | 3.50 cd |
| Mean | 4.31 b | 5.09 a | 1.03 c | ||
| LSD values | Extracts 0.297 | Test bacteria 0.569 | Interaction 0.986 |
Means followed by dissimilaralphabets are significant at P ≤ 0.05.
Minimum inhibitory concentration of Jatropha curcas stem bark extract in methanolic, ethanolic and aqueous extracts against different pathogenic bacteria. Antimicrobial activity of plant extracts was significantly compared with two broad-spectrum antibiotics, ampicillin and streptomycin. DMSO was used as a negative control which was a neutral solvent. Lowest inhibitory concentration was shown by methanolic extract against Escherichia coli; the aqueous extract was unable to inhibit microbial growth at such low concentration.
Determination of minimum Bactericidal Concentration (MBC mgml-1): The MBC of different extracts of J. curcas stem bark given in Table 3 shows that methanolic and ethanolic extract significantly affected the growth of various test bacteria, and the test bacteria also showed a significant variation against these extracts. The interaction between extracts and test bacteria were also found to be significant. It is revealed from the Table 3 that these extracts displayed a fluctuating level of antimicrobial properties (MBC) towards different bacterial strains.
TABLE 3: MINIMUM BACTERICIDAL CONCENTRATION (MBC mgml-1) REGIMES OF STEM BARK EXTRACTS OF J. CURCAS
| Test
Bacteria |
Methanol
(mg ml-1) |
Eethanol
(mg ml-1) |
Aqueous
(mg ml-1) |
ST
(mg ml-1) |
Mean |
| Klebsiella pnemoniae | 8.00 d | 10.00 bc | ND | 2.00 fg | 1.66 bcd |
| Salmonella typhi | 9.00 cd | 10.00 bc | ND | 2.00 fg | 7.00 bc |
| Escherichia coli | 5.00 e | 6.00 e | ND | 0.50 h | 3.83 f |
| Aeromonos | 8.00 d | 9.00 cd | ND | 1.50 gh | 6.16 d |
| Staphylococcus aureus | 9.00 cd | 11.00 b | ND | 2.00 fg | 7.33 b |
| Vibrio cholerae | 10.00 bc | 10.00 bc | ND | 1.00 gh | 7.00 bc |
| Pseudomonas aeruginosa | 6.00 e | 8.00 d | ND | 2.00 fg | 5.33 e |
| Shigella | 9.00 cd | 11.00 b | ND | 1.00 gh | 7.00 bc |
| Staphylococcus eepidermidis | 8.00 d | 10.00 bc | ND | 1.50 gh | 6.00 cd |
| Corynae diptherie | 11.00 b | 13.00 a | ND | 3.00 f | 9.00 a |
| Streptococus penemoniae | 8.00 d | 10.00 bc | ND | 2.00 fg | 6.66 bcd |
| Mean | 8.27 | 9.81 | ND | 1.68 | |
| LSD Values | Extracts 0.4119 | Test Bacteria 0.7888 | Interaction 1.3663 |
Means followed by dissimilar alphabets are significant at P ≤ 0.05.
Minimum bactericidal concentration of Jatropha curcas stem bark extract in methanolic, ethanolic and aqueous extracts against different pathogenic bacteria. Antimicrobial activity of plant extracts was significantly compared with two broad-spectrum antibiotics, ampicillin and streptomycin. DMSO was used as a negative control which was a neutral solvent. Lowest bactericidal concentration was shown by methanolic extract against Escherichia coli; the aqueous extract was unable to completely eradicate bacteria at such low concentration.
The lowest bacterial capacity of methanol and ethanol extracts was compared with standard antibiotic ampicillin and was seemed to be a broad spectrum as its properties were independent on both grams positive and gram-negative bacterial species. The least bactericidal capacity of various extracts of J. curcas for different bacterial species ranged between 1.66 and 9.00 mgml-1. The average minimum bactericidal concentration for the methanol and ethanol extract was 8.27, and 9.81 mgml-1 respectively. Hence, it was observed that the methanolic extract was most potent than the other extracts. The interaction between methanol extract and test bacteria ranged between 5.00 and 11.00 mgml-1, while between ethanol and test bacteria was recorded from 6.00 mgml-1 to 13.00 mgml-1.
Zone of Inhibition in Fungi (mm): The antifungal activity of the methanolic, ethanolic, and water solution of J. curcas were studied against ten different pathogenic fungal strains. Antifungal efficacy of extracts was measured in relations of inhibition of fungal growth. The results of antifungal properties are shown in Table 4, which revealed that various extracts of J. curcas stem bark showed significant antifungal activities towards the selected fungal strains. The maximum zone of inhibition was exhibited by methanolic extract followed by the ethanolic extract. The least inhibitory properties were indicated by the aqueous extract. The calculated zone of inhibition of all the extracts were compared with fluconazole antibiotic. The inhibition zone for various fungal species ranged between 20.75 mm to 14.12 mm where for Candida albicans was maximum (20.75 mm) as compared to other fungal species while the minimum inhibitory area (14.12 mm) was recorded for Microsporum canis.
The average inhibition zone for methanol extract (16.60 mm) was investigated to be most active followed by the average inhibition zone of ethanol extract (15.15 mm). The mean aqueous extract showed the lowest antifungal activity with inhibition zone 12.08 mm. The interaction between different extracts and test microbial species ranged between 8.00 mm to 20.00 mm. The maximum zone of inhibition (20.00 mm) was recorded for Candida albicans with methanol extract and the minimum (8.00 mm) for Microsporum canis with aqueous extract.
TABLE: 4 ZONE OF INHIBITION (MM) OF DIFFERENT FUNGI AS AFFECTED BY STEM BARK EXTRACT OF J. CURCAS
| Test
Fungi |
Methanol
(10 mgml-1) |
Eethanol
(10 mgml-1) |
Aqueous | Fluconazol | Mean |
| (10 mgml-1) | (10 mgml-1) | ||||
| Candida albicans | 20.00G | 19.00 GH | 14.00 M-O | 30.00 A | 20.75 A |
| Candida glaberata | 17.50 H-J | 16.00 J-L | 12.00 P-R | 25.00 DE | 17.62 CD |
| Fusarium solani | 15.00 K-M | 14.00 M-O | 10.25 R | 28.00 B | 16.81 DE |
| Microsporum canis | 14.00 M-O | 12.50 O-Q | 8.00 S | 22.00 F | 14.12 F |
| Aspergillus flavus | 16.00 J-L | 14.00 M-O | 11.00 QR | 24.00 E | 16.25 E |
| Penicillium notatum | 17.00 IJ | 15.00 K-M | 13.12 N-P | 26.00 CD | 17.78 C |
| Aspergillus niger | 15.00 K-M | 13.50 M-P | 12.00 P-R | 27.50 BC | 17.00 C-E |
| Trichophytons longifolia | 19.00 GH | 16.50 I-K | 13.50 M-P | 26.00 CD | 18.75 B |
| Conidiobolus coronatus | 14.50 L-N | 14.00 M-O | 13.00 M-P | 25.00 DE | 16.62 E |
| Rhodotorolla Mucillaginosa | 18.00 HI | 17.00 IJ | 14.00 M-O | 26.00 CD | 18.75 B |
| Mean | 16.60 B | 15.15 C | 12.08 D | 25.95 A | |
| LSD Values 16.00 | Extracts 0.56 | Test bacteria 0.88 | Interaction 1.76 |
Means tracked by dissimilar alphabets are significant at P ≤ 0.05.
The maximum zone of inhibition was exhibited by methanolic extract followed by the ethanolic extract. The least inhibitory (Zone of inhibition) properties was indicated by aqueous extract, compared with fluconazole antibiotic. The inhibition zone for various fungal species ranged between 20.75 mm to 14.12 mm where for Candida albicans was maximum (20.75 mm)
Anti-oxidant Activity:
Polyphenolic Compounds and Antioxidant Activity: In the present study, the qualitative, as well as quantitative phytochemical analysis of ethonolic extract of J. curcas stem bark, shows the presence of polyphenolic compounds which acts as antioxidant agents and scavenge free radicals. 20 The % concentration of phenol (0.7 ± 0.2) % and flavonoids (11.1 ± 0.1) % contributed the anti-oxidant activities of the plant extracts. The maximum antioxidant capacity of the ethanolic extract of J. curcas stem bark extract could be due to the presence of phenolic plant secondary metabolites Table 5 and 6.
TABLE 5: QUALITATIVE ESTIMATION OF SECONDARY METABOLITES OF STEM BARK OF J. CURCAS
| S. no. | Phytochemicals | Positive | Slightly Positive | Negative |
| 1 | Tannins | ++ | - | |
| 2 | Phlobatannins | + | - | |
| 3 | Saponins | ++ | - | |
| 4 | Flavonoids | ++ | - | |
| 5 | Steroids | ++ | - | |
| 6 | Terpenoids | + | - | |
| 7 | Cardiac glycosides | + | - | |
| 8 | Alkaloids | ++ | - | |
| 9 | Anthraquinones | + | - | |
| 10 | Total Phenols | + | - |
The biochemical assay of the ethanolic extract of J. curcas indicated that tannins, saponins, flavonoids, steroids, and alkaloids were positive while the phlorotannins, terpenoids, cardiac glycosides anthraquinones and total phenols were slightly positive.
TABLE 6: QUANTITATIVE ESTIMATION (%) OF SECONDARY METABOLITES STEM BARK OF J. CURCAS
| S.
no. |
Secondary metabolites | Amount of Phytochemicals present (mean ± SD) |
| Stem bark | ||
| 1 | Tannins | 24.1 ± 0.1 |
| 2 | Phlobatannins | 6.0 ± 0.1 |
| 3 | Saponins | 14.1 ± 0.1 |
| 4 | Flavonoids | 11.1 ± 0.1 |
| 5 | Steroids | 19.7 ± 0.1 |
| 6 | Terpenoids | 0.5 ± 0.3 |
| 7 | Cardiac glycosides | 5.0 ± 0.1 |
| 8 | Alkaloids | 12.8 ± 0.2 |
| 9 | Anthraquinones | 1.2 ± 0.3 |
| 10 | Total phenols | 0.7 ± 0.2 |
It is evident from Table 6 that the quantitative estimation of these secondary metabolites showed that tannins were most abundant (24.1 ± 0.1) followed by steroids (19.7 ± 0.1), saponins (14.1 ± 0.1), flavonoids (11.1 ± 0.1) and terpenoids (0.5 ± 0.3).While the total phenols (0.7 ± 0.2), anthraquinones (1.2 ± 0.3), and cardiac glycosides (5.0 ± 0.1) were least in quantity.
DPPH Radical Scavenging Assay: Free radicals scavenging capabilities play a tremendous action in the recovery of acute and chronic healing of wounds 21, 22. The maximum percent DPPH scavenging action was shown by the methanolic extract of J. curcas stem bark extract (90.5%) followed by the aqueous plant extracts (79.5%) while the least scavenging activity was shown by the ethanolic plant extract (77.2%).
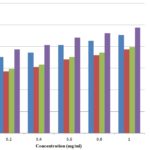
These antioxidant activities of the various extracts were compared with that of standard antioxidant agent BHT (97.5%). The maximum scavenging capacity was recorded at a maximum concentration of 1mg/ml Fig. 1.
FIG. 1: DPPH FREE RADICAL SCAVENGING ACTIVITIES OF THE DIFFERENT EXTRACTS OF JATROPHA CURCAS
Fig. 1 indicated antioxidant activity against DPPH free radicals. Maximum scavenging activities were shown by methanolic extract followed by aqueous extract, and the least antioxidant potential was shown by the ethanolic extract. Antioxidant activities were significantly compared with BHT, a positive control.
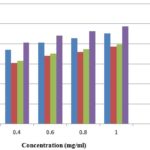
ABTS Radical Scavenging Activity: In this assay, the percent inhibition potential of ABTS free radical of the plant extract was determined. The scavenging activity determined were concentration dependent as shown in Fig. 2. The maximum activities were shown at 1mg/ml.
The maximum activities were shown by methanolic extract (87.0%) followed by ethanolic extract (86.4%) while the least scavenging activities were shown by aqueous extracts (84.8%). The activities of these extracts were significant as compared with standard BHT which was (94.9%). The antioxidant activities of the J. curcas were found to be remarkably high.
FIG. 2: ABTS FREE RADICAL SCAVENGING ACTIVITIES OF THE DIFFERENT EXTRACTS OF J. CURCAS
Fig. 2 indicated the antioxidant activity of methanolic, ethanolic and aqueous extract of Jatropha curcas against ABTS free radicals. Maximum scavenging activities were shown by methanolic extract followed by ethanolic extract, and the least antioxidant potential was shown by the aqueous extract. Antioxidant activities were significantly compared with BHT, a positive control.
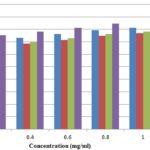
Superoxide Anion Scavenging Activity: Various extracts of stem bark of J. curcas showed percent inhibition of superoxide anion scavenging activities which were significantly comparable with BHT as shown in Fig. 3. The maximum scavenging activities were shown by methanolic extract (80.2%) followed by aqueous extract (78.3%), and the least antioxidant activity was shown by ethanolic extract (75.4%). These activities were measured at a high concentration of 1mg/ml; the findings were also compared with standard BHT (86.69%).
FIG. 3: SUPER OXIDE ANION FREE RADICAL SCAVENGING ACTIVITIES OF THE DIFFERENT EXTRACTS OF JATROPHA CURCAS
Fig. 3 indicated the antioxidant activity of methanolic, ethanolic and aqueous extract of Jatropha curcas against superoxide anion free radicals. Maximum scavenging activities were shown by methanolic extract followed by aqueous extract, and the least antioxidant potential was shown by the ethanolic extract. Antioxidant activities were significantly compared with BHT, a positive control
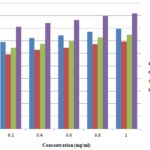
Nitric Oxide Scavenging Activity: The different extract of J. curcas showed excellent inhibitory activities with percent inhibition of nitric oxide. The antioxidant activities studied were concentration dependent; the excellent activities were recorded at a maximum concentration of 1mg/ml. The maximum antioxidant activities were shown by methanolic extracts (79.40%) followed by aqueous extract (74.79%), and the least scavenging potential was shown by the ethanolic extract (69.40%). These activities were significantly compared with standard BHT (91.85%) as shown in Fig. 4.
FIG. 4: NITRIC OXIDE FREE RADICAL SCAVENGING ACTIVITIES OF THE DIFFERENT EXTRACTS OF JATROPHA CURCAS
Fig. 4 indicated the antioxidant activity of methanolic, ethanolic and aqueous extract of Jatropha curcas against nitric oxide free radicals. Maximum scavenging activities were shown by methanolic extract followed by aqueous extract, and the least antioxidant potential was shown by the ethanolic extract. Antioxidant activities were significantly compared with BHT, a positive control
Phytochemical Analysis of J. curcas Stem Bark Extract: It is evident from Table 6 that the quantitative estimation of these secondary metabolites showed that tannins were most abundant (24.1 ± 0.1) followed by steroids (19.7 ± 0.1), saponins (14.1 ± 0.1), flavonoids (11.1 ± 0.1) and terpenoids (0.5 ± 0.3). The least identified plants secondary metabolites were total phenols (0.7 ± 0.2), anthraquinones (1.2 ± 0.3), and cardiac glycosides (5.0 ± 0.1).
DISCUSSION:
Antibacterial Susceptibility Tests:
Zone of Inhibition in Bacteria (mm): The average inhibition zone for methanol extract (25.31mm) was established to be extra valuable than the average inhibition zone of ethanol extract (19.72 mm). The mean aqueous extract showed low antibacterial activity with inhibition zone 7.77 mm ranging between 1-11 mm. It is oblivious from the findings that the methanolic extract was more potent than the rest of the extracts. It may be due to the occurrence of different phenolic and polyphenolic compounds 23. The low antibacterial activity of aqueous extract is concurrence with the earlier study which mentioned that the aqueous extract of Jatropha has little activity against the test bacteria. It may be due to the insolubility of the various organic compounds in water which were responsible for antimicrobial activities 24-28 reported that the methanolic root extract of Jatropha curcas showed antimicrobial potential against those bacteria causing urinary tract infection and sexually transmitted diseases.
Determination of Minimum Inhibitory Concentration (MIC mgml-1): The average least inhibitory quantity for the methanol and the ethanolic suspension was 4.31, and 5.09 mgml-1 respectively. Hence, it was observed that the methanolic suspension was more potent than the other extracts. The previous finding also revealed that different extracts from various parts of the Jatropha curucs exhibit broad-spectrum anti-bacterial property through a different mode of action. Igbinosa 29 investigated the antimicrobial activity of Jatropha stem bark extract in the laboratory and stated that the methanolic and aqueous extract of the selected plant exhibit significant bacteriological properties. Atindihou 30 studied the antimicrobial properties of J curcas leaves and roots and reported that the root and leaves of the selected plant inhibit the growth of various harmful bacteria. Flavonoids are the phytochemical of Jatropha stem bark, are excellent bacteriological in nature, prevent bacterial growth and multiplication by the inhibition of a DNA synthesizing enzyme called DNA gyrase 31. Phenolic compound included Gallic acid and pyrogallol are the excellent antimicrobials 31. Plant subordinate metabolites found in J. curcas seed, exhibiting well defined bacteriological and antifungal activities towards the chronic pathogens including g+ve and g-ve. 32 The least activity of the aqueous solution towards all microbial species revealed in this study is in the settlement with earlier studies who reported that water extract of herb usually investigated reduce or no bacteriological properties 24-28.
Determination of Minimum Bactericidal Concentration (MBC mgml-1): The average minimum bactericidal concentration for the methanol and ethanol extract was 8.27, and 9.81 mgml-1 respectively. Hence, it was observed that the methanolic extract was most potent than the other extracts. The minimum bactericidal concentration observed in this study are in agreement with that of earlier reports 33-38. Previous findings support the antimicrobial activity and medicinal importance of J. curcas plant's parts 29, 36, 39, 40 The antimicrobial activity of J. curcas may be attributed due to the presence of certain phytochemicals which included saponins, tannins, alkaloids, and glycosides 36-37. Similarly igbinosa 29 reported the bactericidal properties of stem bark aqueous extract of J. curcas against a wide range of bacterial isolates excluding Klebsiella pneumonia. The disparity in the different reports may be due to differences in extract preparation and concentrations as well as strain differences and geographical location of the plant material collected.
Zone of Inhibition in Fungi (mm): The maximum zone of inhibition (20.00 mm) was recorded for Candida albicans with methanol extract and the minimum (8.00 mm) for Microsporum canis with aqueous extract. In the present study, various extracts of Jatropha were founded sensitive against all the test organisms which might be due to the existence of diverse phytochemical compounds with pharmacological properties that can be of appreciable therapeutic index. This also supported the earlier investigation 41-42, that medicinal plants with tannin content possesses outstanding toxic property towards bacteria and fungi and may assume pharmaceutical reputation. Aliero 43 reported significant mycelial growth inhibition of A. niger with extracts of A. cordifolia and A. sativum which is an agreement with this investigation.
Anti-oxidant Activity:
Polyphenolic Compounds and Antioxidant Activity: The phytochemical analysis revealed the presence of phenol (0.7 ± 0.2) % and flavonoids (11.1 ± 0.1) % which contributed to the antioxidant activities of the plant extracts. The exact mode of action of these compounds are unknown, but the previous study revealed that the antioxidant capacity of these plants secondary metabolites might be due to the ability of phenolic compounds to absorb and neutralize free radicals and quench the active oxygen species and break down the superoxide and hydroxyl radicals 44. In an ethanolic extract of J. curcas. Flavonoids are the phytochemicals which act as an antioxidant by the inhibition of membrane-bounded enzymes such as the ATPase and phospholipase A2. 45 Along with the antioxidant activity, the phytochemical analysis of J. curcas showed the existence of other compounds which also act as antimicrobial agent46.
The current results revealed that the methanolic plant extract could be used as a potent and natural antioxidant agent. These results show that the plant extracts contain the natural compounds that are capable of donating hydrogen to a free radical to neutralize and remove abnormal electron which is responsible for radical reactivity. The free radical scavenging potential of J. curcas is due to its strong proton donating capability. 47 The findings of the current study differ from the previous work 48 by who stated that the compounds which exhibit ABTS scavenging potency might not contain DPPH scavenging capacity. In this study, the extract was able to inhibit both DPPH and ABTS radical with a similar trend. So, this is concluded from the current study that at higher concentration the plant extracts can treat various pathological abnormalities regarding free radicals. Nitric oxide is produced from sodium nitroprusside in an aqueous or cytoplasmic solution at physiological pH which is reactive free radical and quickly react with oxygen in the reaction to form nitrite. The current plant extract stops nitrite formation by directly competing with oxygen in the reaction. The previous work also support the anti-inflammatory activities of the nitric oxide which recommend the J. curcas for the treatment of inflammation and healing of wounds 49.
Phytochemical Analysis of J. curcas Stem Bark Extract: The phytochemical estimation of these secondary metabolites showed that tannins were most abundant (24.1 ± 0.1) followed by steroids (19.7 ± 0.1). According to Shimada, 50 tannins are the plant's secondary metabolites that have a specific mechanism of action to form irreversible complexes with skin protein as well as with proline-rich proteins that causes aggregation of local tissues showing astringent effects and inhibit protein synthesis. Tannins may also be used to treat swelling, ulceration and corrosive tissues in different parts of the body. Plants which contains tannins as its major constituent can cause local tissue aggregation which is applied for the management of gastrointestinal diseases such as dysentery and duodenal ulceration.51 Therefore the results of the previous investigation justify the use of J. curcas in homeopathic as well as in the allopathic system of medicine. Li and Wang 45 reported that tannins are the plant's secondary metabolites which exhibit anti-tumor activities and can be used as adjuvant therapy in tumor treatment. Thus signifying that J. curcas is a credible birthplace of valuable biologically active compound for the control and prophylaxis of tumor.
Alkaloids which are the big class of photochemical and plants secondary metabolites having surprising therapeutic properties including strong analgesic properties 52. The most important and natural activities of alkaloids are their specific toxicities against a foreign entity. These properties have been extensively deliberated for their prospective use in the control and eradication of various human tumors 53. The anti-inflammatory activity of saponins on the infectious tissues are reported previously so that the saponins can be used as effective anti-inflammatory agent 54. Steroidal compounds are also planted secondary metabolites which are present abundantly in various crude extracts of J curcas stem bark; they are very important because of their correlation with many endocrinal and exocrine hormones 55. Quinlan 56 studied the antimicrobial activities of steroidal compounds and mentioned both as an antibacterial and antifungal agent. Flavonoids are also planted secondary metabolites present in J. curcas stem bark extract exhibiting excellent antimicrobial activities along with a wide range of other pharmacological activities 57.
Ekandayo 58 also reported the same result, who stated that the capability of the organic extract of the bark and leaf of J. curcas to stop the growth and multiplication of the micro-organism is a signal of its antimicrobial efficacy which may be employed in the treatment and control of pathogenic severe infections. The presence of these secondary metabolites in such amount can familiarize the J. curcas stem bark extract as a probable candidate for the management of disorders triggered by the resistant microbes. However, there is a need to carry out a toxicological investigation of the stem bark extract to find out their safety on a human.
CONCLUSION: It is concluded from the current study that the maximum antibacterial, antifungal and antioxidant activities of stem bark extracts were shown by the methanolic extract followed by ethanolic extract and the least activities were shown by the aqueous extract. The phytochemical analysis shows the presence of various plant secondary metabolites among which tannins were founded most abundantly (24.1 ± 0.1). The potent antimicrobial and antioxidant activities of J. curcas stem bark extract suggest that this extract may be used in the treatment of microbial infections and disorders caused by free radicals. However, there is a need to conduct the detailed toxicological evaluation of the bark to determine their safety on a human.
ACKNOWLEDGEMENT: Thanks and sincerest appreciations are also extended to my Supervisor Prof. Dr. Abdul my Co-Supervisor-I Prof. Dr. Ejaz Ahmad Khan, Dr. Muhammad Sadiq, Assistant Professor, Dr. Samiullah Khan for their sincere advice, consistent encouragement, inspiring guidance and constructive criticism throughout the course of these investigations and help in the preparation of this manuscript.
CONFLICT OF INTEREST: Nil
REFFERENCES:
- Sukayana SL, Sudisha P and Niransjana SR: Aftric J Biotech 2009; 8(23): 6677-82.
- Srivastava J, Lambert J and Vietmeyer N: Medicinal Plants: an expanding role in development. The World Bank, Washington, DC, 1996: 18.
- Prusti A, Mishra SR, Sahoo S and Mishra SK: Anti-bacterial activity of some Indian medicinal plants. Ethnobotanical Leaflets 2008; 12: 227-30.
- Beuchat LR, Brackett RW and Doyle MP: Lethality of carrot juice to L. monocytogenes as affected by pH, sodium chloride, and temperature. J Food Prot 1994; 57: 470-80.
- Elujoba AA, Odeleye OM and Ogunyemi CM: Traditional medical development for medical and dental primary health care delivery system in Africa. Afr J Trad, CAM. 2006; 2(1): 46-61.
- Okigbo RN and Mmeka EC: An appraisal of phyto-medicine in Africa. KMITL Sci J 2007; 6(2): 83-93.
- Okigbo RN and Ajalie AN: Inhibition of some human pathogens with tropical plant extracts. International J Molecular Med Advanced Sciences 2005; 1(1): 34-41.
- Gulcin: Comparison of in-vitro antioxidant and antiradical activities of 1-tyrosine and 1-dopa. Amino Acids 2006; 32: 431-38.
- Gulcin: Comparison of in-vitro antioxidant and antiradical activities of 1-tyrosine and 1-dopa. Amino Acids 2007; 32: 551-60.
- Bahman N, Mohammad K and Raza H: In-vitro free radical scavenging activity of five Salvia species. Pak J Pharm Sci 2007; 20: 291-94.
- Osawa T: Protective role of dietary polyphenols in oxidative stress, 1999; 111: 133-39.
- Harborne JB: Phytochemical Methods- A guide to modern techniques of plant analysis. Chapman and Hall, London, 1998: 182-90.
- Irobi ON, Young MM, Anderson WA and Daramola SO: Antimicrobial activity of the bark of Bridelia ferruginea. Int J Pharmacog 1994; 34: 87-90.
- Akinpelu DA and Kolawole DO: Phytochemical and antimicrobial activity of leaf extract of Piliostigma thonningii (Schum.). Sci Focus J 2004; 7: 64-70.
- Spencer ALR and Spencer JFT: Public Health Microbiology: Methods and Protocols. Human Press Inc. New Jersey 2004: 325-27.
- Liyana-Pathiranan CM and Shahidi: Antioxidant activity of commercial soft and hard wheal as affected by gastric PH conductive Journal of Agriculture Food Chemistry 2005; 53: 2433-40.
- Re R, Pellegrini N, Proteggente A, Pannala A, Yang M and Rice-Evans C: Antioxidant activity applying an improved ABTS radical cation dollarization assay. Free Rad. Biology 1999; 26: 1231-37.
- Liyana-Pathiranan CM and Shahidi: Antioxidant activity of commercial soft and hard wheal as affected by gastric pH conductive. Journal of Agriculture Food Chemistry, 2005; 53: 2433-40.
- Garret DC: The Qualitative Analysis of Drugs 1964: 450-58.
- Milliauskas G, Venskutonis PR and Van-Beek TA: Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem 2004; 85: 231-37.
- Shukla A, Rasik AM and Potnaik GK: Depletion of reduced glutathione ascorbic acid, vitamin E and cutaneous healing wound. Free Rod Res 1997; 26: 93-101.
- Mcdaniel DH, Ash K, Lord J and Newman: The accelerated loser is resurfacing wound healing using a triad of topical antioxidants. Dermatol Surg 1998; 24: 661-64.
- Kowalski R and Kedzia B: Antibacterial activity of S. perfoliatum extracts Pharm Biol 2007; 45: 495-500.
- Koduru S, Grierson DS and Afolayan AJ: Antimicrobial activity of Solanum aculeastrum (Solanaceae). Pharmacol Biol 2006; 44: 284-86.
- Aliero AA, Grierson DS and Afolayan AJ: Antifungal activity of S. pseudocapisum. Res J Bot 2006; 1: 129-33.
- Ashafa AOT, Grierson DS and Afolayan AJ: Anti-microbial activity of extract from Felicia muricata Thunb. J Biol Sci 2008; 8(6): 1062-66.
- Aiyegoro OA, Akinpelu DA, Afolayan AJ and Okoh AI: Antibacterial activities of crude stem bark extracts of D. benthamianus Baill. J Biol Sci 2008; 8(2): 356-61.
- Aiyelaagbe OO, Adeniyi BA, Fatunsin OF and Arimah BD: In-vitro antimicrobial activity and phytochemical analysis of J. curcas roots. Int J Pharmacol 2007; 3(1): 106-110.
- Igbinosa OO, Igbinosa EO and Aiyegoro OA: Anti-microbial activity and phytochemical screening of stem bark extract from Jatropha curcas (Linn). Afr J Pharmacy and Pharmacology 2009; 3(2): 058-62.
- Atindehou K, Kone M, Terreaux C, Traore DK and Hostettmann M: Antimicrobial activity of leaf extracts of Indian medicinal plants against clinical and antiparasitic activities of J. curcas. East Africa Med J 2002; 75: 508-11.
- Cushnie T and Lamb A: Antimicrobial activity of flavonoids. Int J Antimicro Ag 2005; 26: 343-56.
- Sparg SG, Light ME and Staden JV: Biological activities and distribution of plant saponins. J Ethnopharmacol 2004; 94: 219-43.
- Gubitz GM, Mittelbach M and Trabi M: Exploitation of the tropical oil seed plant Jatropha curcas L. Bioresources Technology 1999; 67: 73-82.
- Kumar VL and Arya S: Medicinal uses and pharmacological properties of Calotropis procera. Recent Progress in Medicinal Plants 2006; 11: 373-88.
- Kamboj A and Saluja AK: Bryophyllum pinnatum (Lam.) Kurz: Phytochemical and pharmacological profile: A review. Phcog Rev 2009; 3(6): 364-74.
- Arekemase M.O and Oyeyiola GP: Effects of skin coating materials, storage conditions on pH, titratable acidity and vitamin C contents of citrus fruits stored at room and refrigerated temperatures. Journal of Asian Scientific Research, 2011; 1: 376-89.
- Namuli A, Abdullah N, Sieo CC, Zuhainis SW and Oskoueian E: Phytochemical compounds and antibacterial activity of Jatropha curcas Linn. extracts. J Med Pl Res 2011; 5: 3982-90.
- Afzal AFM: Substantiation of the fracture behavior and mechanical properties of sol-gel derived silica packed epoxy networks. Journal of Sol-Gel Science and Technology 2012; 61: 44-48.
- Rachana S, Tarun A, Rinki R, Neha A and Meghna R: Comparative analysis of antibacterial activity of Jatropha curcas fruit parts. Journal of Pharmaceutical and Biomedical Sciences 2012; 15(15): 1-4.
- Omoregie EH and Folashade KO: Broad spectrum antimicrobial activity of Jatropha curcas. Afr J Pharma 2013; 14(10): 11-20.
- Banso A and Adeyemo SO: Evaluation of. Antibacterial properties of tannins isolated from Dichrostachys cinerea. Afr J Biotechnol 2007; 6(15): 1785-87.
- Varaprasad B, Katikala PK, Naidu KC and Penumajji S: Antifungal activity of selected plant extracts against phytopathogenic fungi Aspergillus niger. Indian Journal of Science and Technology 2009; 2(4): 87-90.
- Aliero AA, Grierson DS and Afolayan AJ: Antifungal activity of S. pseudocapisum. Res J Bot 2007; 1: 129-33.
- Duh PD, Tu YY and Yen GC: Antioxidant activity of water extract of Harng Jyur (Chrycanthemum morifolium Ramat). Lebensm. Wiss Technology 1999; 32: 269-77.
- Li D and Wang P: Antifungal activity of Paraguayan plant used in traditional medicine. J. Ethnopharmacol 2003; 76: 93-98.
- Hausteen B: Flavonoids, a class of natural products of high pharmacological potency. Biochem Pharmacy 1983; 29: 200-10.
- Mc daniel DH, Ash K, Lord J and Newman: The accelerated loser is resurfacing wound healing using a triad of topical antioxidants. Dermatol Surg 1998; 24: 661-64.
- Wang ML, Rangarajan M, Shao Y, Lavoie E and Huang HO: 1998.
- Moncada S, Palmer RMJ and Highs EA: Nitric oxide physiology, pathophysiology, and pharmacology. Pharmacological Review, 1991; 43: 109-42.
- Shimada T: Salivary proteins as a defense against dietary tannins. J Chem Ecol 2006; 32: 1149-63.
- Dharmananda S: Gallnuts and the uses of Tannins in Chinese Medicine. In: Proceedings of Institute for Traditional Medicine, Portland, 2003.
- Kam P and Liew CA: Traditional Chinese herbal medicine and anesthesia. Anaesth 2002; 57: 1083-89.
- Nobori T, Miurak K, Wu DJ, Takabayashik LA and Carson DA: Deletion of the cyclin-dependent kinase-4 inhibitor gene in multiple human cancers. Nat 1994; 368(6473): 753-56.
- Just RM, Giner, Cueller MJ, Manez S, Bilia and Rios JL: Anti-inflammatory activity of unusual lupine saponins from B. fruticescens. Planta Medica 1998; 64: 404-7.
- Okwu DE: Evaluation of the chemical composition of indigenous Spices and flavoring agents. Global J Appl Sci, 2001; (3): 455-59.
- Quinlan MB, Quinlan RJ and Nolan JM: Ethnophysiology and herbal treatments of intestinal worms in Dominica, West Indies. J Ethnopharmacol 2002; 80: 75-83.
- Hodek P, Trefil P and Stiborova M: Flavonoids - Potent & versatile biologically active compounds interacting with cytochrome P450. Chem-Biol Int 2002; 139(1): 11-21.
- Ekundayo FO, Adeboye CA and Ekundayo EA: Antimicrobial activities and phytochemical screening of pignut (Jatrophas curcas Linn.) on some pathogenic bacteria. J Med Plants Res 2011; 5(7): 1261-64.
How to cite this article:
Khan N, Shah AH, Khan EA, Sadiq M, Khan S and Baloch N: Phytochemical analysis and biological activities of stem bark extract of Jatropha curcas Linn. Int J Pharmacognosy 2017; 4(5): 155-68. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(5).155-68.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
155-168
737
2060
English
IJP
N. Khan *, A. H. Shah, E. A. Khan, M. Sadiq, S. Khan and N. Baloch
Gomal Center of Biochemistry and Biotechnology, Gomal University, Dera Ismail Khan, Khyber Pakhtunkhwa, Pakistan.
phnaqab87@gmail.com
07 February 2017
20 April 2017
25 April 2017
10.13040/IJPSR.0975-8232.IJP.4(5).155-68
01 May 2017