PHYTOCHEMICAL ANALYSIS AND ANTIOXIDANT PROPERTY OF PREMNA LATIFOLIA
HTML Full TextPHYTOCHEMICAL ANALYSIS AND ANTIOXIDANT PROPERTY OF PREMNA LATIFOLIA
R. Krishnamoorthi * and V. Ratha Bai
PG and Research Department of Zoology, Presidency College (Autonomous), Chennai - 600005, Tamil Nadu, India.
ABSTRACT: Plants have been used for centuries as a remedy for human diseases because they contain phytochemical components of therapeutic values. Hexane, ethyl acetate and ethanolic extract of Premna latifolia were investigated for phytochemical constituent and antioxidant activity. The phytochemical constituent was investigated using standard chemical methods. Further, the study was extended by analyzing the antioxidant potential using DPPH method. The results showed that ethanolic extract showed a significant number of phytoconstituents and better radical scavenging activity at the concentration of 75 µg/ml.
| Keywords: |
Premna latifolia, DPPH, Antioxidant, Phytochemicals
INTRODUCTION: Premna latifolia belongs to the family Verbenaceae, widely distributed in tropical and subtropical and coastal areas. The leaves are diuretic and are used as a folk medicine for treating dropsy 1. Premna latifolia possesses anti-inflammatory activity in the animal models 2. Premna latifolia bark is applied to cure boils. The leaves are diuretic and are used as a folk medicine for treating dropsy 3. Premna latifolia possesses anti-inflammatory activity in the animal models. Premna latifolia bark is applied to cure boils. Traditionally it has been used in the treatment of hepatic disorders 4, antioxidant 5 and anticancer activity 6. Herbal medicines from the plants act as a primary source for the discovery of new drugs. Phytochemicals are defined as bioactive non-nutrient plant compounds reducing the risk of major chronic diseases 7.
The significant phytochemicals such as alkaloids, tannins, glycosides, flavonoids, steroids, tannins, etc., constitute a chemical agent for the approach of new anti-infective agents. Antioxidants are the compounds which help to delay or inhibit the oxidation of lipids and other molecules through the inhibition of either initiation or propagation of oxidative chain reactions 8. Free radicals contribute to more than one hundred disorders in humans including atherosclerosis, arthritis, ischemia, reperfusion injury of many tissues, central nervous system injury, gastritis, cancer and AIDS 9, 10.
MATERIALS AND METHODS:
Collection of Plants: Plants for this study were collected from Chinnapaliyampattu, Tiruvannamalai district and was authenticated by Dr. Rathna Kumar, Department of plant biotechnology, Presidency College, Chennai-05.
Preparation of Extracts: Collected plants were dried at room temperature and ground to make a fine powder. 20 gm of plant powder was well dissolved in 100 ml of solvents (Hexane, Ethyl acetate, and Ethanol) (ratio 1:5). The suspension was filtered by using filter paper of pore size 0.2µm. The filtrate was then air dried, and extracts were collected in sterile vials for further use.
Phytochemical Test: The phytochemical analysis of these extracts was performed using the method adopted by Harborne 11 and Sofowora 12.
Test for Carbohydrates (Molisch’s Test): To 2 ml of plant extract, 1 ml of Molisch’s reagent and a few drops of concentrated sulfuric acid were added. Presence of purple or reddish color indicates the presence of carbohydrates.
Test for Tannins (Ferric Chloride Test): To 1 ml of plant extract, 2 ml of 5% ferric chloride was added. Formation of dark blue or greenish black indicates the presence of tannins.
Test for Saponins (Frothe’s Test): To 2 ml of plant extract, 2 ml of distilled water was added and shaken in a graduated cylinder for 15 min lengthwise. Formation of a 1 cm layer of foam indicates the presence of saponins.
Test for Flavonoids (Shinoda Test): To 2 ml of plant extract, 1 ml of 2N sodium hydroxide was added. Presence of yellow color indicates the presence of flavonoids.
Test for Alkaloids (Mayer’s Test): To 2 ml of plant extract, 2 ml of concentrated hydrochloric acid was added. Then a few drops of Mayer’s reagent were added. The presence of green color or white precipitate indicates the presence of alkaloids.
Test for Quinines: To 1 ml of extract, 1 ml of concentrated sulfuric acid was added. Formation of red color indicates the presence of Quinones.
Test for Glycosides (Molisch’s Test): To 2 ml of plant extract, 3 ml of chloroforms and 10% ammonia solution was added. Formation of pink color indicates the presence of glycosides.
Test for Cardiac Glycosides (Keller – Kiliani Test): To 0.5 ml of extract, 2 ml of glacial acetic acid and a few drops of 5% ferric chloride were added. This was under layered with 1 ml of concentrated sulfuric acid. The formation of a brown ring at the interface indicates the presence of cardiac glycosides.
Test for Terpenoids (Salkowski Test): To 0.5 ml of extract, 2 ml of chloroform was added and concentrated sulfuric acid is added carefully. Formation of red-brown color at the interface indicates the presence of terpenoids.
Test for Triterpenoids: To 1.5 ml of extract, 1 ml of Liebmann–Buchard reagent (aectic anhydride + concentrated sulfuric acid) was added. Formation of blue-green color indicates the presence of triterpenoids.
Test for Phenols (Ferric Chloride Test): To 1 ml of the extract, 2 ml of distilled water followed by a few drops of 10% ferric chloride was added. Formation of blue or green color indicates the presence of phenols.
Test for Coumarins: To 1 ml of extract, 1 ml of 10% NaOH was added. Formation of yellow color indicates the presence of coumarins.
Steroids and Phytosteroids (Libermann -Burchard Test): To 1 ml of plant extract equal volume of chloroform is added and subjected with a few drops of the concentrated sulfuric acid appearance of brown ring indicates the presence of steroids and appearance of the bluish-brown ring indicates the presence of phytosterols.
Phlobatannins: To 1 ml of plant extract a few drops of 2% HCl was added the appearance of red color precipitate indicates the presence of phlobatannins.
Anthraquinones (Borntrager’s Test): To 1 ml of plant extract, a few drops of 10% ammonia solution were added, appearance pink color precipitate indicates the presence of anthraquinones.
Antioxidant Activity: The antioxidant activity of Plant extracts was determined by, the DPPH (1,1-diphenyl-2-picryl-hydroxyl) in-vitro method.
DPPH free Radical Scavenging Activity: The antioxidant activity of hexane, ethyl acetate and ethanolic extracts of Premna latifolia and the standard compound BHT was measured in terms of hydrogen donating radical scavenging ability using the stable DPPH method 13. 1 ml of the extract was added to 3.7 ml of methanol solution.
After centrifugation, the supernatant is collected 200μml of DPPH solution is added. Kept in the dark for 45 min and the resulting decrease in absorbance at 517 nm were recorded against blank using a UV-Vis Spectrophotometer.
The radical scavenging activity on DPPH was expressed as, % DPPH radical-scavenging = [(Absorbance of control - Absorbance of test Sample) / (Absorbance of control)] × 100.
RESULTS: The preliminary phytochemical screening of Premna latifolia showed the presence of plant components such as carbohydrates, flavonoids, quinones and coumarins in hexane extract, carbohydrates, tannins, flavonoids, cardiac glycosides, phenols and coumarins in ethyl acetate extract and carbohydrates, tannins, flavonoids, quinones, cardiac glycosides phenols and coumarins in ethanol extract Table 1.
TABLE 1: PHYTOCHEMICAL ANALYSIS OF PREMNA LATIFOLIA
| S.
no. |
Phytochemical
Tests |
Test
Performed |
Hexane Extract | Ethyl Acetate Extract | Ethanol Extract |
| 1 | Carbohydrates | Molisch’stest | + | + | + |
| 2 | Tannin | Ferric chloride test | - | + | + |
| 3 | Saponin | Frothe’s test | - | - | - |
| 4 | Flavonoids | Shinoda test | + | + | + |
| 5 | Alkaloids | Mayer’s test | - | - | - |
| 6 | Quinones | - | - | - | + |
| 7 | Glycosides | Molisch’s test | - | - | - |
| 8 | Cardiac glycosides | Keller – Kiliani test | + | + | + |
| 9 | Terpenoids | Salkowski test | + | - | - |
| 10 | Phenols | Ferric chloride test | + | + | + |
| 11 | Coumarins | - | + | - | - |
| 12 | Steroids | Libermann – Burchard test | - | + | - |
| 13 | Phlobotanins | - | - | - | - |
| 14 | Anthraquinones | Borntrager’s test | - | - | - |
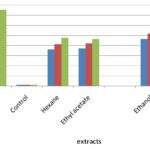
Free Radical Scavenging Activity (DPPH): The stable free radical scavenging activity by the DPPH method is an easy, rapid and sensitive way to survey the antioxidant activity of specific plant extracts. Fig. 1 indicates the percentage of free radicals scavenging activity in various extractions with different concentrations 25 µg, 50 µg and 75 µg of Premna latifolia.
FIG. 1: ANTIOXIDANT ACTIVITY OF PREMNA LATIFOLIA BY DPPH ASSAY
TABLE 2: DPPH ASSAY OF PREMNA LATIFOLIA AGAINST DIFFERENT EXTRACTS
| Concentrations
(µg) |
Control | % of Inhibition | |||
| Hexane | Ethyl Acetate | Ethanol | BHT | ||
| 25 | 0.9593 | 36.07 ± 0.97 | 36.95 ± 0.96 | 46.55 ± 0.98 | 72.36 ± 1.64 |
| 50 | 0.9593 | 41.15 ± 0.62 | 41.97 ± 0.53 | 51.72 ± 0.76 | 76.23 ± 0.85 |
| 75 | 0.9593 | 47.31± 0.43 | 46.36± 0.35 | 57.03± 0.54 | 81.68 ± 0.61 |
In this study percentage inhibition of free radicals was carried out with different extractions of selected plants. Ethanol extract with 75 μg concentration gives a higher percentage (57.03%), hexane extract showed the moderate activity of 47.31% and ethyl acetate extract showed the least activity of 46.36% of free radical scavenging activity. The free radical scavenging activity increases with increase in concentration Table 2, Fig. 1. The percentage inhibition of control was found to be 81.68% which showed higher activity than the extract.
DISCUSSION: The results of the present study reveals that the ethanolic extract of Premna latifolia showed the maximum number of components such as Carbohydrate, tannin is used for the treatment of skin eruption, antimicrobial activity, and bowel condition, flavonoids possess wound healing activity due to the astringent, antioxidant and antimicrobial properties which appear to be responsible for wound contraction and elevated rate of epithelisation 14, quinones, cardiac glycosides, phenols was found to be toxic to the growth and development of pathogens and coumarins in the ethanolic extract when compared to other solvents. The previous study reported that the stem bark shows the presence of iridoid glucosides and geniposidic acid 15, 16. Another report says that stem powder of Premna latifolia shows the presence of carbohydrates, proteins, phenols, oils, fats, terpenoids, steroids, saponins, flavonoids, alkaloids and tannins 17, 18. The DPPH scavenging of the stem extract of Premna latifolia was studied as described by Singh 19, 20.
Thus, the present work also correlates with the aforesaid studies. The significant concentration of antioxidant in the ethanolic extract is due to phytoconstituents. However, the extract was not as effective as the standard antioxidant BHT. Thus the ethanolic extract showed maximum antioxidant activity when compared to other solvents. Hence, the ethanolic extract can act as antioxidant activity and can protect the cells from the free radicals.
CONCLUSION: The present study suggests that the Premna latifolia have maximum number of bioactive components and higher amount of antioxidant potential in the ethanolic extract; therefore the ethanolic extract may act as a significant activity and can be further analyzed for many pathogenic disorders as well as may be helpful in the future for preventing or slowing the progress of diseases involved. However, it is obvious that fewer information was available further to explore this plant; more researchers should be carried out.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Sharma J and Sharma JN: Arthritis in ancient Indian literature. Ind J Hist Sci 1973; 8(1/2): 37-42.
- Mahire NB, Tote MV, Jain AP, Undale VR, and Bhosle AV: Anti-inflammatory effect of Premna latifolia Pharmacology Online 2009; (3): 929-937.
- Kirtikar KR and Basu MD: Indian Medicinal Plants. Periodical express book agency. New Delhi, Edition 2nd, 1991; 3: 1926.
- Shanmugavelu M: Siddha cure for diseases. Tamil Nadu Siddha medical board Publications, Chennai. India 1987.
- Devi K, Anadam R, Devaki T, Apparanatham T and Balakrishnan K: Biomed Res 1998; 19: 339.
- Hymavathi A, Babu KS, Naidu VGM, Ramakrishna S, Diwan PV and Rao JM: Bioorg Med Chem Lett 2009; 19; 5727.
- Liu, Mahmoud RH and Tanabe H: J Nutri 2009; 134: 3479-3485.
- Jaleel CA: Antioxidant potentials and ajmalicine accumulation in Catharathus roseus after treatment with giberellic acid. Colloids and surfaces B: Biointerfaces 2007; 60: (2), 195-200
- Cook NC and Samman S: Flavonoids chemistry metabolism cardioprotective effects and dietary sources. Nutritional Biochemistry 1996; 7: 66-76.
- Kumpulainen JT and Salonen JT: Natural antioxidants and anticarcinogens in nutrition, health and disease. The Royal Society of Chemistry, UK 1997; 178-187.
- Harborne JB: Phytochemical methods: A guide to modern techniques of plant analysis. Chapman and Hall, New York, Edition 3rd, 1973: 279.
- Sofowora A: Medicinal plants and Traditional medicinal in Africa. Sunshine house, Ibadan, Nigeria: Spectrum books Ltd; Screening plants for bioactive agents; Edition 2nd, 1993: 134-156.
- Eberhardt MV, Lee CY and Liu RH: Antioxidant activity of fresh apple. Nature 2000; 405: 903-904.
- Shenoy C, Patil MB, Kumar R and Patil S: Preliminary phytochemical investigation and wound healing activity of Allium cepa (Liliaceae). Int J Pharm Pharm Sci 2009; 2: 167-175.
- Khare CP: Indian medicinal plants. An illustrated dictionary. Springer – Verlag. Heidelberg; Edition 1st, 2007.
- Dutta S, Dey P and Chaudhuri T: Quantification and correlation of the bioactive chemicals of Chroton bonpladinum leaves of the sub-Himalayan region of West Bengal. Asian J Pharm Clin Res 2013; 6: 142-147.
- Singh N and Rajinia PS: Free radical scavenging activity of an aqueous extract of potato peel. Food Chem 2004; 82: 593-597.
- Khare CP: Indian medicinal plants. An illustrated dictionary. Springer – Verlag. Heidelberg; Edition 1st, 2007.
- Dutta S, Dey P and Chaudhuri T: Quantification and correlation of the bioactive chemicals of Chroton bonpladinum leaves of sub Himalayan region of West Bengal. Asian J Pharm Clin Res 2013; 6: 142-147.
- Singh N and Rajinia PS: Free radical scavenging activity of an aqueous extract of potato peel. Food Chem 2004; 82: 593 -597.
How to cite this article:
Krishnamoorthi R and Bai VR: Phytochemical analysis and antioxidant property of Premna latifolia. Int J Pharmacognosy 2015; 2(8): 414-18. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(8).414-18.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
414-418
586
1994
English
IJP
R. Krishnamoorthi and V. R. Bai
PG and Research Department of Zoology, Presidency College (Autonomous), Chennai, Tamil Nadu, India
kmoorthi222@gmail.com
25 June 2015
15 July 2015
11 August 2015
10.13040/IJPSR.0975-8232.IJP.2(8).414-18
31 August 2015