PHYTOCHEMICAL ANALYSIS AND ANTIOXIDANT PROPERTY OF JUSTICIA TRANQUEBARIENSIS
HTML Full TextPHYTOCHEMICAL ANALYSIS AND ANTIOXIDANT PROPERTY OF JUSTICIA TRANQUEBARIENSIS
R. Krishnamoorthi and V. Ratha Bai *
PG and Research Department of Zoology, Presidency College (Autonomous), Chennai - 600005, Tamil Nadu, India.
ABSTRACT: The present study was investigated to determine the possible phytochemical components from the solvents such as hexane, ethyl acetate and ethanolic extract of Justicia tranquebariensis. Among the phytochemical screening of these extracts, ethyl acetate extract showed that the whole plant was rich in carbohydrate, tannin, flavonoids, quinones, cardiac glycosides, phenols, steroids, and coumarins. Further, the study was extended by analyzing the antioxidant potential using DPPH method. Among all the extracts ethyl acetate and ethanolic extract showed better antioxidant activity at the concentration of 75 µg/ml when compared to hexane extract.
| Keywords: |
Justicia tranquebariensis, DPPH, Antioxidant, Phytochemicals
INTRODUCTION: Herbal plants were extensively used to treat several medical disorders. Justicia tranquebariensis belongs to the family Acanthaceae is a small herb distributed in the Peninsular and Deccan region of India. It is recognized by the following names, in Sanskrit ‘Pindi’, in Tamil ‘Tavashoo moorunghie,’ in Telugu it is ‘Pindikonda’ 1, 2. Justicia has been used in the traditional system for the treatment of fever 3, pain 4, inflammation 5, diabetes 6, diarrhoea 7 and liver disease 8. They also possess antitumoral 9, antiviral 10, analgesic and anti-inflammatory 11 activities. Juices of the leaves act as a cooling agent and aperients and also given to children in smallpox. A paste made of the leaves applied externally on the swelling to reduce the pain.
Phytochemicals present in this plant can act as an agent for several disorders like ischemia, asthma, arthritis, cancer, ageing, atherosclerosis, reperfusion injury of many tissues, central nervous system injury, gastritis, cancer and AIDS 12, 13. These phytochemicals naturally possess anti-oxidants. Medically antioxidants help the cells from the prevention of oxidative damage and degenerative diseases 14.
Antioxidants are the compounds which help to delay or inhibit the oxidation of lipids and other molecules through the inhibition of either initiation or propagation of oxidative chain reactions 15. Free radicals are often generated as byproducts of biological reactions or from exogenous factors. The involvements of free radicals in the pathogenesis of a large number of diseases are well-documented Pourmorad 16. Medicinal plants can protect against the harmful effects of ionizing radiation. Natural plant extracts or pure compounds are safe ingredients, which do not have any toxic effects. This paper aimed to determine the possible phytochemical components and antioxidant activity in hexane, ethyl acetate and ethanolic extract of Justicia tranquebariensis.
MATERIALS AND METHODS:
Collection of Plants: Plants for this study were collected from Chinnapaliyampattu, Tiruvannamalai district and was authenticated by Dr. Rathna Kumar, Department of Plant Biotechnology, Presidency College, Chennai - 600005.
Preparation of Extracts: Collected plants were dried at room temperature and ground to make a fine powder. 20 gm of plant powder was well dissolved in 100 ml of solvents (Hexane, ethyl acetate, and ethanol) (ratio 1:5).
The suspension was filtered by using filter paper of pore size 0.2µm. The filtrate was then air dried, and extracts were collected in sterile vials for further use.
Phytochemical Tests: The phytochemical test of these extracts was performed using the method adopted by Harborne 17 and Sofowora 18.
Test for Carbohydrates (Molisch’s Test): To 2 ml of plant extract, 1 ml of Molisch’s reagent and a few drops of concentrated sulfuric acid were added. Presence of purple or reddish color indicates the presence of carbohydrates.
Test for Tannins (Ferric Chloride Test): To 1 ml of plant extract, 2 ml of 5% ferric chloride was added. Formation of dark blue or greenish black indicates the presence of tannins.
Test for Saponins (Frothe’s Test): To 2 ml of plant extract, 2 ml of distilled water was added and shaken in a graduated cylinder for 15 min lengthwise. Formation of a 1 cm layer of foam indicates the presence of saponins.
Test for Flavonoids (Shinoda Test): To 2 ml of plant extract, 1 ml of 2N sodium hydroxide was added. Presence of yellow color indicates the presence of flavonoids.
Test for Alkaloids (Mayer’s Test): To 2 ml of plant extract, 2 ml of concentrated hydrochloric acid was added. Then a few drops of Mayer’s reagent were added. The presence of green color or white precipitate indicates the presence of alkaloids.
Test for Quinines: To 1 ml of extract, 1 ml of concentrated sulfuric acid was added. Formation of red color indicates the presence of Quinones.
Test for Glycosides (Molisch’s Test): To 2 ml of plant extract, 3 ml of chloroforms and 10% ammonia solution was added. Formation of pink color indicates the presence of glycosides.
Test for Cardiac Glycosides (Keller – Kiliani Test): To 0.5ml of extract, 2 ml of glacial acetic acid and a few drops of 5% ferric chloride were added. This was under layered with 1 ml of concentrated sulfuric acid. The formation of a brown ring at the interface indicates the presence of cardiac glycosides.
Test for Terpenoids (Salkowski Test): To 0.5 ml of extract, 2 ml of chloroform was added and concentrated sulfuric acid is added carefully. Formation of red-brown color at the interface indicates the presence of terpenoids.
Test for Triterpenoids: To 1.5 ml of extract, 1 ml of Liebmann–Buchard reagent (aecticanhydride + concentrated sulfuric acid) was added. Formation of blue-green color indicates the presence of triterpenoids.
Test for Phenols (Ferric Chloride Test): To 1 ml of the extract, 2 ml of distilled water followed by a few drops of 10% ferric chloride was added. Formation of blue or green color indicates the presence of phenols.
Test for Coumarins: To 1 ml of extract, 1 ml of 10% NaOH was added. Formation of yellow color indicates the presence of coumarins.
Steroids and Phytosteroids (Libermann-Burchard Test): To 1 ml of plant extract equal volume of chloroform is added and subjected with a few drops of the concentrated sulfuric acid appearance of brown ring indicates the presence of steroids and appearance of the bluish-brown ring indicates the presence of phytosterols.
Phlobatannins: To 1 ml of plant extract a few drops of 2% HCl was added the appearance of red color precipitate indicates the presence of phlobatannins.
Anthraquinones (Borntrager’s Test): To 1 ml of plant extract, a few drops of 10% ammonia solution were added, appearance pink color precipitate indicates the presence of anthraquinones.
Antioxidant Activity: The antioxidant activity of plant extracts was determined by, the DPPH (1,1-diphenyl-2-picryl-hydroxyl) in-vitro method.
DPPH Free Radical Scavenging Activity: The antioxidant activity of hexane, ethyl acetate and ethanolic extracts of Justicia tranquebariensis and the standard compound BHT was measured in terms of hydrogen donating radical scavenging ability using the stable DPPH method 19. 1 ml of the extract was added to 3.7mL of methanol solution. After centrifugation, the supernatant is collected 200 μml of DPPH solution is added. Kept in the dark for 45 min and the resulting decrease in absorbance at 517 nm were recorded against blank using a UV-Vis Spectrophotometer. The radical scavenging activity on DPPH was expressed as,
% DPPH radical-scavenging = Abs of control – Abs of test sample / Abs of control × 100
RESULTS: The preliminary phytochemical screening of Justicia tranquebariensis showed the presence of plant components such as carbohydrates, flavonoids, quinones and coumarins in hexane extract, carbohydrates, tannins, flavonoids, cardiac glycosides, terpenoids, phenols, coumarins and steroids in ethyl acetate extract and carbohydrates, tannins, flavonoids, quinones, cardiac glycosides, phenols and coumarins in ethanol extract Table 1.
TABLE 1: PHYTOCHEMICAL ANALYSIS OF JUSTICIA TRANQUEBARIENSIS
| S.
no. |
Phytochemical
Tests |
Test
Performed |
Hexane Extract | Ethyl Acetate Extract | Ethanol Extract |
| 1. | Carbohydrates | Molisch’stest | + | + | + |
| 2. | Tannin | Ferric chloride test | - | + | + |
| 3. | Saponin | Frothe’s test | - | - | - |
| 4. | Flavonoids | Shinoda test | + | + | + |
| 5. | Alkaloids | Mayer’s test | - | - | - |
| 6. | Quinones | - | + | - | + |
| 7. | Glycosides | Molisch’s test | - | - | - |
| 8. | Cardiac glycosides | Keller – Kiliani test | - | + | + |
| 9. | Terpenoids | Salkowski test | - | + | - |
| 10. | Phenols | Ferric chloride test | - | + | + |
| 11. | Coumarins | - | + | + | + |
| 12. | Steroids | Libermann – Burchard test | - | + | - |
| 13. | Phlobotanins | - | - | - | - |
| 14. | Anthraquinones | Borntrager’s test | - | - | - |
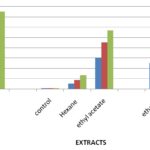
Free Radical Scavenging Assay: The stable free radical scavenging activity by the DPPH method is an easy, rapid and sensitive way to survey the antioxidant activity of specific plant extracts. Fig. 2 indicates the percentage of free radicals scavenging activity in various extractions with different concentrations 25 µg, 50 µg, and 75 µg of Justicia tranquebariensis. In this study, percentage inhibition of free radicals was carried out with different extractions of selected plants. Ethyl acetate extract with 75 μg concentrations gives a higher percentage (56.77%), ethanol extract showed the moderate activity of 36.80% and Hexane extract showed the least activity of 13.66% of free radical scavenging activity. The free radical scavenging activity increases with increase in concentration Table 2, Fig. 1. The percentage inhibition of control was found to be 81.68% which showed higher activity than the extract.
FIG. 1: ANTIOXIDANT ACTIVITY OF JUSTICIA TRANQUEBARIENSIS BY DPPH ASSAY
TABLE 2: DPPH ASSAY OF JUSTICIA TRANQUEBARIENSIS AGAINST DIFFERENT EXTRACTS
| Concentrations
(µg) |
Control | % of Inhibition | |||||
| Hexane | Ethyl acetate | Ethanol | BHT | ||||
| 25 | 0.9593 | 5.39 ± 0.78 | 30.32 ± 1.77 | 25.23 ± 1.25 | 72.36 ± 1.64 | ||
| 50 | 0.9593 | 8.85 ± 0.58 | 45.56 ± 0.45 | 31.74 ± 0.52 | 76.23 ± 0.85 | ||
| 75 | 0.9593 | 13.66 ± 0.22 | 56.77 ± 0.33 | 36.80 ± 0.39 | 81.68 ± 0.61 | ||
DISCUSSION: The results of the present study reveals that the ethyl acetate extract of Justicia tranquebariensis showed the maximum number of components such as carbohydrate, tannin is used for the treatment of skin eruption and bowel condition, flavonoids possess wound healing activity due to the astringent, antioxidant and antimicrobial properties which appear to be responsible for wound contraction and elevated rate of epithelisation 20, cardiac glycosides, terpenoids, phenols was found to be toxic to the growth and development of pathogens, coumarins and steroids in the ethanolic extract when compared to other solvents and steroids previous study reported that presence of phytosterols, flavonoids, glycosides, from the leaf extract of Justicia tranquebariensis 21.
The present work also correlates with the studies above. The ethyl acetate extract showed maximum antioxidant activity when compared to other solvents. Hence, the ethyl acetate extract can act as a maximum scavenging activity and can protect the cells from the free radicals.
CONCLUSION: The present study suggests that the Justicia tranquebariensis have the maximum number of bioactive components and higher amount of antioxidant potential in the ethyl acetate extract, therefore the ethyl acetate extract may act as a significant activity and can be further analyzed for many pathogenic disorders as well as may be helpful in the future for preventing or slowing the progress of diseases involved.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Yoganarasimhan SN: Medicinal plants of India, Regional Research Institute, Banglore, 2000: 2.
- Ainsle W: Materia indica, Longman, Rees, ORMe, Brown and Green, London, 1826; 2: 412.
- Chen CC, Hsin WC, Ko FN, Huang YL, Ou JC and Teng CM: J Nat Prod 1996; 59: 1149.
- Bhattarai NK: Fitoterapia 1993; 64: 483.
- Panthong A and Kanjanapothi D: J Ethnopharmacol 1986; 18: 213.
- Mahabir D: Am J Pub Health. 1997; 1: 174.
- Heinrich M, Kuhnt M, Wright CW, Rimplex H, Philipson JD, Schandelmaier A and WarHurst DC: Journal Ethnopharmacol. 1992; 36: 81.
- Yangfg LL, Yen KY, Kiso Y and Kikino HJ: Ethnopharmacol 1987; 19: 103.
- Fukamiya N and Lu KH: J Nat Prod 1986; 49: 348.
- Asano J, Chiba K, Tad M and Yoshi T: Phytochemistry 1996; 42: 713.
- Lino CS, Taviera ML, Viana GSB and Matos FJA: Phytotherapy Res 1997; 11: 211.
- Cook NC and Samman S: Flavonoids chemistry metabolism cardioprotective effects and dietary sources. Nutritional Biochemistry 19967; 66-76.
- Kumpulainen JT and Salonen JT: Natural antioxidants and anticarcinogens in nutrition, health and disease. (The Royal Society of Chemistry, UK) 1997; 178-187.
- Parr A and Bolwell GP: Phenols in the plant and in man: The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. J Sci Food Agric 2008; 80: 985-1012.
- Jaleel CA: Antioxidant potentials and ajmalicine accumulation in Catharathus roseus after treatment with gibberellic acid. Colloids and surfaces B: Biointerfaces, 2007; 60(2): 195-200.
- Pourmorad F: Antioxidant activity, phenol and flavonoid contents of some selected Iranian medicinal plants. African Journal of Biotechnology 2009; 5(11): 1142-1145.
- Harborne JB: Phytochemical methods: A guide to modern techniques of plant analysis. Chapman and Hall, New York, Edition 3rd, 1973: 279.
- Sofowora A: Medicinal plants and Traditional medicinal in Africa. Sunshine house, Ibadan, Nigeria: Spectrum books Ltd; Screening plants for bioactive agents; Edition 2nd, 1993: 134-156.
- Eberhardt MV, Lee CY and Liu RH: Antioxidant activity of fresh apple. Nature 2000; 405: 903-904.
- Shenoy C, Patil MB, Kumar R and Patil S: Preliminary phytochemical investigation and wound healing activity of Allium cepa (Liliaceae). Int J Pharm Pharm Sci 2009; 2: 167-175.
- Akilandeshwari, S, Mainmaran, S, Valarmathi, R, Karpagam Kumara S and Loganathan SV: Phytochemical observation on Leaf of Justicia tranquebariensis. The Ancient Science of Life. 2001; 20: 60-61.
How to cite this article:
Krishnamoorthi R and Bai VR: Phytochemical analysis and antioxidant property of Justicia tranquebariensis. Int J Pharmacognosy 2015; 2(5): 254-58. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(5).254-58.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
8
254-258
590
5256
English
IJP
R. Krishnamoorthi and V. R. Bai*
PG and Research Department of Zoology, Presidency College (Autonomous), Chennai, Tamil Nadu, India
mvragav444@yahoo.com
29 January 2015
26 March 2015
28 April 2015
10.13040/IJPSR.0975-8232.IJP.2(5).254-58
01 May 2015