PHYTOCHEMICAL ANALYSES, ANTIOXIDANT AND ANTIBACTERIAL ACTIVITIES OF ACMELLA OLERACEA, A VARIETY GROWN IN MIZORAM
HTML Full TextPHYTOCHEMICAL ANALYSES, ANTIOXIDANT AND ANTIBACTERIAL ACTIVITIES OF ACMELLA OLERACEA, A VARIETY GROWN IN MIZORAM
P. B. Lalthanpuii 1, R. Lalawmpuii 2 and K. Lalchhandama * 1
Department of Zoology 1, Pachhunga University College, Aizawl - 796001, Mizoram, India.
Department of Pharmacy 2, Regional Institute of Paramedical and Nursing Sciences, Zemabawk - 796017, Mizoram, India.
ABSTRACT: Acmella oleracea (L.) R.K. Jansen (family Asteraceae) is an interesting plant because of its wide range of medicinal and culinary uses. It has been traditionally used in the treatments of anemia, body ache, cancer, gastric ulcer and infections, gingivitis, gout, inflammations, laceration, malaria, stammering, and worm infection (helminthiasis). Different extracts of the plants were prepared, and the methanol extract showed the highest number of bioactive compounds, including carbohydrates, phytosterols, and tannins. The antioxidant activity was estimated from the rate of scavenging of the free radical, 2,2-diphenyl-1-picrylhydrazyl (DPPH). The plant extract showed similar concentration-dependent scavenging activity as that of butylated hydroxytoluene (BHT). The total antioxidant activity was estimated against ascorbic acid, and it showed a higher value than BHT. The methanol extract was treated with two Gram-negative bacteria Pseudomonas aeuroginosa and Escherichia coli, and two gram-positive bacteria Staphylococcus aureus and Bacillus subtilis. However, no antibacterial activity was observed. Nonetheless, the study provides important information on the basic pharmacological properties of the plant.
| Keywords: |
Acmella oleracea, Antioxidant activity, Medicinal plant, Antimicrobial activity
INTRODUCTION: Acmella oleracea (L.) R.K. Jansen is a perennial flowering herb belonging to the family Asteraceae. Native to Peru, it has been introduced throughout tropical and subtropical regions including Africa, America, Borneo, India, Sri Lanka, and Southeast Asia 1, 2. It has a distinct pungent smell and burning taste upon chewing. Native Americans use it as a spice for its unique aroma. In India, Brazil, and Southeast Asia, it is served as a common vegetable 3.
In different traditional medicine, it is known to have anaesthetic, anticonvulsant, antiseptic, anti-fungal, anti-protozoal, anti-diarrhoeal, analgesic, antiulcer, anti-pyretic, anti-tumor, anti-inflammatory, diuretic, aphrodisiac, and insecticidal activities 4, 5. For its antiseptic and analgesic properties, its extract is used in oral health care for the treatment of sore throat, oral ulcer, gingivitis, and general toothache. It is because of this application that it is given an English vernacular name the toothache plant 6.
A. oleracea is also used for clinical diseases such as anemia, hemorrhage, cancer, dysentery, gastro-intestinal ulcer, rheumatism, scurvy, stammering, and xerostomia and snake bite 7-9. It is also used in infectious diseases such as malaria and helminthiasis 10. Its cytotoxic, antioxidant, and vasorelaxant activities have been attributed to its use in the treatment of blood disorders 8. Its antipyretic activity against Brewer’s yeast-induced pyrexia is credited to its use in a high fever 6. It also has insecticidal activity against the pest Tuta absoluta 11, and vectors of infectious diseases including Aedes aegyptii 12.
In Indian medicine, it is used as an effective aphrodisiac, and is being prescribed for cases of impotence 2. All these medicinal applications have been supported from its pharmacological properties such as anaesthetic, anti-inflammatory, analgesic, antipyretic, anti-obesity and diuretic activities 13-15. The anti-inflammatory activity has been demonstrated in experimental rats 14. It is also experimentally shown to increase the number of macrophages, specialized white blood cells vital for various immunological responses. This supports the use of the plant in the treatment of rheumatism. 16 This study, therefore, aims at chemical analysis and tests for some biological activities of the plant.
MATERIALS AND METHODS:
Collection of Plant Material: A. oleracea was collected from the plantation field in the village of Ngopa, Champhai District, Mizoram, India (located between 23.8861° latitude north and 93.2119° longitude east) in 2015. A voucher specimen was identified and is maintained at the herbarium section of the Department of Botany, Pachhunga University College, Aizawl, Mizoram. The aerial parts, i.e., leaves and flowers, of the plant were dried in a thermostat oven at 45 °C.
Preparation of Plant Extracts: The dried plants were pulverized to a powder using mortar and pestle. The plant powder was subjected to continuous hot extraction in a Soxhlet apparatus using different solvents of increasing polarity, namely hexane, chloroform, and methanol. Hexane extraction was run for 48 h, while chloroform and methanol extraction required 72 h. The extracts were concentrated in a vacuum rotary evaporator (Buchi Rotavapor® R-215). The plant extracts were produced in the form of semi-solid mass and were refrigerated at 4 °C until further use.
Phytochemical Detection: The various chemical components were screened using standard protocols. Alkaloids were tested by Meyer’s test, and Dragendroff’s test; carbohydrates by Wagner’s test, Hager’s test, Molisch’s test, Fehling’s test, Barfoed’s test, and Benedict’s test; phytosterols by Liebermann-Burchard’s test, and Salkwoski reaction; glycosides by Legal’s test, and Keller Killian's test; tannins by FeCl3 test, K2Cr3O7 test, and lead acetate test; saponin by foam test; reducing sugars by Fehling’s test, and Benedict’s test; flavonoids by Shinoda test, and zinc hydrochloride reduction test; and amino acids by Biuret test, and ninhydrin test.
Antimicrobial Activity: The antimicrobial activity was determined using a disk diffusion method. Two Gram-negative bacteria Pseudomonas aeuroginosa and Escherichia coli, and two Gram-positive bacteria Staphylococcus aureus and Bacillus subtilis were used. The bacteria were grown in culture dishes containing Mueller-Hinton agar. The plant extracts were impregnated on absorbent disks (Whatman Antibiotic Assay Discs). The control experiment was maintained containing the bacteria culture only. For standard reference, tetracycline was used. The experiment was prepared in triplicate. The culture dishes were maintained at a 37 ± 1 °C. After 20 h, the size of bacterial growth and the corresponding inhibition zones were noted.
Antioxidant Activity: Estimation of the antioxidant activity followed the method of Blois 17. Briefly, a stable free radical DPPH (2, 2-diphenyl-1-picrylhydrazyl) was used as a substrate. Antioxidant activity was shown by the scavenging of DPPH. 1 ml of 0.1 mM solution of DPPH in methanol and 3 ml of the methanol extract were incubated at 37 ± 1 °C for 30 min. For the standard reference, butylated hydroxytoluene (BHT) was used. Absorbance was measured at 517 nm against control in a UV-Visible spectrophotometer (EvolutionTM, Thermo Scientific). The percentage of inhibition was calculated by comparing the absorbance values of the test samples with those of the controls. The inhibition percentage (I) was calculated using the formula:
I = (Abs control – Abs sample) / Abs control × 100
Total Antioxidant Activity: The total antioxidant activity was determined by phosphomolybdate estimation using ascorbic acid as a standard. 0.1 ml of sample solution was mixed with 1 ml of reagent solution (0.6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate). After incubation at 95 °C, absorbance was measured at 695 nm.
RESULTS: Biochemical detections using various methods show the presence of important phytochemicals in A. oleracea such as phytosterols, tannins, reducing sugars and carbohydrates Table 1. The methanol extract indicated a maximum variety of compound including phytosterols, tannins, reducing sugars and carbohydrates in all the tests, except in Molisch’s test for carbohydrate. The hexane showed the presence of phytosterols in both Liebermann-Burchard’s and Salkwoski tests. However, other important phytochemicals such as alkaloids, flavonoids, and saponins could not be detected in any of the tests used.
TABLE 1: CHEMICAL COMPOSITION OF A. OLERACEA USING DIFFERENT BIOCHEMICAL TESTS
| S. no. | Phytochemicals | Name of test | Hexane | Chloroform | Methanol |
| 1 | Alkaloids | Meyer’s test | - | - | - |
| Dragendroff’s test | - | - | - | ||
| Wagner’s test | - | - | - | ||
| Hager’s test | - | - | - | ||
| 2 | Carbohydrates | Molisch’s test | - | + | - |
| Fehling’s test | - | + | + | ||
| Barfoed’s test | - | - | + | ||
| Benedict’s test | - | + | + | ||
| 3 | Phytosterols | Liebermann Burchard’s test | + | - | + |
| Salkwoski reaction | + | - | + | ||
| 4 | Glycosides | Legal’s test | - | - | - |
| Keller Killian's test | - | - | - | ||
| 5 | Tannin | FeCl3 test | - | - | + |
| K2Cr3O7 test | - | - | + | ||
| Lead acetate test | - | - | + | ||
| 6 | Saponins | Foam test | - | - | - |
| 7 | Reducing sugars | Fehling’s test | - | + | + |
| Benedict’s test | - | - | + | ||
| 8 | Flavonoid | Shinoda test | - | - | - |
| Zinc hydrochloride reduction test | - | - | - | ||
| 9 | Proteins and amino acids | Biuret test | - | - | - |
| Ninhydrin test | - | - | - |
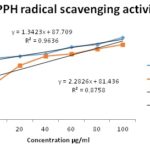
FIG. 1: ANTIOXIDANT ACTIVITY OF A. OLERACEA USING SCAVENGING ACTIVITY OF 2,2-DIPHENYL-1-PICRYLHYDRAZYL (DPPH)
The antioxidant activity was estimated by the method of DPPH free radical scavenging assay using the methanol extract Fig. 1. Increasing concentrations of the plant extract were prepared from 10, 20, 40, 60, 80 to 100 µg/ml. Exactly similar concentrations were used for the reference compound BHT. Both the extract and BHT showed concentration-dependent activity against DPPH, i.e., increased scavenging activity with increased concentration. BHT appeared to be more potent than the plant extract at all concentrations tested. IC50 of standard BHT and the plant extract was calculated from the standard graph. The plant extract showed IC50 of 13.773 mg/ml while for BHT it was 28.098 mg/ml. The total antioxidant activity was estimated as ascorbic acid equivalent, i.e. 85 mg/ml.
Antimicrobial activity was assessed upon two commonly available bacteria, viz. Pseudomonas aeuroginosa and Escherichia coli (both Gram-negative), and Staphylococcus aureus and Bacillus subtilis (both gram-positive). The antibiotic tetracycline was used as a positive treatment. Using the equivalent concentrations (10 and 20 mg/ml) as that of the drug, the methanol extract of the plant did not produce any significant zones of inhibition, all the bacteria grew normally; while the standard drug showed a distinct zone of inhibition.
DISCUSSION AND CONCLUSION: In the present study, the presence of important bioactive compounds in A. oleracea was confirmed. Phytosterols, tannins, reducing sugars, and carbohydrates were detected. The leaves of A. oleracea have been reported to contain alkamides, amides, carbohydrates, tannins, steroids, carotenoids, essential oils, sesquiterpenes and amino acids, 8, 18 phytosterols (e.g. β-sitosterol, stigmasterol, α- and β-amyrins), essential oils (e.g. limonene and β-caryophyllene), sesquiterpenes, α- and β-bisabolene and cadinenes, flavonoid glucoside and a mixture of long chain hydro-carbons 1, 19. The most well-known phytosterols such as β-sitosterol, stigmasterol, and campesterol, are well established to be pharmacologically beneficial for their therapeutic actions in cardio-vascular diseases, colon and breast cancer 20. The methanol extract exhibited concentration-dependent antioxidant activity like that of butylated hydroxytoluene (BHT). Although, its overall activity was lower than that of BHT, it showed higher total activity with an IC50 of 13.773 mg/ml, compared to BHT having an IC50 of 28.098 mg/ml. Free radicals are responsible for physiological oxidative stress, which is the cause of health problems including several cardiovascular, neurodegenerative, cancer and even aging 21. Antioxidants are required to downplay the oxidation process by converting the harmful free radicals to harmless molecules or by destroying them 22. Hence, dietary antioxidants are the principal sources of defense for cellular oxidation 23, 24. Antimicrobial activity was not observed against four bacteria, P. euroginosa and E. coli, S. aureus and B. subtilis. However, is has been reported that closely related species showed antibacterial activity against Klebsiella pneumonia, 25 Streptococcus pyogenes 8, Salmonella typhi 26. But, similar to the present study, Prachayasittikul et al. 8 found no antibacterial activity for Spilanthes acmella. These data coupled with the present study suggest that the antibacterial property of A. oleracea may be highly species specific.
ACKNOWLEDGEMENT: The study is funded by the University Grants Commission’s Major Research Project [MRP-BIOC-2013-36855, sanction F. No. 43-47/2014(SR) of 22/8/2015] to KLC. PBL is a UGC Project Fellow. The authors are grateful to Dr. H. Lalhlenmawia, Head, Department of Pharmacy, RIPANS, for providing all the facilities and technical support.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Tiwari KL, Jadhav SK and Joshi V: An updated review on medicinal herb genus Spilanthes. Zhong Xi Yi Jie He Xue Bao (Journal of Chinese Integrated Medicine) 2011; 9: 1170-1178.
- Prachayasittikul V, Prachayasittikul S, Ruchirawat S and Prachayasittikul V: High therapeutic potential of Spilanthes acmella: a review. EXCLI Journal 2013; 12: 291-312.
- Lim TK: Edible Medicinal and Non-Medicinal Plants: Flowers. Springer Science+Business Media Dordrecht, Netherlands 2014; 7: 163-174.
- Dubey S, Maity S, Singh M, Saraf SA and Saha S: Phytochemistry, pharmacology, and toxicology of Spilanthes acmella: a review. Advances in Pharmacological Sciences 2013: 423750.
- Paulraj J, Govindarajan R and Palpu P: The genus Spilanthes ethnopharmacology, phytochemistry, and pharmacological properties: a review. Advances in Pharmacological Sciences 2013; 510298.
- Chakraborty A, Devi BR, Sanjebam R, Khumbong S and Thokchom IS: Preliminary studies on local anaesthetic and antipyretic activities of Spilanthes acmella in experimental animal models. Indian Journal of Pharmacology 2010; 42: 277-279.
- Duke JA: Duke's Handbook of Medicinal Plants of Latin America. CRC Press, Boca Raton, Florida USA, 2008: 13-14.
- Prachayasittikul S, Suphapong S, Worachartcheewan A, Lawung R, Ruchirawat S and Prachayasittikul V: Bioactive metabolites from Spilanthes acmella Molecules 2009; 14: 850-867.
- Dubey S, Maity S, Singh M, Saraf SA and Saha S: Phytochemistry, pharmacology, and toxicology of acmella: a review. Advances in Pharmacological Sciences 2013; 423750.
- Spelman K, Depoix D, McCray M, Mouray E and Grellier P: The traditional medicine Spilanthes acmella, and the alkylamides spilanthol and undeca‐2e‐ene‐8, 10‐diynoic acid isobutylamide, demonstrate in-vitro and in-vivo antimalarial activity. Phytotherapy Research 2011; 25: 1098-1101.
- Moreno SC, Carvalho GA, Picanço MC, Morais EG and Pereira RM: Bioactivity of compounds from Acmella oleracea against Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae) and selectivity to two non‐target species. Pest Management Science 2012; 68: 386-393.
- Simas NK, Dellamora EDCL, Schripsema J, Lage CLS, de Oliveira Filho AM, Wessjohann L, Porzel A and Kuster RM: Acetylenic 2-phenylethylamines and new isobutyl-amides from Acmella oleracea (L.) RK Jansen, a Brazilian spice with larvicidal activity on Aedes aegypti. Phytochemistry Letters 2013; 6: 67-72.
- Ratnasooriya WD, Pieris KP, Samaratunga U and Jayakody JR: Diuretic activity of Spilanthes acmella flowers in rats. Journal of Ethnopharmacology 2004; 91: 317-320.
- Chakraborty ARKB, Devi RK, Rita S, Sharatchandra KH and Singh TI: Preliminary studies on anti-inflammatory and analgesic activities of Spilanthes acmella in experimental animal models. Indian Journal of Pharmacology 2004; 36: 148-150.
- Wu LC, Fan NC, Lin MH, Chu IR, Huang SJ, Hu CY and Han SY: Anti-inflammatory effect of spilanthol from Spilanthes acmella on murine macrophage by down-regulating LPS-induced inflammatory mediators. Journal of Agricultural and Food Chemistry 2008; 56: 2341-2349.
- Savadi RV, Yadav R and Yadav N: Study on immunomodulatory activity of ethanolic extract of Spilanthes acmella Leaves. Indian Journal of Natural Products and Resources 2010; 1: 204-207.
- Blois MS: Antioxidant determinations by the use of a stable free radical. Nature 1958; 181: 1199-1200.
- Nagashima M and Nakatani N: LC-LS analysis and structure determination of pungent alkamides from Spilanthes acmella flowers. Lebensmittel – Wissenschaft + Technologie 1992; 25: 417–421.
- Sahu J, Jain K, Jain B and Sahu RK: A review on phytopharmacology and micropropagation of Spilanthes acmella. Pharmacologyonline Newslett 2011; 2: 1105-1110.
- Choudhary S and Tran L: Phytosterols: perspectives in human nutrition and clinical therapy. Current Medicinal Chemistry 2011; 18: 4557-4567.
- López-Alarcón C and Denicola A: Evaluating the antioxidant capacity of natural products: A review of chemical and cellular-based assays. Analytica Chimica Acta 2013; 763: 1-10.
- Young IS and Woodside JV: Antioxidants in health and disease. Journal of Clinical Pathology 2001; 54: 176-186.
- Halliwell B: Antioxidants in human health and disease. Annual Review of Nutrition 1996; 16: 33-50.
- Carocho M and Ferreira IC: A review on antioxidants, prooxidants and related controversy: natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food and Chemical Toxicology 2013; 51: 15-25.
- Arora S, Vijay S and Kumar D: Phytochemical and antimicrobial studies on the leaves of Spilanthes acmella. Journal of Chemical and Pharmaceutical Research 2011; 3: 145-150.
- de Alcantara BN, Kobayashi YT, Barroso KF, da Silva IDR, de Almeida MB and Barbosa WLR: Pharma-cognostic analyses and evaluation of the in vitro antimicrobial activity of Acmella oleracea (L.) RK Jansen (Jambu) floral extract and fractions. Journal of Medicinal Plants Research 2015; 9: 91-96.
How to cite this article:
Lalthanpuii PB, Lalawmpuii R and Lalchhandama K: Phytochemical analyses, antioxidant and antibacterial activities of Acmella oleracea, a variety grown in Mizoram. Int J Pharmacognosy 2017; 4(4): 118-22. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(4).118-22.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
118-122
526
1423
English
IJP
P. B. Lalthanpuii, R. Lalawmpuii and K. Lalchhandama *
Department of Zoology, Pachhunga University College, Aizawl, Mizoram, India.
chhandama@gmail.com
13 January 2017
23 March 2017
25 March 2017
10.13040/IJPSR.0975-8232.IJP.4(4).118-22
01 April 2017