PHLORIZIN: A COMPREHENSIVE REVIEW OF ITS PHARMACOLOGY, MECHANISMS, AND THERAPEUTIC POTENTIAL
HTML Full TextPHLORIZIN: A COMPREHENSIVE REVIEW OF ITS PHARMACOLOGY, MECHANISMS, AND THERAPEUTIC POTENTIAL
Nyeme Konyak, Ranjan Kumar Singh *, Neeraj Jain and Tanya Sharma
Department of Pharmaceutical Sciences, Mewar University, Gangrar, Chittorgarh, Rajasthan, India.
ABSTRACT: A naturally occurring dihydrochalcone glycoside, Phlorizin, is mostly found in apple tree species and other Rosaceae family members. Its diverse range of biological actions, including antidiabetic, antioxidant, anti-inflammatory, cardioprotective, nephroprotective, neuroprotective, and anti-obesity properties, has garnered significant scientific attention. The creation of contemporary SGLT2 inhibitor medications used to treat type 2 diabetes mellitus was made possible by phlorizin, which is historically acknowledged as the first inhibitor of sodium–glucose cotransporters (SGLTs) to be discovered. Phlorizin has strong pharmacological activity; however, its fast hydrolysis and poor oral bioavailability limit its therapeutic use. Phlorizin's chemical structure, physicochemical characteristics, biosynthesis, extraction methods, pharmacokinetics, pharmacodynamics, mechanisms of action, therapeutic potentials, toxicity profile, formulation strategies, and future research are all covered in detail in this study. This study provides a comprehensive overview of phlorizin, including its chemical structure, physicochemical properties, biosynthesis, extraction techniques, pharmacokinetics, pharmacodynamics, mechanisms of action, therapeutic potentials, toxicity profile, formulation strategies, and future research prospects. Consolidating existing knowledge and highlighting phlorizin's translational potential in pharmaceutical and biomedical sciences are the goals of this review.
Keywords: Phlorizin, SGLT inhibitors, Antidiabetic agents, Natural products, Pharmacology, Phytochemistry
INTRODUCTION: Natural products continue to be one of the most significant sources of new therapeutic agents and have historically served as the basis for contemporary pharmacology. Natural goods, their derivatives, or synthetic molecules modelled after natural scaffolds are thought to make up around half of all approved medications.
Particularly, plants produce a vast array of secondary metabolites with physiological, ecological, and defence functions; many of these compounds have strong pharmacological effects on people. Polyphenols are a significant class of secondary metabolites that have been shown to have anti-inflammatory, anti-microbial, antidiabetic, cardioprotective, antioxidant, and anticancer effects 1-2.
Flavones, flavanols, flavanones, isoflavones, anthocyanidins, and dihydrochalcones are all subclasses of flavonoids, a broad subclass of polyphenols distinguished by a C6–C3–C6 carbon skeleton. A saturated three-carbon linker between two aromatic rings distinguishes dihydrochalcones, which are very rare in nature but have special biological properties related to metabolic diseases 3. The most researched dihydrochalcone glycoside is phenolizing, which has maintained scientific interest because of its significant impact on glucose metabolism. One of the biggest worldwide public health issues of the twenty-first century is diabetes mellitus. Recent epidemiological data show that type 2 diabetes mellitus accounts for almost 90% of cases, and the prevalence of diabetes is still rising globally. Chronic hyperglycaemia raises morbidity and mortality by causing long-term problems with the kidneys, eyes, neurological system, and cardiovascular system 4. Although there are many antidiabetic medication classes, drawbacks such as weight gain, gastrointestinal intolerance, hypoglycaemia risk, and diminishing effectiveness underscore the necessity for novel treatment approaches.
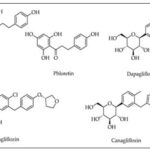
Phlorizin holds a special place in the annals of antidiabetic medication development. Instead of increasing insulin production or enhancing insulin sensitivity, it was the first substance to be demonstrated to lower blood glucose levels by blocking renal glucose reabsorption. The creation of sodium–glucose cotransporter 2 (SGLT2) inhibitors, which are currently a mainstay of type 2 diabetes treatment, was directly influenced by this discovery, which identified the kidney as a feasible pharmaceutical target for glycaemic control 5. Phlorizin has a wide range of biological benefits in addition to its antidiabetic properties, such as antioxidant, anti-inflammatory, lipid-lowering, cardioprotective, nephroprotective, and neuroprotective properties. These pleiotropic effects imply that phlorizin may be useful as a treatment for a number of chronic illnesses linked to metabolic malfunction and oxidative stress 6. Phlorizin's chemical properties, natural sources, historical history, pharmacokinetics, pharmacodynamics, molecular mechanisms, therapeutic potentials, safety profile, and potential for further research are all included in this review's thorough and critical assessment. The structures of phlorizin, phloretin, and some selective SGLT2 inhibitors are given below in the Fig. 1.
FIG. 1: STRUCTURES OF PHLORIZIN, PHLORETIN, AND SOME SELECTIVE SGLT2 INHIBITORS
Overview of Phlorizin: A naturally occurring dihydrochalcone glycoside, phlorizin (also spelled phlorizin), is mostly found in apple trees (Malus domestica) and other Rosaceae family members. Phloretin-2′-O-β-D-glucopyranoside, as it is chemically known, is one of the first known plant-derived inhibitors of renal glucose transport pathways. The substance has traditionally been essential in expanding our knowledge of renal physiology and glucose balance. Phlorizin was first identified as a botanical component in the nineteenth century, but it later became a key pharmacological tool that helped create the contemporary sodium-glucose cotransporter-2 (SGLT2) inhibitors that are now commonly prescribed for the treatment of type 2 diabetes mellitus (T2DM) and cardiovascular protection 7-9.
Phlorizin is structurally composed of a hydrophilic glucose molecule connected to a hydrophobic aglycone moiety, phloretin, by a β-glycosidic connection at the 2′ position. Amphipathic qualities are conferred by this structural arrangement, which affects the drug's pharmacokinetic and biological activity.
Phlorizin functions as a competitive inhibitor of sodium-glucose cotransporters found in the intestinal epithelium and renal proximal tubule. It lowers plasma glucose levels without the help of insulin signalling pathways by preventing the kidneys from reabsorbing glucose, which results in glucosuria 8.
In a number of experimental systems, phlorizin has shown antioxidant, anti-inflammatory, lipid-modulating, cardioprotective, nephroprotective, and neuroprotective qualities in addition to its glucose-lowering activities 10–12.
As a multipurpose bioactive substance with potential uses in metabolic syndrome, cardiovascular disease, obesity, and chronic kidney disease, phlorizin has rekindled interest due to its pleiotropic properties.
Phlorizin itself is not clinically used because of pharmacokinetic limitations, but it was the structural model for selective SGLT2 inhibitors like canagliflozin and dapagliflozin, which were logically developed to get around its lack of selectivity between SGLT1 and SGLT2 and poor oral bioavailability 9-13. Phlorizin thus holds a special place in the nexus of contemporary pharmacotherapy, renal physiology, and phytochemistry.
Historical Background and Discovery: In 1835, the French chemist De Koninck discovered phenol for the first time from apple tree root bark 14. It was not until the late nineteenth century that its physiological effects were completely understood, as it was initially just considered a botanical glycoside. Von Mering reported in 1886 that giving phlorizin to animals caused glucosuria without causing noticeable hyperglycaemia 15. The prevalent knowledge of diabetes pathogenesis at the time was called into question by this contradictory behaviour. The so-called "phlorizin diabetes" model was produced in the early 20th century by subsequent research, when experimental animals experienced severe glycosuria and temporary metabolic changes that resembled diabetes mellitus 16.
Crucially, this model showed that renal glucose reabsorption was not passive diffusion but rather an active, saturable transport process. Phlorizin's mechanism of action was not fully understood until the 1980s, when sodium-dependent glucose transporters (SGLTs) were discovered and cloned. The successful cloning of SGLT1 by Hediger and others in 1987 provided molecular proof of sodium-coupled glucose transport in epithelial tissues 17.
Subsequent analysis of SGLT2 showed that it is mostly expressed in the renal proximal tubule's S1 segment, which is in charge of reabsorbing about 90% of filtered glucose 18.
Phlorizin was later found to be a competitive inhibitor of SGLT1 and SGLT2. However, its limited therapeutic application was caused by its poor oral bioavailability and lack of selectivity. However, its molecular architecture sparked medicinal chemistry research that eventually produced derivatives of C-glucoside with enhanced transporter selectivity and metabolic stability 13-19.
From plant extract to physiological probe to structural prototype for a significant class of contemporary antidiabetic medicines, phlorizin's historical trajectory therefore demonstrates a remarkable progression.
Sources and Natural Occurrence: Phlorizin is typically found in Rosaceae species, especially in pear trees (Pyrus communis), apple trees (Malus domestica), and crab-apple species. The bark, roots, and leaves are where it is most prevalent, whereas the fruit pulp and peel have lesser concentrations 20-21. The source of Phlorizin and Phlorizin containing plant species (i.e., an apple) is shown in the Table 1.
TABLE 1: PHLORIDZIN-CONTAINING PLANT SPECIES
| Plant species | Tissue | Extraction Solvent | References |
| Acanthopanax senticosus | Root | Methanol | [17] |
| Acca sellowiana | Flesh | Methanol | [18] |
| Aspalathus linearis | Leaf | Methanol and Water (1% Formic acid) | [19] |
| Aspalathus pendula | Leaf | Methanol and Water (1% Formic acid) | [19] |
| Berberis jaeschkeana | Flesh | Methanol | [20] |
| Camellia japonica | Leaf | Water | [21] |
| Docyni adcne | Leaf | Methanol and Acetonitrile | [22] |
| Docynia indica | Leaf | Ethyl acetate, n-hexane, n-butanol and Methanol | [23] |
| Eleutherococcus senticosus | Root | Methanol | [24] |
| Fragaria xananassa | Flesh | Acetone | [25] |
| Glycine max | Seed | Ethanol | [26] |
| Hemerocallis hybrida | Flower | Hexane, Ethylacetate and Methanol | [27] |
| Lactuca sativa | Flesh | Water | [28] |
| Lithocarpus polystachyus | Leaf | Ethanol, Methanol | [29-32] |
| Lippia graveolens | Leaf | Methanol | [33] |
| Nelumbo nucifera | Seed | Ethanol | [34] |
| Pisum sativum | Seed | Acetone | [35] |
| Polygonum cuspidatum | Flower | Methanol | [36] |
| Prunus persica | Flesh | Methanol and Water with Formic acid | [37] |
| Psidium guajava | Peel and Flesh | Methanol and Water with Formic acid | [38] |
| Punica granatum | Flesh | [39] | |
| Pyrus pashia | Flesh | Ethanol | [40] |
| Rosa canina | Flesh | Methanol and Water with Formic acid | [41] |
| Rubus occidentalis | Flesh | Methanol | [42] |
| Solanum lycopersicum | Flesh | Methanol | [43] |
| Sechium edule | Flesh | Methanol | [44] |
| Vaccinium vitis-idaea | Flesh | Methanol, Ethanol, Formic acid, Acetone, Ethylacetate, Water, | [45] |
Phlorizin content has been shown to vary significantly according to cultivar, developmental stage, environmental factors, and agricultural practices, according to quantitative assessments. While apple fruit normally contains 20–100 mg/kg fresh weight, apple bark can have concentrations as low as 5–15% of dry weight 20-22.
A very rich and profitable source for extraction is apple pomace, a by-product of making juice 23. Phlorizin serves as a phytoalexin in plants and supports defence systems against herbivores and infections. By adjusting the oxidative equilibrium in plant tissues, it also contributes to UV protection and stress tolerance 24.
Phlorizin is widely consumed through apples, which has led to research into how it might contribute to the well-established cardiometabolic advantages of diets high in apples. Phlorizin's biological activity, pharmacokinetics, and formulation difficulties can only be explained by an understanding of its chemical structure and physical properties. The solubility, stability, membrane permeability, and interaction with biological targets are all influenced by the presence of numerous hydroxyl groups, glycosidic linkages, and aromatic rings.
Chemical Structure and Physicochemical Properties:
Chemical Classification: Phlorizin is a member of the dihydrochalcone class of polyphenolic chemicals, which is a subclass of flavonoids distinguished by the lack of the heterocyclic C-ring that is characteristic of flavones and flavanols. It has a molecular weight of 436.41 g/mol and the formula C21H24O10 25. Dihydrochalcones are made up of two aromatic rings joined by a three-carbon bridge and are produced via the phenylpropanoid pathway. In phlorizin, a β-D-glucopyranoside bond glycosylates the aglycone phloretin (C15H14O5) at the 2′ hydroxyl position.
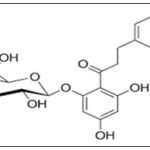
Phlorizin's placement in the flavonoid family is noteworthy because polyphenolic substances are well known for their cardioprotective, anti-inflammatory, and antioxidant properties 26. However, phlorizin's particular high-affinity interaction with sodium-glucose cotransporters sets it apart from other flavonoids. The chemical structure of Phlorizin has been shown in the Fig. 2.
FIG. 2: CHEMICAL STRUCTURE OF PHLORIZIN
Structural Characteristics:
The structural architecture of phlorizin comprises:
- Two phenolic rings (A and B rings)
- Multiple hydroxyl substituents
- A saturated three-carbon linker
- A β-D-glucose moiety
Having several hydroxyl groups gives you the ability to form strong hydrogen bonds and scavenge radicals. These hydroxyl groups contribute to antioxidant activities by giving hydrogen atoms to counteract reactive oxygen species (ROS) 26. Pharmacokinetics is greatly influenced by the glycosidic bond. Glycosylation decreases membrane permeability but increases aqueous solubility. Intestinal β-glucosidases hydrolyse phlorizin in the gastrointestinal tract to produce phloretin, which is less selective for SGLT2 but more lipophilic and easily absorbed 27. Phlorizin's glucose moiety interacts with the sodium-binding pocket of SGLT transporters, according to molecular docking studies, whereas its aromatic rings interact with hydrophobic residues in the transporter channel 28. Its competitive inhibitory behaviour can be explained by this dual interaction.
Stability, Solubility, and Bioavailability: Phlorizin has weak lipid solubility and moderate water solubility. Although it is prone to hydrolysis in alkaline settings and when β-glucosidases are present, it remains chemically stable in acidic situations 27.
Oral bioavailability is low due to:
- Rapid intestinal hydrolysis
- First-pass metabolism
- Limited intestinal permeability
- Efflux transporter activity
The small intestine primarily transforms phlorizin into phloretin after oral treatment. Before entering the bloodstream, phloretin goes through additional hepatic conjugation, including glucuronidation and sulfation 29.
The prolonged pharmacological action in rodent models is limited by the plasma half-life, which varies from 30 to 60 minutes 30. The creation of C-glucoside SGLT2 inhibitors, which substitute a more metabolically stable carbon-carbon bond for the O-glycosidic bond, was directly driven by these pharmacokinetic issues.
Biosynthesis and Natural Extraction Methods:
Phytochemical Pathways in Plants: The phenylpropanoid–flavonoid system, a highly conserved metabolic pathway, is the mechanism by which phenolizing is biosynthesized in plants. This pathway produces a wide range of polyphenolic chemicals that are important in pigmentation, UV protection, stress adaptation, and pathogen defence. Phlorizin, in contrast to other flavonoids, is a member of the dihydrochalcone subclass, which is produced by specific branch-point alterations in the flavonoid biosynthesis cascade 31.
The amino acid L-phenylalanine is the starting point of the biosynthesis pathway. It is deaminated by the enzyme phenylalanine ammonia-lyase (PAL) to produce cinnamic acid. Cinnamate-4-hydroxylase (C4H) subsequently hydroxylates cinnamic acid to yield p-coumaric acid, which 4-coumarate-CoA ligase (4CL) subsequently activates to p-coumaroyl-CoA 32.
At this point, naringenin chalcone, the primary precursor of flavonoids, is produced by chalcone synthase (CHS) catalysing the condensation of p-coumaroyl-CoA with three malonyl-CoA molecules. Chalcone reductase (CHR), a specialized enzyme, alters the intermediate structure in apple species and kindred Rosaceae plants, forming phloretin instead of traditional flavanones 33. UDP-glucose: phloretin 2′-O-glycosyltransferase (P2′GT) glycosylates phloretin, the aglycone of phlorizin, at the 2′-hydroxyl position to produce phlorizin. Because it improves solubility, stability, and intracellular compartmentalization within plant vacuoles, this glycosylation process is essential 34. Phlorizin production is tissue-specific, according to gene expression studies in Malus domestica, with higher expression of CHS and glycosyltransferases in bark and roots than in fruit flesh 35. Environmental stresses like wounding, UV light, and pathogen invasion dramatically boost the enzymes involved in the phenylpropanoid pathway, which increases the buildup of phlorizin 36. The ecological role of phlorizin includes:
- Antimicrobial protection against fungus-related infections
- Adjusting the reactions to oxidative stress
- Preventing herbivores
- Control of signals for plant growth
Numerous glycosyltransferase isoforms have been found through recent transcriptome investigations to contribute to the structural diversity of dihydrochalcones in apple cultivars 37. These results provide opportunities for metabolic engineering techniques to increase the output of phlorizin for use in industrial settings.
Extraction and Isolation Techniques: Phlorizin's sensitivity to oxidation and hydrolysis necessitates careful optimization during its extraction from plant material. While modern extraction techniques increasingly include green chemistry approaches for increased sustainability and efficiency, traditional extraction methods have mostly depended on organic solvent extraction.
Conventional Solvent Extraction: Because of their polarity and compatibility with phenolic chemicals, methanol and ethanol are the most widely used solvents for phlorizin extraction. Although Soxhlet extraction is still a traditional method, it has limitations due to its lengthy extraction durations and potential for thermal damage 38.
Although cold maceration and reflux extraction are also used, the extraction yield is greatly influenced by the solvent composition, which is usually 70–80% ethanol-water mixtures 39.
Ultrasound-Assisted Extraction (UAE): Through cavitation effects, ultrasound waves break down plant cell walls, improving solvent penetration and mass transfer. UAE improves phlorizin recovery while drastically cutting down on extraction time and solvent usage 40.
Extraction Assisted by Microwaves (MAE): Microwave irradiation increases the efficiency of dihydrochalcone extraction by accelerating solvent heating and cellular disintegration. Optimizing microwave power and exposure time is necessary to prevent thermal degradation 41.
Supercritical Fluid Extraction: Ethanol is frequently used as a cosolvent in supercritical CO₂ extraction, which offers an environmentally beneficial, solvent-free substitute. Despite phlorizin's relative polarity, its extraction efficiency at supercritical conditions is increased by the inclusion of modifiers 42.
Purification Methods:
Purification after extraction usually entails:
- Liquid-liquid separation
- Column chromatography using Sephadex LH-20 and silica gel
The gold standard for isolation and purification is still preparative high-performance liquid chromatography (HPLC) using reverse-phase C18 columns 43.
Analytical Methods for Quantification: For pharmaceutical and commercial purposes, precise measurement of phlorizin is crucial.
Liquid Chromatography with High Performance (HPLC): Because aromatic phenolic structures have high absorbance, reverse-phase HPLC with UV detection at 280 nm is frequently used for quantification 44. Separation is improved by gradient elution utilizing acetonitrile–water systems that have been acidified with formic acid.
Mass Spectrometry and Liquid Chromatography (LC-MS/MS): Superior sensitivity and structural confirmation are offered by LC-MS/MS. Commonly used in negative mode, electrospray ionization (ESI) detects the deprotonated molecular ion [M–H] ⁻ at m/z 435 45.
The Capillary Electrophoresis Method: Although sensitivity may be lower than LC-MS techniques, this technology enables quick analysis and little solvent consumption 46.
Methods of Spectrophotometry: Total phenolic tests, like Folin–Ciocalteu, are occasionally employed for initial screening of plant extracts, despite their lower specificity. Overall, the uniformity and repeatability of phlorizin quantification have been greatly enhanced by analytical developments.
Pharmacokinetics of Phlorizin:
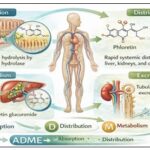
Absorption, Distribution, Metabolism, and Excretion (ADME): Phlorizin's pharmacokinetic profile is typified by its poor oral bioavailability and fast metabolism, which have historically hindered its therapeutic development. The pharmacokinetics of phlorizin (ADME) are shown below in a Fig. 3.
FIG. 3: PHARMACOKINETICS OF PHLORIZIN (ADME)
Absorption: Intestinal lactase-phlorizin hydrolase (LPH), a brush-border β-glucosidase, hydrolyses oral phlorizin extensively, resulting in the production of glucose and phloretin 47. As a result, following oral dosage, intact phlorizin is rarely found in the systemic circulation. Because it is more lipophilic, phenacetin is absorbed by passive diffusion; nevertheless, it does not have the parent compound's transporter selectivity.
Distribution: Phlorizin quickly enters the hepatic and renal organs after intravenous treatment. Moderate protein binding occurs in plasma, mostly through hydrogen bonding and hydrophobic interactions with albumin 48.
Metabolism:
The liver's phase II metabolism of phenacetin includes:
- Glucuronidation
- Sulfation
- Methylation
These conjugates are quickly eliminated and have lower pharmacological activity 49.
Excretion: Urine contains both conjugated metabolites and trace levels of the original substance, indicating that renal excretion is the main clearance pathway 50.
Plasma Half-Life and Metabolic Fate: Phlorizin has a plasma half-life of 30 to 60 minutes after intravenous injection in rodent studies 7-30. Systemic exposure is limited by quick conjugation and hydrolysis. Several glucuronide conjugates of phloretin have been found in plasma and urine by metabolomic profiling 50. Modern SGLT2 inhibitors had to undergo structural changes to improve stability due to the short half-life.
Factors Affecting Pharmacokinetic Profile: Phlorizin pharmacokinetics are influenced by many factors:
- Activity of intestinal β-glucosidase
- Composition of the gut microbiota
- Effects of dietary matrix
- The formulation approach
- The administrative route
By shielding the glycosidic link from enzymatic hydrolysis, nano formulation techniques have shown enhanced systemic exposure 51.
Pharmacodynamics and Mechanisms of Action: Growing evidence indicates that phenazine’s pharmacodynamic profile extends well beyond glucose transport regulation, although it primarily inhibits sodium-dependent glucose transporters to produce its pharmacological effects. Its modes of action include oxidative stress reduction, downstream metabolic reprogramming, transporter-level competitive inhibition, suppression of inflammatory pathways, and alteration of cellular energy sensors. This section provides a detailed mechanistic analysis of these processes.
Inhibition of Sodium–Glucose Cotransporters (SGLT1 and SGLT2):
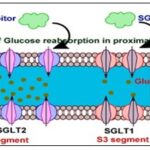
SGLT Inhibition's Molecular Underpinnings: Phlorizin inhibits the sodium-glucose cotransporters SGLT1 and SGLT2 competitively. These sodium-coupled secondary active transporters, which are members of the solute carrier family 5 (SLC5), are responsible for actively absorbing glucose across epithelial membranes 8-17. While SGLT1 is expressed in the kidney's S3 segment and the small intestine, where it mediates glucose absorption from food, SGLT2 is primarily expressed in the S1 segment of the renal proximal tubule and accounts for approximately 90% of glucose reabsorption from the glomerular filtrate 18-52. Phlorizin binds to SGLTs' extracellular glucose-binding pocket. Transporter modelling and structural study revealed that:
- Phlorizin's glucose moiety is located in the glucose recognition site.
- In the transporter cavity, phloretin interacts with hydrophobic residues.
- Competitive binding prevents the necessary conformational shift for sodium-glucose cotransport 28-53. Phlorizin's non-selective profile is impacted by its micromolar inhibition constant (Ki) and similar inhibitory activity against SGLT1 and SGLT2 8. The Sodium-Glucose Cotransporter (SGLT1 and SGLT2) of Phlorizin is shown in the 4.
FIG. 4: SODIUM–GLUCOSE COTRANSPORTERS (SGLT1 AND SGLT2) OF PHLORIZIN
One of the latest and well-liked drugs used to decrease blood sugar in people with type 2 diabetes is sodium-glucose co-transporter (SGLT) inhibitors. Renal glucose is reabsorbed into the bloodstream by the SGLT protein, which is located in the kidney's nephron. 90% of renal glucose reabsorption is carried out by SGLT2, which is found in the proximal renal tubules; the remaining 10% is absorbed by SGLT1. Inhibiting this overactive SGLT protein in the kidneys is a difficult but promising strategy for researchers because very little glucose is eliminated through urine as a result.
Functional Consequences:
Renal glucose reabsorption is decreased by SGLT2 inhibition, which results in:
- Glycosuria, or increased excretion of glucose in the urine
- A decrease in the levels of plasma glucose
- Caloric loss (extrapolated to be between 200 and 300 kcal/day equivalent in people)
Phlorizin is mechanistically different from insulin secretagogues since it decreases glucose without relying on the activity of pancreatic β-cells, in contrast to insulin-dependent treatments 9.
On the other hand, intestinal SGLT1 suppression may hinder the absorption of glucose and result in gastrointestinal disorders. The creation of SGLT2-specific inhibitors was spurred by this lack of selectivity 19.
Effects on Glucose Homeostasis:
The Management of Renal Glucose: Under normal conditions, the human kidney filters around 180 grams of glucose every day. The majority of this is reabsorbed in the proximal tubule via SGLT2 and SGLT1 8. The renal glucose threshold is reduced by phenazine-induced blocking, causing the glucose titration curve to slope downward.
As a result:
- A decrease in plasma glucose levels.
- Reduced requirement for insulin.
- Glucotoxicity is reduced.
Chronic phlorizin therapy was found to improve insulin sensitivity and normalize fasting glucose levels in diabetic animal models while not stimulating insulin production 54.
Impact on Insulin Sensitivity: Phlorizin indirectly improves insulin sensitivity by reducing hyperglycaemia.
- Reduced oxidative stress.
- Reduced formation of advanced glycation end products (AGEs).
- Reduced lip toxicity.
- Insulin receptor signalling has been restored 55.
Diabetic rats treated with phlorizin showed improved Akt activation and insulin receptor substrate-1 (IRS-1) phosphorylation, indicating that downstream insulin signalling cascades had been restored 56.
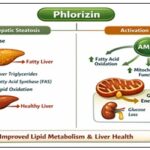
Influence on Lipid Metabolism: Phlorizin affects lipid metabolism by systemic metabolic rebalancing in addition to reducing glucose. It is shown in the Fig. 5.
FIG. 5: LIPID METABOLISM
Reduction in Hepatic Steatosis: Through the increase of sterol regulatory element-binding protein-1c (SREBP-1c), chronic hyperglycaemia stimulates hepatic de novo lipogenesis. Phlorizin indirectly inhibits SREBP-mediated lipogenesis by reducing glucose availability 57.
Research on animals reveals:
- Reduced build-up of triglycerides in the liver
- Decreased expression of fatty acid synthase (FAS)
- Better indicators of lipid oxidation 58.
Activation of AMPK: Phlorizin may activate AMP-activated protein kinase (AMPK), a key regulator of energy balance, according to new research 59. Activation of AMPK encourages:
- Enhanced oxidation of fatty acids
- The suppression of lipogenesis
- Better mitochondrial performance
The disturbed cellular energy balance brought on by the caloric loss caused by glycosuria may be the secondary cause of this impact.
Antioxidant and Anti-Inflammatory Mechanisms:
Scavenging of Reactive Oxygen Species (ROS): Phlorizin can directly neutralize reactive oxygen species by donating hydrogen thanks to its polyphenolic composition. The aromatic rings' hydroxyl groups increase their ability to scavenge radicals 32. The mechanisms of phlorizin as an antioxidant and anti-inflammatory are shown in a Fig. 6.
FIG. 6: ANTIOXIDANT AND ANTI-INFLAMMATORY MECHANISMS
Phlorizin lowers oxidative stress indicators in diabetic animals, such as Malondialdehyde (MDA):
- The generation of superoxide
- Products of lipid peroxidation 18.
NF-kB Signalling Modulation: Nuclear factor kappa B (NF-kB), a transcription factor that controls the expression of pro-inflammatory cytokines, is activated by chronic hyperglycaemia. It has been demonstrated that phlorizin inhibits NF-κB activity, which lowers:
- TNF-α
- IL-6
- Expression of MCP-1 60.
This anti-inflammatory action helps to protect the kidneys and arteries.
Mitochondrial Protection: Excess superoxide is produced when mitochondrial dysfunction brought on by hyperglycaemia occurs. In experimental contexts, phlorizin decreases mitochondrial ROS generation and increases mitochondrial membrane potential 61.
Other Molecular Targets and Emerging Mechanisms:
Modulation of GLUT Transporters: GLUTs, especially GLUT2 and GLUT1, are inhibited by phloretin, the aglycone metabolite of phlorizin 62. Systemic glucose flow is further impacted by this dual inhibitory action.
Interaction of the Renin-Angiotensin System (RAS): According to certain research, SGLT inhibition may have nephroprotective benefits by indirectly suppressing intrarenal RAS activity 63.
Autophagy Regulation: According to preclinical data, phlorizin may improve cellular stress tolerance by modifying autophagy pathways through AMPK–mTOR signalling 64.
Enhancement of Endothelial Function: In diabetic rats, phlorizin decreases endothelial dysfunction indicators and increases endothelial nitric oxide synthase (Enos) activity 65.
Therapeutic Applications and Potentials: Phlorizin's fundamental function as a sodium–glucose cotransporter (SGLT) inhibitor is the main source of its therapeutic significance. Nevertheless, a growing body of experimental data indicates that its biological effects go far beyond glycaemic regulation. Phlorizin is a multifunctional metabolic regulator because of its effects on oxidative stress, inflammation, lipid metabolism, endothelial function, and cellular energy control. Phlorizin is still a very useful pharmacological tool and a prototype for drug research, even though its pharmacokinetic constraints prevent it from being employed in clinical settings.
Antidiabetic Potential:
Mechanistic Foundation: Competitive inhibition of intestinal and renal SGLT1 and SGLT2 transporters causes a decrease in glucose reabsorption and an increase in urine glucose excretion, which is the basis for phlorizin’s antidiabetic action 8-52.
In insulin-resistant conditions, this insulin-independent glucose-lowering mechanism is very crucial. The release of pancreatic β-cell insulin is not stimulated by phlorizin, in contrast to sulfonylureas or insulin treatment. It therefore lowers hyperglycaemia in normoglycemic situations without raising the risk of hypoglycaemia.
Proof from Research on Animals: Historically, the phlorizin-induced diabetes model has yielded important insights into the physiology of renal glucose 16. More significantly, it has been demonstrated that long-term phlorizin administration in streptozotocin (STZ)-induced diabetic rats:
- Return fasting glucose levels to normal.
- Increase the sensitivity of peripheral insulin
- Reduce the malfunction of β-cells caused by glucotoxicity
- Restore the absorption of glucose by skeletal muscle 54.
Rossetti et al. confirmed that hyperglycaemia itself plays a substantial role in insulin resistance by showing that phlorizin treatment of hyperglycaemia restored insulin sensitivity in diabetic rats without changing insulin levels 54.
Consequences for Current SGLT2 Inhibitors: The mechanism of phlorizin directly influenced the creation of selective SGLT2 inhibitors, including canagliflozin and dapagliflozin 13-19.
These drugs improve oral bioavailability and selectivity while maintaining the benefits of decreasing blood sugar. Thus, phlorizin is the structural and conceptual forerunner of one of the most revolutionary medication classes in modern diabetes.
Cardioprotective Effects: The primary cause of death for diabetics is cardiovascular disease. Despite the lack of extensive clinical cardiovascular trials, preclinical data indicate that phlorizin may have cardioprotective qualities.
Enhancement of Endothelial Activity: Through oxidative stress, decreased nitric oxide bioavailability, and the activation of inflammatory cytokines, hyperglycaemia causes endothelial dysfunction 65. In animal models of diabetes, phlorizin has been demonstrated to:
- Boost the expression of endothelial nitric oxide synthase (Enos)
- Lower the generation of vascular superoxide
- Reduce the indicators of lipid Peroxidation 12-61.
Phlorizin indirectly maintains endothelium integrity by lowering glucose and oxidative stress.
Diminished Cardiac Remodelling: According to experimental research, SGLT inhibition lessens heart fibrosis and hypertrophy. Phlorizin therapy reduced left ventricular hypertrophy and enhanced cardiac function metrics in diabetic animals 66.
Effects of Metabolic Heart Disease: Calorie loss brought on by glycosuria encourages moderate ketosis and causes the heart to use fatty acids and ketone bodies instead of glucose, which may increase cardiac efficiency 67. The same physiological basis comes from phlorizin-induced glucosuria, despite the fact that this has been thoroughly investigated with contemporary SGLT2 inhibitors.
Nephroprotective Roles: The kidney is both the primary site of phlorizin action and a major target organ in diabetic complications. The root bark, leaves, and young fruits of apple trees contain phlorizin, also known as phlorizin, a naturally occurring dihydrochalcone flavonoid that functions as a strong, non-selective inhibitor of sodium-glucose cotransporters 1 and 2 (SGLT1/SGLT2). It has important nephroprotective (kidney-protective) functions, especially in reducing diabetic nephropathy and drug-induced acute renal injury. It is regarded as a pioneer in the creation of contemporary SGLT2 inhibitors (gliflozins).
Glomerular Hyperfiltration Reduction: Increased sodium reabsorption in diabetes is facilitated by increased glucose reabsorption, which also activates tubuloglomerular feedback mechanisms that raise intraglomerular pressure and decrease distal sodium delivery 63.
- Phlorizin inhibits SGLT2.
- Decreases salt reabsorption at the proximal level
- Tubuloglomerular feedback is restored.
- The reduction of intraglomerular hypertension
- In diabetic animals, this pathway aids in nephroprotection.
Reduction of Fibrosis and Oxidative Stress: Phlorizin has been demonstrated to lessen:
- Expression of transforming growth factor-β (TGF-β)
Deposition of collagen - Diabetic kidney mesangial growth 68.
These results imply preventive actions against the advancement of diabetic nephropathy.
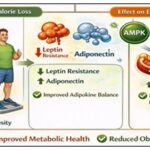
Anti-Obesity and Metabolic Health Effects:
Weight Loss and Calorie Loss: Phlorizin-induced glycosuria results in net caloric loss. Chronic treatment causes a slight decrease in weight and obesity in animal models 69.
Adipokine Modulation: In obese diabetic rodents, phlorizin therapy has been linked to decreased leptin resistance and increased adiponectin levels 70.
Effect on Energy Consumption: Following phlorizin therapy, several studies show improved fatty acid oxidation and higher AMPK activation 59. This implies metabolic reprogramming that goes beyond the simple removal of glucose. As it is shown in the Fig. 7.
FIG. 7: ANTI-OBESITY AND METABOLIC HEALTH EFFECTS
Neuroprotective Effects: Phlorizin may have neuroprotective functions, according to new research.
Neuroinflammation Reduction: Hyperglycaemia exacerbates neuroinflammation and cognitive impairment. Phlorizin reduces inflammatory cytokines and oxidative stress in experimental brain tissue models 71.
Defense against Oxidative Damage to Neurons: ROS scavenging inside cerebral tissues is made possible by its polyphenolic nature, which may help to slow down neurodegenerative processes 26. Despite the lack of direct clinical evidence, molecular similarities with neuroprotection linked to SGLT2 inhibitors merit additional research.
Other Emerging Therapeutic Areas:
Potential Anti-Cancer Effects: According to certain in vitro research, phlorizin and phloretin prevent the growth of cancer cells by obstructing the intake of glucose and the glycolytic metabolism (the Warburg effect) 72.
Antimicrobial and Antiviral Properties: Plant-derived polyphenols, including phlorizin, exhibit antimicrobial properties through membrane disruption and oxidative stress induction in pathogens 24.
Digestive Health: In the intestine, SGLT1 inhibition modifies postprandial glucose excursions and may affect the composition of the gut microbiota 73.
Preclinical and Clinical Studies: For more than a century, phenazine has been the subject of considerable experimental investigation. It has been a key component in the study of renal physiology and a template for the development of SGLT2 inhibitors, despite the fact that its pharmacokinetic constraints have prevented it from moving forward into normal clinical use. In-vitro cellular systems, in-vivo animal disease models, and a few human experimental studies have all been used to assess its effects.
In-vitro Studies:
Models of Glucose Transport in Cells: Characterizing phlorizin's inhibitory effects on sodium–glucose cotransporters (SGLT1 and SGLT2) has been the main focus of in-vitro research. Phlorizin competitively inhibits sodium-dependent glucose absorption in a concentration-dependent manner, according to early studies utilizing brush-border membrane vesicles derived from renal cortex tissue 52. Using Xenopus laevis oocytes transfected with human SGLT1 and SGLT2, further expression investigations verified:
- Direct attachment to glucose-binding sites outside of cells
- Reversible Inhibition
- Competitive kinetics in relation to the concentration of glucose 17-53.
A somewhat stronger affinity for SGLT1 is indicated by the somewhat lower inhibitory constant (Ki) for SGLT1 compared to SGLT2, which helps to explain the gastrointestinal side effects of systemic exposure.
Models of Insulin Signalling and Glucotoxicity: It has been demonstrated that phlorizin therapy restores insulin receptor signalling in cultured skeletal muscle and hepatocyte cell lines exposed to high glucose conditions. Usually, hyperglycaemia causes:
- Reduced phosphorylation of insulin receptor substrate-1 (IRS-1)
- A reduction in Akt activation
- A rise in oxidative stress
- Insulin-mediated glucose uptake routes are restored in experimental settings when phlorizin is used to correct glucose levels 54-56.
Models of Inflammation and Oxidative Stress: In cell-based tests, phenolizing exhibits antioxidant capacity by:
- Reduced generation of reactive oxygen species (ROS)
- NF-κB activation inhibition
- Pro-inflammatory cytokines (TNF-α, IL-6) are downregulated 54.
Phlorizin decreased indicators of oxidative damage and enhanced nitric oxide generation in endothelial cell cultures subjected to hyperglycaemic stress 65.
Studies That Are Anti-Proliferative: Phloretin, an aglycone metabolite of phlorizin, inhibits glucose transporter-mediated uptake (GLUT1) in cancer cell lines, which hinders tumour growth that is dependent on glycolysis 62-72. These results point to a possible metabolic weakness in cells that divide quickly.
In-vivo Animal Models: The most compelling evidence for phlorizin's potential as a treatment comes from research conducted on animals.
Diabetic Models Induced by Streptozotocin (STZ): Because STZ destroys β-cells, diabetic rats have extreme hyperglycaemia. Phlorizin administration in these models:
- Lowers blood glucose during fasting
- The induction of glycosuria
- Enhances sensitivity to insulin
- Reduces the synthesis of glucose in the liver 54.
Rossetti et al. confirmed the key function of glucotoxicity in insulin resistance by showing that phlorizin correction of hyperglycaemia restored insulin sensitivity in peripheral tissues 54.
Type 2 Diabetes Genetic Models: Phlorizin therapy in db./db. mice (a mouse with a leptin receptor deficiency):
- Lower blood sugar levels
- Better β-cell morphology in the pancreas
- Decreased oxidative stress indicators 69.
Models of Cardiovascular Disease: Phlorizin attenuated in animals with diabetes:
- Heart enlargement
- Cardiovascular fibrosis
- Markers of oxidative stress 66
Phlorizin-based models provided early molecular insights, despite the fact that these results are frequently extrapolated from research on selective SGLT2 inhibitors.
Models of Diabetic Nephropathy: Chronic hyperglycaemia leads to glomerular hyperfiltration and renal injury. Phlorizin in animal models:
- A decrease in albuminuria
- Better glomerular architecture
- A reduction in TGF-β expression
- Reduced growth of the mesangial matrix 68
These findings demonstrated proof-of-concept for SGLT inhibition-based renal protection.
Human Studies and Clinical Investigations:
Initial Experimental Research: Early human investigations demonstrated that phlorizin might cause glucosuria when given intravenously, even though it was never intended as a medicinal medication. On the other hand, fast intestinal hydrolysis rendered oral delivery ineffective 7-47. Clinical adoption was hindered by the gastrointestinal symptoms, fast metabolism, and limited oral bioavailability.
Drug Development's Translational Role: The most significant clinical contribution of phlorizin is its function as the structural and molecular model for contemporary SGLT2 inhibitors. Changes in medicinal chemistry resulted in:
- The substitution of a C-glycosidic bond for an O-glycosidic connection
- SGLT2 has higher selectivity than SGLT1.
- Enhanced half-life and oral bioavailability 13-19.
Studies of the structure-activity relationship of phlorizin are directly responsible for the development of dapagliflozin, canagliflozin, and empagliflozin.
Comparative Effectiveness with Modern SGLT2 Inhibitors: Despite having a similar molecular base, phlorizin and contemporary SGLT2 inhibitors differ significantly as it is shown in Table 2.
TABLE 2: COMPARATIVE EFFECTIVENESS WITH MODERN SGLT2 INHIBITORS
| Parameter | Phlorizin | Modern SGLT2 Inhibitors |
| Selectivity | Non-selective (SGLT1 & SGLT2) | Highly selective for SGLT2 |
| Oral Bioavailability | Poor | High |
| Plasma Half-Life | Short | 10–13 hours (average) |
| Clinical Use | No | Widely approved |
| Cardiovascular Outcomes | Preclinical evidence only | Proven reduction in CV events |
SGLT2 selective inhibitors have shown:
- A decrease in significant adverse cardiovascular events
- Reduced development of chronic kidney disease
- A decrease in heart failure hospitalizations 67
These important discoveries support the treatment pathway, which was first discovered by phlorizin research.
Toxicity and Safety Profile: Phlorizin, a naturally occurring dihydrochalcone glycoside originally isolated from Malus domestica bark in the early nineteenth century, was the first identified competitive inhibitor of sodium-glucose cotransporters (SGLTs) 1-2. The discovery of phlorizin has had a profound impact on the conceptual understanding of glucose transport in the kidney and paved the way for the development of a new class of antidiabetic agents. Nevertheless, unlike its modern derivatives, phlorizin itself was never pursued as a therapeutic agent owing to its marked pharmacokinetic drawbacks. However, over the past century, extensive experimental work, mainly conducted in-vitro and in-vivo using animal models, has offered valuable information on its toxicity and safety profile, which remain critical for understanding both its properties and the safety expectations of its optimized variants. Notably, given the fact that the main mechanism of action of phlorizin is based on the inhibition of SGLT1 and SGLT2, the toxic effects of phlorizin are, to a great extent, expected and mechanism-based; however, several other aspects come into play, including its rapid hydrolysis to phloretin in the intestine, off-target interactions, metabolic changes, and polyphenolic structure 74-75-76.
Acute and Chronic Toxicity:
Acute Toxicity: Acute toxicity is defined as adverse physiological consequences due to a single dose or short-term treatment with a compound.
In the context of phlorizin, acute toxicity has been investigated in-depth in rodents and other mammalian models using both oral and parenteral routes of administration. Historically, the early pharmacological studies showed that intravenous phlorizin causes extreme glucosuria with minimal risk of acute hypoglycaemia if the body's own glucose production is preserved. This occurs due to the competitive inhibition of SGLT2 in the renal proximal tubule, which decreases glucose reabsorption and increases urinary glucose excretion 77. Since, this mechanism is insulin-independent and does not directly activate pancreatic β-cell insulin secretion, the danger of acute hypoglycaemia, a potential safety risk for most antidiabetic compounds, is negligible in the presence of normoglycemia 76-77. The primary acute physiological effect of phlorizin administration is osmotic diuresis due to glucosuria. As the concentration of glucose in the urine exceeds the renal threshold, water passively diffuses into the lumen due to:
- Elevated urine output, leading to polyuria;
- Transient electrolyte disturbance, especially sodium and chloride loss, because of the inhibition of coupled sodium-glucose transport;
- Mild dehydration if fluid intake is not sufficient, especially in small rodents, where fluid turnover is very rapid.
These phenomena are not regarded as direct toxic effects of the compound itself but rather as the anticipated physiological consequences of inhibiting a major renal transport mechanism 77-78. In acute rodent models, high doses of phlorizin were not associated with gross organ damage evident at necropsy; there was no evidence of hepatotoxicity or direct cellular toxicity at doses that caused glucosuria. Routine blood chemistry analyses following single-dose administration of the compound have revealed changes suggestive of mild osmotic nephropathy, with elevations in blood urea nitrogen (BUN) and creatinine, but these have been transient and resolved promptly after compound clearance. Histopathological examination of kidney, liver, and heart samples in acute studies has rarely disclosed evidence of necrosis, apoptosis, or inflammatory cell infiltration directly related to phlorizin exposure 79.
Gastrointestinal Side Effects: Indeed, one of the first known limitations – and acute tolerability problems – of phlorizin is its non-selective inhibition of SGLT1 in the small intestine. SGLT1 is known to mediate the active uptake of glucose from the intestinal lumen into enterocytes. The non-selective inhibition of SGLT1 by phlorizin results in an increased luminal glucose concentration, which can result in osmotic water retention in the lumen and an increase in intestinal transit, manifesting as:
- Osmotic diarrhoea;
- Abdominal cramping;
- Flatulence and bloating;
- Transient nausea in some models.
These gastrointestinal side effects may have played a significant role in the early abandonment of phlorizin as a systemic antidiabetic drug. In contrast, contemporary SGLT2 inhibitors were designed to have very high selectivity for SGLT2 over SGLT1, thereby reducing intestinal side effects to a great extent 80.
Chronic Toxicity: Chronic toxicity can be defined as adverse reactions that become apparent after repeated or chronic exposure over a period of weeks to months. In the context of phlorizin, animal models over a period of several months have helped to elucidate the physiological adaptations and toxicities that may be present. In diabetic rodent models (such as streptozotocin-induced diabetes), chronic administration of phlorizin has been shown to produce persistent glucosuria with improved glycaemic control, decreased oxidative stress markers, decreased renal hypertrophy, and reduced glomerular hyperfiltration, the latter indicating restored tubuloglomerular feedback as a result of decreased proximal tubular sodium and glucose reabsorption 81, 82. In these models, chronic administration does not cause significant structural organ damage due to phlorizin itself. Careful histopathological examination of tissues from the kidneys, liver, pancreas, and heart typically shows either normal histology or improvement compared to diabetic controls, indicating decreased metabolic stress 81, 82.
- Nonetheless, it is necessary to recognize certain adaptive reactions. Animals with chronic glucosuria frequently exhibit:
- Slight drops in body weight that correspond to calorie reduction;
- Changes in energy substrate use and increased lipolysis;
- Increased generation of ketone bodies, especially when there is a concurrent insulin shortage or dietary restriction.
Theoretically, increased ketogenesis could predispose to ketoacidosis in situations of acute insulin deficit or protracted fasting, even though these alterations might represent advantageous metabolic remodelling rather than direct toxicity. In fact, rare instances of euglycemic diabetic ketoacidosis have been reported in recent clinical experience with SGLT2 inhibitors, which mimic the glucosuria effects of phlorizin, indicating a mechanistic risk that warrants observation 83. In animal studies, long-term phlorizin administration does not necessarily harm renal parenchyma in terms of renal shape and function; instead, it seems to lessen nephropathic alterations linked to diabetes. At therapeutic levels, hepatic and cardiovascular tissues also show no signs of chronic histological toxicity 81, 82.
Side Effects and Limitations: Even though phlorizin doesn’t directly cause cytotoxicity, its mode of action and metabolic destiny result in drawbacks and restrictions that need to be carefully taken into account in any safety assessment.
Metabolic Consequences of Sustained Glycosuria: Persistent urine glucose excretion, which results in a net caloric deficit, is the most predicted physiological effect of prolonged SGLT inhibition. Under transporter blockade, glucosuria typically causes 200–300 kcal of glucose loss per day, which triggers adaptive metabolic reactions 84.
Among these answers are:
- Increased mobilization and oxidation of lipids from adipose tissue;
- Increased synthesis of ketone bodies in the liver;
- The liver's compensatory reaction to energy loss is the upregulation of gluconeogenic pathways.
These changes are typically adaptive and consistent with better metabolic profiles. (e.g., decreased adiposity, enhanced insulin sensitivity) in controlled experimental models. Enhanced ketogenesis, however, increases the potential risk of ketoacidosis in the context of severe insulin deficit or stress, especially when insulin levels are not high enough to inhibit hepatic ketone production. Clinical similarities with SGLT2 inhibitors indicate that researchers and practitioners should exercise caution when converting glucosuria treatments into states of impaired insulin activity, notwithstanding the paucity of direct evidence of phlorizin-induced ketoacidosis in animal models 83, 85.
Impact on Electrolyte Balance and Hemodynamic: Sodium reabsorption is linked to SGLT2-mediated glucose transport in the proximal tubule. Thus, phlorizin's inhibition of this mechanism results in natriuresis, or increased sodium excretion, which has several physiological repercussions downstream:
- Slight drops in blood pressure
- The compensatory renin-angiotensin-aldosterone system (RAAS) is activated.
- Changes in fluid balance and alterations in plasma volume
Phlorizin therapy regularly results in mild drops in both systolic and diastolic blood pressure in animals, which is indicative of natriuretic and osmotic diuretic effects 78, 86. In hypertension or hypervolemic situations linked to metabolic syndrome, these hemodynamic alterations are typically regarded as beneficial; nevertheless, in normotensive or hypovolemic patients, they would need to be closely watched.
Hydrolysis to Phloretin and Off-Target Effects: Lactase-phlorizin hydrolase quickly hydrolyzes phenol, producing glucose and phloretin in the colon. Phloretin has unique biological effects, such as the following, despite having negligible SGLT inhibitory activity:
- Inhibition of the GLUT1 and GLUT2 facilitative glucose transporters
- Changes in the fluidity of cell membranes
- Interaction between different ion channels
In addition to potentially contributing to gastrointestinal intolerance and other non-specific effects seen in experimental models, these off-target responses make it more difficult to interpret the systemic pharmacology linked to phlorizin 87.
Poor Pharmacokinetic Profile: Phlorizin's low oral bioavailability is a major drawback as a pharmacotherapeutic drug, and this results from:
- Quick hydrolysis in the gut to phloretin;
- Restricted intestinal passive permeability
- Extensive liver metabolism in the first pass (glucuronidation, sulfation)
In rodents, intact phlorizin is rarely detected after an oral dose, while the plasma half-life after parenteral injection is around 30 to 60 minutes 88. Medicinal chemists created stable C-glucoside derivatives, or the contemporary SGLT2 inhibitors, in response to these pharmacokinetic limitations. These compounds prevent fast hydrolysis and permit prolonged systemic exposure 80, 89.
Safety in Long-Term Use:
Evidence from Chronic Animal Models: Phlorizin dosing over a period of weeks to months in diabetic animal models has not shown any appreciable organ harm. Conversely, long-term phlorizin treatment frequently results in improvements in several metabolic stress indicators, such as:
- Decreased indicators of oxidative stress in the liver and kidney;
- Reduced glomerular damage and renal hypertrophy
- Decreased myocardial fibrosis and enhanced cardiac function
In these long-term trials, histopathological assessment of key organs rarely shows direct lethal effects caused by phlorizin. For instance, compared to untreated diabetic controls, renal and hepatic tissues usually show less oxidative damage and inflammatory infiltration, which is consistent with phlorizin's metabolic advantages 81, 82.
Carcinogenicity and Genotoxicity: There is currently no solid proof that phlorizin has genotoxic or carcinogenic qualities. Due to its widespread presence in dietary sources, especially apples and kindred fruits, this polyphenolic molecule is regularly exposed at low levels through human diets without being linked to an increased risk of cancer 79-90. Although phlorizin is only one of a complex mixture of co-occurring phytochemicals, epidemiological studies that link eating apples to a lower risk of cardiovascular disease and metabolic disorders offer encouraging evidence of safety with long-term dietary exposure. Rigorous carcinogenicity studies specifically assessing purified phlorizin at high doses remain limited, and such evaluations would be necessary for any future clinical development of phlorizin-based therapeutics.
Comparison with Modern SGLT2 Inhibitor Safety: In massive randomized controlled trials, contemporary SGLT2 inhibitors (such as empagliflozin and canagliflozin) generated from the phlorizin scaffold have been thoroughly evaluated for human safety. Among their adverse event profiles are:
- Increased risk of genital mycotic infections
- Modest increases in haematocrit;
- Rare cases of euglycemic diabetic ketoacidosis;
- Rare reports of necrotizing fasciitis of the perineum (Fournier’s gangrene)
Rather than being inherent to the molecular scaffold, these side effects are usually seen as mechanism-based effects of natriuresis and glucosuria 83-85-91. Because modern SGLT2 inhibitors were designed to circumvent phlorizin’s pharmacokinetic limitations while retaining transporter blockade, their safety experiences provide useful context for understanding phlorizin’s safety profile and risk expectations.
Formulation Approaches and Drug Delivery: Phlorizin's therapeutic potential is limited by significant pharmacokinetic and formulation issues rather than a lack of biological action. Despite well-established antidiabetic, antioxidant, anti-inflammatory, and cardio-renal protective effects, its intrinsic physicochemical characteristics such as rapid intestinal hydrolysis, limited membrane permeability, low oral bioavailability, and short systemic half-life have historically prevented its direct clinical application 92-93. As a result, formulation techniques that improve absorption, prevent enzymatic degradation, increase stability, and allow for targeted distribution have become crucial elements of translational research in this field. This section provides a thorough foundation for the pharmacological exploitation of phlorizin and its derivatives by reviewing these formulation issues and examining innovative solutions created to address them.
Challenges with Phlorizin Delivery:
Rapid Enzymatic Degradation: Phlorizin's vulnerability to enzymatic cleavage is the biggest obstacle to its efficient distribution. The O-glycosidic bond that connects the aglycone (phloretin) to glucose is quickly hydrolysed in the small intestine by brush-border β-glucosidases and lactase–phlorizin hydrolase (LPH). Before substantial systemic absorption, this hydrolysis converts phlorizin into phloretin and D-glucose 94-95. The anticipated therapeutic effect is effectively nullified by this first-pass metabolism since phloretin lacks the β-glucoside necessary for strong SGLT inhibition. Because of this, pharmacodynamic effects are mostly restricted to the gut lumen and the early post-absorptive phase, and oral phlorizin treatment results in minimal plasma levels of the intact molecule. Modern (C-glycoside) SGLT2 inhibitors, on the other hand, can withstand enzymatic hydrolysis because of the carbon–carbon glycoside bond 96.
Poor Membrane Permeability and Absorption: Because of its numerous hydroxyl groups and glycosidic moiety, phenol is relatively hydrophilic, which restricts its capacity to move across lipid membranes by passive diffusion. Its strong polarity and molecular weight (~436 g/mol) limit oral absorption independent of metabolic breakdown by reducing penetration through enterocyte cell membranes 97. Because phlorizin's recognition moiety is similar to that of glucose, there is little transporter-mediated absorption of it; yet, the majority of glucose transporters preferentially mediate glucose influx over glycoside uptake. Thus, in Caco-2 and MDCK cell monolayer models, phlorizin's permeability coefficients are low, which is in line with its poor oral bioavailability.
First-Pass Metabolism: First-pass hepatic metabolism greatly lowers systemic exposure, even if some intact phlorizin is absorbed. The aglycone moiety is subject to phase II conjugation processes, such as glucuronidation and sulfation, which quickly result in metabolites with decreased biological activity. Rapid systemic clearance and a brief half-life (about 30 to 60 minutes in animal models) are guaranteed by this double hit, which consists of intestinal hydrolysis followed by hepatic metabolism 98.
Non-Selective Transporter Interaction: Intestinal adverse effects (such as diarrhoea and osmotic dysregulation) caused by phlorizin’s non-selective blockage of both SGLT1 and SGLT2, as opposed to primarily SGLT2, make formulation objectives more difficult to achieve. Theoretically, kidney-specific administration would optimize therapeutic efficacy while reducing gastrointestinal interactions, but technically, this has been difficult to do without causing significant systemic exposure.
Novel Formulation Strategies (e.g., Nanocarriers, Complexes): In light of these difficulties, sophisticated formulation techniques have been investigated to improve the stability, bioavailability, and therapeutic payload delivery of phlorizin.
These tactics, which shield the parent molecule from deterioration and enhance its pharmacokinetic profile, span from molecular alterations to complex carrier systems.
Nanoparticle-Based Delivery Systems: Platforms for nanoparticles have become a flexible way to improve absorption, allow for regulated release, and shield labile medications from enzymatic destruction 99-100.
Nanoparticles of Polymers:
- Poly (lactic-co-glycolic acid) (PLGA), a biodegradable polymer, has been widely utilized to encapsulate labile compounds. Polymeric nanoparticles loaded with phenolizing can:
- Protect phlorizin from the activities of luminal β-glucosidase
- Prolong the complete drug’s release
- Improve intestinal absorption by means of paracellular transport and cohesion
- Prevent first-pass metabolism to lower systemic clearance.
Compared to free phlorizin, in-vitro research shows that PLGA-encapsulated phlorizin has far greater stability in simulated gastrointestinal fluids and prolonged release characteristics 99.
Crucially, surface modification of nanoparticles (such as PEGylation) can reduce non-specific uptake by the reticuloendothelial system and further extend circulation duration.
Lipid-Based Nanocarriers: Another interesting approach is provided by solid lipid nanoparticles (SLNs) and lipid vesicles (liposomes). Phlorizin can be embedded in a solid lipid core or lipid bilayer in these formulations to:
- Boost solubilization inside the intestinal environment;
- Avoid first-pass hepatic metabolism by promoting lymphatic absorption
- Assign a capacity for regulated release
Through lipid matrix entrapment, solid lipid nanoparticles in particular stabilize phlorizin, decreasing its vulnerability to hydrolysis and promoting increased bioavailability.
Nano micelles and Polymer–Drug Conjugates: Phlorizin can be sequestered within hydrophobic cores or covalently bonded via labile linkers in amphiphilic polymer micelles and polymer–drug conjugates. These devices may enable site-specific administration by releasing the medication in particular microenvironments (such as tailored pH zones).
Cyclodextrin Inclusion Complex: Cyclodextrins, which are cyclic oligosaccharides with a hydrophobic core cavity, improve the water solubility and stability of poorly soluble medicines by forming inclusion complexes with them 101. In simulated gastrointestinal fluids, phenol-cyclodextrin complexes have demonstrated superior dissolving properties and increased resistance to enzymatic hydrolysis.
The inclusion complex is capable of:
- Lessen the glycosidic bond's exposure to brush-border enzymes;
- Boost medication solubility to increase membrane permeability;
- Prevent oxidative or pH-induced deterioration of phlorizin.
However, to balance complex stability and drug release kinetics, rigorous tuning of the substitution pattern and cyclodextrin type (α, β, γ) is needed.
Strategies for Prodrug and Structural Derivatives: Instead of only altering the delivery system, prodrug design is another effective tactic that entails chemically connecting phlorizin to components that:
- Increase the lipophilicity of
- Prevent enzymatic assault on the glycosidic site
- Encourage uptake mediated by transporters.
For instance, prodrugs that more easily pass through the intestinal epithelium can be made by esterifying hydroxyl groups or conjugating them to amino acids or peptides. These prodrugs are then broken down by systemic enzymes to produce active phlorizin. To guarantee that the released moiety maintains pharmacological effectiveness without producing harmful byproducts, such techniques must be carefully designed.
Specific Renal Delivery: Targeted delivery strategies seek to give phlorizin or its derivatives directly to kidney proximal tubular cells, which display high levels of SGLT2. Though mostly experimental, possible tactics consist of:
- Nanocarriers with ligand modifications that can identify renal proximal tubule receptors;
- Carriers that react to pH or enzymes and release medication into the renal microenvironment;
- Conjugates of antibodies and drugs that are particular to the kidneys.
Theoretically, targeted administration could lessen systemic exposure and the negative gastrointestinal consequences of SGLT1 inhibition.
Enhancing and Bioavailability and Stability: Achieving therapeutically appropriate systemic exposures of the intact molecule while reducing degradation and off-target effects is the ultimate goal of phlorizin formulation research. This means maximizing release and absorption patterns in addition to safeguarding the medication.
Enteric and Controlled Release Systems: Bypassing the acidic gastric environment, enteric coatings can postpone disintegration until the formulation reaches the small intestine. However, enteric coatings alone are not enough to stop hydrolysis since intestinal β-glucosidases are mostly active on the brush edge. By using controlled-release matrices (such as hydrophilic polymers like HPMC) in conjunction with enteric protection, the drug's release rate can be regulated, allowing for gradual delivery and lowering peak local concentrations that might indicate degradative pathways.
Mucoadhesive and Permeation- Enhancing Formulations: Tight connections between intestinal epithelial cells can be momentarily opened by mucoadhesive polymers like chitosan, which increases paracellular trafficking. Using these polymers in formulations can:
- Lengthen the time spent in the GI tract;
- Improve intact phlorizin absorption;
- Limit access to luminal enzymes by being close to the epithelium.
These tactics have the potential to greatly increase oral bioavailability when paired with protective nanoparticles or enzyme inhibitors.
Multifunctional Composite Systems: Modern delivery methods are becoming more and more multipurpose, combining controlled release, targeting, and protection. Among the examples are:
- Core-shell nanocarriers having an internal reservoir of phlorizin shielded by external polymers resistant to enzymes;
- Phlorizin is released by stimuli-responsive systems in reaction to redox gradients or pH variations;
- Particles with surface functionalization that improve absorption by interacting with particular gut receptors.
The convergence of material science, pharmacology, and molecular biology required to overcome the intricate obstacles inherent in phlorizin distribution is demonstrated by such composite techniques.
Biotechnological and Industrial Applications: Phlorizin is a significant illustration of how natural phytochemicals can function as both direct bioactive agents and structural models for the creation of synthesized drugs from a biotechnological standpoint. The creation of contemporary SGLT2 inhibitors, a clinically revolutionary class of antidiabetic medications, was directly influenced by the finding that phlorizin inhibits both SGLT1 and SGLT2 102-103. Phlorizin thus serves as a link between industrial pharmaceutical innovation and the chemistry of natural products. Phlorizin is widely available in apple processing byproducts, especially apple pomace, which supports industrial applications. Apple waste streams are becoming valuable sources of polyphenols, including phlorizin, thanks to the growth of the fruit juice and cider industries 104-105. This allows for sustainable extraction and commercialization techniques. This is consistent with circular bioeconomy models that prioritize environmentally conscious production techniques and waste valorisation.
Furthermore, the viability of adding phlorizin to food, cosmetic, and biomedical formulations has been improved by developments in extraction technologies, chromatographic purification, encapsulation based on nanotechnology, and stabilization techniques 106. The creation of structurally altered derivatives with enhanced pharmacokinetic and therapeutic qualities has also been made easier by biotechnological advancements such as enzyme modification, microbial biotransformation, and synthetic chemical techniques 107.
Notwithstanding these encouraging advancements, issues with intestinal hydrolysis, stability, bioavailability, and large-scale standardization continue to be important factors for industrial translation 108. In order to enhance this compound's commercial and therapeutic potential, research is still being conducted to investigate structural alterations, sustainable sourcing, and optimized delivery systems. All things considered, phlorizin is a remarkable illustration of a molecule produced from plants that has developed from a physiological study tool into a multipurpose substance with extensive biotechnological and economic significance. Its transdisciplinary importance in contemporary life sciences is highlighted by its applications in nutraceuticals, biomedical research, pharmaceutical development, and sustainable agro-industrial processes.
Use in Nutraceutical and Functional Foods: Phlorizin, also known as phlorizin, is a naturally occurring dihydrochalcone that is primarily found in the bark, leaves, and fruit of apples (Malus species). Because of its anti-inflammatory, antioxidant, and glucose transport-modulating qualities, it has garnered a lot of attention in the nutraceutical and functional food industries 109-110. Phlorizin is a dietary polyphenol that is a member of the larger class of flavonoids, which are substances that are well known for their role in promoting cardiometabolic health and preventing chronic diseases 111-112. Phlorizin and similar polyphenols have been partially implicated in epidemiological findings that apples are associated with a lower incidence of cardiovascular disease, type 2 diabetes mellitus, and several types of cancer 113-114. Uses of phlorizin in nutraceutical and functional foods are shown in the Fig. 8.
FIG. 8: PHLORIZIN USE IN NUTRACEUTICAL & FUNCTIONAL FOODS
This property has led to the investigation of phlorizin-enriched extracts from apple pomace and peel as ingredients in glycaemic control–oriented food products 118. Phlorizin is valued in functional food applications because it inhibits sodium–glucose cotransporters (SGLT1 in the intestine and SGLT2 in the kidney), reducing postprandial glucose excursions 115-116. Although its poor oral bioavailability limits systemic pharmacological action, localized intestinal SGLT1 inhibition may contribute to attenuation of dietary glucose absorption when incorporated into food matrices. 117.
Pomace, a byproduct of apple processing, is a sustainable and industrially plentiful source of phlorizin 119. To improve production and stability for food-grade applications, biotechnological extraction and purification techniques such as solvent extraction, membrane filtering, and chromatographic separation have been refined 120. Apple waste streams are valued in accordance with the circular bioeconomy principles, which offer advantages for the economy and the environment 121. Phlorizin demonstrates antioxidant activity from a nutraceutical perspective by scavenging radicals and modifying oxidative stress pathways. 3-14. In experimental settings, it has been demonstrated to decrease the production of reactive oxygen species (ROS), attenuate lipid peroxidation, and alter inflammatory mediators 123. These characteristics lend credence to its possible inclusion in dietary supplements that address obesity, insulin resistance, and metabolic syndrome 124. Stability issues, intestinal hydrolysis by lactase-phlorizin hydrolase, and conversion to phloretin, which could change biological function, are still problems, though 125. To increase stability in functional food matrices, encapsulation techniques such as microencapsulation and Nano formulations have been suggested 126.
Applications in Biomedical Research: Phlorizin has long been a vital research instrument in studies of glucose transport and renal physiology. It has been commonly used to describe renal tubular glucose reabsorption mechanisms since it was discovered to be a glucosuria agent in the 19th century 109-115. SGLT1 and SGLT2 transporters were cloned and characterized as a result of the functional discovery of sodium-dependent glucose cotransport systems made possible by the experimental administration of phlorizin 127-128.
Phlorizin has been used in experimental diabetes studies to regulate hyperglycaemia in animal models without insulin production, which enables researchers to separate the metabolic effects of long-term hyperglycaemia 129. Phlorizin-mediated glycosuria has been shown in diabetic mouse studies to reverse glucotoxicity, enhance insulin sensitivity, and restore pancreatic β-cell responsiveness 130. The validation of renal glucose reabsorption as a therapeutic target was greatly aided by these findings 131.
Additionally, phlorizin has been widely employed in nephrology and cardiovascular studies. It has shown nephroprotective qualities by lowering intraglomerular pressure and hyperfiltration in diabetic animals 132. Its significance in vascular biology research has also increased due to its involvement in regulating oxidative stress and endothelial dysfunction 133. Phloretin, the aglycone metabolite of phlorizin, has been employed as an inhibitor of facilitative glucose transporters (GLUT1 and GLUT2) in addition to metabolic investigations, offering information on tumour metabolism and glucose dependence in cancer cells 134. Phlorizin compounds are still used as molecular probes in oncology research because of the Warburg effect, which makes altering glucose transport pathways of increasing interest 135.
Synthetic Derivatives and Their Significance: Phlorizin's drawbacks, including its non-specific SGLT1 inhibition, fast hydrolysis, and low oral bioavailability, led to intensive medicinal chemistry attempts to create more stable and selective counterparts 136. Clinically accepted SGLT2 inhibitors like dapagliflozin, canagliflozin, and empagliflozin are the result of structural changes that produced C-glucoside derivatives resistant to enzymatic cleavage 137-138. The significance of synthetic derivatives is shown in the Fig. 9.
FIG. 9: SYNTHETIC DERIVATIVES AND THEIR SIGNIFICANCE
Oral absorption and metabolic stability were enhanced by substituting a carbon-carbon bond for the O-glycosidic linkage 137. Furthermore, gastrointestinal side effects were reduced by preferentially targeting SGLT2 over SGLT1 116. These developments created a successful class of treatments for heart failure and type 2 diabetes using a naturally occurring phytochemical scaffold 139. Additionally, combined SGLT1/SGLT2 inhibition and applications in obesity and non-alcoholic fatty liver disease (NAFLD) have been studied for synthetic derivatives 140. Thus, the phlorizin scaffold is a seminal example of drug discovery inspired by natural products 141.
Future Directions and Research Prospects: A paradigm shift in the knowledge and management of metabolic disorders was brought about by the identification of phlorizin as a naturally occurring dihydrochalcone and the structural model for sodium–glucose cotransporter (SGLT) inhibitors. Phlorizin's legacy is fundamental, ranging from its early experimental use to its impact on contemporary pharmacotherapy. Nevertheless, the field of phlorizin science is still developing after decades of research. Unanswered mechanistic questions, chances for novel drug development, and extensive translational pathways spanning the metabolic, cardiovascular, renal, cancer, and nutraceutical domains are all part of the developing landscape. Therefore, to fully utilize the medicinal and scientific potential inherent in phlorizin and its derivatives, future studies must incorporate multidisciplinary techniques spanning from chemical biology and systems pharmacology to clinical research and industrial biotechnology.
Knowledge Gaps:
Characterization of Human Pharmacokinetics and Metabolism: The lack of a thorough, human-specific pharmacokinetic (PK) profile is one of the most enduring knowledge gaps in phlorizin research. Early rodent research showed that lactase-phlorizin hydrolase and brush-border β-glucosidases in the colon quickly hydrolyse phlorizin, producing glucose and the aglycone phloretin 142-143.
The interpretation of systemic effects is complicated by the distinct pharmacological profiles and membrane transport properties of these metabolites. Nevertheless, there is a dearth of thorough human PK data, which includes tissue distribution, absorption, biotransformation pathways, and elimination profiles. Dose extrapolation, bioavailability evaluation, and systemic exposure estimates all crucial for clinical translation and safety assessment become questionable as a result. To produce reliable human PK data, sophisticated analytical methods like physiologically based pharmacokinetic (PBPK) modelling, stable isotopic tracing, and high-resolution LC-MS/MS are required. These methods can highlight possible target organs, off-target effects, and enterohepatic cycling in addition to elucidating plasma kinetics, tissue compartmentalization, and metabolite production rates. Beyond SGLT inhibition, integrated PK-pharmacodynamic (PD) modelling may shed more light on how dynamic metabolite patterns affect biological responses.
Molecular Processes Other Than SGLT Inhibition: Although phlorizin's primary mechanism of action is still the inhibition of SGLT1 and SGLT2, new research suggests that its biological repertoire may encompass more extensive cellular and molecular pathways. In vitro, phlorizin and its aglycone phloretin have shown antioxidant action, regulation of inflammatory transcription factors such as nuclear factor-κB (NF-κB), and effects on mitochondrial function 144, 145.
However, important mechanistic questions are still open:
- Are phlorizin or its metabolites directly responsible for the reported anti-inflammatory activity?
- Does phlorizin stimulate intracellular signalling pathways such sirtuin networks, AMP-activated protein kinase (AMPK), or mammalian target of rapamycin (mTOR) that are not dependent on glucose translocation?
- How much does phlorizin affect mitochondrial biogenesis, metabolic flexibility, and epigenetic regulation?
Systematic mechanistic research utilizing CRISPR-based gene editing, sophisticated cellular models, and omics technologies (transcriptomics, proteomics, metabolomics) is needed to answer these concerns. Such integrative investigations will make it clear if different receptor-independent mechanisms or glycaemic regulation alone are responsible for phlorizin's pleiotropic effects.
Interaction between Gut Microbiota and Metabolites: A new area of study in metabolism is the interaction between the gut microbiota and dietary polyphenols. Gut bacteria convert a large number of polyphenols into bioactive substances with unique biological functions. Because phlorizin is a glycoside, it is especially vulnerable to microbial DE glycosylation, which produces a variety of phenolic metabolites that could affect human physiology 146. Nevertheless, little is known about the precise microbial species in question or the functional importance of the metabolites that are produced. To understand these intricate host-microbe interactions and guide dietary or probiotic therapies that maximize positive effects, multi-omics integration combining metagenomics with metabolomics and host transcriptomics will be essential.
Natural Extract Standardization: Phlorizin has been extracted from apple pomace and other plant sources because of its interest in the industrial and nutraceutical sectors. On the other hand, cultivar, geographic origin, seasonal impacts, and processing conditions all significantly affect the diversity of natural extracts 147.
Reproducibility in research and the creation of commercial products is hampered by this variability. To guarantee consistent efficacy and safety, standardization is required at several levels, such as defined phlorizin content, stability evaluations, and impurity profiling. For industrial applications, regulatory compliance, and clinical evaluation, the development of proven analytical techniques (e.g., HPLC, LC-MS/MS) for phlorizin and related phenolics quantification and fingerprinting is crucial.
Safety and Toxicity in the Long Run: Large-scale clinical trials have provided well-documented safety profiles for modern SGLT2 inhibitors, including the possibility of genital mycotic infections, infrequent instances of diabetic ketoacidosis, and slight haematocrit elevations 143. However, there is a dearth of comparable long-term safety information about human pure phlorizin or phlorizin-rich extracts. There hasn't been a systematic investigation of high-dose chronic exposure, including research on reproductive toxicity, carcinogenicity, and organ-level evaluations. If phlorizin or its optimized derivatives are to be used for long-term therapeutic purposes or incorporated into standardized nutraceutical products outside of the typical food intake, such safety assessments are essential.
Potential for Drug Development:
SGLT Modulators of the Future: The therapeutic potential of targeting renal glucose transport is validated by the effective translation of phlorizin's action into clinically authorized SGLT2 inhibitors, such as dapagliflozin, canagliflozin, and empagliflozin 142-143. Opportunities to use next-generation modulators to broaden this pharmacological class exist:
By focusing on intestinal glucose absorption, selective SGLT1 inhibitors may improve postprandial glucose regulation while lowering systemic adverse effects 148.
Dual SGLT1/SGLT2 inhibitors: Patients with significant postprandial hyperglycaemia or concomitant metabolic dysfunction may benefit from synergistic glycaemic effects from balanced inhibition 149.
Allosteric Modulators: It may be possible to provide more precise control with different safety profiles by designing compounds that modify SGLT activity through non-competitive sites.
For these next-generation drugs, medicinal chemistry techniques that make use of phlorizin's framework, such as C-glucoside bonds that withstand enzyme hydrolysis, continue to be a fruitful design paradigm.
Organ-Specific Delivery with Targets: The creation of organ-targeted delivery systems to target particular tissues with phlorizin or its analogues is an interesting approach.
For instance:
Targeting Renal Proximal Tubules: Drugs may be preferentially delivered to nephron segments that are high in SGLT2 via ligand-mediated systems or nanoparticle carriers.
Focus on the Intestinal Epithelium: By delivering medication locally to enterocytes, SGLT1 inhibition may be maximized while systemic exposure and adverse effects are reduced.
Such focused techniques could be made possible by advanced drug delivery platforms, such as liposomes, polymeric nanoparticles, and stimuli-responsive systems, which would increase the therapeutic index and decrease off-target effects 150.
Extension to Non-metabolic: New preclinical data indicate that various disease pathways may be impacted by glucose transport modulation:
Cancer Metabolism: Phloretin, an aglycone of phlorizin, inhibits specific GLUT isoforms and has demonstrated anticancer effects in-vitro. Tumour cells also commonly overexpress glucose transporters. Compounds that target tumour metabolic weaknesses may be produced by structure-based optimization 151.
Inflammatory Diseases: Oxidative pathways and metabolic stress are closely related to chronic inflammation. In autoimmune or age-related disorders, modulators that combine anti-inflammatory signalling with glucose transport inhibition may be therapeutically useful.
Thorough preclinical examination should be used in translational research to investigate these non-classical indications. The processes of how Organs-Specific Delivery & Non-Metabolic Disease Targets are shown in the Fig. 10.
FIG. 10: ORGANS-SPECIFIC DELIVERY & NON-METABOLIC DISEASE TARGETS
Translational Research Opportunities: Mechanistic biology, pharmacology, regulatory science, and clinical research must all be systematically integrated for phlorizin to be translated from a traditional experimental molecule to a therapeutic or preventive drug with clinical significance. The molecular basis of phlorizin immediately led to the creation of commercially licensed SGLT2 inhibitors that have shown improvements for cardiovascular and renal outcomes 153-154. Although phlorizin itself is not clinically employed due to its limited oral bioavailability and fast hydrolysis 152-155. For upcoming innovation, this historical process offers a potent translational paradigm.
Development of Biomarkers and Precision Medicine: Precision medicine techniques customized to specific metabolic characteristics are among the most promising translational pathways. Significant decreases in hospitalization for heart failure and the advancement of renal disease have been shown in large cardiovascular outcome trials using SGLT2 inhibitors, such as EMPA-REG OUTCOME and DAPA-HF 153-154. Interindividual differences in cardiovascular protection, natriuretic effects, and glycaemic response, however, point to underlying biological heterogeneity.
Future studies in translation should concentrate on determining:
- Drug responsiveness is influenced by genetic variations in SGLT transporters.
- Biomarkers for cardiovascular and renal protection
- Metabolic markers linked to enhanced mitochondrial performance
Patients who are most likely to benefit from transporter modification medications may be stratified using pharmacogenomic analysis and systems-level biomarker profiling 157. These tailored strategies may increase the effectiveness of treatment while reducing side effects.
Including Preventive Metabolic Health Techniques: Phlorizin has considerable translational promise in preventive health contexts due to its origin as a dietary polyphenol and its molecular influence on glucose transport 152. In people with prediabetes, reduced glucose tolerance, or early metabolic dysfunction, standardized phlorizin-rich extracts from apple sources may be assessed.
In both epidemiological and interventional investigations, dietary flavonoids have been linked to better cardiometabolic outcomes 155-156. Controlled clinical studies evaluating:
- Relationships between dose and response
- decrease inglycaemic variability
- Improvements in insulin sensitivity
- Impact on Inflammatory Markers
Would offer the evidentiary base required for the positioning of nutraceuticals. Moreover, integration into lifestyle change initiatives can provide complementary metabolic advantages.
Integration of Multi-Omics and Systems Biology: Integrative systems biology frameworks are becoming more and more important in modern translational science. Metabolomics, proteomics, and transcriptomics are examples of multi-omics platforms that might clarify global biological reactions to SGLT regulation beyond glucose reduction. For example, changes in substrate use, increased ketone body synthesis, and modified myocardial energetics have all been associated with SGLT2 inhibition 153-154.
How these metabolic adjustments lead to cardiovascular and renal advantages may become clearer with systems-level modelling. Furthermore, physiologically based pharmacokinetic (PBPK) modelling can improve formulation techniques, simulate tissue distribution, and improve dose prediction 157. Computational modelling combined with omics data will enhance translational predictability and enable logical medication design.
Additionally, integrated computational models may direct logical drug design and forecast treatment outcomes.
Studies of the Microbiome-Host Interaction: Recent studies emphasize how the gut bacteria regulate polyphenol metabolism and its systemic effects 156. Phlorizin is deglycosylases in the intestinal lumen 155 and compounds produced by microbes may have further biological effects.
Opportunities for translation include:
- Finding markers of the microbiota linked to enhanced metabolic response
- Creating probiotic strains with phlorizin to create symbiotic formulations
- Using targeted metabolomics to characterize microbial metabolites.
Pathways for Clinical and Regulatory Development: A thorough clinical examination is necessary for translation to be successful. The significance of large-scale randomized controlled trials in proving cardiovascular and renal protection is demonstrated by lessons learned from the development of SGLT2 inhibitors 153-154.
The following should be included in future translational development of derivatives based on phlorizin or structurally optimized:
- Pharmacokinetic and safety phase I trials
- Phase II research motivated by mechanistic biomarkers
- Trials of cardiometabolic outcomes
Commercialization methods and evidentiary criteria will be determined concurrently by regulatory classification, whether as a pharmaceutical, nutraceutical, or functional food additive.
CONCLUSION: Phlorizin is a naturally occurring dihydrochalcone glycoside that is mostly present in the fruit, leaves, and bark of apple trees. Because of its varied pharmacological actions and function as a predecessor to contemporary sodium–glucose cotransporter (SGLT) inhibitors, it has garnered significant scientific attention over the past few decades. This thorough analysis demonstrates that phlorizin is a potentially bioactive substance with substantial therapeutic promise because of its many biological characteristics, which include antidiabetic, antioxidant, anti-inflammatory, cardioprotective, neuroprotective, and anticancer effects.
Phlorizin's capacity to block sodium-glucose cotransporters (SGLT1 in the gut and SGLT2 in the kidney) is one of its most significant pharmacological characteristics. Phlorizin lowers blood glucose levels by reducing intestinal and renal glucose absorption by inhibiting these transporters. The scientific basis for the creation of contemporary SGLT2 inhibitor medications used to treat type 2 diabetes mellitus was established by this process. Phlorizin exhibits potent antioxidant activity by scavenging reactive oxygen species (ROS) and lowering oxidative stress in addition to its glucose-lowering actions. This is important for preventing metabolic disorders and chronic diseases. By modifying inflammatory signalling pathways and cytokine production, phlorizin also demonstrates anti-inflammatory qualities. Its preventive effects against metabolic syndrome, obesity-related comorbidities, and cardiovascular illnesses are partly due to these mechanisms. Additionally, new research indicates that phlorizin may prevent neurodegenerative diseases by lowering oxidative damage and inflammation in neural regions. New directions for medicinal study are also opened by its possible anticancer activity, which is associated with the regulation of cell proliferation and death pathways.
Despite these encouraging pharmacological effects, phlorizin's therapeutic use is limited by a few factors. The substance has a comparatively low oral bioavailability and is quickly degraded into phloretin in the colon by lactase-phlorizin hydrolase, which could change its biological effects. Its systemic therapeutic efficacy may also be diminished by stability problems and fast metabolism. Recent research is investigating cutting-edge delivery methods such as Nano formulations, encapsulating methods, and structural alterations to enhance its stability, bioavailability, and pharmacokinetic profile in order to overcome these obstacles.
From the standpoint of nutraceuticals and functional foods, phlorizin is a useful natural substance that can be obtained sustainably from apple peel and pomace. In addition to adding value to agricultural waste, this is consistent with sustainable food production systems and the circular bioeconomy. Phlorizin-rich extracts may help avoid chronic diseases, promote metabolic health, and regulate blood sugar levels when added to functional meals or dietary supplements.
To sum up, phlorizin is a versatile natural substance with substantial pharmacological significance and therapeutic potential. It is a promising option for future medication development and nutraceutical applications due to its varied modes of action, especially in glucose metabolism, oxidative stress management, and inflammatory modulation. To completely clarify its therapeutic efficacy, safety, and long-term advantages, more study is required, including carefully planned clinical trials and sophisticated formulation techniques. Further research into phlorizin may eventually aid in the creation of innovative medicinal substances and functional food items meant to enhance human health and fend against chronic illnesses.
ACKNOWLEDGEMENT: No acknowledgement to be included.
CONFLICT OF INTEREST: No conflict of interest to be declared.
REFERENCES:
- Newman DJ and Cragg GM: Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J Nat Prod 2020; 83(3): 770–803.
- Harvey AL, Edrada-Ebel R and Quinn RJ: The re-emergence of natural products for drug discovery in the genomics era. Nat Rev Drug Discov 2015; 14(2): 111–129.
- Ehrenkranz JRL, Lewis NG, Kahn CR and Roth J: Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Zheng Y, Ley SH and Hu FB: Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol 2018; 14(2): 88–98.
- Bailey CJ: Renal glucose reabsorption inhibitors to treat diabetes. Trends Pharmacol Sci 2011; 32(2): 63–71.
- Vinayagam R and Xu B: Antidiabetic properties of dietary flavonoids: a cellular mechanism review. Nutr Metab 2015; 12: 60.
- Ehrenkranz JR, Lewis NG, Kahn CR and Roth J: Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Wright EM, Loo DDF and Hirayama BA: Biology of human sodium glucose transporters. Physiol Rev 2011; 91(2): 733–794.
- Abdul-Ghani MA, Norton L and DeFronzo RA: Role of sodium-glucose cotransporter 2 inhibitors in treatment of type 2 diabetes. Diabetes Care 2011; 34(2): 219–225.
- Gosch C, Halbwirth H and Stich K. Phloridzin: biosynthesis, distribution and physiological relevance in plants. Phytochemistry 2010; 71(8–9): 838–843.
- Tsao R and Yang R: Optimization of a new mobile phase to know the complex and real polyphenolic composition: toward a total phenolic index. J Agric Food Chem 2003; 51(21): 6347–6353.
- Yokozawa T and Nakagawa T: Inhibitory effects of phlorizin on diabetic oxidative stress. Biol Pharm Bull 2006; 29(11): 2226–2230.
- Meng W, Ellsworth BA and Nirschl AA: Discovery of dapagliflozin. J Med Chem 2008; 51(5): –1149.
- De Koninck J: Recherches sur la phloridzine. Ann Chim Phys 1835; 59: 91–99.
- von Mering J. Zur Wirkung der Phloridzin. Z Klin Med 1886; 10: 175–185.
- Chasis H, Jolliffe N and Smith HW: The action of phlorizin on renal function. J Clin Invest 1933; 12(6): 1083–1090.
- Hediger MA, Coady MJ, Ikeda TS and Wright EM: Expression cloning of the Na+/glucose cotransporter. Nature 1987; 330(6146): 379–381.
- Kanai Y, Lee WS, You G, Brown D and Hediger MA: The human kidney low affinity Na+/glucose cotransporter. J Clin Invest 1994; 93(1): 397–404.
- Nomura S, Sakamaki S and Hongu M: Discovery of canagliflozin. J Med Chem 2010; 53(17): 6355–6360.
- Vrhovsek U, Rigo A, Tonon D and Mattivi F: Quantitation of polyphenols in apples. JAFC 2004; 52(21): 6532–6538.
- Gosch C, Halbwirth H and Stich K: Distribution of phloridzin in apple tissues. Phytochemistry 2009; 70(18): 2112–2118.
- Escarpa A and González MC: High-performance liquid chromatography determination of phenolics. J Chromatogr A 2001; 897(1–2): 161–170.
- Lu Y and Foo LY: Antioxidant and radical scavenging activities of polyphenols from apple pomace. Food Chem 2000; 68(1): 81–85.
- Treutter D: Significance of flavonoids in plant resistance. Environ Chem Lett 2006; 4(3): 147–157.
- National Center for Biotechnology Information. PubChem Compound Summary for CID 6072, Phlorizin.
- Rice-Evans CA, Miller NJ and Paganga G: Structure-antioxidant activity relationships. Free Radic Biol Med 1996; 20(7): 933–956.
- Crespy V, Aprikian O and Morand C: Bioavailability of phloretin and phlorizin in rats. J Nutr 2001; 131(12): 3227–3230.
- Faham S, Watanabe A and Besserer GM: The crystal structure of a sodium galactose transporter. Science 2008; 321(5890): 810–814.
- Manach C, Williamson G, Morand C, Scalbert A and Rémésy C: Bioavailability of polyphenols. Am J Clin Nutr 2005; 81(1): 230-242.
- Ehrenkranz JR: Pharmacokinetics of phlorizin. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Vogt T: Phenylpropanoid biosynthesis. Mol Plant 2010; 3(1): 2–20.
- Dixon RA and Paiva NL: Stress-induced phenylpropanoid metabolism. Plant Cell 1995; 7(7): 1085–1097.
- Dare AP: Dihydrochalcone biosynthesis in apple. Plant Physiol 2013; 161(3): 1251–1264.
- Gosch C: Biosynthesis of phloridzin in apple. Phytochemistry 2010; 71(8–9): 838–843.
- Halbwirth H: Tissue-specific gene expression in apple phenylpropanoid pathway. Plant Sci 2006; 170(4): 819–827.
- Treutter D: Significance of flavonoids in plant resistance. Environ Chem Lett 2006; 4(3): 147–157.
- Li X: Transcriptomic analysis of apple polyphenol biosynthesis. BMC Plant Biol 2012; 12: 33.
- Wang J: Extraction optimization of phlorizin. Food Chem 2016; 204: 1–7.
- Escarpa A and González MC: Determination of phenolics. J Chromatogr A 2001; 897(1–2): 161–170.
- Chemat F: Ultrasound-assisted extraction. Ultrason Sonochem 2017; 34: 540–560.
- Mandal V: Microwave-assisted extraction. Phcog Rev 2007; 1(1): 7–18.
- Herrero M: Supercritical fluid extraction of bioactives. J Chromatogr A 2010; 1217(16): 2495–2511.
- Lin LZ: LC-MS identification of apple polyphenols. J Agric Food Chem 2005; 53(10): 3804–3813.
- Vrhovsek U: Quantitation of polyphenols. J Agric Food Chem 2004; 52(21): 6532–6538.
- Stalmach A: LC-MS analysis of apple phenolics. Rapid Commun Mass Spectrom 2011; 25(15): 2141–2148.
- Sun J: Capillary electrophoresis of flavonoids. Electrophoresis 2005; 26(20): 3944–3951.
- Crespy V: Intestinal metabolism of phlorizin. J Nutr 2001; 131(12): 3227–3230.
- Manach C: Bioavailability of polyphenols. Am J Clin Nutr 2005; 81: 230-242
- Day AJ: Polyphenol conjugation and metabolism. Drug Metab Dispos 2000; 28(7): 756–763.
- Walle T: Absorption and metabolism of flavonoids. Free Radic Biol Med 2004; 36(7): 829–837.
- Zhai X: Nanocarrier delivery of phlorizin. Mol Med Rep 2018; 17(3): 4195–4202.
- Wright EM: Renal Na+-glucose cotransporters. Am J Physiol Renal Physiol 2001; 280(1): 10–18.
- Sala-Rabanal M: Molecular determinants of SGLT transport inhibition. J Biol Chem 2012; 287(50): 42625–42635.
- Rossetti L: Correction of hyperglycemia with phlorizin normalizes insulin sensitivity. J Clin Invest 1987; 79(5): 1510–1515.
- Brownlee M: Biochemistry of diabetic complications. Nature 2001; 414(6865): 813–820.
- Kahn BB and Flier JS: Obesity and insulin resistance. J Clin Invest 2000; 106(4): 473–481.
- Shimano H: SREBP regulation of lipid metabolism. Prog Lipid Res 2001; 40(6): 439–452.
- Yokozawa T: Phlorizin and lipid metabolism. Biol Pharm Bull 2006; 29(11): 2226–2230.
- Hardie DG: AMPK signaling. Nat Rev Mol Cell Biol 2016; 17(9): 495–510.
- Vessal M: Anti-inflammatory effects of phlorizin. J Med Plants Res 2010; 4(2): 169–174.
- Nishikawa T: Hyperglycemia-induced mitochondrial superoxide. Nature 2000; 404(6779): 787–790.
- Walle T: Inhibition of GLUT transport by flavonoids. Mol Pharmacol 2003; 63(3): 677–684.
- Vallon V: Renal effects of SGLT inhibition. Curr Opin Nephrol Hypertens 2011; 20(1): 36–41.
- Kim J: AMPK and autophagy. Mol Cell 2011; 44(2): 203–215.
- Tesfamariam B: Free radicals in diabetic endothelial dysfunction. Free Radic Biol Med 1994; 16(3): 383–391.
- Lee TM: SGLT inhibition and cardiac remodeling. Cardiovasc Res 2017; 113(12): 1443–1453.
- Ferrannini E: Mechanisms of cardiovascular benefit of SGLT2 inhibition. Circulation 2016; 134(10): 752–772.
- Vallon V: Renoprotective mechanisms of SGLT inhibition. Kidney Int 2014; 86(4): 693–700.
- Rossetti L: Chronic phlorizin treatment in diabetic rats. J Clin Invest 1987; 79(5): 1510–1515.
- Yamamoto T: Effects of SGLT inhibition on adipokines. Metabolism 2011; 60(5): 638–646.
- Duarte AI: Polyphenols in neuroprotection. Neurochem Int 2012; 61(3): 391–402.
- Liu Y: Phloretin inhibits cancer cell growth. Oncol Rep 2010; 23(3): 715–720.
- Powell DR: SGLT1 inhibition and intestinal glucose handling. Diabetes 2013; 62(10): 3404–3412.
- Wright EM, Loo DD and Hirayama BA: Biology of human sodium glucose transporters. Physiol Rev 2011; 91(2): 733–794.
- Ehrenkranz JR, Lewis NG, Kahn CR and Roth J: Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Chasis H, Jolliffe N and Smith HW: The action of phlorizin on renal glucose excretion. J Clin Invest 1933; 12(6): 1083–1090.
- Rossetti L, Smith D, Shulman GI, Papachristou D and DeFronzo RA: Correction of hyperglycemia with phlorizin normalizes insulin sensitivity in diabetic rats. J Clin Invest 1987; 79(5): 1510–1515.
- Wright EM: Renal Na+-glucose cotransporters. Am J Physiol Renal Physiol 2001; 280(1): 10–18.
- Boyer J and Liu RH: Apple phytochemicals and their health benefits. Nutr J 2004; 3: 5.
- Washburn WN: Evolution of sodium–glucose cotransporter 2 inhibitors. J Med Chem 2009; 52(7): 1785–1794.
- Yokozawa T and Nakagawa T: Inhibitory effects of phlorizin on diabetic oxidative stress and renal injury. Biol Pharm Bull 2006; 29(11): 2226–2230.
- Mogensen CE: Glomerular hyperfiltration in diabetes. Kidney Int 1971; 1(1): 7–17.
- Zinman B, Wanner C and Lachin JM: Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373(22): 2117–2128.
- Abdul-Ghani MA and DeFronzo RA: Sodium–glucose cotransporter inhibitors: mechanisms and clinical applications. Diabetes Care 2011; 34(2): 219–225.
- Neal B, Perkovic V and Mahaffey KW: Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017; 377(7): 644–657.
- DeFronzo RA and Norton L: Sodium–glucose cotransporter 2 inhibitors. Diabetes Care 2017; 40(5): 78–e79.
- Crespy V, Aprikian O and Morand C: Bioavailability of phloretin and phlorizin in rats. J Nutr 2001; 131(12): 3227–3230.
- Dahlqvist A: Assay of intestinal disaccharidases. Biochem J 1964; 92(1): 8–14.
- Tsao R, Yang R, Young JC and Zhu H: Polyphenolic profiles in apples. J Agric Food Chem 2003; 51(21): 6347–6353.
- Giacco F and Brownlee M: Oxidative stress in diabetic complications. Circ Res 2010; 107(9): 1058–1070.
- Pfützner A: SGLT2 inhibitor safety in large clinical trials. Clin Res Cardiol 2018; 107(1): 13–22.
- Wright EM, Loo DD and Hirayama BA: Biology of human sodium glucose transporters. Physiol Rev 2011; 91(2): 733–794.
- Ehrenkranz JR, Lewis NG, Kahn CR and Roth J: Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Dahlqvist A: Assay of intestinal disaccharidases. Biochem J 1964; 92(1): 8–14.
- Crespy V, Aprikian O and Morand C: Bioavailability of phloretin and phlorizin in rats. J Nutr 2001; 131(12): 3227–3230.
- Washburn WN: Evolution of sodium–glucose cotransporter 2 inhibitors. J Med Chem 2009; 52(7): 1785–1794.
- Tsao R, Yang R, Young JC and Zhu H: Polyphenolic profiles in apples. J Agric Food Chem 2003; 51(21): 6347–6353.
- DeFronzo RA and Norton L: Sodium–glucose cotransporter 2 inhibitors: mechanisms and clinical applications. Diabetes Care 2017; 40(5): 78–e79.
- Zhai X, Peng R and Cao J: Nanocarrier delivery of phenolic compounds, including phlorizin. Mol Med Rep 2018; 17(3): 4195–4202.
- Costa C: Advances in nanosystems for delivery of natural products: phlorizin as a model. Pharmaceutics 2019; 11(12): 628.
- Loftsson T and Brewster ME: Pharmaceutical applications of cyclodextrins: basic science and product development. J Pharm Pharmacol 2010; 62(11): 1607–1621.
- Chao EC and Henry RR: SGLT2 inhibition—a novel strategy for diabetes treatment. Nat Rev Drug Discov 2010; 9(7): 551–559.
- Meng W: Discovery of dapagliflozin. J Med Chem 2008; 51(5): 1145–1149.
- Schieber A: Apple pomace as a source of bioactive compounds. Trends Food Sci Technol 2001; 12: 401–413.
- Mirabella N: Current options for waste valorization. Waste Manag 2014; 34: 256–268.
- McClements DJ: Nanoemulsions in food systems. J Food Sci 2012; 77: 1–10.
- Newman DJ and Cragg GM: Natural products as sources of new drugs. J Nat Prod 2016; 79: 629–661.
- Day AJ: Dietary flavonoid glycosides are hydrolysed by lactase-phlorizin hydrolase. FEBS Lett 2000; 468: 166–170.
- Ehrenkranz JRL, Lewis NG, Kahn CR, Roth J. Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Tsao R and Yang R: Optimization of a new mobile phase to know the complex and real polyphenolic composition of apples. J Chromatogr A 2003; 1018: 29–40.
- Scalbert A and Williamson G: Dietary intake and bioavailability of polyphenols. J Nutr 2000; 130: 2073–2085.
- Manach C: Bioavailability of polyphenols. Am J Clin Nutr 2005; 81: 230–242.
- Boyer J and Liu RH: Apple phytochemicals and their health benefits. Nutr J 2004; 3: 5.
- Hyson DA: A comprehensive review of apples and apple components. Adv Nutr 2011; 2: 408–420.
- Wright EM: Biology of human sodium glucose transporters. Physiol Rev 2011; 91: 733–794.
- Rieg T and Vallon V: Development of SGLT inhibitors. Diabetologia 2018; 61: 2079–2086.
- Powell DR: Selective SGLT1 inhibition. Diabetes Ther 2013; 4: 29–41.
- Schieber A: Apple pomace as a source of bioactive compounds. Trends Food Sci Technol 2001; 12: 401–413.
- Vendruscolo F: Apple pomace as industrial waste. J Food Eng 2008; 85: 49–55.
- Lu Y and Foo LY: Polyphenolics of apple pomace. Food Chem 2000; 68: 81–85.
- Mirabella N: Current options for waste valorization. Waste Manag 2014; 34: 256–268.
- Lu Y and Foo LY: Antioxidant activities of polyphenols. Food Chem 2001; 72: 327–332.
- Testa R: Antioxidant properties of polyphenols. Oxid Med Cell Longev 2011; 2011: 1–9.
- Hanhineva K: Impact of dietary polyphenols on carbohydrate metabolism. Int J Mol Sci 2010; 11: 1365–1402.
- Day AJ: Flavonoid glycoside hydrolysis. FEBS Lett 2000; 468: 166–170.
- McClements DJ: Nanoemulsions in food systems. J Food Sci 2012; 77: R1–R10.
- Hediger MA and Rhoads DB: Molecular physiology of SGLTs. Physiol Rev 1994; 74: 993–1026.
- Wright EM: Renal Na-glucose cotransporters. Am J Physiol Renal Physiol 2001; 280: 10–18.
- Rossetti L: Correction of hyperglycemia with phlorizin. J Clin Invest 1987; 79: 1510–1515.
- Rossetti L, Giaccari A and DeFronzo RA: Glucose toxicity. Diabetes Care. 1990; 13:610–630.
- DeFronzo RA: SGLT2 inhibitors and renal glucose transport. Diabetes Care 2012; 35: 215–225.
- Vallon V: SGLT inhibition and nephroprotection. J Am Soc Nephrol 2011; 22: 104–112.
- Sena CM: Oxidative stress in diabetes. Free Radic Biol Med 2013; 65: 219–230.
- Oka Y: Phloretin as GLUT inhibitor. J Biol Chem 1990; 265: 1926–1931.
- Vander Heiden MG: The Warburg effect. Science 2009; 324: 1029–1033.
- Chao EC and Henry RR: SGLT2 inhibition strategy. Nat Rev Drug Discov 2010; 9: 551–559.
- Meng W: Discovery of dapagliflozin. J Med Chem 2008; 51: 1145–1149.
- Abdul-Ghani MA, Norton L and DeFronzo RA: SGLT2 inhibitors. Endocr Rev 2011; 32: 515–531.
- Zinman B: Empagliflozin cardiovascular outcomes. N Engl J Med 2015; 373: 2117–2128.
- Ferrannini E: Dual SGLT1/2 inhibition. Diabetes Care 2014; 37: 1800–1807.
- Newman DJ and Cragg GM: Natural products as drug sources. J Nat Prod 2016; 79: 629–661.
- Ehrenkranz JRL, Lewis NG, Kahn CR and Roth J: Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Zinman B: Empagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015; 373(22): 2117–2128.
- McMurray JJV: Dapagliflozin in heart failure. N Engl J Med 2019; 381(19): 1995–2008.
- Day AJ: Flavonoid glycoside hydrolysis by lactase-phlorizin hydrolase. FEBS Lett 2000; 468: 166–170.
- Crespy V: Phlorizin and phloretin bioavailability in rats. J Nutr 2001; 131(12): 3227–3230.
- Rowland M: Physiologically based pharmacokinetics. Nat Rev Drug Discov 2011; 10: 664–673.
- Powell DR: SGLT1 inhibition and postprandial glucose control. Diabetes 2013; 62(10): 3404–3412.
- Ferrannini E: Dual SGLT inhibition. Diabetes Care 2014; 37: 1800–1807.
- Torchilin VP: Multifunctional nanocarriers. Nat Rev Drug Discov 2005; 4: 145–160.
- Vander Heiden MG: Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 2009; 324(5930): 1029–1033.
- Ehrenkranz JRL, Lewis NG, Kahn CR and Roth J: Phlorizin: a review. Diabetes Metab Res Rev 2005; 21(1): 31–38.
- Zinman B, Wanner C and Lachin JM: Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373(22): 2117–2128.
- McMurray JJV, Solomon SD and Inzucchi SE: Dapagliflozin in patients with heart failure and reduced ejection fraction. NEJM 2019; 381(21): 1995–2008.
- Day AJ, Cañada FJ and Díaz JC: Dietary flavonoid and isoflavone glycosides are hydrolysed by the lactase site of lactase-phlorizin hydrolase. FEBS Lett 2000; 468(2-3): 166–170.
- Crespy V, Aprikian O and Morand C: Bioavailability of phloretin and phloridzin in rats. J Nutr 2001; 131(12): 3227–3230.
- Rowland M, Peck C and Tucker G: Physiologically based pharmacokinetics in drug development and regulatory science. Nat Rev Drug Discov 2011; 10(9): 664–673.
- Kumari R, Singh RK and Garg A: Herbal plants used in the treatment of PCOS: A comprehensive review. Int J Pharma Prof Res (IJPPR) 2024; 15(1): 57-71.
- Ranjan Kumar Singh, Ajay Garg and Khushboo Shrimali: Botanical description, Photochemistry, Traditional Uses, and Pharmacology of the “Wonder Plant” Kalanchoe pinnata (Linn.) Pers: An Updated Review/J Pharmaceut Med Chem 2022; 8(1): 11-24.
- Singh RK, Garg A, Shrimali K, Jain V, Sinha SK and Pradhan J: A brief review on Solanum xanthocarpum along with phytochemical and pharmacological profile. Adv Pharm J 2022; 23-31.
- Singh RK, Garg A, Shrimali K, Jain V, Sinha SK and Pradhan J: A brief review on Solanum xanthocarpum along with phytochemical and pharmacological profile. Adv Pharm J 2022; 23-31.
- Ranjan Kumar Singh, Satpal Kushwaha, Mantun Prasad Gupta, Ajay Garg, Vaibhav Saxena and Firoz Ahmed: Pharmacological Assessment of Medicinal Flora with Antidiarrheal Properties: Integrating Traditional Wisdom and Contemporary Therapeutics. IJPRMP [Internet]. 2025 Dec. 31 [cited 2026 Apr. 21]; 1(3):01-8. Available from: https://www.informativejournals.com/journal/index.php/Journal_Medicinal_Plants/article/view/215.
How to cite this article:
Konyak N, Singh RK, Jain N and Sharma T: “Phlorizin: a comprehensive review of its pharmacology, mechanisms, and therapeutic potentials”. Int J Pharmacognosy 2026; 13(5): 337-67. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(5).337-67.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
1
337-367
2837 KB
10
English
IJP
Nyeme Konyak, Ranjan Kumar Singh *, Neeraj Jain and Tanya Sharma
Department of Pharmaceutical Sciences, Mewar University, Gangrar, Chittorgarh, Rajasthan, India.
rxsingh41800557@gmailcom
18 April 2026
02 May 2026
08 May
10.13040/IJPSR.0975-8232.IJP.13(5).337-67
15 May 2026