PHARMACOGNOSTICAL, PHYSICO-CHEMICAL AND PRELIMINARY PHYTOCHEMICAL ANALYSIS OF WHOLE PLANT OF PIPER LONGUM
HTML Full TextPHARMACOGNOSTICAL, PHYSICO-CHEMICAL AND PRELIMINARY PHYTOCHEMICAL ANALYSIS OF WHOLE PLANT OF PIPER LONGUM
Fathima Suman *, Minhaj, I. Arthi, S. Neethu Krishnan and V. K. Riyama Shirin
Department of Pharmacognosy and Phytochemistry 1, 3, 4, 5, College of Pharmaceutical Sciences, Government Medical College Kannur, Kerala, India.
ABSTRACT: This study deals with the Pharmacognostical physicochemical and phytochemical evaluation of the whole plant of Piper longum L. Pharmacognostical studies includes macroscopic, microscopic, and physicochemical evaluation. Physicochemical constants showed the values of total ash (10%), acid insoluble ash (1%), water-soluble ash (32%), sulphated ash (22%), nitrated ash (18.5%), hot extraction (11.2%), water-soluble extractive (4.8%), ethanol-soluble extractive (12.4%), ether soluble extractive (5.6%), loss on drying (29%), swelling index (0.33) and mucilage content (0.8%) w/w. The preliminary phytochemical screening of the alcoholic extract showed alkaloids, phenolic compounds, tannins, mucilage, starch, steroid, and flavanoids. This study helps to development of different standard parameters of the plant, which help the plant identification and authentication.
Keywords: Piper longum L., Whole plant, Pharmacognostic, Phytochemical, Extract
INTRODUCTION: Medicinal plants have great economic value in the Indian subcontinent. The documentation of conventional knowledge, particularly the medicinal uses of plants in history, has provided many important drugs of the modern-day 1. Among historical civilizations, India has been recognized to be a rich repository of medicinal plants. The woodland of India is the major repository of a large number of aromatic and medicinal plants; raw materials are collected for the manufacture of drugs and perfumery products. About 8,000 herbal remedies have been reported
and used in AYUSH systems in India. Ayurveda, Unani, Siddha, and Folk (tribal) treatments are the major systems of indigenous medicines. Among these systems, Ayurveda and Unani Medicine are the most advanced and broadly practiced in India 2. More than seven hundred species are present in the genus Piper; they grow in the subtropical and tropical rain forests. The indigenous system of medicine uses the majority of the piper species in India 3.
Piper longum L. is a small shrub with large roots in woody nature; the stem is numerous, creeping, jointed, and thickening at the nodes. The leaves are of alternate nature, spreading, without stipules, and with blades varying greatly in size. The fruits grow as fleshy spikes of 2.5-3.5 cm long and 5 mm thickness, oblong in shape, blunt, and blackish-green in colour. It is seen in the hotter parts of India, central Himalayas to Assam, the khasi and the mikir hills, the lower hills of Bengal, and the evergreen forests of the Western Ghats from konkan to Travancore. Its pharmacological actions include antibacterial, hypoglycemic, antispasmodic, cough suppressor, antigiardial, immuno stimulatory, hepatoprotective, analeptic, anti-inflammatory, insecticidal, antimalarial, CNS stimulant, antitubercular, anthelmintic, antinarcotic, antiulcerogenic 4.
MATERIALS AND METHODS:
Collection and Authentication of Plant: Piper longum L. was collected from Malappuram District, Kerala, during November 2020. It was an authentic filled by Dr. P. Sreeja, Department of Botany and Research Centre Sir Syed College Taliparamba, Kannur, Kerala. The specimen voucher has been deposited in the Department of Pharmacognosy, College of Pharmaceutical Sciences, Government Medical College, Kannur, Kerala.
Pharmacognostical Studies:
Macroscopic Evaluation: Macroscopic study was carried out by standard methods to determine the different external characters of stem, leaf, and root. The morphological characters of plants include size, shape, colour, odour, taste, appearance, which were studied 5.
Microscopic Evaluation:
Transverse Section 6: Microscopic studies were carried out by preparing thin sections of leaf, stem and root of Piper longum L. The fresh or dried parts of the plant were cut into thin sections using a razor blade.
Select the thin section of the material. Stain it using phloroglucinol and HCl, wash the excess stain with water. First, it is observed under the compound microscope after staining and mounting, and then the section is observed with the aid of Leica M80 Stereomicroscope with camera Lucida .
Powder Microscopy: The dried parts of the plant were crushed separately to form a powder and then passed through the sieve no.40 to get uniform powder mass. Then the powder was stained with phloroglucinol in the presence of hydrochloric acid, and the lignified structures were viewed under microscope 7.
Quantitative Leaf Microscopy
Vein- Islet Number 8: Take pieces of leaf lamina with an area of 4 square millimeters from the central portion of the lamina. Clear the pieces by heating in a test tube containing chloral hydrate solution and prepare a mount in glycerol solution. Place stage micrometer on the microscope stage and examine with 6 × eyepiece and 4 ×objective. Construct a square using the line, representing 4 square millimeters of area.
Place the slide with the cleared leaf piece on the microscope and draw in the veins and veinlets within the square. Count the number of vein-islets within the square, including those overlapping on two adjacent sides and excluding those intersected by the other two sides. The obtained result is the number of vein-islets in 4 square millimeters area.
Vein Termination Number 9: A piece of the leaf was cleared by boiling in chloral hydrate solution, and camera lucida and drawing board were arranged, and 1mm line was drawn with the help of stage mm. A square was constructed on this line in the center of the field. Using the slide, the number of veinlet termination present within the square was counted. The average number of veinlet termination numbers from the four squares to get the value for 1 square mm was found as vein termination number.
Stomatal Index 8: Place leaf fragments of about 5 × 5 mm in size in a test tube containing chloral hydrate solution and heat it for about 15 min. Transfer a fragment to a slide and mount the lower epidermis uppermost and put a small drop of glycerol-ethanol solution on one side of the coverslip to prevent the preparation from drying. Examine with a 6 × eyepiece and 40 × objectives to which a microscopical drawing apparatus is attached. Mark on the drawing paper across for each epidermal cell and a circle for each stoma. Calculate the result as follows:
S × 100/ Stomatal index = E + S
Where S = the number of stomata in a given leaf area; and E = the number of epidermal cells, including trichomes in the same area of the leaf. For each sample of the leaf, make not fewer than ten determinations and calculate the average index
Physicochemical Parameters 10, 11, 12:
Ash Values:
Total Ash: About 2 g of the powdered drug was weighed accurately and spread AS a fine layer at the bottom of the tared silica crucible. The crucible was incinerated at a temperature not exceeding 450 °C until free from carbon. The crucible was cooled and weighed. The entire procedure was repeated till a constant weight was observed. The percentage of total ash was calculated concerning the air-dried drug.
Acid Insoluble Ash: The ash obtained in the total ash was boiled with 25 ml of hydrochloric acid for 5 min. The insoluble ash was collected on an ash-less filter paper by filtration and washed with hot water. The insoluble ash was transferred to a tared silica crucible together with ash-less filter paper and ignited at a temperature not exceeding 600 °C, cooled, weighed. The procedure was repeated till a constant weight was observed. The percentage of acid insoluble ash was calculated with reference to the air-dried drug.
Water Soluble Ash: The ash obtained as described in total ash was boiled with 25 ml of hot water for 5 min, filtered on an ashless filter paper, and washed with hot water. The insoluble ash was transferred to a tared silica crucible and ignited at 600 °C. The procedure was repeated to get a constant weight. The weight of the insoluble matter was subtracted from the weight of the total ash. The difference in weight was considered for water-soluble ash. The percentage of the water-soluble ash was calculated with reference to the air-dried drug.
Sulphated Ash 13: A silica or platinum crucible was heated to redness for 10 min, allowed to cool in a desiccator, and weighed. 1 to 2 g of the test drug substance accurately weighed in the crucible was ignited gently at first until the substance was thoroughly charred.
The residue was cooled and moistened with 1 ml of sulphuric acid, heated gently until white fumes no longer evolved, and then ignited at 800 + 25 °C until all the black particles disappeared. The ignition was conducted in a place protected from air currents. The crucible was allowed to cool, and then a few drops of sulphuric acid was added and ignited as before.
It was allowed to cool and then weighed. The operation was repeated until two successive weighings did not differ by more than 0.5 mg.
Nitrated Ash: A silica or platinum crucible was heated to redness for 10 min, and procedure as mentioned above in the determination of sulphated ash was carried out. Instead of sulphuric acid, nitric acid was used here, and the rest of the procedure was the same as mentioned.
Extractive Values: Extractive value is a measure of the content of the drug and its nature.
Cold Maceration: The dried powdered plant was extracted with water, ethanol, petroleum ether, and chloroform using the maceration process. The powdered plant material was weighed for about 5 g and then transferred into a 250 ml conical flask. The conical flask was filled with different solvents for 100 mL separately. The flask was kept aside for 24 h at room temperature with continuous agitation for the first 6 h, then allowed to stand for 18 h. The mixtures were then filtered through Whatman filter paper. After that, only 25 mL of the filtrate was taken and kept above the heating mantle for drying at a constant temperature. The final extractive values were calculated in percentage
Hot Percolation: 5 g of the drug was taken into a conical flask, and 100 ml of water was added to it. This mixture was stirred gently and warmed in a water bath for 30 min. The solution was shaken gently at intervals. Then the solution was taken from the water bath and cooled, and filtered through a cotton plug. 25 ml of the filtrate was taken and evaporated to dryness. The amount of the extract was weighed and multiplied by 4 to get the actual value.
Moisture Content: About 1 g of the drug was taken and powdered. A glass-stoppered bottle was dried for 30 min under the same conditions to be employed in the determination, and the weight of the bottle was taken. The sample was transferred into the bottle and weight of the bottle with the contents was noted. The sample was distributed evenly and was placed in the drying chamber (Oven). The stopper was removed and left it in the chamber. The drying was carried out by heating to 100-105 °C.
Then, the bottle was removed from the oven, and the bottle was closed promptly. The bottle was allowed to cool to room temperature and weighed. The experiment was repeated to get a constant value.
Foaming Index: About 1g of the weighed plant material reduced to a coarse powder was transferred to a 500 ml conical flask containing 100 ml of boiling water and boiled moderately for 30 min. Then, it was cooled and filtered into 100 ml volumetric flask, and sufficient water was added through the filter to dilute the volume to 100 ml. The above decoction was placed into 10 stoppered test tubes (ht. 16 cm, dia. 16 mm) in a series of successive portions of 1, 2, 3 up to 10 ml. The volume of the liquid in each tube was adjusted with water to 10 ml. The tubes were stoppered and shaken in a lengthwise motion for 15 seconds of 2 frequencies per second. The tubes were allowed to stand for 15 min. The tubes were allowed to stand for 15 min, and the height of the foam was measured.
Swelling Index: 1 g of the drug was transferred to a 100 ml stoppered cylinder containing 90 ml of water. After shaking well for 30 seconds, it was allowed to stand for 24 h, shaking gently on three occasions during this period. Sufficient water was added to produce 100 ml. It was mixed gently for 30 seconds and allowed to stand for 5 h. The final volume was measured. The determination was repeated 3 times.
Mucilage Content: 5 g of the powdered drug was taken and 100 ml of water was added to obtain the aqueous extract of the powder. It was filtered through cotton, and the mucilaginous filtrate was collected. 10 ml of this mucilage solution was mixed with 25 ml of absolute alcohol to precipitate the mucilage.
This mucilage precipitate was collected by filtration with a tared filter paper. The filter paper was dried along with the mucilage. After drying the filter paper was carefully weighed and the mucilage content was calculated from the weight.
Preliminary Phytochemical Screening 10, 14: The coarsely powdered dried whole plant of Piper longum L. was extracted with ethyl alcohol by hot extraction process (Soxhlet) for 4 h. After completion of extraction, the solvent was removed by distillation and concentrated. Chemical tests were carried out for extract of Piper longum L. whole plant in order to identify the presence of various phytochemical constituents.
RESULTS:
Morphology:
Stem: Cut pieces of the stem having distinct internodes and swollen nodes with a number of small rootlets and root scars; stout, cylindrical shape, 0.2 to 0.6 cm thickness, aromatic odour, reddish-brown to grey colour, pungent taste.
Leaves: Leaves are simple, stipulate, alternate, and petiolate or nearly sessile. Lower ones broadly ovate, cordate; upper one oblong, ovate, all entire, smooth, thin with reticulate venation, veins are raised beneath of leaf.
FIG. 1 (A) PIPER LONGUM L. PLANT (B) ROOT, LEAF AND STEM OF PIPER LONGUM L. (C) DRIED FRUIT
Root: The roots are long, cylindrical 0.4-0.6 cm thick. The exterior of the root is reddish-brown to muddy, it was longitudinally wrinkled and having roots and root scars on the surface. Aromatic odour, pungent to acrid taste.
Fruit: Fruit greenish-black to black, cylindrical in shape, 2.5 to 5 cm long and 0.4 to 1 cm thickness, consisting of minute sessile fruits, they arranged around an axis. The surface is rough and composite; broken surface shows a central axis, and 6 to 12 fruit lets arranged around an axis: taste, pungent producing numbness on the tongue. Odour is aromatic.
Microscopy:
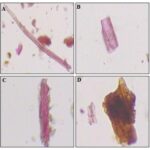
Transverse Section of Root: Transverse section shows the outer cork, middle cortex, and central stellar region. The transverse section of the root is almost circular with the regular outline. The outermost ie cork appears as a strip that is brown coloured. It consists of 3 to 5 rows of thin-walled, rectangular-slightly tangentially elongated cork cells. The cortex is wide and parenchyma except for small thick-walled cells. The cortical cells are large, thin-walled and rounded to oblong, having large intercellular space. Cell walls of the peripheral rows are slightly thickened but not lignified. Most of the cells are heavily loaded with spherical to oval-shaped starch grains. Many secretory cells are present as scattered in the cortex. Pith is occupied by 4 to 6 wedge-shaped radiating strips of vascular tissues (stellar region) having wider ends towards the outside.
The cells of pith are polygonal, thin-walled, and have starch grains. Outside the pith, evenly spaced 6 groups of primary xylem bundles are present. In each vascular strip, the xylem is composed of xylem vessels and parenchyma surrounded with woody fibers. These xylem vessels are arranged in 3 to 4 radial rows. Few thick-walled xylem parenchyma cells are there, along with wood fibers. A strip of cambium consisting of one to two rows of narrow thin-walled rectangular cells is present between the xylem and phloem. The phloem is composed of many sieve tubes with their companion cells that can be distinctly made out towards the inner region of the phloem and small thin-walled polygonal phloem parenchyma cells.
The cells at the outer convex side are slightly compressed and appear tangentially elongated. One or two groups of stone cells are present at the peripheral region of the phloem. The outer border of the phloem is limited by a row of pericyclic cells found just inner to the endodermis. Medullary rays are four to six broad wedges shaped medullar rays with their wider ends at the periphery and alternating with the radiating bands of vascular tissue. Each ray had 10-15 cells in width and extended from pith up to endodermis. The ray cells are all thin-walled cubical to slightly radially elongated and arranged in regular radial rows. Most of the cells are heavily loaded with starch grains similar to those present in the cortex. Some cells contain minute crystals of calcium oxalate and a few cells have oil globules.
Transverse Section of Stem: Shows a single-layered epidermis of round or oval-shaped, followed by a continuous ring of collenchymatous and round to oval thin-walled, parenchymatous cells; vascular bundles show peripheral and medullary arrangement, separated from each other by a wavy strip of sclerenchyma forming a ring, enclosing pith; bundles collateral and arranged in rings, having a sclerenchymatous sheath of pericyclic cap over phloem. Xylem wedge-shaped, 2-4 layered, endarch. Phloem is 4-8 layered. Starch grains simple and compound having 2-7 components, round to oval, measuring 3-14 µ in dia., present abundantly throughout the section. Non-glandular unicellular trichomes are present frequently.
Transverse Section of Leaf: The midrib is biconvex, abaxial semi-circled with no ridges and furrows, adaxial is angular. The hypodermis is 2 layered in both sides. The transverse section of the dorsiventral leaf shows the presence of single collateral vascular bundle. Stomata is hypostomatic and is cycloptic. The midrib and lamina portion's epidermis is uniseriate and is covered with thin and smooth cuticle. 2 layered palisade tissues and 3-5 layers of spongy parenchyma with no or small intercellular space is observed. Trichomes are present only in the midrib region of the leaf and are 1-3 cellular glandular trichomes confined only in the abaxial surface.
Transverse Section of Fruit: Catkin consists of 6 - 12 fruits arranged like a circle on an axis. Fruits have an outer epidermal layer of irregular cells filled with brown content and covered with a thick cuticle layer. Mesocarp consists of larger cells, collapsed, irregular in shape, and thin-walled. Several stone cells are present as singles or as groups. Endocarp and seed coat are fused to a deep zone; this outer layer is composed of thin-walled cells and is colorless; the inner layer is composed of tangentially elongated cells, having reddish-brown content. Most of the endocarp is filled with starch grains, round to oval measuring 3 - 8 µ diameter.
FIG. 2: (A) TRANSVERSE SECTION OF LEAF (B) STOMATA ON THE LOWER SIDE OF LEAF (C) TRANSVERSE SECTION OF ROOT (D) STELLAR REGION OF ROOT (E) TRANSVERSE SECTION OF STEM (F) VASCULAR BUNDLES OF STEM (G) FRUIT WITH ENDOCARP, MESOCARP, PERICARP AND GROUND TISSUE (H) VASCULAR BUNDLE OF THE FRUIT
Powder Microscopy:
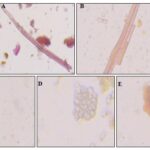
Stem: Powder is coarse, fibrous, greyish brown coloured with characteristic odour and acrid taste. Powder analysis shows the presence of fibres, xylem vessel, medullary rays and cork cell.
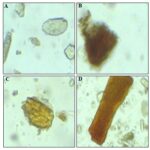
Root: Powder is coarse, fibrous, brownish-white coloured with a pungent and characteristic odour.
Powder microscopy shows the presence of cork cell, simple and compound starch grains with tannin, pitted vessels, fragments of lignified fibres and sclerides.
Leaf: The leaf powder of leaf is fine, dark green in colour, fibrous, pungent, heating in taste and with a characteristic odour. Powder microscopy shows the presence of unicellular and multicellular trichomes, epidemis with cell wall, parenchymatous cell.
Fruit: The powder of fruit is dark brown in colour, pungent and heating in taste.
The microscopical study of powder shows oval to elongated stone cells, calcium oxalate, brown content and oleoresin.
FIG. 3: POWDER MICROSCOPY OF STEM (A) FIBRE (B) PITTED XYLEM VESSEL (C) MEDULLARY RAYS (D) CORK CELL
FIG. 4: POWDER MICROSCOPY OF ROOT (A) SCLEROID (B) TRACHEIDS AND VESSELS WITH MEDULLARY RAYS (C) STARCH GRAINS (D) PARENCHYMATOUS CELLS WITH RESIN (E) CORK
Quantitative Leaf Microscopy: The quantitative leaf microscopy of Piper longum L. gives the following constants. Vein islet number – 12, Vein termination number -9, Stomatal index – 28.1
FIG. 5: POWDER MICROSCOPY OF LEAF (A) (B) TRICHOMES (C) EPIDERMIS WITH CELL WALL (D) PARENCHYMATOUS CELL
FIG. 6: POWDER MICROSCOPY OF FRUIT (A) CALCIUM OXALATE (B) BROWN CONTENT (C) OLEORESIN (D) STONE CELL
Physicochemical Parameters
| Piper longum L. | |
| Ash values | |
| (a) Total ash | 10% w/w |
| (b) Acid insoluble ash | 1% w/w |
| (c) Water soluble ash | 32% w/w |
| (d) Sulphated ash | 22% w/w |
| (e) Nitrated ash | 18.5% w/w |
| Extractive values | |
| (a) Hot extraction | 11.2% w/w |
| (b) Water soluble extractive | 4.8% w/w |
| (c) Ethanol soluble extractive | 12.4% w/w |
| (d) Ether soluble extractive | 5.6% w/w |
| Loss on drying | 29% w/w |
| Foaming index | Nil |
| Swelling index | 0.33 |
| Mucilage content | 0.8% w/w |
Phytochemical Screening
| Test for constituents | Piper longum L. |
| Carbohydrates | - |
| Starch | + |
| Gums | - |
| Mucilage | + |
| Proteins | - |
| Amino Acids | - |
| Fats and Oils | - |
| Steroids | + |
| Glycosides | - |
| Phenolic compounds | + |
| Terpenoids | - |
| Flavanoids | + |
| Alkaloids | + |
| Tannins | + |
DISCUSSION: In this study, all the important Morphological, microscopic, pharmacognostic and phytochemical properties of different parts of Piper longum L. was characterized by using standard guidelines. The morphological study states that the stem pieces having distinct internodes and swollen nodes with a number of small rootlets and root scars, cylindrical shape with 0.2-0.6 cm thickness, reddish-brown to grey colour, aromatic odour, and pungent taste.
The leaves are simple, alternate, stipulate, petiolate, or nearly sessile; lower leaves are broadly ovate and cordate. Upper one oblong, ovate, all entire, smooth, thin with reticulate venation; veins raised beneath. The roots are long, cylindrical 0.4-0.6cm thick. The exterior of the root is reddish brown to muddy, it was longitudinally wrinkled and having roots and root scars on the surface. Aromatic odour, pungent to acrid taste. Transeverse sections of different parts shows the presence of cork, cortex, and stealer region (root); vascular bundles, stomata, and trichomes (leaf); epidermis, collenchymatous cell, parenchymatous cell, and vascular bundles (stem); catkin have fruit inside consist mesocarp and endocarp (fruit). Powder microscopy revealed the presence of various components in the different parts of plants like fiber, starch grains, cork cells, calcium oxalate etc Fig. 3, 4, 5, 6. Physicochemical parameter reveals the characters of the plant. Primary phytochemical screening shows the major presence of compounds like alkaloids, flavonoids, and steroids, which may the reason of the majority of the pharmacological action of the plant Piper longum L.
CONCLUSION: The present study attempted to perform the pharmacognostic, phytochemical and physicochemical characterization of the whole plant of Piper longum L. The ethanolic extract was used for the phytochemical screening of Piper longum L.. This study helps in the identification and authentication of plants in future studies.
ACKNOWLEDGEMENT: Nil
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Fabricant DS and Farnsworth NR: The value of plants used in traditional medicine for drug discovery Environ. Health Perspect 2001; 109: 69-75.
- Jain M, Bhandari A, Bhandari Ankansha and Patel P: Isolation, characterization and in-vitro Antiurolithiatic activity of cerpegin alkaloid from ceropegiabulbosavar lushii root. Int J Drug Dev & Res 2012; 4(4): 154-60.
- Parthasarathy U, Saji K V, Jayarajan K and Parthasarathy VA: Biodiversity of Piper in South India - application of GIS and cluster analysis. Current Science 2006; 91(5): 652-58.
- Sadhana Singh, ApurvaPriyadarshi, Brijesh Singh and Poonam Sharma: Pharmacognostical and phytochemical analysis of Pippali (Piper longum). The Pharma Innovation Journal 2018; 7(6): 286-89.
- Abdullahi MN, Ilyas N, Hajara I and Kabir YM: Pharmacognostic evaluation of the leaf of Microtrichiaperotitii DC. Asteraceae J Pharmacogn Phytother 2018; 10(4): 76-84.
- Rani S, Gahlaut K and Kumar A: Pharmacognostical of Cressacreticalinn, study and establishment of quality parameters of whole plant. Der Pharmacia Lettre 2018; 10(2): 36-51.
- Srilakshmi P: Determination of physicochemical properties and morphological observation in Syzygium Cumuni leaf galls. Am J Phytomed Clin Ther 2014; 2: 174-79.
- The Ayurvedic Pharmacopoeia of India. Part-I Volume VI. First edition. Government of India Ministry of Health and Family Welfare Department of AYUSH 2008.
- Dinesh Kumar, Karunesh Kumar and Om Prakash: Pharmacognostic evaluation of leaf and root bark of HolopteleaintegrifoliaRoxb. Asian Pac J Trop Biomed 2012; 2(3): 169-75.
- Khandelwal KR: Practical pharmacognosy- techniques and experiments. Nirali Prakashan 8th Edition 2001.
- Brain KR. and Turner TD: The practical evaluation of phyto pharmaceuticals. Wright Sciente Bristol 1975; 4-35.
- World Health Organization Expert Committee Quality Control Methods for Medicinal Plant Materials. WHO Geneva 9: 22-34.
- Harborne JB: In. phytochemical methods- a guide to modern techniques of plant analysis, 3 edition. Chapman & Hall 1998.
- Kokate CK: In practical pharmacognosy. Vallabha Prakashan New Delhi 107-103.
How to cite this article:
Suman FP, Minhaj, Arthi I, Krishnan NS and Shirin RVK: Pharmacognostical, physico-chemical and preliminary phytochemical analysis of whole plantof Piper longum. Int J Pharmacognosy 2021; 8(9): 413-22. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(9).413-22.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
398-407
1438 KB
1134
English
IJP
P. Fathima Suman *, Minhaj, I. Arthi, S. Neethu Krishnan and V. K. Riyama Shirin
Department of Pharmacognosy and Phytochemistry 1, 3, 4, 5, College of Pharmaceutical Sciences, Government Medical College Kannur, Kerala, India.
fsumanp@gmail.com
05 August 2021
17 September 2021
19 September 2021
10.13040/IJPSR.0975-8232.IJP.8(9).413-22
30 September 2021