PHARMACOGNOSTIC INVESTIGATIONS AND PRELIMINARY PHYTOCHEMICAL STUDIES OF INDIGOFERA TINCTORIA LINN.
HTML Full TextPHARMACOGNOSTIC INVESTIGATIONS AND PRELIMINARY PHYTOCHEMICAL STUDIES OF INDIGOFERA TINCTORIA LINN.
Dhanapal Venkatachalam
Department of Pharmacognosy, Sanjo College of Pharmaceutical Studies, Velappara, Palakkad - 678702, Kerala, India.
ABSTRACT: Objective: To study detailed pharmacognostic profile and preliminary phytochemical investigation of the leaves of Indigofera tinctoria Linn. commonly known as ‘indigo’ belongs to the family Fabaceae. It is distributed through the hotter parts of India. Medicinal uses include the juice of the leaves as a prophylactic against hydrophobia, and as a decoction for blennorrhagia; plant extract as the treatment for epilepsy, nervous disorders, bronchitis, and as an ointment for sores, old ulcers, and hemorrhoids; and roots for hepatitis, scorpion bites, and urinary complaint. Methods: Leaf of I. tinctoria Linn. was studied by macroscopical, microscopical, quantitative microscopy, physicochemical, phytochemical analysis of leaf powder of the plant and other methods for standardization recommended by WHO. Results: Macroscopically the leaves with 9-13 leaflets and broken pieces of rachis, opposite, oblanceolate with very short mucronate tip, pale greenish black, mucronate apex, cuneate base, smooth texture, characteristic odor and taste. Leaflets 1-2.5 cm long and 0.3-1.2 cm wide. Microscopically, the leaflet appears wavy in T.S. view; prominent adaxial and abaxial thickening, the vascular bundle is simple and wide, angular thick-walled xylem elements, the thick arc of phloem. Sclerenchymatous bands occur on the adaxial side, cylindrical with thick walls epidermal cells. Small, collateral vascular strand, anamocytic stomata, thin-walled lignified sclerenchyma cells are wide on the adaxial side. Preliminary phytochemical studies of the powder showed the presence of flavonoids, glycosides, tannins, terpenoids, mucilage, and saponins. Conclusion: The results of the study can serve as a valuable source of information and provide suitable standards for identification of this plant material in future investigations and applications.
| Keywords: |
Indigofera tinctoria, Macroscopy, Microscopy Phytochemical evaluation
INTRODUCTION: Medicinal plants are playing a very active role in traditional medicines for the treatment of various ailments 1. However, a key obstacle, which has hindered the promotion in the use of alternative medicines in the developed countries, is no evidence of documentation and absence of stringent quality control measures.
There is a need for the record of all the research work carried out on traditional medicines in the form of documentation. With this drawback, it becomes extremely important to make surety about the standardization of the plant and parts of the plant to be used as a medicine.
In the process of standardization, we can use different techniques and methodology to achieve our goal in a stepwise manner like pharmacognostic and phytochemical studies. These steps and processes are helpful in identification and standardization of the plant material. Correct characterization and quality assurance of starting material is an essential step to ensure reproducible quality of herbal medicine, which will help us to justify its safety and efficacy 2, 3, 4, 5. The World Health Organization (WHO) estimates that more than 80% of the populations in developing countries rely on traditional medicine for their primary health care 6. The value of ethnomedicine and traditional pharmacology is these days achieving great appreciation in modern medicine, as the search for new potential medicinal plants is frequently based on an ethnomedicinal basis I. tinctoria Linn. is a leguminous plant which is widespread across tropical regions around the globe, as it had been cultivated and highly valued for centuries as the main source of indigo dye, leading to its common names ‘true indigo’ and ‘common indigo’ before commercial synthetic indigo production came into use in 1897 and reduced the world’s total plant-derived indigo production to 4% by 1914.
Medicinal uses include the juice of the leaves as a prophylactic against hydrophobia, and as a decoction for blennorrhagia; plant extract as a treatment for epilepsy, nervous disorders, bronchitis, and as an ointment for sores, old ulcers, and hemorrhoids; and roots for hepatitis, scorpion bites, and urinary complaints. Pharmacognostic studies on leaves are not adequate necessitating the present investigation. The current work aims to contribute to solving the problems of controversial drugs prevalent in Ayurveda besides helping in laying down Pharmacopoeial standards. Therefore, keeping above view in mind various macroscopic, histological and physiochemical and quantitative microscopical studies and preliminary phytochemical investigation of leaves of I. tinctoria Linn. was carried out in the present study.
MATERIALS AND METHODS:
Collection and Authentication: I. tinctoria Linn. leaf was collected, from in and around Palakkad, Kerala, India and authenticated by a Taxonomist and the authenticated specimen is deposited in the Department of Pharmacognosy, Sanjo College of Pharmaceutical studies, Palakkad. Authentication specimen number is SCPS/P.COG/008/2018 the fresh leaves were kept for shade drying.
The dried specimen was powdered using a mechanical grinder and passed through 60 mesh sieve to get the powder of desired coarseness. Powdered material was preserved in an airtight container.
Pharmacognostic Standardization: Organoleptic characters such as shape, size, color, odor, the taste of leaf was determined. Microscopic studies were carried out by preparing a thin hand section of the leaf with chloral hydrate solution, stained with phloroglucinol- hydrochloric acid (1:1) and mounted in glycerine 10. Histochemical studies and powder microscopy were carried out to know about the inclusions and detailed anatomical characters of the material 7.
Quantitative Microscopy and Physico-chemical Evaluations: The vein islet number, vein terminal number, stomatal number, stomatal index were determined on fresh leaves using standard procedure 8, 9, 10. The parameters were done to evaluate the proceedings of total ash; water-soluble ash; acid insoluble ash and sulfated ash were calculated as per Indian Pharmacopoeia 11. Extracts of the powdered leaf were prepared with different solvents for the study of extractive value. Fluorescence analysis was also carried out for the powder and extract as per standard procedure 12.
Extraction of Plant Material: For preliminary phytochemical analysis, the extract was prepared by weighing 1 kg of the dried powdered leaf was subjected to hot successive continuous extraction with different solvents as per the polarity, petroleum ether, benzene, chloroform, ethanol and aqueous. The extracts were filtered in each step using Whatman filters paper. The filtrate was concentrated using a rotary evaporator at low temperature (40-45 ºC) and pressure. The presence or absence of the primary and secondary phyto-constituents was detected by usual prescribed methods 13, 14.
Powder Analysis: Preliminary analysis of the powder of the leaf powder of I. tinctoria Linn. with different chemical reagents was carried out microscopically 15, 16.
RESULTS:
Macroscopical Characters of the Leaf: 17 The plant occurs in the form of leaflets and broken pieces of rachis, leaflets 1-2.5 cm long and 0.3-1.2 cm wide, opposite, membranous, rounded and apiculate, oblanceolate with very short mucronate tip green but drying greenish black. Mucronate apex, cuneate base, smooth texture, characteristic odor, and bitter taste. Leaf was 1.65 cm length and 1.34 cm width.
Anatomy of Leaf:
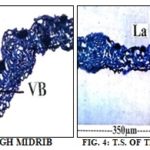
Leaf: The leaflets appear waxy in the transactional view. The midrib and lateral view are prominent with adaxial and abaxial thickening Fig. 3 and 4. The midrib is 150 µm thick and 180 µm wide. The adaxial epidermis of the midrib consists of vertically elongated rectangular or horizontally cylindrical thin-walled cells.
LV-Lateral vein, La – Lamina, Ads – Adaxial side, VB- Vascular bundle, MR – Midrib
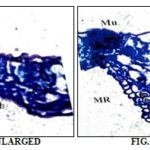
La –Lamina, MU –Mucilage, MT- Mesophyll tissue, LM – Leaf margin, AdE – Adaxial epidermis, AbE- Abaxial epidermis
Sc- Sclerenchyma, X – Xylem, Ph – Phloem
The abaxial epidermis is there with papillate small thick-walled cells. The vascular bundle is single and wide. It consists of a few compact radial rows of wide angular thick-walled xylem elements and a thick arc of phloem elements on the lower end of the xylem. Sclerenchymatous bands occur on the adaxial side there is a wide and thick conical mass of sclerenchyma cells which extend up to the adaxial epidermis. The adaxial sclerenchyma cells are wide, thick-walled, and lignified.
The palisade zone extends up to the lateral sides of the sclerenchyma mass Fig. 8. The lateral vein is somewhat similar to the midrib Fig. 7. The vein is conically projecting both on the adaxial and abaxial sides. The epidermal cells on both adaxial and abaxial sides are vertically cylindrical or horizontal with thick walls. There is a small, collateral vascular strand comprising a group of wide thick-walled xylem elements and a small cluster of phloem elements. Thick and wide masses of sclerenchyma cells are situated on the adaxial and abaxial ends of the vascular strand Fig. 7. The lateral vein is 120 µm thick and 70 µm wide.
Lamina: The lamina consists of the thick and wavy epidermal layer of thin-walled cells. Some of the epidermal cells are dilated and filled with large mass of mucilage Fig. 6. The mesophyll consists of an adaxial layer of single vertically elongated palisade cells and 2 to 3 layer of loped and loosely arranged spongy parenchyma cells.
The thickness of the lamina is 70 µm. The marginal part of the lamina is semicircular without any modification of the lamina. It is comparable to the remaining part of the lamina Fig. 5.
Powder Microscopy: Powder characteristics revealed the presence of epidermal cells with starch grains, covering trichomes, epidermal cells with trichomes, Anamocytic stomata, vessel elements.
Quantitative Microscopy: The quantitative microscopy such as vein-islet number, vein- terminal number, stomatal number, and stomatal index were determined, and the results were tabulated Table 1.
Physicochemical Features: The powdered drug was evaluated for its physicochemical parameters like total ash values, acid-insoluble ash, water-soluble ash, and loss on drying, and the results were tabulated Table 2.
TABLE 1: QUANTITATIVE EVALUATION OF THE CRUDE DRUG OF THE LEAF OF I. TINCTORIA LINN.
| S. no. | Plant constants | Values |
| 1 | Vein islet no. | 8/sq mm |
| 2 | Vein termination no. | 6/sq mm |
| 3 | Stomatal number (upper) | 16.16 |
| 4 | Stomatal number (lower) | 28.66 |
| 5 | Stomatal index (upper) | 6.311 |
| 6 | Stomatal index (lower) | 8.703 |
TABLE 2: PHYSICOCHEMICAL EVALUATION OF THE CRUDE DRUG OF LEAF OF I. TINCTORIA LINN.
| S. no. | Physical Evaluation | %w/w |
| 1 | Total ash | 5.52 |
| 2 | Acid-insoluble ash | 4.61 |
| 3 | Water soluble ash | 3.58 |
| 4 | Loss on drying | 0.3 |
Fluorescence Analysis of the Extracts: The extracts were prepared as per their polarity in hot successive extraction technique, and they were treated with reagents, and the color changes were observed under Ultra Violet light, and the results were tabulated Table 3.
TABLE 3: FLUORESCENCE ANALYSIS OF LEAF OF I. TINCTORIA LINN.
| S. no. | Sample | Colour in Day Light | Colour in UV Light |
| 1 | Petroleum ether extract | Pale
green |
Dark
green |
| 2 | Benzene extract | Green | Light green |
| 3 | Chloroform extract | Brownish green | Green |
| 4 | Ethanol extract | Green | Dark Green |
| 5 | Aqueous
extract |
Brownish
green |
Yellowish green |
Extractive Values: The extracts were prepared according to the polarity, and they were concentrated, and their values were calculated concerning air-dried drug, and the results were tabulated Table 4.
TABLE 4: EXTRACTIVE VALUES OF THE LEAF OF WITH I. TINCTORIA LINN. DIFFERENT SOLVENTS
| S. no. | Sample | Extractability (%) |
| 1 | Petroleum ether extract | 3 |
| 2 | Benzene extract | 3.2 |
| 3 | Chloroform extract | 2.4 |
| 4 | Ethanol extract | 2.2 |
| 5 | Aqueous extract | 20 |
Preliminary Phytochemical Analysis: The leaf powder and various extracts such as petroleum ether extract, benzene extract, chloroform extract, ethanol extract, and aqueous extract were subjected to preliminary phytochemical screening for their presence or absence of the constituents, and the results were tabulated Table 5.
TABLE 5: PRELIMINARY PHYTOCHEMICAL TESTS FOR DRUG POWDER AND VARIOUS EXTRACTS OF LEAF OF I. TINCTORIA LINN.
| S.
no. |
Test | Drug powder | Petroleum ether extract | Benzene extract | Chloroform extract | Ethanol extract | Aqueous extract |
| 1. | Sterols | - | + | - | - | - | - |
| 2. | Terpenoids | + | - | - | - | + | + |
| 3. | Carbohydrates | + | - | - | - | + | + |
| 4. | Flavanoids | + | - | - | - | + | + |
| 5. | Proteins | - | - | - | - | - | - |
| 6. | Alkaloids | - | - | - | - | - | - |
| 7. | Glycosides | + | - | - | - | + | + |
| 8. | Saponins | + | - | - | - | - | - |
| 9. | Tannins | + | - | - | - | + | + |
| 10. | Mucilages | - | - | - | - | + | + |
| 11. | Volatile oil | + | - | - | - | + | + |
+ indicates positive reaction, - indicates negative reaction.
DISCUSSION: Our study has focused on examining pharmacognostic and preliminary phytochemical studies of leaves of I. tinctoria Linn. Normalization of the macroscopic and microscopic characteristics of the I. tinctoria drug remains essential in other to identify and avoid falsification. Microscopically, the leaf showed the presence of palisade, mesophyll, covering trichome, adaxial epidermis, adaxial epidermis, anamocytic stomata, mucilage, sclerenchyma, vascular strand are the diagnostic features noted from the anatomical study.
Organoleptic characteristics are important in drugs because they play a role in the detection of adulterated or substituted drugs 18. Thus leaves pale greenish black in color, characteristic odor, bitter taste. The powdery appearance of the crushed leaves, with a coarse texture. The micrograph performed on the powder has highlighted several characteristic elements namely: epidermal cells with starch granules, covering trichomes, epidermal cells with trichomes, anamocytic stomata, vessel elements are diagnostic substances for drugs of plant origin. These diagnostic elements are consistent with botanical standards and WHO guidelines 19, 20.
The study of physicochemical parameters such as moisture content and ash values are useful as it determines the physiological and non-physiological state of ash, this will help to determine the possibility of microbial growth and lastly contaminant or impurities. The moisture content of the drug studied had a rate of 0.3 ± 0.1, which is below 10%. This result complies with the standards established by the International Pharmacopoeia, because this water content rate, prevent oxidation reactions, fermentation, and give less chance to microbial growth and contamination in drugs 21.
Therefore, for the proper conservation of drugs made from the leaves of I. tinctoria, it would be desirable to use those whose water content is less than or equal to 10%. The determination of total ash gave us a rate of 5.52 ± 0.03. This value indicates the level of minerals in drugs. Insoluble ash in hydrochloric acid gave a rate of 4.61 ± 0.02. Indeed, the ash insoluble in hydrochloric acid tells us about the contamination of the drug by siliceous elements 22. This result is in agreement with Srikanth et al., 23 who found the rate of 0.97% and 0.5% respectively. The maximum extractive value was found in aqueous (20%), followed by ethanol (7.2%), benzene (3.20%), chloroform (2.4%). All the extracts of the drug were subjected to different tests for detecting the presence of various phytoconstituents present in the drug, which revealed the presence of flavonoids, glycosides, saponin, tannins, terpenoids, and mucilage.
Preliminary phytochemical analysis indicated a high percentage of quercetin and flavonoids, and this may be one of the reasons behind the pharmacological activity of the plant. These parameters, which are being reported for the first time in this plant, are significant towards establishing the pharmacognostic standards for future identification and authentication of genuine plant material. Though I. tinctoria is a highly reputed drug used in Ayurveda. Barring the anatomical details and preliminary phytochemical screening, rest of the pharmacognostical parameters gives us a clue that it can be cashed economically as well to improve the standard of health in the developing countries.
CONCLUSION: WHO has emphasized the need to ensure quality control of the raw materials used for Ayurvedic medicines by using modern techniques and by applying suitable parameters and standards. In the present study various standardization parameters such as macroscopy, microscopy (histochemical and powder), physicochemical standards, preliminary phyto-chemical investigation, which are being reported for the first time in this plant and could be helpful in authentication and preparation of a suitable monograph for the proper identification of I. tinctoria Linn. for the future.
ACKNOWLEDGEMENT: The authors are thankful to the Director and Principal of Sanjo College of Pharmaceutical studies, Vellapara, Palakkad for providing facilities to carry out the present research.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Mohammad Saleem TS, Christina AJ, Chidambaranathan N, Ravi V and Gauthaman K: Hepatoprotective activity of Annona squamosal on an experimental animal model. Int J Appl Res Nat Prod 2008; 1: 7.
- Ahmad I, Aqil F and Owais M: Modern Phytomedicine: turning medicinal plants into drugs. New York: John Wiley & Sons; 2006.
- Willow JH: Traditional herbal medicine research methods: identification, analysis, bioassay and pharmaceutical and clinical studies. New York: John Wiley & Sons; 2011.
- Benzie IF and Wachtel-Galor S: Herbal medicine: bimolecular and clinical aspects, oxidative stress and disease. CRC Press, Florida, Edition 2nd, 2011: 499.
- Odugvemi T: A textbook of medicinal plants from Nigeria. Nigeria: Tolu Odugbemi; 2008.
- Nathiya S, Santhi N and Kalaiselvi S: A comparative study on the ontogenic expression of antioxidants and secondary metabolites in Withania somnifera. Int Res J Pharm 2012; 3(1): 2010-2015
- Johansen DA: Plant microtechnique. McGraw- Hill, New York, USA; 1940.
- Indian Pharmacopoeia: Controller of Publication, Delhi, India, Vol. 2, 1995: A-54.
- Horbone JB: Phytochemical methods-A guide to modern techniques of plant analysis, Chapman and Hall, London, 1998: 42, 129, 203.
- Kokate CK: Practical Pharmacognosy. Vallabh Prakashan, Delhi, Edition 4th, 1994: 115.
- Wallis TE: Practical Pharmacognosy. J and A Churchill Ltd., London, Edition 6th, 1955: 139-140, 173-174, 180-184.
- Wallis TE: Analytical Pharmacognosy. J and A Churchill Ltd., London, Edition 3rd.
- Trease GE and Evans WC: Pharmacognosy. Saunders publisher London, Edition 15th, 2004: 137-44.
- Khandelwal KR: Practical Pharmacognosy techniques and experiments. Nirali Prakashan, Pune, 2002.
- Reddy YSR, Venkatesh S and Ravichandra T: Pharma-cognostical studies on tinctoria bark. Pharmaceutical Biology 1999; 37: 291-295.
- Pratt PR and Chasse ER: Fluorescence powder vegetable drugs in particular to develop systems of identification. Journal of American Pharmaceutical Association 2014; 38: 324-331.
- Easu K: Anatomy of seed plants. John Wily and Sons, Newyork 1979: 550
- Fouraste I: Le contrôle des Plantes médicinales. Actualités Pharmaceutiques 1990; 278: 55-58.
- Kumar S, Kumar V and Prakash O: Microscopic evaluation and physicochemical analysis of Dillenia indica Asian Pac J Trop Biomed 2011; 1: 337-340.
- Nasreen S and Radha R: Assessment of quality of somnifera Dunal (Solanaceae): Pharmacognostical and physicochemical profile. Int J Pharm Sci 2011; 3(2): 152-155.
- Organisation de l’unité africaine/commission scientifique technique et de la recherche (OUA/CSTR). Pharmacopée africaine, méthodes générales d’analyses. Publisher, Lagos (Nigéria), Edn 1, 1998: 254.
- Sambo MH: Etude du traitement traditionnel du diabète par une recette et les écorces de tronc de Manilkara multinervis Dub (Sapotaceae). Th Pharm., Univ.de Bamako, Mali, 2005; 125:
- Srikanth K, Vikram G, Archana P, Rajinikanth M, and Ram SN: Pharmacognostic and phytochemical investigations in potatorum L. F. J of Pharm and Phyt 2013; 2(4): 46-51.
How to cite this article:
Venkatachalam D: Pharmacognostic investigations and preliminary phytochemical studies of Indigofera tinctoria Linn. Int J Pharmacognosy 2018; 5(11): 732-37. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(11).732-37.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
732-737
747
1323
English
IJP
D. Venkatachalam
Department of Pharmacognosy, Sanjo College of Pharmaceutical Studies, Velappara, Palakkad, Kerala, India.
vddpaul@gmail.com
26 September 2018
21 October 2018
23 October 2018
10.13040/IJPSR.0975-8232.IJP.5(11).732-37
01 November 2018