PHARMACOGNOSTIC ANALYSIS AND PHYTOCHEMICAL ANALYSIS OF THE MEDICINAL PLANT CANTHIUM PARVIFLORUM LAM.
HTML Full TextPHARMACOGNOSTIC ANALYSIS AND PHYTOCHEMICAL ANALYSIS OF THE MEDICINAL PLANT CANTHIUM PARVIFLORUM LAM.
Shweta J. Sabannavar * and A. S. Chitra
Department of Botany, Mount Carmel College (Autonomous), Bangalore - 560052, Karnataka, India.
ABSTRACT: The crude extract from the leaf of Canthium parviflorum Lam. in different solvents were subjected to physicochemical, fluorescence analysis, phytochemical and antimicrobial study. The microscopic analysis revealed that in C. parviflorum there is the presence of starch grains. The ash value was 5.81% in C. parviflorum. The fluorescent analysis at 366 nm of C. parviflorum powder extracted with alcohol showed orange-colored fluorescence with other it was no fluorescence. The extraction value with different solvents exhibited that C. parviflorum had maximum extractive value with chloroform followed by benzene, ethanol and petroleum ether. The phytochemical screening of the various extract of C. parviflorum revealed that the benzene extract showed the presence of alkaloids, tannins, flavonoids, saponins, and steroids. The chloroform extracts of the leaf showed the presence of alkaloids, tannins, flavonoids, saponins, and steroids. Anti-bacterial screening in C. parviflorum using stem revealed that the ethanol solvent showed maximum inhibition against gram-positive bacteria (Staphylococcus aureus) and chloroform solvent showed maximum inhibition in the case of gram-negative bacteria (Serratia marcescence).
| Keywords: |
Phytochemical, Fluorescence analysis, Anti-bacterial screening
INTRODUCTION: Medicinal plants are the richest bio-resource of drugs of traditional systems of medicine, nutraceuticals, food supplements, folk medicines, pharmaceutical intermediates and chemical entities for synthetic drugs 1. Drugs from the plants are easily available, less expensive, safe, and efficient and rarely have side effects. The plants which have been selected for medicinal use over thousands of years constitute the most obvious choice of examining the current search for therapeutically effective new drugs such as anticancer drugs, antimicrobial drugs 2, antihepatotoxic compounds.
According to the World Health Organization (WHO), medicinal plants would be the best source to obtain a variety of drugs. However, such plants should be investigated to understand their properties, safety, and efficiency 3 better.
The detection of active principles in medicinal plants plays a strategic role in the phytochemical investigation of crude plant extracts and is very important in regards to their potential pharmacological effect 4. The knowledge of the chemical constituents of plants is desirable because such information will be value for the synthesis of complex chemical substances. Phytochemicals are the natural bioactive compounds found in plants. These phytochemicals work with nutrients and fibers to form an integrated part of the defense system against various diseases and stress conditions 5. The most important of these bioactive constituents of plants are alkaloids, tannins, flavonoids, steroid, terpenoid, carbohydrate, and phenolic compounds. The majority of these bioactive compounds are alkaloids, followed by sesquiterpenes, diterpenes, triterpene saponins, triterpene aglycones, flavonoids, sterols, coumarins, quinines and monoterpenes 6. Extraction (as the term is pharmaceutically used) is the separation of medicinally active portions of plant tissues using selective solvents through standard procedures. The ash and extractive values of crude drugs help in the identification and determination of its purity and quality 7. The products so obtained from plants are relatively complex mixtures of metabolites, in liquid or semisolid state or (after removing the solvent) in dry powder form, and are intended for oral or external use. Extraction methods used pharmaceutically involves the separation of medicinally active portions of plant tissues from the inactive/inert components by using selective solvents. During extraction, solvents diffuse into the solid plant material and solubilize compounds with similar polarity.
A large number of phytochemicals belonging to several chemical classes have been shown to have inhibitory effects on all types of microorganisms in-vitro 8. This study investigates the fundamental scientific bases for the use of medicinal plants by defining the crude phytochemical constituents present in these plants. The present work leads to phytochemical analysis and antimicrobial analysis of Canthium parviflorum Lam.
FIG. 1: CANTHIUM PARVIFLORUM LAM.
MATERIALS AND METHODS: The fresh plant materials of Canthium parviflorum Lam. Fig. 1 were collected from markets from Bangalore, Karnataka, India. The leaves of C. parviflorum Lam were cut into small fragments and shade dried till uniform and smooth. The dried plant materials were powdered by using a blender. The final uniform powder was used for the study. Physio-chemical standards such as ash, extractive values were determined as per the standard Indian Pharmacopoeia methods 9. Antimicrobial activity was also carried out using gram-positive and negative bacteria.
Powder Microscopic Analysis: For examining characters of the powder, sufficient amount of leaf powder was mixed in Chloral-hydrate solution on a slide and covered with a coverslip, warmed over a low flame for a short time.
Starch: To examine the presence of starch, two specimens were taken, one in an Iodine solution and the other in the water. With Iodine solution starch turned blue. The shape and structure of starch grains were observed in water, and their size was measured.
Fixed Oil: To examine the presence of fixed oil, a specimen was prepared in a solution of Sudan III, droplets of fixed oil were colored orange-pink.
Determination of Ash: The ash remaining on ignition of medicinal plant materials was determined by three different methods which measure the total ash, acid-insoluble ash, and water-soluble ash. The total ash method was designed to measure the total amount of material remaining after ignition. This includes both "physiological ash," which was derived from the plant tissue itself, and "non- physiological ash, which was the residue of the extraneous matter (e.g., sand)
Total Ash: About 2 gm of accurately weighed, ground plant sample was taken in a previously weighed silica dish, previously ignited and weighed. The ground dry sample was scattered in a fine even layer on the bottom of the dish and gradually increased the heat 500-600 ºC until it was white, indicating the absence of carbon. In a desiccator, it was cooled and weighed. If carbon-free ash cannot be obtained in this manner, the crucible was cooled and moistened with nitrate. Dried on a water-bath, then on a hot plate and ignite to constant weight.
The residue was allowed to cool in a suitable desiccator for 30 min, later weighed without delay. The percentage of ash concerning the air-dried plant sample was calculated.
Fluorescence Analysis: The plant powder was treated with alcohol, water, 0.1 N Sodium hydroxide and dilute hydrochloric acid. They were subjected to fluorescence analysis in daylight and UV- light (254 nm and 365 nm).
Extraction Value: The powdered plant sample Canthium parviflorum Lam. of 10 g was packed and sealed in a filter paper placed and inside a thimble into the main chamber of the Soxhlet extractor. The Soxhlet is then placed on a flask containing the extraction solvent. The solvent used for extraction is chloroform, petroleum ether, ethanol, and benzene. About 250 ml of the solvent was loaded. Previously the dried flask was weighed with pumice pieces to get empty weight. The extractor was then attached to the condenser. The water supply was then switched on for water circulation, six cycles of extraction were carried out for 8 h at 60 ºC with required solvent. The solvent was removed and was dried in an oven at 100 ºC to a constant weight.
Calculation for the percentage of extract:
Preliminary Phytochemical Screening: The screening of the alcoholic and aqueous extracts of the plant material was carried out for qualitative determination of the groups of organic compounds present in them 10.
Alkaloids:
a) Dragendorff’s Test: A 2 ml of alcoholic or aqueous extract of the sample was dissolved in 5 ml of distilled water, 2 M hydrochloric acid was added until an acid reaction occurred, then 1 ml of Dragendorff’s reagent was added, an orange or orange-red precipitate was formed immediately.
b) Hager’s Test: A few drops of Hager’s reagent added to 1 ml of alcoholic extract of the sample taken in a test tube, Formation of yellow precipitate confirmed the presence of alkaloids.
c) Wagner’s Test: 1 ml of alcoholic extract of the drug was acidified with 1.5% v/v of hydrochloric acid, and a few drops of Wagner’s reagent was added. A yellow or brown precipitate was formed.
d) Mayer’s Test: A few drops of Mayer’s reagent was added to 1 ml of acidic aqueous extract of the drug. The white or pale yellow precipitate was formed.
Flavonoids: In a test tube containing 0.5 ml of alcoholic extract of the drug, 5-10 drops of dilute hydrochloric acid was added followed by a small piece of magnesium. In the presence of flavonoids a pink, reddish pink or brown color is produced.
Millon’s Test: A small quantity of aqueous extract of the plant sample was dissolved in 1ml of distilled water, and 5-6 drops of Millon’s reagent was added. A white precipitate was formed which turns red on heating, indicating the presence of proteins.
Resins: The ethanol extract was dissolved in acetone, and the solution was poured into distilled water. The presence of resins was indicated by turbidity.
4. Saponins: In a test tube containing about 5ml of an aqueous extract of the sample and a drop of Sodium bicarbonate solution was added, the mixture was vigorously shaken and left for 3 min. A honeycomb-like forth was formed.
Steroids:
(a) Liebermann-Burchard's Test: To 1 ml of petroleum ether extract of the sample in chloroform 2 ml of acetic anhydride solution was added followed by 1 ml of concentrated sulphuric acid. A greenish color was developed which turned to blue.
(b) Salkowiski Reaction: To 2 ml of the chloroform extract of the drug 1 ml of concentrated sulphuric acid was added carefully, from the side of the tube. A red color was formed in the chloroform layer.
Tannins: To 1- 2ml of an extract of the drug a few drops of 5% FeCl3 solution was added. A green color indicates the presence of gallotannins while brown color tannins.
Anti-bacterial Analysis: The gram-positive organisms like Staphylococcus aureus and gram-negative organism like Serratia marcescens was isolated from local isolates and maintained at the Microbiology laboratory of Mount Carmel College (Autonomous), Bangalore to use as the test organisms for the study. The chloroform, petroleum ether, ethanol and acetone extracts of C. parviflorum were tested against the above-said organisms to test the antibiotic properties of the chosen extracts of the plant.
The nutrient agar media was prepared according to the composition and sterilized for 15 minutes at 121 ºC temperature & 15 lbs pressure, in an autoclave. The nutrient agar is a microbial growth medium commonly used for the routine cultivation of non-fastidious bacteria. The test bacterial cultures used in work were subcultured regularly on nutrient agar slants. They were maintained by refrigeration, throughout the work period. A loopful of test organisms were dissolved in a test tube containing 5 ml of distilled water to obtain seed inoculums.
Agar Diffusion Method: Using a sterile swab the seed inoculum of the respective test organism was inoculated onto labeled plates. The plates were kept aside for 15-20 min, and then with a sterile cork borer, one central well was bored. The same method was followed for all the other test organisms. The extract (100 µl) was added into the well of a plate already inoculated with a test organism. Then the plates were incubated at 37 ºC for 24 h. This was repeated thrice, and the average of the three zones of inhibition for the test organism was measured.
RESULTS: Plant, when subjected to standardization, will avoid any ambiguity in the identity of the plant. The present study was focused on microscopic analysis, preliminary phytochemical screening and in-vitro antibacterial activity of Canthium parviflorum Lam. The microscopic analysis revealed that C. parviflorum there is the presence of starch grains and trichome Fig. 2. The ash values were found to be 5.81% in C. parviflorum. The powder from the leaf of C. parviflorum fluoresced under ultraviolet light at 366 nm, powder extracted with alcohol showed orange-colored fluorescence. Powder that extracted with water and 0.1 N Sodium hydroxide and diluted hydrochloric acid had no fluorescence Table 1.
It was found that C. parviflorum has maximum extractive value with chloroform followed by benzene, ethanol. The petroleum ether had minimum extractive value Table 2.
Preliminary phytochemical screening of the various extracts of the leaves in C. parviflorum with different solvents for the presence of alkaloids, flavonoids, steroids, tannin, saponins, and resins was tested and presented in Table 3. The petroleum ether extract of the leaf showed the presence of alkaloids, flavonoids and steroids, tannins, and saponins. The benzene extract of the leaf showed the presence of alkaloids, tannins, flavonoids, saponins, and steroids. The chloroform extracts of the leaf showed the presence of alkaloids, tannins, flavonoids, saponins, and steroids. The ethanol extracts showed the presence of alkaloids, tannins, and steroids.
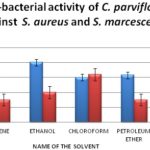
The leaf extract of C. parviflorum was also tested for their antibacterial activity against S. aureus (gram-positive) and S. marcescence (Gram-negative). The maximum zone of inhibition was observed in ethanol extract in case of gram-positive bacteria. The zone of inhibition was high with chloroform extract in case of gram-negative bacteria observed in Fig. 3. The minimum zone of inhibition was observed in chloroform solvent against gram-positive. Petroleum ether and benzene solvents showed the lowest zone of inhibition with negative bacteria.
FIG. 2: PRESENCE OF STARCH GRANULES AND TRICHOME
TABLE 1: FLUORESCENCE ANALYSIS
| Powdered drug | UV 366nm | |
| Powder extracted with alcohol | Orange color | |
| Powder extracted with water | No fluorescence | |
| Powder extracted with 0.1N sodium hydroxide | No fluorescence | |
| Powder extracted with dilute hydrochloric acid | No fluorescence | |
TABLE 2: EXTRACTION VALUE
| Name of solvent | Extraction value |
| Chloroform | 484.2 |
| Petroleum ether | 147.21 |
| Ethanol | 177.42 |
| Benzene | 210 |
TABLE 3: PHYTOCHEMICAL SCREENING OF CANTHIUM PARVIFLORUM LAM.
| Test | Chloroform | Petroleum ether | Ethanol | Benzene |
| Alkaloids | + | + | + | + |
| Flavonoids | + | + | + | - |
| Resins | - | - | - | - |
| Saponins | + | + | + | - |
| Steroids | + | + | + | + |
| Tannins | + | + | + | + |
FIG. 3: ANTIBACTERIAL ACTIVITY OF CANTHIUM PARVIFLORUM AGAINST STAPHYLOCOCCUS CITRUS (GRAM POSITIVE) AND SERRATIA MARCESCENCE (GRAM NEGATIVE)
DISCUSSION: The powder analysis carried out in C. parviflorum revealed the presence of trichome, parenchyma cells containing starch granules, vascular bundle). The total ash is particularly important in the evaluation of purity of drugs, i.e. the presence or absence of foreign organic matter such as metallic salts and or silica. The ash value can also detect the nature of the material added to the drug for adulteration.
Fluorescence analysis is the phenomenon exhibited by various chemical constituents present in the plant material under UV light. This can be used to characterize crude drugs. Thus, the crude drug is often assessed qualitatively and forms an important parameter of pharmacognostical evaluation 11. Many phytocompounds fluorescences when suitably illuminated. The fluorescence color is specific for each compound. A non-fluorescent compound may fluoresce if mixed with impurities that are fluorescent. The C. parviflorum powder extracted with alcohol showed orange color indicating the purity of the sample which could be made upon different color observed.
Extractive values are primarily useful for the determination of exhausted or adulterated drug 12. The variance in the extractive value may be possible due to the presence of a specific compound, according to the solubility, soil condition, atmospheric condition and water content of the sample. It was found that C. parviflorum had maximum extraction with chloroform and least extraction in petroleum ether. Plant synthesizes a wide variety of chemical compounds, which can be sorted by their chemical class, biosynthetic origin, and functional groups into primary & secondary metabolites. Presence or absence of certain compounds in an extract is determined by the color reactions of the compounds with specific chemicals which act as dyes. This procedure is a simple preliminary pre-requisite before going for the detailed phytochemical investigation.
Phytochemical screening of the various extracts of C. parviflorum with different solvents exhibited the presence of alkaloids, flavonoids, steroids, tannin, saponins, and resins. Alkaloids have been associated with medicinal uses for centuries, and one of their common biological properties is their cytotoxicity 13. Saponins have the property of precipitating and coagulating red blood cells. Some of the characteristics of saponins include the formation of foams in aqueous solutions, hemolytic activity, cholesterol binding properties and bitterness 14.
The samples tested were negative for resins in all the solvents. Steroids present in the plant extract are of special interest in cancer studies. Steroids have considerable values in aiding the control of cancer in man. Steroids in modern clinical studies have supported their role as anti-inflammatory and analgesic agents 15.
Significant antioxidant and diuretic activity was exhibited by extracts of Canthium leaves, the chloroform fraction mainly contains Triterpenoids, and so this activity was because of Triterpenoids. As triterpenoids are reported as a good antiulcer and antioxidant compound. The presence of tannins could be extremely helpful as a diuretic agent. Medically, tannins are used as antidotes to poisoning by alkaloids depending on their capacity to form insoluble tannates.
Tannins, phenolics, saponins, alkaloids, and flavonoids have been linked or suggested to be involved with antibacterial and anti-viral activity while tannins and flavonoids are thought to be responsible for antidiarrheal activity 16.
The remedial values of tannins include application on burns to heal the injury and on cuts to stop bleeding. Tannins ability to form a strong ‘leather’ resistance on the exposed tissues helps in protecting the wounds from being affected further. Tannin has several industrial uses as preservatives. So, the identification of tannins in medicinal plants screened could be very advantageous in healing various diseases, and these plants could be potentially used for a varied range of applications. So the presence of this type of phytochemical compounds in the screened medicinal plants has a wide range of applications and could be certainly used for a variety of applications. The leaf extract of C. parviflorum revealed a maximum zone of inhibition in ethanol extract with gram-positive bacteria. The zone of inhibition was high with chloroform extract in case of gram-negative bacteria. Petroleum ether and benzene solvents showed the lowest zone of inhibition with negative bacteria.
The activity is higher in these two solvents compared to other solvents may be due to the additive effect of antibacterial property of the solvent themselves along with the plant extract. In general, gram-positive bacteria are more resistant than gram-negative bacteria. The resistance is due to the difference in the cell wall composition. The gram-positive bacterium has its cell wall made up of several peptidoglycan layers joined together forming a thick and rigid structure. By contrast, gram-negative bacteria have only a thin peptidoglycan layer.
The cell wall of a gram-positive bacterium has teichoic acids, which mainly consist of alcohols and phosphate. In the gram-negative bacteria, the outer membrane acts as a great barrier to much environmental substance. Presence of thick murine layer in the cell wall prevents the entry of inhibitors. The outer membrane is permeable to nutrients due to the presence of porins, proteins that form channels toward the cytoplasm 18.
CONCLUSION: The present study reveals to be an important medicinal plant has it exhibited the presence of alkaloids, flavonoids, steroids, tannin, saponins, and resins.
ACKNOWLEDGEMENT: I would like to thank Principal of Mount Carmel College for providing the laboratory facilities to carry out the research.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Ncube NS, Afolayan AJ and Okoh AI: Assessment techniques of antimicrobial properties of natural compounds of plant origin: current methods and future trends. African Journal of Biotechnology 2008; 7(12): 1797-1806.
- Phillipson JD and Wright CW: Plants With Antiprotozoal Activity: Tease and Evans, Pharmacognosy, WB Saunders Company 1996; 14: 612.
- Arunkumar S and Muthuselvam: Analysis of phytochemical constituents and antimicrobial activities of Aloe vera against clinical pathogens. World J Agril Sc 2009; 5(5): 572-576.
- Pascual ME, Carreter ME, Slowing KV and Villar A: Simplified screening by TLC of plant drugs. Pharmaceutical Biology 2002; 40(2): 139-143.
- Koche D, Shirsat R and Imran S: Phytochemical screening of eight traditionally used ethnomedicinal plants from Akola district (MS) India. International Journal of Pharma and Bio Sciences 2010; 1(4):
- Pascaline J, Charles M, Lukhoba C and George O: Phytochemical constituents of some medicinal plants used by the Nandis of South Nandi district Kenya. Journal of Animal & Plant Sciences 2011; 9(3): 1201-1210.
- Kokate CK, Purohit AP and Gokhale SB: Pharmacognosy 2010; Nirali prakashan, Mumbai India 64.
- Cowan MM: Plant products as antimicrobial agents. Clin Microbiol Rev 1999; 564-582.
- Anon: "The Wealth of India - Raw Materials" Vol n C., CSR. New Delhi 1950; 66.
- Sofowara AE: Medicinal plants and traditional medicine in Africa. Vol. 2. Spectrum Books Ltd, Ibadan 1993: 288.
- Dominic VJ and Madhavan M: Pharmacognostic and preliminary phytochemical screening of the rhizome of Anaphyllum wightii schott, an endemic and threatened genus of the Western ghats. Journal of Herbal Medicine and Toxicology 2012; 6(2): 55-60.
- Shrivastwa S and Leelavathi S: Int J Pharm Sci Review and Research 2010; 3: 114-118.
- Nobori T, Miurak K, Wu DJ, Takabayashik LA and Carson DA: Deletion of cyclin-dependent kinase-4 inhibitor gene in multiple human cancers. Nature 1994; 46: 753-56.
- Okwu DE: Phytochemicals and vitamin content of indigenous species of south-eastern Nigeria. J Sustain Agric Environ 2004; 6(1): 30-37.
- Singh AP: Short Review: Distribution of Steroid like Compounds in Plant Flora. Pharmacognosy Magazine 2006; 2(6): 87-89.
- Enzo AP: Traditional plants and herbal remedies used in the treatment of diarrheal disease: Mode of action, quality, efficacy and safety considerations. In: Ahmad I, Aqil F, Owais M, editors. Modern Phytomedicine Turning Medicinal Plants into Drugs, WILEY- VCH Verlag GmbH & Co. KGaA, Weinheim 2007; 248-60.
- Lena G: Phenolic compounds, antioxidant activity and in vitro inhibitory potential against key enzymes relevant for hyperglycemia and hypertension of commonly used medicinal plants, herbs and spices in Latin America. Bio Resour Technol 2010; 10: 4676-89.
- Wheelis M: Principles of Modern Microbiology, Published by Jones & Bartlett Publishers
How to cite this article:
Sabannavar SJ and Chitra AS: Pharmacognostic analysis and phytochemical analysis of the medicinal plant Canthium parviflorum Lam. Int J Pharmacognosy 2017; 4(3): 92-98. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(3).92-98.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
92-98
644
1818
English
IJP
S. J. Sabannavar * and A. S. Chitra
Department of Botany, Mount Carmel College (Autonomous), Bangalore, Karnataka, India.
shwetsab@gmail.com
31 December 2016
22 January 2017
24 February 2017
10.13040/IJPSR.0975-8232.IJP.4(3).92-98
01 March 2017