NOVEL DRUG DELIVERY SYSTEMS OF HERBAL MEDICINE
HTML Full TextNOVEL DRUG DELIVERY SYSTEMS OF HERBAL MEDICINE
Pankaj A. Tagad *, Atish. J. Shelake, Krushna A. Palve, Rushikesh A. Kale and Santosh S. Bansode
Dr. Kolpe Institute of Pharmacy, Kolpewadi, Kopargaon, Maharashtra, India.
ABSTRACT: Over the past several years, great advances have been made in developing novel drug delivery systems (NDDS) for plant actives and extracts. Various novel herbal formulations such as polymeric nanoparticles, nanocapsules, liposomes, phytosomes, animations, microsphere, transfersomes, and ethosomes have been reported using proactive and plant selections. The novel formulations are reported to have remarkable 0 advantages over conventional formulations of plant actives and extracts, which include a cement of solubility, bioavailability, protection from toxicity, enhancement of pharmacological activity, enhancement of stability, improved tissue macrophages distribution, sustained delivery, and protection from physical and chemical degradation. Phytosome is a patented technology developed by a leading maker of drugs and nutraceuticals to incorporate standardized plant extracts or water-soluble phytoconstituents into phospholipids to produce lipid‑compatible molecular complexes. The herbal drugs can be used in a more upright course with enhanced efficacy by incorporating them into modern dosage forms. The present review highlights the current status of developing novel herbal formulations. It summarizes their preparation method, type of active ingredients, size, entrapment efficiency, route of administration, biological activity, and applications of novel formulations.
Keywords: Liposomes, Nanoparticles, Phytosome, Ethosomes, Emulsion, Marketed Herbal Novel Drug Delivery Formulations
INTRODUCTION: In the past few decades, considerable attention has been Concentrated on the evolution of novel drug delivery. The novel carriers should ideally fulfill two prerequisites. Firstly, it should deliver the drug at a Rate directed by the body's needs throughout Treatment. Secondly, it should channel the active entity of the herbal drug to the site of action. Conventional dosage forms, including prolonged-release dosage forms, cannot meet any of these. In phyto formulation research, developing nanosystem (NDDS) for herbal drugs.
Dosage forms (polymeric nanoparticles and nanocapsules, Liposomes, solid lipid nanoparticles, phytosomes Nanoemulsion, etc.) have several advantages for herbal drugs, including enhancement of solubility and bioavailability, pro-M Tection from toxicity, enhancement of pharmacological activity, enhancement of stability, improving tissue macrophages distribution, sustained delivery, protection from physical and chemical degradation, etc.
Thus the nano-sized novel drug delivery systems of herbal drugs have a potential future for Enhancing the activity and overcoming problems associated with plant medicines. Liposomes, which are biodegradable and essentially non-toxic vehicles, can encapsulate hydrophilic and hydrophobic materials 1. Liposome-based drug Delivery systems offer the potential to enhance the therapeutic Index of anti-cancer agents, either by increasing the drug concentration in tumor cells and/or by decreasing the exposure in normal tissues exploiting enhanced permeability and retention effect phenomenon and utilizing targeting strategies 2. The main advantages of using liposomes include: i) the high biocompatibility, ii) the easiness of preparation, iii) the chemical versatility that allows the loading of hydrophilic, amphiphilic, and lipophilic compounds, and iv) the simple modulation of their pharmacokinetic properties by changing the chemical composition of the bilayer components 3. Delivery of agents to the reticuloendothelial system (RES) is easily achieved since most conventional liposomes are trapped By the RES 1. Applying novel approaches can also improve the efficacy of herbal cosmetic formulations on the Human body 4.
Similarly, the other vesicular systems like Nanoemulsion, ethosomes, and transferases are highly useful assemblies and find various advantages in the delivery of herbal medicines; some are summarized in the present article. The phytosome process has also been applied to many popular herbal extracts, including Ginkgo biloba, grape seed, Hawthorn, milk thistle 5, green tea, and ginseng. These herbal extracts' Flavonoid and terpenoid components lend themselves quite well for directly binding to Phosphatidylcholine. Phytosome is produced by binding Individual components of herbal extracts to phosphatidyl Choline, resulting in a dosage form that is better absorbed. Thus, produces better results than the conventional herbal Extracts 6.
The results indicate that the absorption of silybin from silybin phytosome is approximately seven times greater Compared to the absorption of silybin from regular milk Thistle extract 5. Drugs can be embedded or dissolved in Nanoparticles and can also be adsorbed or coupled on the Surface 7. Encapsulating drugs within NPs can improve the solubility and pharmacokinetics of drugs and, in some cases, enable further clinical development of new chemical entities that have stalled because of poor pharmacokinetic properties 8. The major carrier materials of nanoparticles are synthetic Biodegradable high molecular polymers and natural polymers. The former usually includes poly-α-cyanoacrylate alkyl Esters, polyvinyl alcohol, polylactic acid, and polylactic-Glycolic acid. The latter is usually divided into two classes: Proteins (albumin, gelatin and vegetable protein) and Polysaccharides (cellulose, starch and its derivatives, alginate, Chitin, and chitosan, etc.) 9. In this article, an attempt has been made to touch upon Different aspects related to the development of novel herbal formulations, including method of preparation, type of active ingredient, entrapment efficiency, and applications, etc.
Types of Novel Herbal Drug Delivery Systems: Several approaches in case of new herbal drug delivery system include different types of expressions such as mouth‑dissolving tablets, liposomes, phytosomes, pharmacosomes, museums, nanoparticles, microspheres, transfersomes, ethosomes, transdermal drug delivery system (TDDS), and proniosomes are discussed Mouth‑dissolving tablets: Asoka Lifescience Limited launched Res‑Q, the world’s first Polyherbal mouth‑dissolving tablet, fast mouth‑dissolving drug. It induces a new drug delivery system that imparts increased efficacy. In the Ayurvedic medicine segment; is the inaugural Attempt to make medicines more effective in managing chronic Ailments. Res‑Q is a polyherbal medicine highly effective for Lung problems and other respiratory ailments such as asthma. This unique mouth‑dissolving drug delivery system ensures that the drug reaches the blood immediately and bypasses the first-pass Metabolism. It dissolves in the mouth by mixing with the saliva and gets absorbed. This Res‑Q provides relief from Respiratory distress within 15 min. Hence, the product resembles Sorbitrate's efficacy, a revolutionary Mouth-dissolving drug used in cardiac distress 10.
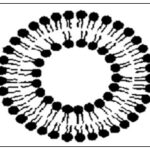
Liposomes: The liposomes are spherical particles that encapsulate a Fraction of the solvent, in which they freely diffuse (float) into their interior. They can have one, several, or multiple Concentric membranes. Liposomes are constructed of polar Lipids characterized by having a lipophilic and Hydrophilic group on the same molecules 11. Upon Interaction with water, polar lipids self-assemble and form Self-organized colloidal particles. Simple examples are deter-Gents; components form micelles, while polar lipids with Bulkier hydrophobic parts cannot associate micelles with High curvature radii but form bilayers that can self-close into liposomes or lipid vesicles. A cross-section of a liposome Fig. 1 depicts the hydrophilic heads of the amphiphile orienting towards the water compartment. In contrast, the lipophilic tails orient away from the water towards the center of the vesicle, thus forming a bilayer.
Consequently, water-soluble compounds are entrapped in the water compartment and aggregate in the lipid section. Uniquely, liposomes can encapsulate both hydrophilic and Lipophilic materials. Liposomes usually formed from phospholipid’s have been used to change the pharmacokinetics Profile of drugs, herbs, vitamins, and enzymes. A Variety of herbal liposomal formulations has been studied, summarized in Table 1. Because of their unique properties, liposomes can enhance the performance of Products by increasing ingredient solubility, improving Ingredient bioavailability, enhancing intracellular uptake, and altering pharmacokinetics and biodistribution 12 and in-vitro and in-vivo stability. Liposomes as a drug delivery system can improve drugs' therapeutic activity and safety, mainly by delivering them to their site of action and maintaining Therapeutic drug levels for prolonged periods 13–14. Milk thistle (Silybummarianum) is one of the few herbal Drugs whose excellent pharmacological profile readily provides proof of clinical efficacy 15.
Meanwhile, silymarin is poorly absorbed (20–50%) from the gastrointestinal tract 16. That causes the effects of silybin, one of the main active Flavonoids commonly found in dried fruits of silymarin, to be greater after parenteral than oral administration 17.
FIG. 1: CROSS-SECTION OF A LIPOSOME 4
TABLE 1: HERBAL FORMULATIONS IN LIPOSOMAL DRUG DELIVERY SYSTEMS
| Plants/constituents | Therapeutic category | Applications with respect to liposomal technology | Reference |
| Ampelopsin | Anticancer | Improved therapeutic efficacy | 18 |
| Capsaicin | Analgesic | Prolong action, permeation enhancement | 19 |
| Curcumin | Anticancer | Long systemic residence time and high entrapment efficiency | 20 |
| Paclitaxel | Anticancer | pH sensitivity and improved entrapment efficiency | 21 |
| Usnic acid | Antimycobacterial | Prolong action and solubility enhancement | 22 |
| Wogonin | Anticancer | Prolong duration of action | 23 |
| Quercetin and rutin | Hemoglobin | Enhancement of Hemoglobin binding | 24 |
| Garlicin | Lungs | Increase efficiency | 25 |
| Catechins | Antioxidant and chemopreventive | Increased permeation through skin | 26 |
| Breviscapin | Cardiovascular diseases | Sustained delivery of breviscapine | 27 |
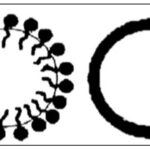
Nanoparticles: In recent years, the nanonization of herbal medicines has attracted much attention; 28 of them are illustrated in Table 2. Nanoparticles and Nanoemulsions Fig. 2 are Colloidal systems with particles varying in size from 10 nmTo 1000 nm 29, 30. Nanoparticle systems with mean particle Size well above the 100 nm standard have also been reported In literature, including nanonizedcurcuminoids 31, pacli-Taxel 32 and praziquantel 33 which have a mean particle Size of 450, 147.7, and even higher than 200 nm, respectively. IN addition, nanoparticles could also be defined as being Submicronic (b1 lm) colloidal systems 34. The nanospheres Have a matrix type structure in which the active ingredient is Dispersed throughout (the particles), whereas the Nano capsules have a polymeric membrane and an active ingredient Core. Nanonization possesses many advantages, such as increasing compound solubility, reducing medicinal doses, and improving the absorbency of herbal medicines compared with the respective crude drugs preparations 35.
FIG. 2: CROSS-SECTION OF (A) NANOEMULSION AND (B) BIOPOLYMERIC NANOPARTICLES 4
TABLE 2: HERBAL NANOPARTICULATE DRUG DELIVERY SYSTEM
| Plants/constituents | Therapeutic category | Applications Wrt Nanotechnology | Reference |
| Triptolide nanoparticle | Anti‑inflammatory | Enhance the penetration of drugs through the stratum corneum by increased hydration | 36 |
| Artemisinin nanocapsules | Anticancer | Sustained drug release | 37 |
| Texel nanoparticle | Anticancer | Enhance the bioavailability and sustained drug release | 38 |
| Berberine nanoparticle | Anticancer | Sustained drug release | 39 |
| Curcuminoids solid lipid Nanoparticle | Anticancer | Prolonged release of the curcuminoids | 40 |
| Camptothecin encapsulated Nanoparticle | Anticancer | Prolonged blood circulation and high accumulation in tumors | 41 |
| Naringenin nanoparticle | Hepatoprotective | Improved the release of NAR and improved its solubility | 42 |
| Breviscapine nanoparticle | Cardiovascular and cerebrovascular | Prolong the half‑life and decrease RES uptake | 43 |
| Tetrandrine nanoparticle | Lung | Sustained drug release | 44 |
| Glycyrrhizic acid nanoparticle | Anti‑inflammatory, antihypertensive | Improve the bioavailability | 45 |
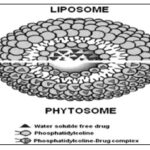
Phytosome: Over the past century, phytochemical and phytopharmacological sciences established the compositions, biological Activities, and health-promoting benefits of numerous plant Products. Most of the biologically active constituents of plants are polar or water-soluble molecules. However, water-soluble Phytoconstituents (like flavonoids, tannins, terpenoids, etc.) are poorly absorbed either due to their large molecular size, Which cannot absorb by passive diffusion, or due to their poor Lipid solubility, severely limiting their ability to pass across The lipid-rich biological membranes, resulting poor bioavailability 46. It has often been observed that the isolation and Purification of the constituents of an extract may lead to a Partial or total loss of specific bio-activity for the purified Constituent the natural constituent synergy becomes lost. Very often, the chemical complexity of the crude or partially purified extract seems essential for the active constituents' bioavailability. When taken orally, some Constituents may be destroyed in the gastric environment. As Standardized extracts are established, poor bioavailability often limits their clinical utility due to above said reasons. It has been observed that complexation with certain other clinically useful nutrients substantially improves the bioavailability of such extracts and their individual constituents. The nutrients so helpful for enhancing absorption are the Phospholipids. Phytosome is a patented technology developed by a leading manufacturer of drugs and nutraceuticals to incorporate standardized plant extracts or water-soluble Phytoconstituents into phospholipids to produce lipid-compatible molecular complexes, called phytosomes and so vastly improve their absorption and bioavailability 47 Table 3. In liposomes no chemical bond is formed; the Phosphatidylcholine molecules surround the water-soluble substance. Hundreds or even thousands of Phosphatidylcholine molecules may surround the water-soluble compound. In contrast, with the phytosome process, the Phosphatidylcholine and the plant components actually form A 1:1 or a 2:1 molecular complex depending on the substance (s) complexed, involving chemical bonds Fig. 3. Phospholipids are complex molecules that are used in all known life Forms to make cell membranes. In humans and other higher animals, the phospholipids are also employed as natural digestive aids and as carriers for both fat-miscible and water-miscible nutrients. They are miscible in water and Lipid environments and are well absorbed orally. Phytosomes are more bioavailable than conventional herbal extracts due to their enhanced capacity to cross the lipoidal Bio membrane and finally reach the systemic circulation.
FIG. 3: DIFFERENCE BETWEEN LIPOSOME AND PYTOSOME 58
TABLE 3: APPLICATIONS WRT PHYTOSOMAL TECHNOLOGY
| Plants/constituents | Therapeutic category | Applications Wrt Phytosomal technology | Reference |
| Ginkgo biloba | Cardioprotective, antioxidant activity | Flavonoids of GBP stabilize the ROS | 48 |
| Ginsenosides | Nutraceutical, immunomodulator | Increase absorption | 49 |
| Curcumin | Antioxidant, anticancer | Increase antioxidant activity and increase bioavailability | 50 |
| Quercetin | Antioxidant, anticancer | Exerted better therapeutic efficacy | 51 |
| Epigallocatechin | Nutraceutical, systemic antioxidant, anticancer | Increase absorption | 52 |
| Naringenin | Antioxidant activity | Prolong duration of action | 53 |
| Silybin | Hepatoprotective, antioxidant for liver and skin | Absorption of silybin phytosome from silybin is approximately 7 times greater | 54 |
Ethosomes: Newer advancements in patch technology have led to the development of ethosomal patch, which consists of the drug in ethosomes. Ethosomal systems are made up of soya Phosphatidylcholine, ethanol, and water. They may form Multilamellar vesicles and have a high entrapment capacity For particles of various lipophilicities. The elastic vesicles and transfersomes have also been used as drug carriers for a range of small molecules, peptides, proteins and vaccines 55. Ethosome has a high deformability and entrapment efficiency and can penetrate through the skin completely and improve Drug delivery through the skin. Likened to other liposomes, the Physical and chemical properties of ethosomes make the legal Transfer of the drug through the stratum corneum into a deeper Skin layer efficiently or even into the blood circulation 56. This property is important as the topical drug carrier and Transdermal delivery system. Moreover, the ethosomes carrier Also can provide an efficient intracellular delivery for both Hydrophilic and lipophilic drugs 57 percutaneous absorption of matrine an anti-inflammatory herbal drug, is increased 58. It also permits the antibacterial Peptide to penetrate the Fibrocyte easily 59. From the review of the literature it has been observed that, only three clinical trials have been conducted on ethosomal Systems in human volunteers. Horwitz et al. carried out a Pilot, double-blind, randomized clinical study to compare the efficacy of an ethosomal acyclovir preparation and commercially available acyclovir cream (Zovirax®) in treating recurrent herpes labialis in 40 human volunteers. The results revealed that the ethosomal acyclovir preparation performed Betterthan Zovirax cream and showed significant improvement in all the evaluated clinical parameters, such as the time of crust Formation and disappearance and pain parameters. The efficacy of ethosomal gel of clindamycin phosphate and salicylic acid was evaluated in a pilot clinical trial of 40 acne patients treated with the gel twice daily for 8 weeks. Volunteers treated with Ethosomal gel showed considerable improvement in acne Condition, with fewer comedies, pustules, and total lesions compared to placebo. Ethosomal Preparation of prostaglandin E1 was evaluated in a pilot clinical Study in patients with erectile dysfunction. It was observed that 12 of 15 tested patients had improved peak systolic velocity and penile rigidity. Erection duration was 10–60 min. There was no reported adverse skin reactions associated with the Treatment in any of the aforementioned clinical trials 60 Table 7 shows the Clinical data of ethosomes 61.
TABLE 4: THERAPEUTIC WRT NANOTECHNOLOGY
| Plants/constituents | Applications category | Therapeutic Wrt Nanotechnology | Reference |
| Capsaicin transfersomes | Analgesic | Increase skin penetration | 62 |
| Colchicine transfersomes | Antigout | Increase skin penetration | 63 |
| Vincristine transfersomes | Anticancer | Increase entrapment efficiency and skin | 64 |
| Matrine ethosomes | Anti‑inflammatory | Improve the percutaneous permeation | 65 |
| Ammonium glycyrrhizinate ethosomes | Anti‑inflammatory | Increase of the in vitro percutaneous permeation | 66 |
Emulsion: Emulsion refers to a non-homogeneous dispersion system that is composed of two kinds of liquids unable to dissolve each Other, one of which disperse in the other one in the form of Droplets 67. Generally, the emulsion is composed of oil phase, Water phase, surfactant and sub-surfactant. Its appearance is Translucent to transparent liquid. The emulsion can be classified as Intoordinary emulsion (0.1–100 μm), micro-emulsion (10– 100 nm), sub-micro-emulsion (100–600 nm), etc. Table 4 Among them, the micro-emulsion is also called Nanoemulsions, and the sub-micro-emulsion is also called lipid emulsion.
As a Drug delivery system, emulsion distributes in-vivo in the Targeted manner due to its affinity to the lymph. In addition, the drug can be sustained release in a long time because the drug is packaged in the inner phase and kept off direct touch with the Body and tissue fluid 68. After the oily drugs or lipophilic Drugs being made into O/W or O/W/O emulsion, the oil droplets Are phagocytosised by the macrophage and get a high Concentration in the liver, spleen, and kidney in which the Amount of the dissolved drug is very large. While water-soluble drug is produced into W/O or W/O/W emulsion, it can be easily Concentrated in the lymphatic system by intramuscular or Subcutaneous injection. The size of the emulsion particle has an Impact on its target distribution. Apart from its targeted sustained release, producing the Herbal drug into emulsion will also strengthen the stability of The hydrolyzed materials, improve the penetrability of drugs to The skin and mucous, and reduce the drugs’ stimulus to tissues. So far, some herbal drugs, such as camptothecin, Brucea Javanica oil, coixenolide oil and zedoary oil, have been made into emulsions.
For example, Zhou et al. 69 studied the Influence of the elemenum emulsion on the human lung Adenocarcinoma cell line A549 and protein expression. Results showed that the elemenum emulsion has a significant Inhibition on the growth and proliferation of the A549 in-vitro and it showed a time and dose-dependent relationship. Elemenum emulsion is a type of new anti-cancer drug with Great application prospects. Furthermore, it has no marrow Inhibition and no harm to the heart and liver.
TABLE 5: THERAPEUTIC WRT NANOTECHNOLOGY
| Plants/constituents | Applications category | Therapeutic Wrt Nanotechnology | Reference |
| Self‑nanoemulsifying Zedoary | Hepatoprotection anticancer | Improved aqueous | 70 |
| essential oil | Anti‑inflammatory | dispersibility, stability and oral dispersibility, bioavailability | 71 |
| Triptolide microemulsion | Anti-inflammatory | Enhance the penetration of drugs through the stratum corneum by increased hydration | 72 |
| Docetaxel submicron Emulsion | Anticancer | Improve residence time | 73 |
| Berberine nanoemulsion | Anticancer | Improve residence time and absorption | 74 |
| Silybin nanoemulsion | Hepatoprotective | Sustained release formulation | 75 |
| Quercetin microemulsion | Antioxidant | Enhance penetration into stratum corneum and epidermis | 76 |
Marketed Herbal Novel Drug Delivery Formulations: Two companies dominate the market for these systems, Namely, Cosmetochem and Indena. For herbal drug delivery, Cosmetochem launches Herbasec® technology in markets which are liposomal preparations of various herbal Ingredients such as extracts of White tea, Green tea, white Hibiscus, Gurana, and Aloe Vera. These extracts are used in Cosmetics because of their anti-oxidant effects to prevent aging. Indena patented the technology of phytosomes® and launched many products in the market with Diverse therapeutic benefits. Indena commercializes the plant Constituents/extracts of liquorice (18 ß – glycy-rrhetinic acid), Ammi visnaga (visnadin), Centella asiatica (triterpenes), G. biloba (ginkgo flavonglucosides, ginkgolides, bilobalide), Hawthorn flower (vitexin‑2″‑O‑rhamnoside), milk thistle (silymarin and Silybin), horse chestnut (escin ß‑sitosterol), Terminalia sericea (sericoside), Panax ginseng (ginsenosides), Grape seed (polyphenols), Green tea (polyphenols), etc 77.
CONCLUSION: Herbal medications have been widely employed all over the globe since ancient times and have been acknowledged by doctors and patients for their better therapeutic value as they cause fewer adverse effects compared with modern Medications. The drugs of Ayurvedic origin can be utilized in a more upright course with enhanced efficacy by incorporating modern dosage forms.
However, phytotherapeutics needs a scientific approach to render the components in a new way to Increase patient compliance and avoid repeated administration. This can be accomplished by designing NDDS for herbal Ingredients. NDDS not only reduces the repeated administration to overcome noncompliance but also helps increase the Therapeutic value by reducing toxicity and increasing bioavailability. Recently, pharmaceutical scientists.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Medina OP, Zhu Y and Kairemo K: Targeted liposomal drug delivery in Cancer. Curr Pharm Des 2004; 10: 2981 9.
- Sharma G, Anabousi S, Ehrhardt C and Kumar MNVR: J Drug Target 2006; 14: 301–10.
- Chanchal D and Swarnlata S: J Cosmet Dermatol 2008; 7: 89–95.
- Barzaghi N: Eur J Drug Metab Pharmacokinet 1990; 15(4): 333–8.
- doctormurray.com/articles/phytosomes html.
- Yuan DF, Yi YM. Her Med 2003; 22: 113–4.
- Alexis F, Basto P, Levy-Nissenbaum E, Radovic-Moreno AF, Zhang LF and Pridgen E: Chem Med Chem 2008; 3: 1839–43.
- Xiao YL and Li B: Chine Trad Herb Drugs 2002; 33: 385-8.
- Parakh SR and Gothoskar AV: Review of mouth dissolving tablet Technologies. Pharmaceutical Technology. Duluth, MN: Advanstar Communications 2003; 47 52.
- Xiao YL and Li B: Chine Trad Herb Drugs 2002; 33: 385–8.
- Lasic DD: ‘Liposomes: from physics to applications’. Elsevier, AmsterDam/London, New York, Tokyo 1993.
- Abou El Wafa AA, Mursi NM and El-Shaboury KM: A pharmaceuticl study on certain ocular drug delivery systems. MS Thesis. Cairo Univercity Cairo 2003.
- Weiss R and Fintelmann V: Herbal medicine. 2nd Stuttgart, New York Thieme 2000.
- Blumenthal M, Goldberg A and Brinkmann J: Herbal medicine. Integrative Medicine Communications. Newton 2000.
- Carini R, Comogoliom A, Albano A and Poli G: Biochem Pharmacol 1992; 43: 2111–5.
- Terreno E, Castelli DD, Cabellab C, Dastru W, Saninoa A and Stancanellob J: Paramagnetic liposomes as innovative contrast agents for magnetic resonance (MR) molecular imaging applications. Chem Biodivers 2008; 5: 1901 2. Doi.org/10.1002/cbdv.200890178.
- Wen Z, Liu B, Zheng Z, You X, PuY and Li Q: Preparation of liposomes Entrapping essential oil from Atractylodes macrocephala Koidz by Modified RESS technique. Chem Eng Res Des 2010; 88: 1102 7.
- Hong W, Chen DW, Zhao XL, Qiao MX and Hu HY: Preparation and study in-vitro of long circulating nanoliposomes of curcumin. Zhongguo Zhong Yao Za Zhi 2008; 33: 889 92.
- Li DC, Zhong XK, Zeng ZP, Jiang JG, Li L and Zhao MM: Application of targeted drug delivery system in Chinese medicine. J Control Release 2009; 138: 103 12.
- Priprem A, Watanatorn J, Sutthiparinyanont S, Phachonpai W and Muchimapura S: Anxiety and cognitive effects of quercetin liposomes in rats. Nanomedicine 2008; 4: 70 8.
- Gupta VK, Karar PK, Ramesh S, Misra SP and Gupta A: Nanoparticle Formulation for hydrophilic and hydrophobic drugs. Int J Res Pharm Sci 2010; 1: 163 9.
- Chen Y, Lin X, Park H and Greever R: Study of artemisinin nanocapsules as anticancer drug delivery systems. Nanomedicine 2009; 5: 316 22.
- Lira MC, Ferraz MS, da Silva DG, Cortes ME, Teixeira KI and Caetano NP: Inclusion complex of usnic acid with β cyclodextrin: Characterization and nanoencapsulation into liposomes. J Incl Phenom Macrocycl Chem 2009; 64: 215 24.
- Sun P, Den SH and Yu WP: Evaluation of garlicin liposomes. J Shan Univ TCM 2007; 31: 37–9.
- Fang J, Hwang T and Huang Fang C: Effect of liposome encapsulation of Tea catechins on their accumulation in basal cell carcinomas. Int J Pharm 2006; 310: 131 8.
- Zhinan M, Huabing C, Ting W, Yajiang Y and Xiangliang Y: Eur J Pharm Biopharm 2003; 56: 189–96.
- Ratnam DV, Ankola DD, Bhardwaj V, Sahana DK, Kumar MN: J Control Release 2006; 113: 189–207.
- Alle´mann E, Gurny R and Doelker E: Eur J Pharm Biopharm 1993; 39: 173–91.
- Tiyaboonchai W, Tungpradit W and Plianbangchang P: Int J Pharm 2007; 337: 299–306.
- Arica YB, Benoit JP and Lamprecht A: Drug Dev Ind Pharm 2006; 32: 1089–94.
- Mainardes RM and Evangelista RC: Int J Pharm 2005; 290: 137–44.
- Brigger I, Dubernet C and Couvreur P: Adv Drug Deliv Rev 2002; 54: 631–51.
- Mei Z, Chen H, Weng T, Yang Y and Yang X: Solid lipid nanoparticle and Microemulsion for topical delivery of triptolide. Eur J Pharm Biopharm 2003; 56: 189 96.
- Youfang C, Xianfu L, Hyunjin P and Richard G: Evaluation of artemisnin Nanoparticles. Nanomed Nanotechnol Biol Med 2009; 5: 316 22.
- Fu RQ, He FC, Meng DS and Chen L: Taxol PLA nanoparticles. ACTA Acad Med Mil Tertiae 2006; 28: 1573 4.
- Lin AH, Li HY, Liu YM and Qiu XH: Characterisation of berberine Nanoparticles. Chin Pharm 2007; 18: 755-7.
- Mukerjee A and Vishwanatha JK: Formulation, characterization and Evaluation of curcumin loaded PLGA nanospheres for cancer therapy. Anticancer Res 2009; 29: 3867 75.
- Min KH, Park K, KimYS, Bae SM, Lee S and Jo HG: Hydrophobically Modified glycol chitosan nanoparticles encapsulated camptothecin Enhance the drug stability and tumor targeting in cancer therapy. J Control Release 2008; 127: 208 18.
- Liu M, Li H, Luo G, Liu Q and Wang Y: Pharmacokinetics and Biodistribution of surface modification polymeric nanoparticles. Arch Pharm Res 2008; 31: 547 54.
- Feng LY, Tzu HW, Liang TL, Thau MC, Ch Feng LY, Tzu HW, Liang TL, Thau MC and Chun CL: Preparation and Characterization of Cuscuta chinensis Pharm Res 2009; 26: 893 902.
- Xiaoyan A, Jun Y, Min W, Haiyue Z, Li C and Kangde Y: Preparation of chitosan gelatin scaffold containing tetrandrine loaded Nano aggregates and its controlled release behavior. Int J Pharm 2008; 350: 257 64.
- Hou J and Zhou SW: Formulation and preparation of glycyrrhizic acid solid Lipid nanoparticles. ACTA Acad Med Mil Tertiae 2008; 30: 1043 5.
- Manach C, Scalbert A and Morand C: Am J Clin Nutr 2004; 79: 727–47.
- Bombardelli E, Curri SB, Loggia DR, Del NP, Tubaro A and Gariboldi P: Fitoterapia 1989; 60: 1–9.
- Semalty A, Semalty M and Rawat MS: The phyto phospholipid Complexes phytosomes: A potential therapeutic approach for herbal Hepatoprotective drug delivery. Pcog Rev 2007; 1: 369 74.
- Vandana SP and Suresh RN: Cardioprotective activity of Ginkgo biloba Phytosomes in isoproterenol induced myocardial necrosis in rats: A biochemical and histoarchitectural evaluation. Exp Toxicol Pathol 2008; 60: 397 404.
- Bhattacharya S: Phytosomes: Emerging strategy in delivery of herbal Drugs and nutraceuticals. Pharm Times 2009; 41: 9 12.
- Maiti K, Mukherjee K, Gantait A, Saha BP and Mukherjee PK: Curcumin phospholipid complex: Preparation, therapeutic evaluation and pharmacokinetic study in rats. Int J Pharm 2007; 330: 155 63.
- Maiti K, Mukherjee K, Gantait A, Ahamed HN, Saha BP and Mukherjee PK: Enhanced therapeutic benefit of quercetin – Phospholipid complex in Carbon tetrachloride–induced acute liver injury in rats: A comparative Study. Iran J Pharmacol Ther 2005; 4: 84 90.
- Maiti K, Mukherjee K, Gantait A, Bishnu PS and Mukherjee PK: Enhanced Therapeutic benefit of quercetin phospholipid complex in carbon Tetrachloride induced acute liver injury in rats: A comparative study. J Pharm Pharmacol 2006; 58: 1227 33.
- Yanyu X, Yunmei S, Zhipeng C and Qineng P: The preparation of Silybin phospholipid complex and the study on its pharmacokinetics in Rats. Int J Pharm 2006; 307: 77 82.
- Song YM, Ping QN, Wu ZH. Preparation of silybin nano emulsion and its pharmacokinetics in rabbits. J Chin Pharm Univ 2005; 5: 427 31.
- Vicentini FT, Simi TR, Del Ciampo JO, Wolga NO, Pitol DL and Iyomasa MM: Quercetin in w/o microemulsion: in-vitro and in-vivo skin penetration and efficacy against UVB induced skin damages Evaluated in-vivo. Eur J Pharm Biopharm 2008; 69: 948 57.
- Aggarwal G, Garg A and Dhawan S: Transdermal drug delivery: Evolving Technologies and expanding opportunities. Indian J Pharm Educ Res 2009; 43: 251-9.
- Dayan N and Touitou E: Carriers for skin delivery of trihexyphenidyl HCl: Ethosomes vs. liposomes. Biomaterials 2000; 21: 1879-85.
- Touitou E, Godin B, Dayan N, Weiss C, Piliponsky A and Levi Schaffer F: Intracellular delivery mediated by an ethosomal carrier. Biomaterials 2001; 22: 3053-9.
- Zhaowu Z, Xiaoli W, Yangde Z and Nianfeng L: Preparation of matrine Ethosome, its percutaneous permeation in-vitro and anti inflammatory Activity in-vivo in rats. J Liposome Res 2009; 19: 155 62.
- Zheng Y, Hou SX, Chen T and Lu Y: Preparation and characterization of Transfersomes of three drugs in-vitro. Zhongguo Zhong Yao Za Zhi 2006; 31: 728 31.
- Zhaowu Z, Xiaoli W, Yangde Z and Nianfeng L: Preparation of matrine Ethosome, its percutaneous permeation in-vitro and anti inflammatory Activity in-vivo in rats. J Liposome Res 2009; 19: 155 62.
- Zheng Y, Hou SX, Chen T and Lu Y: Preparation and characterization of Transfersomes of three drugs in vitro. Zhongguo Zhong Yao Za Zhi 2006; 31: 728 31.
- Xiao Ying L, Luo JB, Yan ZH, Rong HS and Huang WM: Preparation and in-vitro and in-vivo evaluations of topically applied capsiacin Transfersomes. Zhongguo Zhong Yao Za Zhi 2006; 31: 981 4.
- Singh HP, Utreja P, Tiwary AK and Jain S: Elastic liposomal formulation For sustained delivery of colchicine: In-vitro characterization and in-vivo Evaluation of anti gout activity. AAPS J 2009; 11: 54 64.
- Paolino D, Lucania G, Mardente D, Alhaique F and Fresta M: Ethosomes For skin delivery of ammonium glycyrrhizinate: In-vitro percutaneous Permeation through human skin and in-vivo anti inflammatory activity on human volunteers. J Control Release 2005; 106: 99 110.
- Ma B, Duan X and Wang Z: Clinical and experimental study on Shuanghua Aerosol in treating infantile upper respiratory tract infection. Zhongguo Zhong Xi Yi Jie He Za Zhi 2000; 20: 653-5.
- Zhang Y. Chin Tradition Herbal Drugs 2006; 37: 641–7.
- Lu MF, Cheng YQ, Li LJ and Wu JJ: Mater Rev 2005; 19: 108–10.
- Zhou X, Li LY and Guo ZJ: Chin Clin Oncol 2004; 9: 229–34.
- Lu MF, Cheng YQ, Li LJ and Wu JJ: Progress of study on passive targeting of drug delivery system. Mater Rev 2005; 19: 108 10.
- Zhou X, Li LY and Guo ZJ: Application of Targeted Drug Delivery system in Chinese Medicine. Chin Clin Oncol 2004; 9: 229 34.
- Zhao Y, Wang C, Chow AH, Ren K, Gong T and Zhang Z: Self nanoemulsifying drug delivery system (SNEDDS) for oral delivery of zedoary essential oil: Formulation and bioavailability studies. International Journal of Pharmaceutics 2010; 383: 170 7.
- Zhinan M, Huabing C, Ting W, Yajiang Y and Xiangliang Y: Solid lipid Nanoparticle and microemulsion for topical delivery of triptolide. Eur J Pharm Biopharm 2003; 56: 189 96.
- Li L, Wang DK, Li LS, Jia J, Chang D and Ai L: Preparation of docetaxel Submicron emulsion formation for intravenous administration. J Shenyang Pharm Univ 2007; 12: 736 9.
- Sun HW and Ouyang WQ: Preparation, quality and safety evaluation of berbarine nano emulsion for oral application. J Shangh Jiaotong Univ (Agric Sci) 2007; 1: 60 5.
- Pinto JF: Site specific drug delivery systems within the gastro intestinal Tract: From the mouth to the colon. Int J Pharm 2010; 395: 44 52.
- Li L, Wang DK, Li LS, Jia J, Chang D and Ai L: Preparation of docetaxel Submicron emulsion formation for intravenous administration. J Shenyang Pharm Univ 2007; 12: 736 9.
- Sun HW and Ouyang WQ: Preparation, quality and safety evaluation of berbarine nano emulsion for oral application. J Shangh Jiaotong Univ (Agric Sci) 2007; 1: 60 5.
- Pinto JF: Site specific drug delivery systems within the gastro intestinal Tract: From the mouth to the colon. Int J Pharm 2010; 395: 44 52.
- Pinto JF: Site specific drug delivery systems within the gastro intestinal Tract: From the mouth to the colon. Int J Pharm 2010; 395: 44 52.
How to cite this article:
Tagad PA, Shelake AJ, Palve KA, Kale RA and Bansode SS: Novel drug delivery systems of herbal medicine. Int J Pharmacognosy 2023; 10(6): 315-23. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.10(6).315-23.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
315-323
679 KB
853
English
IJP
Pankaj A. Tagad *, Atish. J. Shelake, Krushna A. Palve, Rushikesh A. Kale and Santosh S. Bansode
Dr. Kolpe Institute of Pharmacy, Kolpewadi, Kopargaon, Maharashtra, India.
tagadpankaj03@gmail.com
05 June 2023
18 June 2023
20 June 2023
10.13040/IJPSR.0975-8232.IJP.10(6).315-23
30 June 2023