NEUROCHEMICAL SCREENING OF PTEROSPERMUM ACERIFOLIUM IN ANIMAL MODELS
HTML Full TextNEUROCHEMICAL SCREENING OF PTEROSPERMUM ACERIFOLIUM IN ANIMAL MODELS
Ankur Choubey * and Gopal Garg
School of Pharmacy, Suresh Gyan Vihar University, Jaipur - 302017, Rajasthan, India.
ABSTRACT: Pterospermum acerifolium is a common to plant in India is considered carminative, stimulant and emmenagogue. In the present study, the ethanol extract of the bark of Pterospermum acerifolium has been evaluated for Neurochemical study. Treatment with ethanolic extract of Pterospermum acerifolium was found to significantly decrease the serum levels of adrenocorticotropic hormone (ACTH), corticosterone (CORT) and -endorphin (-EP) as well as the brain and serum level of norepinephrine (NE). Furthermore, ethanolic extract of Pterospermum acerifolium was able to significantly reverse the chronic stress by decreasing the brain and serum levels of the monoamine neurotransmitters dopamine (DA), 5-hydroxytryptamine (5-HT). The results obtained from this study suggested that the memory-enhancing effect of ethanolic extract of Pterospermum acerifolium was mediated through regulations of neurochemical and neuroendocrine systems. From the present study, it was concluded that herbal drugs could be potentially used to control the state of CNS disorders. Further investigations are, however, necessary to explore mechanism(s) of action involved in these pharmacological activities.
| Keywords: |
Pterospermum acerifolium, Adrenocorticotropic, Corticosterone (CORT), Dopamine (DA), 5-Hydroxytryptamine (5-HT)
INTRODUCTION: India has an antediluvian heritage of traditional medicine. Materica Medica of India provides lots of information on the folklore practices and traditional aspects of the therapeutically consequential natural product. Indian traditional medicine is predicated on phytochemical, pharmacological & allied approaches including instrumental techniques like chromatography, microscopy, and others. There is accumulating evidence suggesting medicinal plants are illimitable reservoirs of drugs.
The astounding structural diversity among their active components makes them a utilizable source of novel therapeutic compounds. Researchers with interest in natural products have intensified their effort towards scientific evaluation of traditional medicines. Healthcare system from time immemorial, particularly among tribal communities. In India, about 2000 drugs used are of plant origin 5. Plant resources are depleting globally at an alarming rate, and some economically and medicinally paramount plant species will anon be extinct. Medicinal plants are now under great pressure due to their exorbitant accumulation or exploitation. 1
It is well established that the disruption of the hypothalamus-pituitary-adrenal (HPA) axis, a central pathway to the entire endocrine system, is often central to most health problems, syndromes, diseases, and even aging itself. Hyperactive status of the HPA axis can result in increasing levels of corticotropin-releasing hormone (CRH), adrenocorticotropic hormone (ACTH), and glucocorticoids in the hypothalamus, pituitary, and adrenal cortex, respectively.
On the one hand, stress can also alter the physiological homeostasis which can result in various neuronal, endocrine, and visceral dysfunctions. Furthermore, stress is also known to alter cognitive functions, such as memory, and it has been linked to the pathophysiology of mood and anxiety disorders 2-8. A central feature of the stress response is the activation of the HPA axis which can increase plasma levels of glucocorticoids. As a consequence of their profound effects on neurons, glucocorticoids can influence behavior, mood, and memory process.
Neurotransmitter systems are also involved in learning and memory processes, and a substantial part of learning and memory impairments is due to changes in neurotransmission 9-18. It is well established that neurotransmitters can interfere with learning acquisition and memory.
In this context, the memory dysfunction described in abnormal Savda syndrome could involve excessive production of corticotropin-releasing hormone (CRH), adrenocorticotropic hormone (ACTH), and glucocorticoids in the hypothalamus, pituitary, and adrenal cortex, respectively, under the stress condition. Pterospermum acerifolium is traditionally used to reduce CNS disorders. Generally, plants possess many pharmacological actions, such as Hepatoprotective, antioxidant, anti-inflammatory, anthelmintic, antimicrobial since they contain numerous constituents of active chemicals in it based on the claim by traditional healers that the plant is effective in the treatment of central nervous system (CNS) diseases.
The present project is done to explore the potential of herbal drugs for the treatment of CNS disorders with a view to performing phytochemical investigation assess neurochemical screening 19-22.
EXPERIMENTAL:
Plant Material: Plants materials P. acerifolium bark were collected from the local market of Bhopal, (M.P.) during May-July 2012. The specimens were identified and authenticated by Dr. Zia ul Hassan, Assistant professor, Department of Botany, Saifia College of Science & Education, Bhopal and their herbarium was deposited. These collected specimens were chosen for the extraction process and assessment of neurobehavioural activity.
Extraction: The plant materials so collected were cleaned properly and washed with distilled water to remove dust particles and dried in the shade. The dried drugs were coarsely powdered and then exhaustively extracted with 50% ethanol in Soxhlet apparatus for 72 h. The ethanolic extracts so obtained were freed of solvent under vacuum (Yield: 9.33 %)
Qualitative Analysis: The Ethanol extracts were screened for the presence of secondary metabolites. Tests was carried out for carbohydrates, reducing sugars, tannins, polyphenols, lipids, flavonoids, ketones, alkaloids, steroids and triterpenes.
In-vivo Study:
Animals for the Experiment: Swiss albino rats were obtained from animal house VNS Institute of Pharmacy with due permission from the Institutional animal ethical committee (Registration Number. 778/03/c/cpcsa).
Acute toxicity studies were conducted by using albino mice of either sex weighing between 20 and 25 gm and healthy adult male albino rats weighing between 150, and 200 gm were selected for the antiurolithiatic activity. The animals were acclimatized to standard laboratory conditions (temperature: 25 ± 2 ºC) and maintained on a 12-h light: 12-dark cycle. They were provided with regular rat chow (Lipton India Ltd., Mumbai, India) and drinking water ad libitum.
Acute Toxicity: In an acute toxicity study of Allium Sativum plant extract, animals were given single doses of the drug. The Swiss albino rats were divided into groups. All animals fed with standard rat pelleted diet (Lipton India Ltd. pellets) and had free access to tap water ad libitum. Acute toxicity studies were performed according to the OECD guidelines.
The doses selected for the study were 50 mg/kg, 100 mg/kg, 200 mg/kg, 300 mg/kg, 400 mg/kg for one day. Three animals were taken for each dose. It was observed that the extract doesn’t produce any significant effect on the behavior of rats. The animals were observed for 3 h after dose administration and also after 24 and 48 h 19-22 - 3.3 Estimation of Neurochemicals.
Estimation of Adrenocorticotropin (ACTH), Corticosterone (CORT), and β Endorphin (β-EP): The blood was collected and centrifuged at 4 ºC; the serum was stored at −80 ºC before assay. Serum levels of ACTH, CORT, and β-EP were determined using the ELISA kit (obtained from R&D Systems). The sensitivity of the assay was 1.0 ng/mL. Intra-assay and interassay coefficients of variation were less than 4.85% and 6.08%, respectively. The test was performed according to the manufacturer’s specification.
Reagents: Adrenocorticotropin (ACTH), cortico-sterone (CORT), and β-endorphin (β-EP) kits were obtained from R & D Systems, USA. Norepinephrine (NE, purity ≥ 97%), dopamine (DA, purity ≥ 99%), 5-hydroxytryptamine (5-HT, purity ≥ 99%), 3, 4–dihydroxyphenylalanine (DOPAC, purity ≥ 99%), and 3,4-dihydroxybenzylamine (DHBA, purity ≥ 98%) were obtained from Sigma Co., Ltd., USA. All other reagents were of analytical grade.
Surgical:
Hemostatic Sponge: AbGel, Absorbable gelatin sponge USP, Srikrishna Laboratories, Mumbai, India.
Sterile Sutures: Ethicon 4-0, Non-absorbable surgical sutures USP. Mersilk (Braided silk black). Ethicon 4-0, Absorbable surgical sutures USP (Catgut), Johnson and Johnson, India.
Surgical Needle: Curved surgical needles were obtained from Pricon Surgicals, New Delhi, India.
Equipment: Stereotaxic Frame: Inco Ambala Materials and Methods 65.
Drug Administration: Wistar rats were divided into four groups of six rats each. Group, I served as a normal group. Animals of Group II serves as a model group and III-V group were administered orally with respective test drugs, between 7:30 am -9:30 am daily during 14 days. These doses were calculated according to the conversion table of equivalent effective dose ratios from human to animals based on the body surface area. Food was withdrawn from the animals 2 h prior to drug administration but the water was allowed freely. The PTetoh pretreatment groups (Gr. III, IV, and V) received the same electric foot-shock one hour after drug administration (8:30 am - 10:30 am).
- Normal group (0.5% Sodium carboxyl methyl cellulose (CMC-Na) solution (20 mL/kg, b.w.)
- Model group
- Ethanolic extract of Pterospermum acerifolium (2.53 g/kg)
- Ethanolic extract of Pterospermum acerifolium (5.06 g/kg g/kg)
- Ethanolic extract of Pterospermum acerifolium (10.12 g/kg)
Measurements of Monoamine Neurotransmitters by HPLC-FCD: Norepinephrine (NE), Dopamine (DA), Serotonin or 5-hydroxytryptamine (5-HT) Level of monoamine neurotransmitters (NE, DA, 5-HT, and) in serum and brain were measured by HPLC coupled with a fluorescence detector (FCD). Mice were sacrificed immediately after exposure to the stress. Blood was sampled into EDTA-containing tubes at 10:00 am and separated in a refrigerated centrifuge at 10,000×g for 10 min at 4 ºC. The serum was stored at −80 ºC until assayed. After blood collection, the brains were quickly removed, frozen in liquid nitrogen, and stored at −80 ºC until assayed. To determine serum monoamine neurotransmitter levels, an equal volume of 0.1M HCl was added to the serum samples containing 200 μg/mL of DHBA as an internal standard 19-22.
The samples were then shaken and mixed for 1.5 min in ice water. One drop of concentrated HCl was then added to the solution and mixed in ice water for another 1.5 min and then centrifuged at 3000 rpm, 4 ºC for 10 min. The samples of brain tissue were homogenized in an ice water solution of 0.1M HCl. Then, 0.1M HCl solution was added to the samples (1 μL/1mg tissue) containing 200 μg/mL of DHBA as an internal standard and centrifuged at 18000 rpm, 4 ºC for 10 min. The samples were filtered through 0.45 μm microfilters (MFS Inc., USA).
Aliquots (10 μL) of supernatant were injected into a reverse phase HPLC column (condition: Agilent 110180 high-voltage pump coupled to a fluorescence detector, chromatographic column ZORBAX ODB C18 4.6 mm × 150 mm × 5 mm, voltage 121V, and wavelength 360 nm). All the brain samples were weighed on an electronic scale before HPLC analysis, and the results were expressed as ng of monoamine/mg of wet weight tissue.
Statistical Analysis: Statistical evaluation of the data was done by Student’s t-test. (Graph PAD Instat software, Kyplot). A value of p<0.05 was considered to be significant.
RESULTS AND DISCUSSION:
Qualitative Analysis: The preliminary phytochemical screening of extracts revealed the presence of various phytoconstituents. The ethanolic extract of P. acerifolium revealed the presence of alkaloids, glycosides, sterols, flavonoids, tannins, phenolic compounds, carbohydrates, fats, and oils.
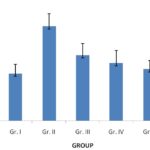
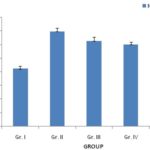
Effects of P. acerifolium on the Serum Levels of ACTH, CORT, and β-EP in Chronic Stress Mice: Table 1 showed that the serum levels of ACTH, CORT, and β-EP were increased (P < 0.01) in the chronic stress mice (Gr. II) when compared to the normal group (Gr. I).
Oral administration of P.acerifolium at doses of 2.53 g/kg, 5.06 g/kg, and 10.12 g/kg (Gr. III, IV, and V) for 14 days was found to decrease of the levels of ACTH, CORT, and β-EP in the serum when compared to the model group (Gr. II).
TABLE 1: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF ACTH
| GROUP | ACTH (pg/mL) |
| Gr. I | 16.0 ± 3.12 |
| Gr. II | 32.10 ± 4.14* |
| Gr. III | 22.25 ± 3.89** |
| Gr. IV | 19.60 ± 4.21** |
| Gr. V | 17.51 ± 2.84** |
*Results are given as means ±SEM, when compared to the normal group (Gr. I), P<0.01.
**Results are given as means ± SEM, when compared to the model group (Gr. II), P<0.05.
FIG. 1: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF ACTH
TABLE 2: EFFECTS OF P.ACERIFOLIUM ON THE SERUM LEVEL OF CORT
| GROUP | CORT (pg/mL) |
| Gr. I | 12.18 ± 4.9 |
| Gr. II | 29.18 ± 4.5* |
| Gr. III | 19.90 ± 5.6** |
| Gr. IV | 21.50 ± 3.34 ** |
| Gr. V | 21.40 ± 2.1 ** |
*Results are given as means ±SEM, when compared to the normal group (Gr. I), P<0.01.
**Results are given as means ±SEM, when compared to the model group (Gr. II), P<0.05.
TABLE 3: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF β-EP
| GROUP | β-EP (pg/mL) |
| Gr. I | 153.17 ± 27.19 |
| Gr. II | 255.21 ± 23.12* |
| Gr. III | 200.32 ± 34.25** |
| Gr. IV | 193.10 ± 19.65** |
| Gr. V | 176.00 ± 20.56** |
*Results are given as means ±SEM, when compared to the normal group (Gr. I), P<0.01.
**Results are given as means ±SEM, when compared to the model group (Gr. II), P<0.05.
FIG. 2: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF β -EP
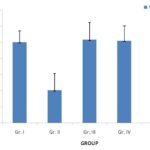
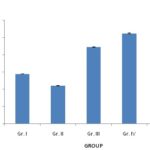
Effects of P. acerifolium on the Contents of Monoamine Neurotransmitters of Brain and Serum in the Chronic Stress Mice: Figures showed an increase (P<0.05) of NE level in the serum of the chronic stress mice (Gr. II) and a decrease of the serum levels of DA when compared to the normal group (Gr. I). Oral administration of P. acerifolium during 14 days at doses of 2.53 g/kg, 5.06 g/kg, and 10.12 g/kg (Gr. III, Gr. IV, and Gr. V) was found to decrease the NE levels (P<0.01), while the levels of DA, Fig. 4 and 5 showed similar results with an increase of the NE level (P<0.01) but a decrease in the levels of DA (P<0.01) in the brain of the chronic stress mice (Gr. II), when compared to the normal group (Gr. I). All doses of P. acerifolium (2.53 g/kg, 5.06 g/kg, and 10.12 g/kg) were found to reduce the concentration of NE in the brain (P<0.01) when compared to the stress mice (Gr. II). In contrast, only the dosages of 5.06 g/kg and 10.12 g/kg of P. acerifolium (Gr. IV and V) could raise the levels of DA in the brain (P<0.05) of the stress mice (Gr. II).
TABLE 4: EFFECTS OF P.ACERIFOLIUM ON THE SERUM LEVEL OF NE (ng/mL)
| Group | NE (ng/mL) |
| Gr. I | 422.22 ± 20.19 |
| Gr. II | 695.55 ± 23.10 |
| Gr. III | 625.32 ± 30.25** |
| Gr. IV | 600.11 ± 18.65** |
| Gr. V | 400.01 ± 20.56** |
FIG. 3: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF NE (ng/mL)
TABLE 5: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF 5-HT (ng/mL)
| GROUP | 5-HT (ng/mL) |
| Gr. I | 100.5 ± 14.19 |
| Gr. II | 40.50 ± 21.10∗ |
| Gr. III | 103.5 ± 31.25** |
| Gr. IV | 102.11 ± 18.65** |
| Gr. V | 104.01 ± 21.56** |
FIG. 4: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF 5-HT (ng/mL)
TABLE 6: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF DA (ng/mg)
| GROUP | DA (ng/mg) |
| Gr. I | 2900.25 ± 21.19 |
| Gr. II | 2212.50 ± 18.10∗ |
| Gr. III | 4450.5 ± 27.25** |
| Gr. IV | 5250.11 ± 38.65** |
| Gr. V | 5650.01 ± 27.56** |
FIG. 5: EFFECTS OF P. ACERIFOLIUM ON THE SERUM LEVEL OF DA (ng/mg)
CONCLUSION: The preliminary phytochemical screening of extracts revealed the presence of various phytoconstituents. The ethanolic extract of P. acerifolium revealed the presence of alkaloids, glycosides, sterols, flavonoids, tannins, phenolic compounds, carbohydrates, fats, and oils. Figures and table showed that the serum levels of ACTH, CORT, and β-EP were markedly increased (P<0.01) in the chronic stress mice (Gr. II) when compared to the normal group (Gr. I). Oral administration of P. acerifolium at doses of 2.53 g/kg, 5.06 g/kg, and 10.12 g/kg (Gr. III, IV, and V) for 14 days caused a decrease of the levels of ACTH, CORT, and β-EP in the serum when compared to the model group (Gr. II).
Figures and table showed an increase (P<0.05) of NE level in the serum of the chronic stress mice (Gr. II) and a decrease of the serum levels of DA when compared to the normal group (Gr. I). Oral administration of P. acerifolium during 14 days at doses of 2.53 g/kg, 5.06 g/kg, and 10.12 g/kg (Gr. III, Gr. IV, and Gr. V) was able to decrease the NE levels (P<0.01), while the levels of DA, Fig. 5 and 6 showed similar results with an increase of the NE level (P<0.01) but a decrease in the levels of DA (P<0.01) in the brain of the chronic stress mice (Gr. II), when compared to the normal group (Gr. I).
All doses of P. acerifolium (2.53 g/kg, 5.06 g/kg, and 10.12 g/kg) were found to reduce the concentration of NE in the brain (P<0.01) when compared to the stress mice (Gr.II). In contrast, only the dosages of 5.06 g/kg and 10.12 g/kg of P. acerifolium (Gr. IV and V) could raise the levels of DA in the brain (P<0.05) of the stress mice (Gr. II).
ACKNOWLEDGEMENT: I am thankful to my mentor Gopal Garg and my research institute VNS Institute of Pharmacy, Bhopal for Research Project.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Abramson, Modeling psychopathology in the laboratory, Psychopathology: Experimental Models. San Francisco. 1978: 1-26.
- Adamec, Long-lasting, selective, anxiogenic effects. Physiology and Behaviour 2004; 83: 401-410.
- Akiskal, Interaction of biologic and psychologic factors. Acta Psychiatrica Scandium. 1985; 71: 131-139.
- Davidson: Achieving remission with venlafaxine and fluoxetine. Depression Anxiety 2002; 16: 4-13.
- Davis: Long-lasting effects of acute stress on the neurochemistry. Psychopharmacology 118: 267-272.
- Deakin: Roles of serotoninergic systems in the escape, Psychopharmacology Vol 2. New York: Academic Press 1983: 149-193.
- Distinct roles of 5HT subsystems in panic, anxiety and depression. (Eds.), Biol. Psych., vol. 1. Elsevier, Amsterdam 1991: 305-307.
- Deakin: 5-HT, anti-depressant drugs and the psychosocial origins of depression. Journal of Psychopharmacology 1996; 10: 31-38.
- Deakin: Social stress, steroid levels, 5-HT and depression. Journal of Psychopharmacology 1997; 11: A93-A97.
- Deakin: Depression and antisocial personality disorder. Journal of Neural Transmission Supplementum 2003; 64: 79-93.
- Deb: The rate of psychiatric illness one year after traumatic brain injury. American Journal of Psychiatry 1999; 156: 374-378.
- Denenberg VH: Open-field behavior in the rat: what does it mean? Annals of the New York Academy of Science 1969; 159: 852-859.
- Edwards: The psychological impact of a cancer diagnosis on families. Psycho-oncology 2004; 13: 562-576.
- Edwards: Olfactory bulb removal: effects on sexual behavior and partner-preference in male rats. Physiology and Behaviour 1990; 48: 447-450.
- Evans: Mood disorders in the medically ill. Biological Psychiatry 2005; 58: 175-189.
- Fann: Sertraline in the treatment of major depression. Journal of Neuro-psychiatry Clinical Neuroscience 2000; 12: 226-32.
- Fedoroff: Depression in patients with acute traumatic brain injury. American Jour of Psychiatry 1992; 149: 918-923.
- Feinstein: The pre-therapeutic classification of co-morbidity in chronic disease. Journal of Chronic Diseases 1970; 23: 455-468.
- Ferré: Struggling and flumazenil effects in the swimming test are related to the level of anxiety in mice. Neuropsychobiology 1994; 29: 23-27.
- Fehr: Serotonergic polymorphisms in patients suffering from alcoholism. Progress in Neuropsychopharmacology and Biology Psychiatr 2001; 25: 965-982.
- Fernandez-Ruiz: Olfactory dysfunction in hereditary ataxia and basal ganglia disorders. Neuroreport 2003; 14: 1339-1341.
- Feske: Anxiety as a predictor of response to interpersonal psychotherapy. Depression and Anxiety 1998; 8: 135-141.
How to cite this article:
Choubey A and Garg G: Neurochemical screening of Pterospermum acerifolium in animal models. Int J Pharmacognosy 2015; 2(1): 38-43. doi: 10.13040/IJPSR.0975-8232.2(1).38-43.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
7
38-43
894
1963
English
IJP
A. Choubey * and G. Garg
School of Pharmacy, Suresh Gyan Vihar University, Jaipur, Rajasthan, India.
chaubey.ankur03@gmail.com
01 October 2014
21 October 2014
24 December 2014
http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(1).38-43
01 January 2015