NANOBOTS AND SMART DRUG DELIVERY: BRIDGING THE GAP BETWEEN NANOTECHNOLOGY AND MEDICINES
HTML Full TextNANOBOTS AND SMART DRUG DELIVERY: BRIDGING THE GAP BETWEEN NANOTECHNOLOGY AND MEDICINES
Kartik Mondal, Satpal Kushawha *, Tanya Sharma and Ranjan Kumar Singh
Faculty of Pharmaceutical Sciences, Mewar University, Gangrar, Chittorgarh, Rajasthan, India.
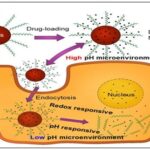
ABSTRACT: Nanobots and smart drug delivery system is one of the radical convergences of nanotechnology and mainstream medicine with unprecedented precision, efficiency and control in therapeutic intervention. Conventional methods of delivering drugs are usually limited by low bioavailability, non-selective distribution, systemic toxicity and variable drug levels that can result in a low level of efficacy and side effects. Conversely, nanobots, engineered nanoscale systems, with the ability to navigate the biological world, and smart drug delivery systems, offer targeted, controlled, and stimuli-responsive therapeutic solutions, thereby overcoming most of these issues. Nanobots are usually made of biocompatible polymers, metals, lipids or hybrid composites and may be functionalised with ligands, antibodies or peptides to bind to a particular cellular or molecular target. It is a type of nanoscale machines that are developed to do complicated jobs, such as the delivery of drugs to target sites, biosensing, diagnostics, and even microsurgery. This is because their capacity to cross biological barriers, including the blood-brain barrier, and concentration at disease sites can result in site-directed drug release, which causes minimal harm to healthy tissues. Moreover, the development of nanorobotics has provided the possibility of integrating propulsion systems, including magnetic, chemical or acoustic control, that will permit external or autonomous navigation in the human body. Smart drug delivery systems are used alongside nanobots, and add stimuli-responsive capabilities to controlled drug delivery in response to particular physiological factors, such as pH, temperature, enzymes, or redox environment. As an example, the tumor microenvironment is frequently acidic with an increased activity of enzymes and can be targeted by pH-responsive or enzyme-responsive nanocarriers to deliver anticancer drugs to the tumor. Likewise, diabetic management systems responsive to glucose have been established, which allows the real-time release of insulin, depending on the level of glucose in the bloodstream. These smart systems improve therapeutic effects and lessen the dosing rate and systemic side effects.
Keywords: Nanobots, Smart drug delivery, Nanotechnology, Targeted therapy, Controlled release, Stimuli-responsive systems, Nanocarriers, Precision medicine, Theranostics, Drug targeting, Biocompatibility, Nanorobotics, Biomedical applications, Cancer therapy, Drug delivery systems etc
INTRODUCTION: Modern day healthcare has been revolutionized by nanotechnology which has provided innovative solutions to the constraints of the conventional therapeutic strategies.
Nanobots are one of the recent innovations that have emerged as a potentially useful technology in drug delivery systems.
Through the introduction of smart features like controlled drug delivery, excellent targeting, and continuous monitoring, the nanobots have created a new potential in the treatment of complex medical conditions like cancer, neurological diseases, and cardiovascular diseases-related disorders 1. In recent years, research in the field of nanomedicine has produced encouraging results. The science that integrates chemistry, medicine, and nanotechnology is called nanomedicine 2. Targeted drug delivery is currently a very important issue in current medicine, with the intention of overcoming the shortcomings of the traditional drug delivery systems. Conventional approaches Often have the problem of low bioavailability through systemic distribution of the agent and non-specific toxicity (such as healthy tissues as well as cancerous cells) resulting in unwanted side effects and poor therapeutic effect 3. As a case in point, in chemotherapy, the cytotoxic material gets distributed throughout the body, so alongside cancerous cells, there are healthy tissues, causing unwanted side effects and low treatment effect On the same note, drugs used in the neurological treatment are usually more difficult to penetrate the blood-brain barrier, and therefore, it is important to have sophisticated drug delivery models that can guarantee the release of drugs to the specific location and minimal harm to the normal tissues 4.
FIG. 1: NANOBOTS AND SMART DRUG DELIVERY
Nanobots are a possible solution to these challenges as they offer more accurate drug delivery, efficacy of therapy, and controlled drug release. Nanobots have advanced navigation systems and can passively move along complicated biological routes and target particular locations. They can be programmed to deliver therapeutic proteins in their optimum doses and reach the site of action. This regulated release system has greatly minimized systemic toxicity and increased drug 5 bioavailability moreover, developments in nanobots designs have added functionalities like magnetic control systems, bio-hybrid models and sensor-based monitoring, which has further enhanced the accuracy of the treatment. Although there has been a lot of advancements in nanobots technology, there are still some challenges. The problems with mass production, long-term biocompatibility, and the accuracy of control in dynamic physiological conditions need to be studied. Also, issues relating to immune responses, regulatory acceptance, and ethical concerns have to be considered before clinical adoption can be fully achieved. Research is underway to refine the propulsion mechanisms, surface modifications to promote enhanced stability, and devise safe biodegradation strategies to ensure optimal therapeutic performance 6.
The analysis will aim to provide a detailed discussion on the application of nanobots in the process of smart drug delivery, particularly focusing on the recent developments in clinical potential, and future development. This review will address current limitations and seek novel solutions that can help to transform nanotechnology into modern medicine by showing the potential the nanobots have to revolutionize the future of personalized medicine and enhance healthcare outcomes 7.
Micro and Nanoscale Robots for Targeted Drug Delivery: Recent advances in Micro and Nanoscale Electromechanical Platforms have made possible the development of implantable robotic systems that can be used to perform specialized tasks including accurate drug and gene delivery.
Following the explosive developments in nanotechnology, scientists have turned to developing Nanorobots that are controlled by an internal or external power source, machine intelligence (MI), and sensing. These factors state of the art robotic systems show features like information processing, signal betweenness, movement control, and cellular therapeutic treatments, which guarantee specific drug dispersion.
This method greatly improves the effectiveness of treatment and minimizes the side effects that come with traditional treatments. Moreover, nanorobots can be very useful in the detection of dangerous substances and theragnostic uses 8.
The bio nanorobot control system works on a set of pre-programmed rules, such as navigation, target recognition, obstacle avoidance, detection and attachment, drug release, task execution and self-deactivation to ensure safe excretion. Molecular manufacturing is AI-controlled to increase control over nanorobot movement and behaviour. The autonomous and cooperative behaviour of these nanorobots in targeted delivery of drugs has been successfully demonstrated using computational models and simulations Bio-nanorobots work through biological actuation mechanisms to move through the blood after administering them intravenously. Numerous novel nanorobotics drug delivery systems have been developed such as Micro scale robotic swimmers that have the ability to deliver drugs precisely. Temporal micro robotic systems that would deliver. Therapeutic payloads with precision via Nanoscale communication architectures and remotely controlled microscopic robots with the ability to penetrate deep tissue and treat chronic diseases 9.
FIG. 2: MICRO AND NANOSCALE ROBOTS FOR TARGETED DRUG DELIVERY
Nanorobots in Autism Therapy and Healthcare: In the case of people with autism spectrum disorder (ASD), difficulties in social interaction, emotional regulation, and verbal communication may occur. The challenges in the interpretation of the body language, maintaining eye contact and conveying emotions affect their relationship-building. As there is no cure to autism at the moment, the emphasis is on improving the quality of life. In a variety of therapeutic interventions, Therapeutic interventions assisted with robotics have come to the forefront against therapy using animals, mainly because of increased control and reduced chances of allergies or infections. These robots interact with patients in sensory, emotional, cognitive, and tactile-kinaesthetic ways, facilitating verbal and nonverbal communication 10.
Besides autism therapy, robots have found their way into other fields of healthcare:
Long-term Patient Care: Robotic systems are used in nursing homes to help serve patients with chronic diseases, disabilities and injuries.
Home-Based Healthcare: Robots enable medical services at home reducing the necessity to visit the hospital.
Hospital-Based Treatment: Robotic technologies facilitate the process of diagnosis and treatment, such as surgical and non-surgical.
Emergency Care: Robotic-assisted systems will aid in the diagnosis and treatment of acute medical conditions that need urgent treatment.
Palliative and Hospice Care: Robots offer solace and supportive care to the critically ill patients.
Substance Abuse Rehabilitation: Robotics are involved in counselling, laboratory testing, and medication management to people that have gone through an addiction healing process 11.
Delivery of Drugs with Micro- and Nanorobots: Current drug administration methods are mostly based upon overall systemic delivery, which is frequently lacking in both precision of localisation of drug delivery and deep tissue penetration.
Micro and nanorobots have become the promising solutions in drug delivery, as intelligent carriers that could be precisely navigated and released. Such miniature robotic systems possess a number of benefits, such as increased therapeutic efficacy and decreased systemic toxicity. There are several models that have been constructed with biological parts like bacteria, red blood cells (RBCs) and stem cells. These natural biocompatible microrobots pass on the biocompatibility of nature and thus immune detection is evaded. But external magnetic fields, or ultrasound waves are usually necessary to control their movement 12. One recent development in this area was reported by Shao et al. who developed autopropelled Autonomous micromotors by fusing neutrophils with chemotactic behaviour with Nanoparticles made of mesoporous silica (MSNs). These micromotors efficiently tracked chemical attraction gradients induced by E. coli thus improving the specific delivery of therapeutic molecules. An alternative novel method is the incorporation of magnetic nanoparticles (MNPs) inside the RBCs and allows the controlled movement by using ultrasound energy. It was demonstrated that through the loading of therapeutic agent (Doxorubicin (Dox), quantum dots (QDs), and MNPs) onto the RBC-based microrobots, the drug can be effectively delivered to the cell without causing cytotoxicity. Also, magneto-aerotactic bacteria such as Magneto coccus marinus MC-1 have been studied with regard to their ability to promote the dispersion of therapeutic agents in hypoxic tumour environments 13.
Decades of laboratory studies have proven the effectiveness of micro and nanorobots in the delivery of drugs. Wu et al. developed polymeric nanomotors (based on multilayers) that were loaded with Doxorubicin and were able to deliver the drug to tumour spots 14.
FIG. 3: DELIVERY OF DRUGS WITH MICRO- AND NANOROBOTS
Active Drug Targeting with Micro and Nanorobots: There has been a remarkable breakthrough in the development of micro and nanorobots as the movement toward experimental research towards potential clinical applications has been achieved. These microrobots loaded with drugs can be directed with external stimuli such as magnetic forces, pH changes, high-frequency sound waves and near-infrared electromagnetic radiation to make sure the drugs are released at the precise point. To improve drug retention and stability, researchers have devised sophisticated drug loading methods, such as physical adsorption and layer-by-layer encapsulation.
The use of catalytic nanorobots to transport drugs is one of the most promising events in this sector. Catalytic nanorobots of Pt nanowires proposed by Wang et al. use the magnetic interactions to deliver Fe3O4 based drug carriers. The use of self-electrophoresis propulsion, however, has limitations in high ionic environment. To overcome this problem, scientists have made micro and nanobots that are propelled by bubbles, which enables the robots to move more effectively through biologically important fluids. Wu et al. developed autonomously mobile polymeric nanorobots with platinum (Pt) and Fe3O4 nanoparticles which were able to release drugs under the influence of ultrasound irradiation 15.
Finding A Way to Overcome the Difficulties of Passive Drug Delivery: The conventional methods of drug delivery rely on circulation and diffusion that frequently lead to an ineffective targeting and low therapeutic outcome (. On the contrary, micro and nanorobots offer improved navigation, directional propulsion as well as deep tissue penetration,) which greatly enhances the drug bioavailability at the target site Traditional chemotherapy often causes extensive toxicity due to its inability to target. To overcome these difficulties, scientists have considered two main options: creating drugs that specifically attack cancer cells and using micro/nanorobots to deliver drugs precisely 16.
One such promising method of cancer therapy is synthetic lethality, whereby a particular genetic mutation in cancer cells causes them to be very sensitive to targeted therapies. Even though this approach has worked in some types of cancer, it is also limited in applicability because of the heterogeneity of the tumour and resistance to drugs. On the other hand, micro and nanorobots are a more versatile approach as they enhance the retention and penetration of drugs in tumour tissues.
In contrast to passive nanoparticles, microrobots are active in search of diseased locations and penetrate high-density tissue architecture and deliver drugs to the target site with high precision in space and time. Moreover, these nanorobots reduce the chances of the therapeutic agents being diluted in the body fluids hence high concentration of drugs in the treatment site. Micro and nanorobot-assisted drug delivery has been shown to be successful in proof-of-concept studies using cellular and animal models. Such developments signify the possibility of transforming targeted therapy and overcoming the shortcomings of the traditional passive drug delivery systems
Micro and nanorobots are a groundbreaking change in targeted drug delivery. The robotic carriers are intelligent and provide accurate navigation, controlled drug release, and improved penetration, overcoming the issue of systemic toxicity and inefficient targeting. Through the use of biological elements, external propulsion systems and novel drug-loading technologies, scientists have proven their potential in both in vitro and in vivo experiments. As this area advances, micro and nano robots will be the basis of improved, customized therapeutic strategies, which will eventually improve patient outcomes in diverse therapeutic practices 17.
Nanorobots in Medicine: Transforming Healthcare Applications:
Enhancing Blood Functions: Nanorobots are introducing remarkable innovations in blood treatments. (2023) Reciprocates are artificial red blood cells which have the potential to carry oxygen effectively, which is of great benefit to patients with oxygen deficiency. They also aid in the removal of toxic gases like the carbon monoxide in the blood. Simultaneously, artificial white blood cells (microbivores) are extremely effective immune fighters, destroying dangerous bacteria and viruses more rapidly than standard antibiotics. The other important innovation is the clottocytes which are artificial platelets which speed up the clotting of the blood to aid in excessive bleeding which is controlled in an effective manner 18.
Innovations in Dentistry: Denti robots are specialized nanorobots that are designed to eliminate plaque without disrupting the natural balance of oral bacteria. These microscopic instruments are used in precisely aligning the teeth in orthodontic treatments. Moreover, they also contribute to decreasing tooth sensitivity; they close uncovered nerve endings, which relieves pain
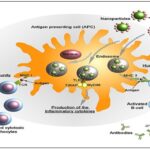
Cancer Therapy Advancements: Nanorobots are used in the context of targeted therapeutic payload delivery with respect to cancer treatment. Nanorobots that utilize bacteria can deliver chemotherapy agents directly to the malignant biological units, enhancing the efficacy of the treatments, and minimizing adverse reactions. Nanorobots operated by DNA detect markers of cancer cells and deliver medication only on the diseased site. An alternative groundbreaking strategy includes utilizing thrombin-releasing nanorobots, that cause clot formation within tumour blood vessels, ultimately cutting off their nutrient supply and hindering tumour growth 19.
Nanotechnology to Manage HIV: Nanorobots are being examined in their applications in fighting HIV by repairing and restoring immune cells which have been damaged by the virus. This is to stimulate the natural immune control of the body, which is a promising new avenue in the future treatment of HIV.
Precise Surgical Assistance: Nanorobots are also assisting surgical procedures to be more precise. These microscopes will be able to detect anomalies and help in fine-tuning surgeries on a cellular level, which will minimize the risk posed by conventional surgical procedures. Their accuracy reduces difficulties, resulting in better patient outcomes.
Applications in Neurology: Nanotechnology is becoming a very important aspect in nerve regeneration and neurosurgery. Nanorobots can help in repairing nerve damage providing a potential advantage in spinal cord injuries. Nanoscale surgical devices, commonly known as nano knives, are useful in brain surgery to allow the creation of a perfect incision and specific treatment. Thedevices are also useful in identifying brain aneurysms through the analysis of molecular biomarkers, thereby minimizing the cost of diagnosing 20. These shortcomings can be effectively addressed by using encapsulated polyphenols instead of free molecules. The applications of polyphenolic chemical nanoencapsulation technology in the food and pharmaceutical industries, as well as its safety and biocompatibility, are the main topics of this study 21. The stability, bioavailability, and targeted administration of phytochemicals are being positively impacted by recent advancements in nanocarrier systems, such as polymeric nanoparticles and antibody-conjugated formulations. Additionally, new pharmaceutical approaches for PCOS control have been made possible by our growing understanding of the manipulation of gut microbiota, functional genomics, and proteome interactions 22. Creating novel and successful treatments requires a thorough grasp of the molecular mechanisms controlling regulated and controlled drug distribution 23.
Gene Therapy Integration: Nanorobots are promising when it comes to gene therapy as they detect and fix defective genes. This could bring the genetic disorders breakthrough and better the management of hereditary diseases
Diabetes Management: The nanorobotic systems can now be used to monitor glucose levels and provide the correct insulin intake. These small micro-sensors constantly monitor blood sugar and provide real-time information to external surveillance systems, which allow patients to keep glycaemic control under control
Evaluation of In-vivo Nanorobot Activities: Although the majority of nanorobot researches have been performed in-vitro, the first in-vivo experiments have yielded promising results. One of the earlier studies to investigate chemically driven micromotors within a living organism was a revolutionary study conducted by Gao et al. Their distribution, retention, cargo delivery efficiency, and toxicity in the stomach of a mouse was analysed in this study. With the development of zinc-based micromotors, the study proved that the propulsion propelled by acids greatly increased their adhesiveness and retention in the stomach lining. These micromotors were able to dissolve in gastric acid with time as they effectively release their therapeutic agents without leaving any toxic residues.
FIG. 4: EVALUATION OF IN-VIVO NANOROBOT ACTIVITIES .0 EVALUATION OF IN-VIVO NANOROBOT ACTIVITIES
Further in-vivo investigations explored nanorobots' potential in targeting and destroying tumours by restricting blood supply. These were run on mice and pigs whereby nanorobots were injected intravenously into the blood of mice with breast cancer, melanoma, lung cancer, and ovarian cancer. It took the nanorobots just a few hours before they could find and enclose the tumour locations. At 24 hours, there was evident tissue damage as a result of blockage of blood flow. Blood clotting was observed within 48 hours and full clot formation was observed after 72 hours in all tumours. Although it was feared that nanorobots could destroy healthy tissues, there is no evidence that that has happened and so they are precise in targeting the cancerous cells.
Two main ways through which nanorobots can be eliminated in the body can be identified. The former is natural excretion and they can be safely discharged without any build-up. The other technique is the active scavenger system that has been created to eliminate them effectively. Alternatively, centrifugationbased filtration can be used to pull out nanorobots. During this procedure, the blood of a patient is directed through a special device, and aural transmitters command nanorobots to keep the blood in a neutral state of buoyancy. Consequently, other blood elements will be filtered outwards and the nanorobot-rich plasma will be filtered through a 1micrometer pathway membrane, which in effect removes the other components. The purified Plasma is then combined with elements in blood and then returned into the body without any harm 24.
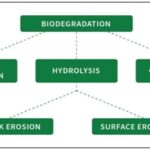
Degradation of Nanorobots: The degradation of nanorobots is a crucial aspect of their design and application in biomedical systems, as it directly impacts their safety, biocompatibility, and clinical feasibility. Once they have fulfilled their target purpose, e.g., delivering drugs to a specific location or performing some form of diagnostic sensing, the nanorobots will have to be safely disassembled and removed without introducing any kind of toxicity or chronic build-up in the body. Thus, the behavior of degradation of nanorobots needs to be learnt and controlled in order to make them a successful part of medicine.
FIG. 5: DEGRADATION OF NANOROBOTS
There are many materials used to make nanorobots, and they may be biodegradable polymers (PLGA and chitosan), lipids, metals or hybrid nanocomposites. Out of these, biodegradable materials are the most desirable since they can be broken down into non-toxic by-products which can be disposed of naturally out of the body. The degradation may or may not be via a number of different mechanisms, such as hydrolysis, enzymatic degradation, oxidation and environmental stimuli such as changes in pH or temperature 25.
One of the most common ways of degradation is hydrolytic degradation, especially of polymer based nanorobot. Water molecules in this process dissolve the chemical bonds in the polymer matrix and the nanorobot structure is disintegrated gradually. An example is a polymer such as poly(lactic-co-glycolic acid) (PLGA), which breaks down to lactic acid and glycolic acid, which is further broken down by the natural biochemical processes.
Enzymatic degradation includes breaking down nanorobots through the action of certain enzymes in the body. Protein-based, lipid-based, or DNA-based nanorobots may be degraded by enzymes like proteases, lipases, or nucleases, respectively. This process is especially beneficial to targeted degradation, where some enzymes are overexpressed in diseased tissues, meaning that nanorobot can be site-specifically broken down upon delivery of drugs to these locations.
Oxidative degradation is the process of structural damage and fragmentation of nanorobot materials when they are in contact with reactive oxygen species (ROS). It is commonly found in inflammatory or cancerous conditions with increased levels of ROS. Though the degradation of nanorobots by oxidative degradation can facilitate the degradation process, it is important to recognize that too much ROS can lead to inadvertent damage to the surrounding tissues, and so, design needs to be carefully considered.
Stimuli-responsive degradation an advanced approach, nanorobots is designed to respond to certain internal or external stimuli by degrading. As an example, pH sensitive nanorobots can be degraded in tumor acidic environments, and temperature sensitive systems can be used in response to local heat. Likewise, externally stimulated triggers (light, magnetic fields, ultrasound, etc.) can trigger degradation at specific sites and times, allowing better control of drug release and clearance.
The byproducts of nanorobots once degraded should be effectively removed out of the body. The main excretion pathways are renal (kidney) and hepatobiliary (liver) excretion of smaller and larger or hydrophobic particles, respectively. Surface characteristics, size, and shape of degradation products have a profound impact on their clearance pathways. Idealistically, nanorobots are supposed to breakdown to particles small enough to be filtered by the kidneys and excrete through urine.
Although biodegradable nanorobots have their benefits, there are still difficulties with full and reliable degradation. Uncompleted degradation can cause the formation of residual material in body parts like the liver or spleen that can cause chronic toxicity. Moreover, other metallic or inorganic nanomaterials might not be easily degraded, which is a concern regarding their permanence in the body. Thus, a lot of research is dedicated to creation of fully biodegradable and bioresorbable nanorobot systems.
The rate of degradation is also another factor that should be put into careful consideration to be able to match the therapeutic needs. Quick degradation can cause rapid release of the drug, and the slow degradation can cause a longer exposure time and toxicity. Designing nanorobots with adjustable degradation profiles is crucial to achieving the best possible therapeutic outcomes 26.
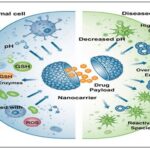
Safety and Biocompatibility of Nanorobots: The safety and biocompatibility of nanorobots is one of the most important aspects that define their successful usage in biomedical applications. Nano robots being introduced into the human body are expected to play well with the biological systems without creating toxicity, immunogenicity and without resulting in any long term negative effects. These properties need to be ensured before nanorobots can be accepted to be used in clinics.
The notion of biocompatibility denotes that the nanorobots are able to carry out the intended purpose without causing any adverse reactions in the body. This is heavily based on the materials that they are made of. Polymers (e.g., PLGA, chitosan), lipids, and some proteins are also commonly used as biocompatible and biodegradable materials since they can be safely broken down by non-toxic byproducts. Surface modification methods, e.g. polyethylene glycol (PEG) coating are frequently employed to increase compatibility, reducing protein adsorption and preventing clearance by the immune system 27.
Cytotoxicity or the possibility of nanorobots damaging or killing cells is one of the main safety issues. The chemical composition, size, shape, or surface charge of nanorobots can result in cytotoxic effects. Smaller nanoparticles, although advantageous in cellular penetration, can react more violently with cellular components, which can cause a break in membranes or intracellular activity.
Thus, long-term in-vitro and in-vivo experiments are performed to determine cell viability, oxidative stress and inflammatory responses.
The other significant factor is the immune response elicited by nanorobots. Human immune system might treat nanorobots as foreign bodies, and, therefore, they can be easily eliminated by the immune system via phagocytosis by the macrophages. Not only does this decrease the effectiveness of therapeutic effects but can also result in inflammation or hypersensitivity reactions. In order to avoid this, nanorobots are commonly engineered to be stealthy, by the use of hydrophilic coatings or biomimetic surfaces that facilitate avoiding immune responses and extend their stay in the bloodstream.
Another important aspect is the hemocompatibility, especially in case of nanorobots, which are injected. These systems should not interfere with the normal blood functions like coagulation, platelet activity, or red blood cell integrity. Thrombosis or hemolysis are some of the complications caused by inadequate hemocompatibility. Thus, extensive blood compatibility analyses are carried out to make sure that nanorobots could safely circulate without any adverse hematological effects.
The problem of nanotoxicology is not only limited to direct cellular effects but also to the long-term effects on the organs and tissues. The filtration and clearance processes of the body can lead to the accumulation of nanorobots in crucial organs like liver, spleen, kidneys and lungs. Such piling up may even cause organ toxicity in the event that the materials are not efficiently biodegradable. Histopathological and biochemical marker studies are crucial in determining the health of the organs after nanorobot delivery.
Nanorobots size, shape, and surface properties are important factors that affect their safety profile. An example is that the spherical nanoparticles tend to be less toxic than the rod-shaped or irregular shape. Similarly, surface charge plays a role in cellular interaction, with highly charged particles often exhibiting higher toxicity. It is important to optimize these physicochemical properties in order to enhance biocompatibility.
Moreover, other significant determinants of safety include dose and length of exposure. Even biocompatible materials may prove to be toxic when in large doses or when exposed over a long period of time. Thus, one of the most important design considerations of nanorobots is to identify the most effective dose that is simultaneously effective and not harmful. Ethical and regulatory factors are also critical towards safety. Nanorobots would have to be subjected to a lot of preclinical testing before clinical application since they would have to be subjected to a series of clinical trials to test their safety and 28.
FIG. 6: SAFETY AND BIOCOMPATIBILITY OF NANOROBOTS
Advantages of Nanorobotics over Conventional Medical Techniques: Nanorobotics is a ground breaking innovation in contemporary medicine and it possesses many benefits over the traditional methods of medicine. Although effective to a certain degree, traditional methods of diagnosing and treating have a number of limitations that include lack of specificity, systemic side effects, invasive procedures and unsatisfactory delivery of drugs. Nanorobotics provides solutions to these problems by proposing very precise, controlled and intelligent systems that can operate at the level of cells and molecules 29.
Targeted drug delivery is one of the greatest benefits of nanorobotics. Traditional ways of administering drugs spread drugs in the body, both in diseased and normal tissues. Such non-selective distribution frequently causes side effects and a decrease in therapeutic effectiveness. Conversely, nanorobots may be designed to contain targeting ligands that bind to particular cells or receptors, allowing the nanorobots to deliver drugs to the disease site. This comes in handy especially in the treatment of cancer where the nanorobots can target tumor cells with minimal harm to the healthy tissues.
The other significant benefit is the decreased side effects and toxicity. Nanorobots are also able to deliver drugs to the target site meaning that the dosage is very minimal in most cases as opposed to traditional treatment. This topical administration is much less systemic and the number of adverse effects is minimized. As an example, nanorobots can reduce the unwanted side effects like loss of hair, nausea and immunosuppression caused by chemotherapy drugs.
Another important advantage of nanorobotics is the minimally invasive treatment. Conventional surgeries are often characterized by extensive tissue damage, increased recovery time and increased chances of infection. Nanorobots are extremely small, which means that they can easily get through blood vessels and tissues to carry out the functions of delivering drugs, clearing clots, or even carrying out a microsurgery without making large incisions. This results in quicker recovery, less pain and better patient comfort.
Controlled and sustained drug release is also possible with Nanorobotics. The traditional methods of drug delivery can lead to variations in the concentration of the drug in the blood and the need to take drugs often. Nanorobots can be programmed to deliver drugs gradually, either in the long-term or in response to certain stimuli, like pH, temperature, or enzymes. This guarantees maintenance of therapeutic levels and enhances treatment 30.
Real-time monitoring and diagnostics are another valuable benefit. Nanorobots may be fitted with sensors and imaging agents that enable continuous check up on physiological conditions. This allows detection of diseases at an early stage, monitoring the course of treatment in realtime, and timely corrections to the treatment. Precision medicine is based on such capabilities, whereby treatment is customized to the needs of a particular patient.
The capability to traverse biological barriers is a special quality of nanorobots. Traditional drug delivery methods frequently fail to overcome obstacles like the blood brain barrier and thus cannot be used to treat neurological diseases. Nanorobots because of their size (nanoscale) and high-performance design bypass these obstacles and can deliver drugs to the previously inaccessible regions, creating new opportunities in the treatment of diseases such as brain tumors and neurodegenerative disorders.
Nanorobotics also accommodates the use of multifunctionality where the same system can be used to carry out various functions such as delivery of drugs, imaging and sensing. This combination of diagnostic and treatment capabilities is referred to as theranostics and can be more efficient and make the implementation of several separate tests unnecessary 31.
FIG. 7: ADVANTAGES OF NANOROBOTICS OVER CONVENTIONAL MEDICAL TECHNIQUES
Challenges and Limitations in Nanorobot Development and Implementation: Although nanorobots have great potential in targeted drug delivery and precision medicine, the process of their development and deployment is fraught with several scientific, technical, regulatory, and ethical issues. These restrictions have to be overcome so as to have safe, effective and wide-spread clinical adoption. Biocompatibility and toxicity is one of the main challenges. Nanorobots are in direct contact with biological systems and any reaction of the biological system is negative and can affect the safety of the patient. Materials and nanorobots can cause cytotoxicity, oxidative stress, or inflammation. Moreover, the non-biodegradable elements may be stored in such vital organs as the liver, spleen, kidneys, and cause long-term toxicity. Making sure that nanorobots are completely biocompatible and break down into harmless byproducts is a key area of research 32.
The other important limitation is the recognition and clearance of immune system. The immune system of the body frequently recognizes nanorobots as foreign bodies and destroys them by other means like phagocytosis. This decreases their circulation time and precludes their reaching target sites. Although surface modifications like PEGylation can help evade immune detection, achieving complete immune compatibility is still challenging.
Accurate navigation and control in the human body is a complicated matter as well. Although external guidance techniques like magnetic fields, ultrasound or light have been promising, it is challenging to control nanorobots in deep tissues or complex physiological settings. Their movement and targeting accuracy can be influenced by factors like the dynamics of blood flow, tissue barriers, and biological variability.
There are further challenges in terms of manufacturing and scalability. Nanorobot creation needs sophisticated nanotechnology methods, which can be complicated and expensive. This is because it is hard to achieve consistency, reproducibility, and large-scale production, which restricts commercialization. Furthermore, the stability in the course of storage and transportation is another complication.
Another significant constraint is the energy supply and propulsion. Nanorobots can move by means of external energy sources or by means of chemical reactions. Nonetheless, it is difficult to have a safe and reliable source of energy in the human body. The chemical propulsion techniques can add toxic byproducts, and the external control techniques may possess a low penetration depth or accuracy 33.
The barriers to nanorobot translation to clinical use are regulatory and approval barriers that have great influence. Safety, efficacy, pharmacokinetics and long-term impact data is needed in large quantities by regulatory agencies. There are no standardized evaluation procedures of nanorobots, which also complicates the approval process. Clinical trials take time, are costly and slow the adoption of these technologies by patients.
Ethical and safety issues are also pivotal. Patient privacy, possible misuse, and long-term biological impacts are questions that emerge with the utilization of nanorobots. To illustrate, there is a fear of uncontrolled conduct, unwanted interactions with biological systems, or problems in retrieval of the nanorobots after use. The need to address these ethical issues is critical to popular acceptance and conscientious execution.
The other significant issue is the issue of limited knowledge of the long-term effects. Due to the novelty of nanorobotics, little data is available on the long-term effects of nanorobots on human health and the environment. Prolonged exposures, possible bioaccumulation and interactions with complex biological systems need to be investigated with long term studies.
Other barriers include cost and access. Nanorobot-based therapies are costly to develop and implement, which means that they are not accessible to a wider population. This will require cutting the cost of production and enhancing affordability in order to be widely adopted 34.
Lastly, there is the complexity of interdisciplinary nature that contributes to the challenges. The development of nanorobots involves many disciplines, such as nanotechnology, biology, medicine, engineering, and computer science. It may be challenging to coordinate these disciplines and to put their knowledge into practice. In order to maximise therapeutic efficacy and minimise drug accumulation in unwanted parts of the body, modern drug delivery systems (DDS) are created using state-of-the-art technology to quickly administer medications across the body to a specific target location. As a result, they have a considerable influence on how diseases are managed and treated. Compared to conventional techniques, modern drug delivery systems (DDS) offer a number of advantages 35.
CONCLUSION: Nanobots and smart drug delivery systems are an innovative breakthrough in the sphere of contemporary medicine as they practically combine nanotechnology and therapeutic sciences. In this work, it has become clear that these technologies provide revolutionary solutions to much of the constraints that are involved with traditional drug delivery means. Nanorobots can greatly improve therapeutic outcomes through delivery of therapeutic agents in a controlled and efficient manner, reducing any adverse effects and thereby improving treatment outcomes. Among other things, nanorobotics has contributed to making it possible to target the cellular and molecular level with precision. Nanorobots, unlike the conventional drug delivery systems that release drugs distributed in a non-specific manner throughout the body, can be designed to locate and target certain disease locations. Not only does this focused strategy enhance the effectiveness of the drugs used but it also minimizes systemic toxicity, which is especially useful in the treatment of diseases like cancer. This precision is further improved by the implementation of smart drug delivery systems that allow the release of drugs in a controlled manner in response to a certain physiological stimulus, e.g., pH, temperature, or enzyme activity.
A major advancement towards personalized medicine is the theranostics of nanorobots, integrating diagnostic and therapeutic capabilities into robots. The multifunctional systems can be used to monitor disease progression and treatment response in real-time and the professionals can customize the therapies to meet the needs of a specific patient. This change to precision medicine can transform healthcare by not focusing on general approaches to treatment but focusing on more individualized and effective treatment approaches.
However, despite these encouraging developments, there are obstacles to the creation and application of nanorobots. Biocompatibility, toxicity, and immune system interaction issues, and long-term safety concerns should be thoroughly considered in order to guarantee their successful use in clinical practice. There are also technical issues like accurate navigation, controlled propulsion and mass production that are a major obstacle. The regulatory and ethical factors also make the translation of nanorobots between laboratory experiments and actual medical practice more complicated. A second important point that is brought out in this paper is the significance of degradation and clearance mechanisms. To be safely used in the human body, nanorobots should break down into non-toxic substances and be effectively removed and not accumulate and harm the body. The development of biodegradable materials and stimuli-responsive systems is aiding in solving these issues, yet more studies are required to obtain the best safety profile.
In-vivo assessment of nanorobot behavior is essential in comprehending their behavior in complex biology. In order to validate their performance and make sure they are reliable, comprehensive research on biodistribution, pharmacokinetics, and therapeutic efficacy is necessary. Further development of imaging and monitoring systems will also help us to learn more about these systems and optimise them.
In the future, the future of nanorobotics in medicine is very bright. The capabilities of nanorobots will continue to increase with emerging technologies in the fields of artificial intelligence, machine learning, and advanced biomaterials, and they will allow more intelligent, adaptable, and highly efficient therapeutic systems. With interdisciplinary collaboration ever-increasing, the blend of nanotechnology with other fields of science will probably introduce novel ways of solving some of the most difficult medical ailments.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kumbhar PR, Kumar P, Lasure A, Velayutham R and Mandal D: An updated landscape on nanotechnology-based drug delivery, immunotherapy, vaccinations, imaging, and biomarker detections for cancers: recent trends and future directions with clinical success [Internet]. Vol. 18, Discover Nano. Springer US; 2023. Available from: https://doi.org/10.1186/s11671-023-03913-6
- Pavan J, Mm C, Rao P, Singh RK, Garg A and Rajeswari T: A comprehensive review on blood brain delivery methods using nanotechnology. Trop J Pharm Life Sci 2024; 11(6): 43–52.
- Mahima A and Vaibhav G: Nanorobots in Medicine: Advancing Healthcare through Molecular Engineering: A Comprehensive Review. IgMin Res 2024; 2(11): 938–49.
- Mehta J, Pathania K and Pawar SV: Recent overview of nanotechnology based approaches for targeted delivery of nutraceuticals. Sustain Food Technol 2025; 3(4): 947–78.
- Ashtekar KS, Monde AA, Nilangekar P, Bagul U, Nazirkar M and Kokare C: A review on nanotechnology based ophthalmic drug delivery system. J Drug Deliv Ther 2023; 13(12): 262–9.
- Sathish Kumar A, Joshna N, Saha GC, Saha H and Masum Billah M: A review of recent advancements in nanotechnology for medical drugs delivery. Res J Pharm Technol 2024; 17(4): 1891–4.
- Dhyani A, Bhatt J, Singh N and Dhyani A: Development and Applications of Micro-and Nanorobotics in Drug Delivery. Int J Appl Pharm 2023; 15(5): 60–7.
- Kim M, Nicholas JD, Puigmarti-Luis J, Nelson BJ and Pané S: Targeted drug delivery: from chemistry to robotics at small scales. Annu Rev Control Robot Auton Syst 2025; 8(1): 379–405.
- Suhail M, Khan A, Rahim MA, Naeem A, Fahad M and Badshah SF: Micro and nanorobot-based drug delivery: an overview. J Drug Target 2022; 30(4): 349–58.
- Bhat RS, Alkhudhairy R, Alshehri AA and Singh R: Nanotechnology - an innovative approach to cope with the distinctive challenges linked with Autism Spectrum Disorder. Int J Autism Challenges Solut 2024; 1(1): 28–38.
- Pennisi P, Tonacci A, Tartarisco G, Billeci L, Ruta L and Gangemi S: Autism and social robotics: A systematic review. Autism Res 2016; 9(2): 165–83.
- Das T and Sultana S: Multifaceted applications of micro/nanorobots in pharmaceutical drug delivery systems: a comprehensive review. Futur J Pharm Sci 2024; 10(1).
- Verma MK, Bala MD, Khoza PB, Papo TR and Shozi ML: Core-shell nanoparticles for water purification: Advances in photocatalytic and antimicrobial applications. Next Mater 2025; 9(9).
- Pinto S, Hosseini M, Buckley ST, Yin W, Garousi J and Gräslund T: Nanoparticles targeting the intestinal Fc receptor enhance intestinal cellular trafficking of semaglutide. J Control Release 2024; 366(2023): 621–36.
- Hu M, Ge X, Chen X, Mao W, Qian X and Yuan WE: Micro/nanorobot: A promising targeted drug delivery system. Pharmaceutics 2020; 12(7): 1–18.
- Mishra A, Singh PK, Chauhan N, Roy S, Tiwari A and Gupta S: Emergence of integrated biosensing-enabled digital healthcare devices. Sensors and Diagnostics 2024; 3(5): 718–44.
- Rana N, Singh SK, Banu NA, Hjazi A, Vamanu E and Singh MP: The Ethnopharmacological properties of green-engineered metallic nanoparticles against metabolic disorders. Med 2023; 59(6).
- Ebrahimnejad P, Mohammadi Z, Babaei A, Ahmadi M, Amirkhanloo S, Asare-Addo K, Sharma P and Jain A: Magnetic nanorobots. Advanced Functional Materials 2024; 34: 2401234. DOI: 10.1002/adfm.202401234. Crit Rev Ther Drug Carrier Syst 2024; 41(1): 35–84.
- Dinesh Kuma: HKPG. Intranasal Niosomal Gel for Brain Drug Delivery: A Promising Strategy for Quetiapine Fumarate Targeting. J Neona Surg 2025; 14(10): 777–88.
- Vishnoi M, Aditya, Sindhu VL, Pallavi H, Siddiqua A and Khayum A: A Comprehensive review on edible and medicinal mushrooms diversity and multifunctionality in Global Food Systems for the Circular Bioeconomy. Microbiol Res J Int 2026; 36(1): 54–81.
- Kumar JP, Rao CMMP, Singh RK and Garg A: A brief review on encapsulation of natural poly-phenolic compounds. Adv Pharm J 2024; 9(2): 33–9.
- Singh RK, Sharma K and Gupta MP: Exploring nanotechnology with traditional herbal pharmacology: nano carrier - based phytochemical delivery in PCOS Treatment. In: Deep Science Publishing 2025; 49–78.
- Kumar PJ, Singh RK, Chennu MM Prasada Rao RT and AG: Recent developments in quercetin-loaded nanoparticles for cancer targeting. Trop J Pharm Life Sci 2025; 12(2): 11–23.
- Wang B, Zhang Y and Zhang L: Recent progress on micro- and nano-robots: Towards in-vivo tracking and localization. Quant Imaging Med Surg 2018; 8(5): 461–79.
- Li W, Wang J, Xiong Z and Li D: Micro/nanorobots for efficient removal and degradation of micro/nanoplastics. Cell Reports Phys Sci 2023; 4(11): 1–26.
- Arvidsson R and Hansen SF: Environmental and health risks of nanorobots: An early review. Environ Sci Nano 2020; 7(10): 2875–86.
- Rajendran S, Sundararajan P, Awasthi A and Rajendran S: Nanorobotics in medicine: a systematic review of advances, challenges, and future prospects with a focus on cell therapy, invasive surgery, and drug delivery. Precis Nanomedicine 2024; 7(1): 1221–32.
- How we got here : the quest for higher AI 2018;
- Yadav VK, Tahira S, Jain J, Ganesan S, Kumar R and Gupta R: Nanotechnology-Based Strategies for Glioblastoma: Diagnostic and Therapeut Adv 2026; 1–50.
- Kumar A, Shahvej SK, Yadav P, Modi U, Yadav AK and Solanki R: Clinical Applications of Targeted Nanomaterials. Pharmaceutics 2025; 17(3).
- Guleria M, Malhan A, Singh V, Singh H, Jain SK, Gupta S, Jain R and Kumar P: Controlled drug release systems using nanotechnology. Journal of Drug Targeting 2023; 31: 400–420. Curr Pharm Des 2026; 1–22. Available from: http://www.ncbi.nlm.nih.gov/pubmed/41676975
- Giri G, Maddahi Y and Zareinia K: A brief review on challenges in design and development of nanorobots for medical applications. Appl Sci 2021; 11(21).
- Sayad U, Rahman SU, Ali A, Khalil K, Ullah A and Ullah S: Nanorobotic Approaches Against Multidrug-Resistant Infections: Design, Principle, Mechanistic Innovation, Translational Challenges and Biomedical Applications Nanorobotic Approaches Against Multidrug-Resistant Infections:
- Guo L, Lan Q and Liu F: Advancements and challenges of nanorobots in surgical medicine : design, applications, and interdisciplinary integration 2026; (March): 1–21.
- Kumar JP, Singh RK, Mm C, Rao P and Tanniru R: Polymeric nano-carriers for drug delivery systems in cancer therapy - an exhaustive review. Trop J Pharm Life Sci 2025; 12(2): 33–44.
How to cite this article:
Mondal K, Kushawha S, Sharma T and Singh RK: “Nanobots and smart drug delivery: bridging the gap between nanotechnology and medicines”. Int J Pharmacognosy 2026; 13(5): 392-04. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(5).392-04.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
392-405
1778 KB
7
English
IJP
Kartik Mondal, Satpal Kushawha *, Tanya Sharma and Ranjan Kumar Singh
Faculty of Pharmaceutical Sciences, Mewar University, Gangrar, Chittorgarh, Rajasthan, India.
satpaldec1998@gmail.com
18 April 2026
03 May 2026
09 May 2026
10.13040/IJPSR.0975-8232.IJP.13(5).392-04
15 May 2026