MORPHO-ANATOMICAL AND PHYTOCHEMICAL CHARACTERISATION OF TRADITIONALLY USED PLANT RUELLIA TUBEROSA L. LEAVES AND ROOTS
HTML Full TextMORPHO-ANATOMICAL AND PHYTOCHEMICAL CHARACTERISATION OF TRADITIONALLY USED PLANT RUELLIA TUBEROSA L. LEAVES AND ROOTS
S. Dutta *, K. Hazra, S. Ghosal, D. Paria, J. Hazra and M. M. Rao
Central Ayurveda Research Institute of Drug Development, 4 CN Block, Sector-V, Bidhannagar, Calcutta - 700091, West Bengal, India.
ABSTRACT: The objective of the present study is to develop the pharmacognostic and phytochemical profiles of Ruellia tuberosa L. leaf and root. The present study considers the detail investigation of macroscopy, microscopy, preliminary qualitative phytochemical analysis, physicochemical evaluations, and chromatography profiling. The microscopical investigation was adopted to identify the cellular powder characters of leaves and roots, including diacytic stomata, few sessile glandular trichomes, profuse oil globules, exfoliating cork, few small schizogenous oil cavities, cystolith, etc. Physicochemical parameters reveal the data of quality, purity while phytochemical screening reflects the presence of various secondary metabolites. The chromatographic fingerprint data represent the authenticity of plant sample. Data obtained from botanical and chemical screening in combination may be considered as standard for identification and authentication of leaves and roots and may be helpful in developing pharmacopoeial standards.
| Keywords: |
Ruellia tuberosa, Microscopy, Cytomorphology, Physicochemical, Phytochemical, Chromatography
INTRODUCTION: Ruellia tuberosa L., (family: Acanthaceae) plant having traditional medicinal value commonly known as Blue-bell, Spearpod, Minnieroot or Snapgragon root 1. The other names of the plant, such as cracker plant or popping pod, are for dried pods that pop while rubbed with spit or contact with water 2. It is a small biennial to perennial plant identified with funnel-shaped striking violet bracteate flowers on dichotomous few-flowered cymes, 12-20 mm long linear hispid calyx lobes, 4-6 cm long corolla tube abruptly expanded above, 12-15 mm wide 2-2.5 mm in diameter purple suborbicular lobes, sessile subcylindrical puberulent capsule (fruit) 2 cm (0.8 in) long having more or less 20 seeds per locule, thick fusiform tuberous roots in cluster 3,
Erect widely branched stem up to 50 cm tall, mostly basal, finely pubescent ovate to oblong petiolate leaves with alate 1.5 cm long petioles and 4-6 × 1.5-2.5 cm pubescent leaf blades with cuneate base, obtuse apex, undulated margin.
Ruellia tuberosa is found in moist and shady environments. It is native to Central America but presently habitating in many places of tropical south and southeast Asia, preferably in grasslands and roadsides - often as a weed in cultivated fields, and also in xerophile and ruderal habitats 4.
Several studies 5, 6, 7 showed that this plant contains steroids, terpenoids, long-chain aliphatic compounds, and flavonoids. In folk medicine 8 and Ayurvedic medicine 9, it is believed to be diuretic, anti-diabetic, antipyretic, analgesic, antihypertensive, and gastroprotective. In the treatment of gonorrhea, its uses are mentioned 10. It is also used as a natural dye for textiles 11. In India, it is used for kidney stone disorders 12. Antimicrobial activity 13 of the plant leaves is reported. The present study aimed at development of standardization and authentication parameters of this plant which is not included in API.
MATERIALS AND METHODS:
Material and Reagents: The work has been carried out by using the chemicals, reagents and solvents of Emplura grade of Merck and aluminum supported Thin Layer Chromatography plates were purchased from E. Merck Pvt. Ltd. (Mumbai, India).
Plant Materials Collection and Authentication: The flowering twig, leaves, and roots of Ruellia tuberosa were freshly collected from the natural habitat of Salt Lake area, Kolkata (22°31′31.8″ N: 88°21′59.4″ E), West Bengal in the month of May 2019 and authenticated in Department of Pharmacognosy, Central Ayurveda Research Institute for Drug Development, Kolkata, a herbarium was prepared and deposited in the Department, available for reference.
Plant Sample Processing: The plant materials, leaves, and roots were washed thoroughly with aqueous ethanol, each of them dried at ambient temperature (20-23 °C). A small portion of both the fresh and air-dried plant samples was used for macroscopic, organoleptic, and microscopic (Transverse section) studies. The rest of both plant materials were pulverized separately with a grinder (National SM 2000) to obtain fine powder (sieved in 60 #) used for powder microscopy, analysis of physicochemical and phytochemical features. The coarse powder (sieved in 25 #) of both the plant materials was used for chromatographic examinations. The whole and powdered plant samples were stored at room temperature in airtight, light-resistant containers as per standard guidelines 14.
Macroscopy of Plant Material: The morpho-logical and organoleptic parameters viz. texture, shape, size, color, odor, etc. of the whole plant material were noted mainly by naked eye observation 14, 15 with the help of simple microscope Olympus OIC DM.
Powder Microscopy (Transverse Section): Leaf and root samples were separately taken and transversely cut to obtain sections using hand razor. Few fine transverse sections were selected and treated with different ethanolic gradations (30%, 50%, 70%, 90% and absolute), stained in safranin, light green, etc. and mounted on slides with Canada balsam separately for both the samples followed by observation under the binocular compound microscope (Olympus OIC-07964) at 10× and 40× magnifications 14, 15.
The photomicrographs of different cellular structures and inclusions were taken using Magcam DC14 camera attached to an Olympus CX21i trinocular compound microscope.
Powder Microscopy of Cytomorphological Features: Fine dried powdered samples i.e. leaf and root (~2 g) were separately treated with different solutions i.e., aqueous saturated chloral hydrate (for maceration), 50% glycerin, phloroglucinol in conc. HCl (for staining lignified tissues) and 0.02 N iodine reagent (for starch grains), mounted on slides with 50% glycerin following a standard protocol and observed under the binocular compound microscope (Olympus OIC-07964) at 10× and 40× magnifications 14, 15. The photomicrographs of different cellular structures and inclusions were taken using Magcam DC14 camera attached to an Olympus CX21i trinocular compound microscope.
Fluorescence Analysis: The coarsely powdered samples i.e. leaf and root (~ 0.5 g each) were treated separately with different (18 in number) reagents (5 ml each) such as, acids and alkaline solutions along with other solvents (including distilled water) inside clean test tubes, which were shaken well and allowed to stand for about 24 h. The individual solutions were observed under normal daylight and UV (254 nm and 365 nm) light for their characteristic colors and compared with the standard color chart 16.
Physicochemical Evaluation: The physico-chemical constant like ash values, loss on drying, extractive values and pH value of the plant material were determined by using coarse powder as per standard guidelines 5.
Extractability was studied with different solvents like hexane, acetone, chloroform, ethyl acetate, methanol, ethanol, water, and aqueous ethanol. Extractions were performed by conventional cold and hot extraction method 14, 15.
Phytochemical: The finely powdered plant materials both leaves and roots were subjected to Soxhlet extraction for 1 h, with methanol and extracts were evaporated to dryness and used for screening the presence of secondary metabolites 17.
Fingerprint Analysis by High-Performance Thin Layer Chromatography (HPTLC): The methanol extract of the plant materials gave the maximum extractive value; the same was used for the fingerprinting analysis. For this, the coarsely powdered plant material (1 g) was extracted with methanol (25 ml) using a Soxhlet apparatus. The extract was filtered, and the final volume made up to 20 ml using methanol and used for the fingerprinting analysis by High-Performance Thin Layer Chromatography (HPTLC).
The extract (2 μL) was applied in the form of 8 mm band, 15 mm from the bottom of a 5 × 10 cm preactivated aluminum supported precoated silica gel 60F254 plate, with the help of ATS-4 applicator attached to a CAMAG HPTLC system. The plate was developed in a pre-saturated twin trough chamber using the mobile phase as hexane: ethyl acetate: acetone: 1,4-dioxan: formic acid (4:3:2:1:0.5, v/v) to a distance of 8 cm, dried for 5 min in ambient air. Images of the developed plate were captured under 254 nm and 366 nm UV light. Densitometric scanning 18 of the developed plate at 254 nm was performed. An image was also captured using visible light after derivatizing the plate with an aqueous 20% sulphuric acid 19.
Fingerprint Analysis by High-Performance Liquid Chromatography (HPLC): This was carried out with an HPLC equipment (Agilent model Infinity 1260), equipped with quaternary LC-2010 AHT VP pumps, a variable wavelength programmable UV/VIS detector, SPD-10AVP column oven, and Class-VP software for analysis. The chromatographic separation was performed using a Phenomenex C18 (250 mm × 4.6 mm, 5 μm particle sizes) column at 25 ºC. The optimized mobile phase was found to be methanol: water (0.1% aqueous orthophosphoric acid) 80:20 (v/v) at a flow rate of 0.5 ml/min. An autosampler with injection volume 20 μl was used for sample loading and the peaks were detected at 254 nm UV.
RESULTS AND DISCUSSION:
Morphological Characters:
Leaf: The fresh Leaves are opposite, petiolate, petiole with up to 2 cm long petiole, lamina elliptic-oblong or oblong-obovate to oblanceolate, 4-7 cm long, 1.2-2.5 cm wide, nearly glabrous, dark-green, sub-acute or obtuse or rounded at apex, cuneate to attenuate at base, minutely hairy on both the sides margin nearly is smooth-edged or entire to slightly undulated. Leaves are arranged oppositely along the stem Fig. 1a, b.
Root: Fresh Roots are elongated, slender, fusiform, tuberous finger-like, thick and cylindrical, 2.5 cm. to 8 cm. long, 0.2 to 0.4 cm. thick, off white to rusty brown in color, present in clusters.
FIG. 1: MORPHOLOGY OF FRESH R. TUBEROSA WHOLE PLANT, LEAVES AND ROOTS
Microscopy (Transverse Section):
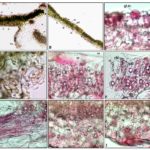
Leaf: Dorsoventral T.S through lamina shows single-layered upper and lower epidermis with thin cuticle, diacytic stomata on lower epidermis, glandular sessile trichomes and few covering trichomes, collenchymatous hypodermis, ground cortical parenchyma with prominent double-layered compact palisade and loosely arranged spongy mesophyll tissue zone with chlorophyll and oil globules; spiral xylem vessels associated with sclerenchymatous fibers present discontinuing the mesophyll tissue. In midrib, a crescent-shaped collateral vascular zone consisting of xylem strands, phloem, and sclerenchymatous fibers, etc. embedded in ground.
FIG. 2: MICROSCOPY (T.S.) OF R. TUBEROSA LEAF
a, b: T.S. through midrib and lamina showing upper (ue) and lower epidermis (le), hypodermis (hyp) , palisade (pal) and spongy (sp) parenchyma, vascular bundle (vb) and ground tissue (gr); c: the epidermis (ep) with thin cuticle (cu) showing sessile glandular trichome (gl.tr.) and mesophyll tissue (ms); d: epidermis (ep) showing diacytic stomata (st) with subsidiary cells (sc); e, f: palisade parenchymatous (pal) zone showing profuse oil globules (og) and chlorophyll (chl); g: vascular zone showing spiral xylem vessels (sp.xv.) with a tuft of fibers (fb); h, i: Spongy tissue (spg) having oil globules (og) and chlorophyll (chl) attached with lower epidermis (le).
Root: T.S shows outermost exfoliating cork cell layers arranged in parallel rows containing brownish cell content, single phelloderm layer, collapsed epidermis, collenchymatous hypodermis and ground cortical parenchyma followed by a ring shaped wavy vascular tissue zone consisting of phloem, exarch xylem stands alternating with sclerenchymatous fibers, bi to triseriate medullary ray cells encircling the wide parenchymatous pith region. Prismatic crystals of calcium oxalate, cystoliths, and few small schizogenous cavities (sz) containing oil globules are scattered in the cortex and central pith region Fig. 2b.
FIG. 3: MICROSCOPY (T.S.) OF R. TUBEROSA ROOT
a, b: Outermost exfoliating cork layers (crk) composed of cork cells arranged in parallel rows containing brownish cell content followed by single cell layered phelloderm (phl), collapsed epidermis(ep), collenchymatous (col) hypodermis and cortex (ctx) as ground tissue containing few oil containing cavity or oil glands (og); c: Cortex (ctx) and vascular zone (vs) encircling the wide pith region (pi); d, e: Cortex (ctx) with parenchymatous cells (par) containg few prismatic crystals (cr) and few oil glands (og); f, g,h,i: Vascular region showing xylem vessels (xv) of two types i.e. metaxylem (mx) and protoxylem (px), fibre (fb), medullary rays (mr), phloem (ph), very few small schizogenous cavity (sz) containing oil and pith (pi).
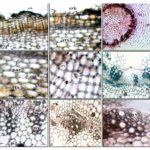
Powder Microscopy (Cytomorphological Features): Leaf: Fine powder dark green in colour with no salient taste and odour, shows the presence of groups of an epidermal cell with diacytic stomata, uniseriate nonglandular trichome, sessile and stalked glandular trichome, aseptate fibres with reticulate striations on thick fiber wall, parenchyma with dense cell contents and chlorophyll, few fragments of dark reddish-brown crystalline mass and prismatic crystals of Ca-oxalate Fig. 3a.
Root: Fine powder grayish brown to rusty brown in color with minute creamish flakes having no salient taste and odour, shows the presence of fragmented thick-walled irregular brown cork cells, aseptate fibers, spiral vessels in groups, number of Cystolith (cys) deposition inside specialized lithocyst cells associated with parenchyma, fragments of vascular tissue, prismatic crystals of Ca-oxalate, cystoliths and very few fragmented squarish to polygonal medullary ray cells Fig. 3b.
FIG. 4: POWDER MICROSCOPY OF R. TUBEROSA LEAF
a: Prismatic crystals of Ca-oxalate ; b: Uniseriate nonglandular trichome; c: Glandular trichome with oval gland and stalk; d: Groups of epidermal cell with diacytic stomata; e: Aseptate fibers; f: Reticulate striations on thick fibre wall; g: Sessile glandular trichome on an epidermal layer (longitudinal & dorsal view); h, i: Parenchymatous cells with cell contents and chlorophyll; i: Reddish-brown crystalline mass.
FIG. 5: POWDER MICROSCOPY OF RUELLIA TUBEROSA ROOT
a: Fragmented cork cells in the group; b: Aseptate fibre with tapering end; c: Spiral vessels in groups; d, e, f: Cystolith (cys) deposition inside lithocyst cells (lst); g: Squarish to polygonal medullary ray cells; h: Broken aggregated cystoliths or calcium carbonate crystals; i: Fragmented portion of vascular tissue with vessels and fibers.
Fluorescence Analysis: Dried powders of leaves and roots were separately treated with different reagents reveals the presence of chromophoric compounds in them. Lesser fluorescence was observed under normal daylight and short UV (254 nm) light for root powder, indicating a very small amount of chromophores in the sample. The fluorescence characters are noted in Table 1 and Table 2.
TABLE 1: FLUORESCENCE ANALYSIS OF R. TUBEROSA LEAF POWDER
| S. no. | Fluorescence Analysis Reagents | Visible/Day
Light |
Short UV
(254 nm) |
Long UV
(366 nm) |
| 1 | 1N HCl | Pink | Brownish with a greenish tinge | Light pinkish tinge ‘+ ve’ |
| 2 | 1N NaOH | Greenish brown | Light pinkish tinge | No color |
| 3 | 1N NaOH + Methanol | Leafy greens | Brownish tinge | Light pinkish tinge ‘+ ve’ |
| 4 | 50% KOH | Yellowish-brown | Brownish black | Blackish grey |
| 5 | 50% H2SO4 | Black | No color | Fade bluish tinge |
| 6 | Conc. H2SO4 | Black | Fade pinkish tinge | No color |
| 7 | Conc. HNO3 | Yellow straw | Fade pinkish tinge | Pale bluish tinge ‘+ ve’ |
| 8 | Acetic acid | Light green | Pinkish tinge | Bluish tinge |
| 9 | 50% HNO3 | Light pink | Fade bluish tinge | No color |
| 10 | Iodine solution | Reddish violet | No color | No color |
| 11 | Distilled water | Opaque solution | Light pale brownish tinge | Bluish tinge |
| 12 | Chloroform | Rusty green | No color | No color |
| 13 | Acetone | Green | Pale brownish tinge | Bluish tinge |
| 14 | Ammonia | Reddish-brown | Brownish tinge | Light blue |
| 15 | Ethanol | Light green | Fade blue | No color |
| 16 | Toluene | Yellowish green | No color | No color |
| 17 | K2Cr2O7 | Rusty brown | Yellowish green | Black |
| 18 | FeCl3 | Blackish brown | Brownish with greenish tinge | Pale blackish-grey |
TABLE 2: FLUORESCENCE ANALYSIS OF R. TUBEROSA ROOT POWDER
| S. no. | Fluorescence Analysis Reagents | Visible/Day
Light |
Short UV
(254 nm) |
Long UV
(366 nm) |
| 1 | 1N HCl | No color | Pale creamish with a pink tinge | White |
| 2 | 1N NaOH | No color | Pale bluish gray | Gray ‘-ve’ |
| 3 | 1N NaOH + Methanol | No color | Greyish | No color |
| 4 | 50% KOH | Pink | Brownish black | Blackish grey |
| 5 | 50% H2SO4 | Greyish | Bluish gray | Pale creamish with a gray tinge |
| 6 | Conc. H2SO4 | Black | Fade pinkish tinge | No color |
| 7 | Conc. HNO3 | Black | Fade pinkish tinge | Pale bluish tinge |
| 8 | Acetic acid | Blue | Pinkish tinge | Bluish tinge |
| 9 | 50% HNO3 | No color | Pale creamish with a yellow tinge | Bright cream |
| 10 | Iodine solution | Yellow | No color | No color |
| 11 | Distilled water | No color | White | White |
| 12 | Chloroform | No color | Light pinkish yellow | White |
| 13 | Acetone | Greyish | Bluish gray | Faint grayish |
| 14 | Ammonia | No color | Pale bluish gray | Creamy white |
| 15 | Ethanol | No color | White | Creamy white |
| 16 | Toluene | No color | No color | Pearl white |
| 17 | K2Cr2O7 | Orange | Yellowish | Dark grey |
| 18 | FeCl3 | Rust brown | Brownish | Pale grey |
Physicochemical: As demonstrated in Table 3, it was revealed that total ash, water-soluble ash, acid insoluble ash values were comparatively higher in R. tuberosa roots. The extractive values of different solvents for the plant samples revealed maximum and least extraction by hexane and methanol, respectively. Based on the best extractive yield in methanol, the same was used for the subsequent fingerprinting analyses.
TABLE 3: PHYSICO-CHEMICAL EVALUATION OF R. TUBEROSA LEAF AND ROOTa
| Physicochemical
Parameters |
Results in Percentage | |||
| Leaf | Root | |||
| Loss on drying (LOD) | 13.41 ± 0.13 | 12.34 ± 0.17 | ||
| Total ash value | 11.67 ± 0.12 | 12.33 ± 0.15 | ||
| Water soluble ash value | 7.15 ± 0.19 | 3.53 ± 0.13 | ||
| Acid insoluble ash value | 2.43 ± 0.11 | 2.34 ± 0.28 | ||
| Sulphated Ash | 2.29 ± 0.09 | 2.65 ± 0.22 | ||
| pH value (10% aq. suspension) | 6.09 ± 0.03 | 6.12 ± 0.09 | ||
| Extractive values | Cold extraction | Hot extraction | Cold extraction | Hot extraction |
| Hexane | 3.12 ± 0.13 | 3.24 ± 0.04 | 3.79 ± 0.04 | 6.23 ± 0.34 |
| Acetone | 5.38 ± 0.11 | 6.13 ± 0.08 | 8.95 ± 0.04 | 13.65 ± 0.23 |
| Chloroform | 9.35 ± 0.16 | 9.12 ± 0.12 | 15.67 ± 0.05 | 16.92 ± 0.14 |
| Ethyl acetate | 9.27 ± 0.09 | 8.76 ± 0.03 | 17.32 ± 0.32 | 17.46 ± 0.21 |
| Methanol | 8.31 ± 0.31 | 11.76 ± 0.05 | 17.71 ± 0.07 | 21.07 ± 0.13 |
| Alcohol | 7.19 ± 0.33 | 6.45 ± 0.13 | 12.61 ± 0.05 | 10.98 ± 0.21 |
| Water | 5.23 ± 0.21 | 5.94 ± 0.07 | 12.34 ± 0.03 | 6.54 ± 0.19 |
| Hydroalcoholic (1:1) | 6.37 ± 0.43 | 7.12 ± 0.04 | 11.23 ± 0.06 | 8.87 ± 0.43 |
aValues are expressed as Mean ± S.D.
Phytochemical: The results of the phytochemical screening are noted in Table 4, which reveals the presence of alkaloids, steroids, phenolics, glycosides, tannins, etc.
TABLE 4: PHYTOCHEMICAL SCREENING OF R. TUBEROSA LEAVES AND ROOTS
| Phytochemical class | Leaves | Roots |
| Alkaloids | + | + |
| Steroids | + | + |
| Triterpenoids | - | + |
| Flavonoids | + | + |
| Tannins | + | + |
| Glycoside | + | + |
| Phenolic | + | + |
| Saponins | + | + |
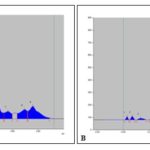
High-Performance Thin Layer Chromatography (HPTLC): The HPTLC experimental condition was optimized by using pre-activated and precoated TLC silica gel 60 F254 plates and different combinations of polar and apolar solvents as the mobile phases (data not shown). Best result was obtained with Hexane: Ethyl acetate: Methanol (5:3:2, v/v) as the mobile phase, which showed nine bands at Rf values of 0.02, 0.13, 0.24, 0.35, 0.45, 0.48, 0.57, 0.69 and 0.76 for leaves and ten bands at Rf values of 0.03, 0.07, 0.13, 0.27, 0.39, 0.49, 0.54, 0.65, 0.73 and 0.79 for roots when visualized under UV at 254 nm. Densitometric scanning at 254 nm of the developed plates gives the relative ratios of the peaks, which are noted in Table 5. Their densitometric scanned pictures as fingerprint profiles are represented in Fig. 8. The pictorial representation of the developed plate of leaves and root methanolic extracts are given in Fig. 6 and 7.
FIG. 6: HPTLC PROFILES OF R. TUBEROSA LEAVES METHANOL EXTRACT
FIG. 7: HPTLC PROFILES OF R. TUBEROSA ROOTS METHANOL EXTRACT
FIG. 8: DENSITOGRAM DISPLAY OF HPTLC PROFILES OF R. TUBEROSA METHANOL EXTRACT OF (A) LEAVES AND (B) ROOTS AT 254 nm
TABLE 5: RELATIVE RATIOS OF THE HPTLC PEAKSa OF R. TUBEROSA LEAVES AND ROOTS
| Leaves | Roots | ||
| Rf values | Relative ratio (%) | Rf values | Relative ratio (%) |
| 0.02 | 3.99 | 0.03 | 0.54 |
| 0.13 | 0.52 | 0.07 | 0.77 |
| 0.24 | 3.36 | 0.13 | 0.70 |
| 0.35 | 0.55 | 0.27 | 1.48 |
| 0.45 | 7.76 | 0.39 | 12.76 |
| 0.48 | 0.51 | 0.49 | 30.02 |
| 0.57 | 6.76 | 0.54 | 18.29 |
| 0.69 | 9.51 | 0.65 | 10.76 |
| 0.76 | 7.05 | 0.73 | 5.30 |
| - | - | 0.79 | 19.37 |
aThe peaks were recorded by visualizing the chromatogram spots at 254 nm
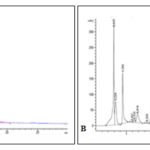
High-Performance Liquid Chromatography (HPLC): HPLC method was developed for the best separation of the chemical constituents of the R. tuberosa leaves and roots methanol extracts. Separated peaks were detected under UV (254 nm). The HPLC fingerprint analysis in Fig. 9.
It shows ten peaks for leaves and eight peaks for roots, and their corresponding retention times with areas under the curves are noted in Table 6 and Table 7.
FIG. 9: HPLC CHROMATOGRAM OF (A) LEAVES AND (B) ROOTS METHANOLIC EXTRACT
TABLE 6: RELATIVE RATIOS OF THE HPLC PEAKSa R. TUBEROSA LEAVES
| Peak Retention time (Minute) | Relative ratio (%) |
| 2.546 | 0.46 |
| 3.831 | 6.3 |
| 3.933 | 5.52 |
| 4.354 | 2.80 |
| 6.139 | 0.86 |
| 7.428 | 1.12 |
| 8.630 | 19.07 |
| 12.539 | 22.77 |
| 13.168 | 28.91 |
| 17.861 | 12.01 |
TABLE 7: RELATIVE RATIOS OF THE HPLC PEAKSa R. TUBEROSA ROOTS
| Peak Retention time (Minute) | Relative ratio (%) |
| 3.224 | 25.40 |
| 4.390 | 32.70 |
| 5.931 | 0.71 |
| 6.310 | 2.47 |
| 6.819 | 13.13 |
| 8.329 | 1.35 |
| 9.29 | 16.94 |
| 10.967 | 7.57 |
aThe peaks were recorded by detecting the chromatogram at 254 nm
CONCLUSION: The present investigations furnished a set of qualitative and quantitative phytopharmacognostic characters along with the HPLC fingerprinting profile of R. tuberosa leaf and roots. These data can serve as diagnostic tools for the establishment of quality standards, authentication, and identification of the medicinally important plant and help in compiling a suitable monograph of this.
ACKNOWLEDGEMENT: Authors are thankful to the Director General for providing continuous support and encouragement
CONFLICTS OF INTEREST: Nil
REFERENCES:
- Martin FW, Ruberte RM and Meitzner LS: Edible leaves of the tropics. Echo Publisher, First Edition 1998.
- Standley PC and Steyermark JA: Flora of Guatemala. McMillan, First edition 1976.
- Mors WB, Rizzini CT and Pereira NA: Medicinal Plants of Brazil. Reference Publications, First Edition 2000.
- Huxley A: The New RHS Dictionary of Gardening. MacMillan, 1992.
- Samy MN, Khalil HE, Wanas AS, Kamel MS, Sugimoto S and Matsunami K: Screening of Medicinal properties of selected Plants. Chemistry of Natural Compound 2013; 49: 175-6.
- Phakeovilay C, Disadee W, Sahakitpichan P, Sitthimonchai S, Kittakoop P, Ruchirawat S and Kanchanapoom T: Chemical constituents of Ruellia tuberosa J of Natural Medicine 2013; 67: 228-33.
- Suriyavathana M and Sivanarayan V: Studies of selected medicinal plants of southern plateau. International Journal of Pharma Science Review and Research 2013; 18: 72-76.
- For cafe (www.saintlucianplants.com) Retrieved 16 March 2018.
- Panda H: Handbook on Ayurvedic Medicines with formulae processes and their uses. National Institute of Industrial Research, First Edition 2002.
- Lans CA: Ethnomedicine as used in Trinidad and Tobago for urinary problems and diabetes mellitus. J Ethnobiol Ethnomed 2006; 2: 45-49.
- Effect of Chitosan and Mordants on Dyeability of Cotton Fabrics with Ruellia tuberosa Linn" (PDF). cmu.ac.th. Retrieved 16 March 2018.[permanent dead link].
- Supawana K, Supawatchara S, Thanapat S and Churdsak J: Phytochemical study of Ruellia tuberosa chloroform extract: antioxidant and anticholinesterase activities. Der Pharmacia Lettre 2016; 8: 238-44.
- Arirudran B, Saraswath A and Vijayalakshmi K: Antimicrobial activity of Ruellia tuberosa (Whole Plant). Pharmacognosy Journal 2011; 3: 91-5.
- Anonymous: Quality control methods for herbal materials. World Health Organization, First Edition 2011.
- Anonymous: The Ayurvedic Pharmacopoeia of India. Department of Health and Family Welfare, Govt. of India, First Edition 1992.
- Chase CR and Pratt R: Fluorescence of powdered vegetable drugs with particular reference to development of a system of identification. J Am Pharm Assoc Am Pharm Assoc 1949; 38: 324-31.
- Harpreet S, Amrita M and Arun KM: Pharmacognostical and physicochemical analysis of Cleome viscosa seeds. Pharmacognosy Journal 2017; 9: 372-7.
- Reich E and Schibili A: High Performance Thin Layer Chromatography. Thieme Publication, First Edition 2006.
- Stahl E: Thin Layer Chromatography, A Laboratory handbook. Springer (I) Pvt. Ltd., Second Edition 2005.
How to cite this article:
Dutta S, Hazra K, Ghosal S, Paria D, Hazra J and Rao MM: Morpho-anatomical and phytochemical characterisation of traditionally used plant Ruellia tuberosa L. leaves and roots. Int J Pharmacognosy 2019; 7(1): 12-22. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232. IJP.7(1).12-22.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
12-22
889
1104
English
IJP
S. Dutta *, K. Hazra, S. Ghosal, D. Paria, J. Hazra and M. M. Rao
Department of Pharmacognosy, Central Ayurveda Research Institute for Drug Development, Kolkata, West Bengal, India.
mailtosreya.27@gmail.com
21 December 2019
25 January 2020
29 January 2020
10.13040/IJPSR.0975-8232.IJP.7(1).12-22
31 January 2020