MEDICINAL PLANTS SUPPORT THE AMYLIN-SUPPRESSED VIABILITY OF ISLET β-CELLS
HTML Full TextMEDICINAL PLANTS SUPPORT THE AMYLIN-SUPPRESSED VIABILITY OF ISLET Β-CELLS
S. G. Sharoyan *, A. A. Antonyan, H. A. Harutyunyan and S. S. Mardanyan
H. Buniatyan Institute of Biochemistry of Armenian National Academy of Sciences, 5/1 P. Sevak Str., Yerevan 0014, Republic of Armenia.
ABSTRACT: The proliferation of β-cells is the main contributor to the mass cell maintenance in the pancreatic islets. In our earlier work the suppression of β-cell growth by aggregated peptide hormone of the pancreas, amylin, has been proved. The in-vitro protection of the cells from aggregated amylin-associated death by several plant extracts and their constituents has been shown. In the present work, the development of β-cells was suppressed preliminary by aggregated amylin. Then, the ethanol extracts of rose petals (Rosa damascena), melilot (Melilotus officinalis), leaves of grape (Vitis vinifera) and sorrel (Rumex Confertus) and the phenol glycoside fraction of rose petals were added to the cultivation medium, and the growth of cells suppressed by aggregate amylin was observed. The promotion of cells viability was registered with the trypan blue exclusion test and the DNA-comet analysis. In the other series of experiments, the reverse of pre-formed amylin aggregates in the presence of several plant preparations was registered using Thioflavin T fluorescence. The significant linear correlation (r = 0.75, p=0.02) between two propensities of plant: to inhibit amylin aggregation (observed in our previous work) and to dissociate the peptide aggregates was observed. Probably, both of these properties of plant preparations are conditioned by their specific interaction with amylin molecule and elucidate the perspective of studied plants in the protection of pancreatic islet β-cells against amylin-induced lost.
| Keywords: |
Amylin, Dissociation of fibrils, DNA Comet, Islet β-cells, Plant constituents, Plant extracts
INTRODUCTION: Pancreatic islets consist of 60-80% beta cells, which secrete insulin, a hormone of profound importance in the regulation of carbohydrate, fat and protein metabolism. Beta cells death and dysfunction result in an insufficient amount of insulin that leads to high glucose levels in the blood, a metabolic disorder known as Diabetes mellitus (DM). The increase in beta cell mass can take place either through an increase in the cell number by neogenesis and proliferation (hyperplasia), or through an increase in the cell volume (hypertrophy) 1.
The primary mechanism of beta cell mass expansion in the postnatal period is replication 2, 3, 4. An essential source of new pancreatic beta cells in adult mice is shown being the proliferation of preexisting cells 2, 5, 6. Many studies aiming to establish new therapeutic applications for DM are targeted at understanding and regulating the beta cells replication as a significant contributor to the increase of adult beta-cell mass 7. It is crucial to find the factors maintaining cell proliferation. This knowledge could lead to the development of new methods to increase the cell mass in DM.
Amylin is a secretory product of pancreatic β-cells 8, 9. It is a regulatory peptide functioning in the islet β-cells and participating in the regulation of glucose metabolism along with insulin. Amyloid deposits of amylin were found in the islets of β-cells in DM in humans and some mammals. Their cytotoxicity is one of the causes of β-cell loss in type 2 DM 10. There are many publications evidencing the perspective of natural compounds like flavonoids, polyphenols, etc. in the prevention of amylin amyloidosis 11-14, considering them as effective remedies for β-cells maintenance and DM treatment.
Armenia has a rich history in the field of phytotherapy. Herbal medicine developed thanks to medieval scholars Mkhitar Heratsi, Amirdovlat Amasiatsi, Grigor Magistros and many others 15, 16. Many medicinal plants have been disseminated from here throughout Europe. Some edible plants widespread in Armenia are used in folk medicine. Earlier we have demonstrated the effective inhibition of dipeptidyl peptidase IV and adenosine deaminase activities, elevated in diabetes, by several aqueous extracts of Armenian Highland plants 17. In our next work, the cytotoxicity of aggregated amylin to pancreatic islet β-cells has been proved 18.
Then, the in vitro protection of cell viability by ethanol extracts of leaves of sorrel, sea buckthorn, grape, bryonia white roots, thistle seeds, melilot, rose petals, as well as some of their fractions, has been studied in the presence and absence of aggregated amylin. We have also observed the in-vitro inhibition of amylin aggregation by several plant preparations 18. The obtained positive effects allowed us recommending some of these plants and their constituents as probable sources for developing antidiabetic therapeutics. In this work, the ability of ethanol extracts of rose petals (Rosa damascena), melilot (Melilotus officinalis), leaves of grape (Vitis vinifera and sorrel (Rumex Confertus) and the phenol glycoside fraction of rose petals to destabilize the conformation of pre-aggregated amylin was observed. The same plant preparations were added to the cultivation medium of β-cells after a two-day suppression of their growth by aggregated amylin. The DNA-comet analysis after the following cultivation during 2 days manifested the protection of DNA against amylin-induced damage.
MATERIAL AND METHODS:
General: Thioflavin T (ThT), RPMI-1640 and supplements were purchased from Sigma Ltd, USA; amylin – from ‘‘Gene Cust’’, Luxembourg. All the other chemicals were of the highest purity.
Spectral measurements were performed on Specord M-40 UV-VIS spectrophotometer (Germany) and spectrofluorometer Perkin-Elmer MPF-44A (USA), using quartz cuvettes with light path 0.5 and 1cm. The light and fluorescence microscopes model BH- 2RFCA, Olympus, with a digital camera, model c35AD-4, Olympus was used.
Plant Material: The collection and the ethanol extract preparation of GL, RP, SL and M, their fractionation and characterization were done as described in our previous works 18, 19. A voucher specimen has been deposited in the herbarium of the Botanical Department of Yerevan State University (Dr. Narine Zaqaryan).
Amylin Preparation: The preparation of amylin solution, the formation of its aggregates and the evaluation of the aggregation state of the peptide, using the fluorescence (λex = 430 nm and λem = 485 nm) were described previously 18. The dissociation of pre-aggregated amylin was studied incubating it in 20 mM phosphate buffer, pH 7.4, containing 0.02% NaN2 (w/v) for 3 days with (or without) the plant preparation at definite concentration.
Islet Cells Isolation and Cultivation: The isolation, cultivation of mouse pancreatic β-cells in the presence and absence of aggregated amylin, as well as the study of the influence of plant preparations on the viability of cells are described in our previous work 18. The experiments involving the laboratory animals were approved by Ethics Committee of Yerevan State Medical University after M. Heratsi, No 7-26.04.2012: Research is not contrary to the Directive 2001/20/EC of The Legal Aspects of Research Ethics and Science in European Community.
DNA Comet Assay: DNA Comet assay was used for qualifying the genotoxicity of aggregated amylin and the ability of plant preparations to protect the DNA integrity in its presence. Alkaline Comet assay was conducted as described by Venturi et al. 20 with minor modifications. The cell suspension was centrifuged at 100 ´ g for 3 min, the obtained cell pellets were re-suspended in 75 μl of 0.5% low melting point agarose in PBS and embedded on microscope slides. Cells were lysed for 2 h at 4 °C in solution containing 2.5 M NaCl; 100 mM Na2EDTA; 10 mM TRIS; 1% Triton X-100, and the slides were placed in a horizontal gel electrophoresis tank, containing fresh electrophoresis solution (1 mM Na2EDTA, 300 mM NaOH, pH > 13). The DNA was allowed to unwind for 20 min, and the electrophoresis was conducted for 30 min at 26 V, 300 mA.
Then, the slides were washed in 0.4 M TRIS, pH 7.5 neutralization buffer, stained with ethidium bromide (2 μg/ml) and analyzed immediately at ´80 magnification using a fluorescence microscope. One hundred randomly selected cells from each slide were scored using the Comet Assay Software Project (CASP, http://www.casp.sourceforge.net) image analysis software. The next parameters: tail length (TL; µm), tail DNA (TD; %) and Extent tail moment (TM; arbitrary units) were used as a metrics of DNA damage. Tail moment was defined as: TM = TL´TD/100 21.
Statistical Analysis: The data, obtained at least in three independent experiments, were analyzed using the software In-Stat, version 3 for Windows (Graph Pad Software, Inc., San Diego, CA, USA). The results were expressed as means ± SE.
RESULTS AND DISCUSSION:
Maintenance of Suppressed Viability of β-cells: In our recent work, the higher toxicity of aggregated amylin compared with its fresh solution to the primary culture of pancreatic islet β-cells has been proved. Then, the in-vitro protection of the cell viability by ethanol extracts of leaves of sorrel and grape, Bryonia white roots, thistle seeds, melilot, rose petals, as well as some of their fractions in the presence of aggregated amylin, has been observed 18. Moreover, several plant preparations have been provided with the ability to inhibit the in vitro aggregation of amylin, and the high correlation between these two propensities of plant preparations has been demonstrated.
In the present work, the experiments were implemented to observe the ability of plant preparations to support the development of β-cells, primarily suppressed by aggregated amylin.
Two cell samples were prepared. The first, in a usual cultivation media, was used as a control. The second sample contained 8 µM aggregated amylin. After cultivation for two days, the second sample (viability suppressed down to 43.9 ± 9.5% of control) was divided into six parts and into five of them, RP PhG fraction and the extracts of SL, GL, RP, and M were added up to the final concentration 200 µg/ml of the dried plant material. Preliminary, we have shown these four extracts as being reach in flavonoid compounds 19, known as antioxidants 22 provided by significant therapeutic value 23. After the addition of indicated plant preparations, the cultivation was continued for the next two days in the seven samples: the control, the part of the amylin-containing sample without plant preparations and five identical to the last one containing the added plant preparations.
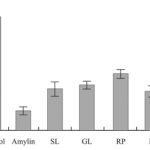
FIG. 1: ALIVE CELLS AFTER CULTIVATION IN THE ABSENCE AND PRESENCE OF PLANT PREPARATIONS
In Fig. 1, the averaged results of three independent experiments are shown. The results are expressed as a percentage of the number of living cells in the control sample. Four-Day cultivation resulted in the decreasing of the amylin-containing sample down to on average 17.1 ± 3.6 % of the control sample. In the identical samples, cultivated during the last two days with the plant preparations along with the amylin, the cell counts were 34-50% of the control. The presence of plant preparations in the aggregate amylin-containing medium preserved the viability of cells in the level 2-3 times higher than that in the sample containing only amylin. The one-tailed P values of the difference between the results for plant preparation containing samples from that of amylin sample, estimated in the unpaired t-test, were: 0.056 (SL), 0.005 (GL), 0.001 (RP), 0.066 (M), 0.012 (RP PhG). In general, they might be considered as rather significant.
DNA-Comet: DNA-comet analysis of islet β-beta cells was performed after the above-described experiment.
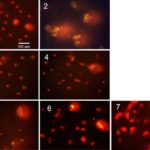
FIG. 2: DNA-COMET PICTURES FOR β-CELLS, CULTIVATED DURING 4 DAYS: 1 - CONTROL, WITHOUT ANY ADDITIONS, 2-7 – THE CULTIVATION MEDIUM CONTAINED AGGREGATED AMYLIN (8µM); 3-7 – IN THE LAST 2 DAYS, THE CULTIVATION MEDIUM BESIDES AMYLIN CONTAINED, RESPECTIVELY, SL, GL, RP, M EXTRACTS AND THE RP PHG FRACTION
In Fig. 2, the alkaline DNA-comet pictures for β-cells cultivated for 4 days without any additions (1, control) and in the presence of 8 µM aggregated amylin (2) are shown. The pictures 3-7 represent the β-cell samples identical to sample 2, cultivated during last 2 days in the presence of, respectively, the ethanol extracts of SL (3), GL (4), RP (5) and M (6) and the RP PhG fraction (7). The final concentration of all plant preparations were 200 µg/ml.
The results of DNA-comet were analyzed using CASP image analysis software. The defined parameters are listed in Table 1. All three parameters: Tail length TL, µm, Tail DNA TD, % and Extent tail moment TM, a.u. for sample 2 are very significantly (P<0.001) higher than those for the control sample. These results evidence the cell DNA damage and prove the genotoxicity of aggregated amylin towards β-cells.
TABLE 1: THE PARAMETERS OF DNA-COMET ANALYSIS OF β-CELLS
| Sample | Statistic | TL, µm | TD, % | TM, a.u. |
| Control | aM | 7±1.29 | 9.95±3.2 | 1.01±0.38 |
| amylin | aM | 64.4±7.11 | 51±5.6 | 31.5±4.5 |
| bP1 | P<0.001 | P<0.001 | P<0.001 | |
| SL | aM | 13.6± 1.34 | 25.44± 3.2 | 3.78± 0.67 |
| bP1 | P>0.05 | P<0.01 | P>0.05 | |
| cP2 | P<0.001 | P<0.01 | P<0.001 | |
| GL | aM | 8.1±1.8 | 11.54±2.4 | 1.25±0.58 |
| bP1 | P>0.05 | P>0.05 | P>0.05 | |
| cP2 | P<0.001 | P<0.001 | P<0.001 | |
| RP | aM | 11.3±2.44 | 17.45±4.4 | 2.45±0.9 |
| bP1 | P>0.05 | P>0.05 | P>0.05 | |
| cP2 | P<0.001 | P<0.001 | P<0.001 | |
| M | aM | 11.8±1.4 | 11.84±1.9 | 1.49±0.35 |
| bP1 | P>0.05 | P>0.05 | P>0.05 | |
| cP2 | P<0.001 | P<0.001 | P<0.001 | |
| RpPhG | aM | 20.5±3.6 | 24.94±6.1 | 7.04±2.5 |
| bP1 | P > 0.05 | P > 0.05 | P>0.05 | |
| cP2 | P < 0.001 | P< 0.05 | P < 0.05 |
aM – mean values, bP1 - the statistical significance of difference from control sample 1
cP2 - the statistical significance of difference from aggregated amylin containing sample 2
The most of the parameters for the samples 3-7 differ from those for control not significantly (P>0.05), but rather significantly from those for sample 2 (mainly P<0.001, at least P<0.05). These results confirm the ability of the used plant preparations to facilitate the integrity of DNA, damaged in the presence of aggregated amylin.
Disaggregation: As one of the possible ways for protection by plant preparations of amylin-suppressed β-cell viability could be considered their ability to destabilize and dissociate the peptide aggregates. Using the Thioflavin T fluorescence test, we studied the influence of several plant preparations on the conformation state of preliminary aggregated amylin. As in the case of amylin aggregation research, described in our previous work 18, this study was limited by fluorescence properties of plant preparations too. Some of preparations quenched the ThT 430/485 fluorescence; the others were provided with their fluorescence in this spectral region. Therefore, taking into account the results of the preliminary screening, in the study of disaggregation of preformed amylin aggregates only the weakly affecting preparations/ concentrations were used. The preliminary aggregated amylin was incubated for 3 days in the presence (or absence, control) of the plant preparation in definite concentration, as described in the materials and methods.
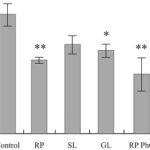
FIG. 3: THE DECREASE OF AMYLIN AGGREGATION STATE UPON INCUBATION OF PRELIMINARY AGGREGATED PEPTIDE IN THE PRESENCE OF PLANT PREPARATIONS
In Fig. 3 the aggregation states of pre-aggregated amylin samples are presented after incubation during 3 days in the presence of the extracts of RP, SL, and GL, the fractions enriched by phenol glycosides from rose petals (RP PhG) and by flavonoids from sorrel leaves (SL Fl). The aggregation state of each sample is expressed as a percentage of the pre-aggregated amylin incubated at the same conditions without any plant preparation (100%). The concentration of all plant preparations in the incubation medium was 7.5µg/ml. The highest degree of the aggregation decrease was registered in the presence of the extract of RP (p<0.01), and the fractions RP PhG (p<0.01) and SL Fl (p<0.05).
The low disaggregation of aggregated amylin was registered in the presence of M extract, coumarin fractions from SL and RP (not shown). Let’s note that only very low concentrations of these preparations were studied due to their fluorescence limitations.
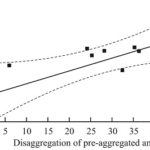
Correlation of the Ability of Plant Preparations to Dissociate the Preformed Aggregates of Amylin with their Capability to Inhibit the Peptide amyloidosis: In our previous work, the in-vitro inhibition of amylin aggregation by several plant preparations has been described 18. Moreover, the comparison of IC50 values in the inhibition of amylin aggregation and the protection of β-cells revealed the high linear correlation between IC50 values of five preparations: extracts of GL and SL, and SL Fl, RP PhG and RP Cm1 fractions (r = 0.9945; p=0.0005). We assumed that these two abilities of each plant preparation – to protect β-cells from aggregated amylin and to prevent aggregation of the peptide, are the interdependent properties. In the present work, we compared the early studied ability of plant preparations to inhibit the process of the peptide aggregation 18 with the observed in the current research their ability to reverse the preformed aggregates of amylin. We performed the correlation analysis between these two features for the GL and SL extracts, and the SL Cm1, RP Cm1, SL AD, SL Fl, RP PhG, RP PhG fractions under the identical concentrations. In Fig. 4 the obtained linear correlation curve is shown.
FIG. 4: THE LINEAR CORRELATION CURVE BETWEEN THE ABILITIES OF PLANT PREPARATIONS TO REVERSE THE PRE-AGGREGATED AMYLIN AND TO INHIBIT ITS AGGREGATION; THE VALUES ARE EXPRESSED AS PERCENTAGE OF CONTROLS
The evaluated coefficient r = 0.75 (p = 0.020) manifests a rather significant correlation between the compared features of the studied plant preparations. Taking into account the demonstrated in the previous work high correlation between the abilities to protect β-cells from the toxicity of aggregated amylin and to inhibit the amylin aggregation, we can conclude the interdependence of three propensities of plant preparations: to inhibit the amylin aggregation, to destabilize the pre-formed aggregates of the peptide, and to protect the β-cells from toxic action of aggregated amylin.
CONCLUSION: The ethanol extracts of rose petals (Rosa damascena), melilot (Melilotus officinalis), leaves of grape (Vitis vinifera) and sorrel (Rumex confertus) and the phenol glycoside fraction from the ethanol extract of rose petals supported in-vitro the aggregate amylin suppressed the growth of islet β-cells.
The DNA-comet analysis proved the genotoxicity of aggregated amylin to β-cells and demonstrated the protection by plant preparations of DNA integrity against the aggregated peptide. The most of studied plant preparations destabilized the pre-formed cytotoxic aggregates of amylin in-vitro. A significant correlation (r = 0.75, p=0.02) was observed between the abilities of plant preparations to dissociate the preformed aggregates of amylin and to inhibit the peptide aggregation. Probably, three properties of plant preparations: to protect β-cells against killing by aggregated amylin, to inhibit the peptide aggregation and to dissociate its preformed aggregates are conditioned by specific interaction between amylin molecule and plant constituents: phenol glycosides, flavonoids, coumarins, etc. These specific interactions may be considered as mechanisms of promotion of β-cells development assuming that in these processes the direct action of plant constituents on the amylin molecule is responsible.
The obtained results allow recommending the use in the diet by persons at risk group the plants with shown effectiveness. Besides, the widely utilized in the world rose petals and melilot, and the user in the Armenian (oriental) cuisine leaves of grape and sorrel could be considered as sources for antidiabetic agents directed to increase in beta cell mass.
ACKNOWLEDGEMENT: This work was supported by State Committee of Science of Armenian Ministry of Education and Science, in the frame of the research projects № SCS «11-1f105» and «13-1F186».
CONFLICT OF INTEREST: The authors have declared that there is no conflict of interest.
REFERENCES:
- Ballian N, Hu M, Liu SH and Brunicardi FC: Proliferation,hyperplasia, neogenesis, and neoplasia in the islets of Langerhans. Pancreas 2007; 35: 199-206.
- Georgia S and Bhushan A: Beta-cell replication is the primary mechanism for maintaining postnatal beta cell mass. J Clin Invest 2000; 4114: 963-968.
- Dor Y, Brown J, Martinez OI and Melton DA: Adult pancreatic beta-cells are formed by self-duplication rather than stem-cell differentiation. Nature 2004; 429: 41-46.
- Bouwens L and Rooman I: Regulation of pancreatic beta-cell mass. Physiol Rev 2005; 85: 1255-1270.
- Brennand K, Huangfu D and Melton D: All beta cells contribute equally to islet growth and maintenance. PLoSBiol 2007; 5: e163.
- Teta M, Rankin MM, Long SY, Stein GM and Kushner JA: Growth and regeneration of adult beta cells do not involve specialized progenitors. Dev Cell 2007; 12: 817-826.
- Yesil P and Lammert E: Review, Islet dynamics: A glimpse at beta cell proliferation. Histol Histopat 2008; 23: 883-895.
- Westermark P, Wernstedt C, Wilander E, Hayden DW, O’Brien TD and Johnson KH: Amyloid fibrils in human insulinoma and islets of Langerhans of the diabetic cat are derived from a neuropeptide-like protein also present in normal islet cells. Proceedings of National Academy of Sciences USA 1987; 84: 3881-3885.
- Cooper GJS, Leighton B, Dimitriadis GD, Parry-Billings M, Kowalchuk JM, Howland K, Rothbard JB, Willis AC and Reid KBM: Amylin found in amyloid deposits in human type 2 diabetes mellitus may be a hormone that regulates glycogen metabolism in skeletal muscle. Proceedings of National Academy of Sciences USA 1988; 85: 7763-7767.
- Westermark P, Andersson A and Westermark GT: Islet amyloid polypeptide, islet amyloid, and diabetes mellitus. Physiological Reviews 2011; 91: 795–826.
- Aarabi MH and Mirhashemi SM: The role of two natural flavonoids on human amylin aggregation. African Journal of Pharmacy and Pharmacology 2012; 6: 2374-2379.
- Meng F, Abedini A, Plesner A, Verchere CB and Raleigh DP: The Flavanol (−)-epigallocatechin 3-gallate inhibits amyloid formation by islet amyloid polypeptide, disaggregates amyloid fibrils and protects cultured cells against IAPP induced toxicity. Biochemistry 2010; 49: 8127-8133.
- Mirhashemi SM and Aarabi MH: Effect of two herbal polyphenol compounds on human amylin amyloid formation and destabilization. Journal of Medicinal Plants Research 2012; 6: 3207-3212.
- Cheng B,Gong H, Li X, Sun Y, Chen H, Zhang X, Wu Q, Zheng L and Huang K: Salvianolic acid B inhibits the amyloid formation of human islet amyloid polypeptide and protects pancreatic beta-cells against cytotoxicity. Proteins 2013; 81: 613-621.
- Torosian A. The Armenian medical herbs. Hayastan, Yerevan, Armenia 2006.
- Vardanian SA. Histiore de la médicineen Arménie.del’antiquité á nosjours. Imprimé par L’ Europenne d’Edition, Paris
- Mardanyan S, Sharoyan S, Antonyan A and Zakaryan N: Dipeptidyl peptidase IV and adenosine deaminase inhibition by Armenian plants and antidiabetic drugs. International Journal of Diabetes and Metabolism 2011; 19: 69-74.
- Sharoyan SG, Antonyan AA, Harutyunyan HA and Mardanyan SS: Inhibition of amylin fibril formation and protection of Islet β-Cells by medicinal plants. Int. J Pharmacognosy 2015; 2: 234-241.
- Antonyan A, Sharoyan S, Harutyunyan H, Movsisyan N, Sargisova Y, Stepanyan H, and Mardanyan S: Cytotoxicity of some edible plants toward ehrlich ascites carcinoma cells. Research J of Med Plant 2014; 8: 20-31.
- Venturi M, Hambly RJ, Glinghammar B, Rafter JJ and Rowland IR: Genotoxic activity in human fecal water and the role of bile acids: a study using the alkaline comet assay. Carcinogenesis 1997; 18: 2353-2359.
- Marczynski B, Rihs HP, Rossbach B, Hölzer J, Angerer J, Scherenberg M, Hoffmann G, Brüning T and Wilhelm M: Analysis of 8-oxo-7,8-dihidro-2'-deoxyguanosine and DNA strand breaks in white blood cells of occupationally exposed workers: comparison with ambient monitoring, urinary metabolites and enzyme polymorphisms. Carcinogenesis 2002; 23: 273-281.
- Beecher GR: Overview of dietary flavonoids: nomenclature, occurrence and J Nutr 2003; 133: 3248S-3254S.
- Baptista FI, Henriques AG, Silva AM, Wiltfang J and da Cruz e Silva OA: Flavonoids as therapeutic compounds targeting key proteins involved in Alzheimer’s disease. ACS Chem. Neurosci 2014; 5: 83-92.
How to cite this article:
Sharoyan SG, Antonyan AA, Harutyunyan HA and Mardanyan SS: Medicinal plants support the amylin-suppressed viability of islet β-cells. Int J Pharmacognosy 2015; 2(9): 448-53. doi: 10.13040/IJPSR.0975-8232.2(9).448-53.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
5
448-453
773
1528
English
IJP
S. G. Sharoyan *, A. A. Antonyan, H. A. Harutyunyan and S. S. Mardanyan
H. Buniatyan Institute of Biochemistry of Armenian National Academy of Sciences, Republic of Armenia.
biochem@ipia.sci.am
03 August 2015
03 September 2015
17 September 2015
biochem@ipia.sci.am
30 September 2015