LC/MS IDENTIFICATION OF PHYTOCONSTITUENTS OF THE METHANOLIC EXTRACT OF THE AERIAL PARTS OF ANTIGONON LEPTOPUS (HOOK & ARN)
HTML Full TextLC/MS IDENTIFICATION OF PHYTOCONSTITUENTS OF THE METHANOLIC EXTRACT OF THE AERIAL PARTS OF ANTIGONON LEPTOPUS (HOOK & ARN)
Nelly Ashraf Ramadan
Giza Opthalmology Hospital, Giza, Egypt.
ABSTRACT: Antigonon leptopus Hook & Arn (family Polygonaceae), also called: Chain of love, Queen’sWreath, and Mexican creeper is a fast-growing vine with stems reaching up to 20 feet long. It has heart-shaped, green leaves climbing by tendrils which wrap around many types of supports. It is native to Mexico and commonly found in tropical Asia, Africa, the Caribbean, and the Americas. It possesses anticoagulant activity, analgesic, anti-thrombin, anti-inflammatory, anti-diabetic and antidepressant activities. Aerial parts of the vine have been used as hepatoprotective and for spleen disorders. During the present study, LC/MS experiment was conducted on the methanolic extract of the aerial parts of the vine. Results revealed the identification of total fifty compounds; flavonoids, phenolics, anthocyanins, and glycosides. Compounds were characterized by their retention times, mass spectra with comparison to online mass bank database. From the results, Antigonon leptopus can be incorporated in pharmaceutical supplements used to aid free radical-mediated disease; cancer, diabetes, inflammation and liver diseases.
| Keywords: |
Antigonon leptopus Hook and Arn, Vine, Polygonaceae, LC/MS, Flavonoids, Phenolics, Hepatoprotective, Anti-diabetic, Anti-inflammatory
INTRODUCTION: Despite the tremendous progress in the development of new synthetic drugs during the 20th century, plant-based drugs never totally lost their importance in the treatment of several ailments. Antigonon leptopus, is one of the species of belonging to the buckwheat family, Polygonaceae. It is native to Mexico, India and commonly found in tropical Asia, Africa, the Caribbean, and the Americas. It is known as the coral vine or Mexican creeper. It is a fast-growing vine with the stems reaching 20 feet long. It climbs by tendrils which wrap around many types of supports.
Long sprays of showy pink flowers appear in the summer and fall rapidly. Hot prepared tea from the aerial portion of A. leptopus is used for the prevention and treatment of cough, sore throat, flu, and menstrual pains. The flowers are used to treat high blood pressure. The leaves possess anticoagulant, analgesic, anti-thrombin, anti-inflammatory, and anti-diabetic activities. Also, they have been used as hepatoprotective, treating asthma, liver and spleen disorders 1-9. There is no such previous study on the exploration of bioactive compounds using LC/MS (Liquid Chromatography -Mass Spectrometry) of the methanolic extract of the aerial parts of the vine.
MATERIALS AND METHODS:
Plant Collection: Samples of Antigonon leptopus used in this study were collected from “El-Zohreya” Park, Cairo, Egypt during the flowering stage (Spring 2012).
A flowering branch was kindly authenticated by Mrs. Terase Labib, plant taxonomist of Orman Garden, Giza, Egypt. The voucher specimen has been deposited in the herbarium of Department of Pharmacognosy, Faculty of Pharmacy, Cairo University “20.12. 15.1”.
Preparation of the Methanolic Extract: The air-dried powder of the aerial parts of A. leptopus (400 gm) was extracted using methanol 70 % with percolation (cold extraction technique) till exhaustion (4 × 4 L). The solvent was evaporated under reduced pressure at 60 ˚C, using a rotary evaporator and the residue was used for successive liquid-liquid fractionation. Total weight of the dried methanol residue was 60 g (15% w:w). The air-dried methanolic extract (60 g) was suspended in distilled water (100 ml) and subjected to liquid-liquid fractionation with solvents of increasing polarity; petroleum ether (5 × 500 ml), methylene chloride (4 × 500 ml), ethyl acetate (6 × 500 ml) and n-butanol (3 × 500 ml) respectively. The solvents were evaporated in each case under reduced pressure at 45 °C using rotary evaporator. The dried solvent-free extractives were weighted.
Preliminary Phytochemical Screening: The methanolic extract was tested for carbohydrates, tannins, flavonoids, saponins, sterols, alkaloids, anthraquinones and cardiac glycosides 10, 11.
LC-MS Analysis: Electrospray ionization (ESI) interfaced bruker daltonic esquire-LC amazon SL ion trap Mass Spectrometer (Bremen, Germany) and Dionex ultimate 3000 (Germany); composed of the quaternary pump with an online degasser. A thermo-statted column compartment, photodiode array detector (DAD), an autosampler and Hystar software were used. (Environmental Studies and Research Institute, El-Sadat City, Egypt). Experimental conditions of LC/MS system were as follows: Dionex bounded silica C18 column, dimensions: 4.6 × 150 mm 3 um. The mobile phase was acetonitrile + 0.1% water (injector temperature set at 38 °C). The carrier gas was N2 with a flow rate of 12 ml/min, and the injection volume was 20μl. The detector temperature was set at 350 °C.
Sample Preparation: 0.9 gm of the crude methanolic prepared was dissolved in 1 ml methanol. The sample was filtered by teflon 0.22 membrane filter, then 100 µm of the sample was dissolved in 1ml of methanol to carry the analysis.
RESULTS AND DISCUSSION:
Preliminary Phytochemical Screening: The preliminary phytochemical study revealed that the methanolic extract of Antigonon leptopus contains carbohydrates, tannins, flavonoids, saponins, sterols, and anthraquinones.
LC-MS Analysis: The results of the LC/MS analysis revealed the identification of fifty compounds from the methanolic extract of Antigonon leptopus. The compounds were flavonoids, flavones, flavonols, flavanones, bi-flavonoid, phenolics, glycosides, anthocyanins, and amino acids. Two types of experiment were carried out to achieve the objectives of this study.
TABLE 1: COMPOUNDS IDENTIFIED FROM LC/MS ANALYSIS OF METHANOLIC EXTRACT OF ANTIGONON LEPTOPUS
| S. no. | RT
(min) |
Molecular weight | Molecular
formula |
Compound
identification |
Class /
biological use |
Area % | References |
| 1 | 0.1 | 316 | C16H12O7 | Rhamnetin
|
Flavonol
Hepatoprotective -reduces liver enzymes |
0.2 | Mass bank ID:
PR020009 |
| 2 | 0.2 | 228 | C14H12O3 | Resveratrol
|
Polyphenol “Stilbenoid”
-Cardioprotective |
0.7 | Mass bank ID:
CE000097 |
| 3 | 0.4 | 493 | C23H25O12 | Malvidin-3-O-beta-D-glucoside | Anthocyanin
-Antioxidant |
7 | Mass bank ID:
PR020065 |
| 4 | 0.4 | 610 | C27H30O16 | Luteolin-3’,7-di-O-glucoside | Flavone
-Antioxidant |
0.2 | Mass bank ID:
PR020059 |
| 5 | 1.2 | 286 | C16H14O5 | Sakuranetin
|
Flavanone
-Antimicrobial -Antimutagenic |
0.1 | Mass bank ID:
PR020021 |
| 6 | 1.3 | 655 | C29H35O17 | Malvin
|
Anthocyanin
-Antioxidant |
0.3 | Mass bank ID:
PR020064 |
| 7 | 1.5 | 331 | C17H15O7 | Neohesperidin dihydrochalcone
|
Flavanone
-Decreases gastric mucus and ulcers |
0.1 | Mass bank ID:
PR020039 |
| 8 | 1.8 | 302 | C15H10O7 | Quercetin
|
Flavonoid
-Antioxidant -Prevents lipid peroxidation |
4.2 | Mass bank ID:
CE000171 |
| 9 | 1.8 | 300 | C16H12O6 | Kaempferide
|
Flavonol
-Antioxidant |
0.2 | Mass bank ID:
PR020013 |
| 10 | 1.9 | 450 | C21H22O11 | Marein
|
Phenol “Chalconoid”
-Antioxidant |
1.8 | Mass bank ID:
PR020071 |
| 11 | 1.9 | 464 | C21H20O12 | Isoquercitrin
|
Flavonoid
-Antioxidant -Antinflammatory -Antimicrobial |
4 | Mass bank ID:
PR040188 |
| 12 | 2.1 | 331 | C17H15O7 | Malvidin
|
Anthocyanin
-Antioxidant |
0.7 | Mass bank ID:
PR020010 |
| 13 | 2.8 | 448 | C21H20O11 | Quercetrin
|
Flavonoid
-Antiviral -Antioxidant -Hypoglycaemic -Hypolipidaemic |
2.1 | Mass bank ID:
PR020079 |
| 14 | 2.9 | 594 | C27H30O15 | Saponarin | Flavone
-Hypoglycaemic -Hepatoprotective -Antioxidant |
0.8 | Mass bank ID:
PRO030006 |
| 15 | 3.1 | 165 | C9H11NO2 | L-Phenyl alanine | Amino-acid
Essential amino acid -Treats depression, arthritis |
0.2 | Mass bank ID:
KO003668 |
| 16 | 3.3 | 204 | C11H12N2O2 | L-tryptophan
|
Amino-acid
-Essential amino acid -Treats anxiety, depression and premenstrual syndrome |
0.5 | Mass bank ID:
KNA001015 |
| 17 | 3.3 | 448 | C21H20O11 | Luteolin-7-O-glucoside
|
Flavone
-Antioxidant -Antinflammatory -Antibacterial |
0.2 | Mass bank ID:
PR040140 |
| 18 | 12.1 | 318 | C15H10O8 | Myricetin
|
Flavonoid
-Antioxidant -Protects against DNA damage |
0.2 | Mass bank ID:
PR020006 |
| 19 | 15.9 | 862 | C42H38O20 | Sennoside A
|
Senna
Glycoside -Purgative |
3 | Mass bank ID:
TY000200 |
| 20 | 16.4 | 286 | C15H10O6 | Luteolin
|
Flavonoid
-Antiallergic -Antinflammatory |
0.5 | Mass bank ID:
TY000143 |
| 21 | 19.5 | 608 | C28H32O15 | Flavocommelin
|
Flavone
-Antioxidant |
0.5 | Mass bank ID:
TY000133 |
| 22 | 19.7 | 286 | C15H10O6 | Flavanone
|
Flavanone
-Antimicrobial |
0.1 | Mass bank ID:
PR020003 |
| 23 | 20.9 | 462 | C22H22O11 | Diosmetin-7-O-beta-D-glucopyranoside | Flavone
-Antioxidant -Anticancer -Phytoestrogen fights breast cancer |
0.8 | Mass bank ID:
TY000189 |
| 24 | 21.3 | 300 | C16H12O6 | Chrysoeriol
|
Flavone
-Antioxidant -Antinflammatory |
1.1 | Mass bank ID:
TY000126 |
| 25 | 21.4 | 284 | C16H12O5 | Acacetin | Flavone
-Dietary estrogen -Antidiabetic “α-glucosidase inhibitor” |
3.1 | Mass bank ID:
TY000176 |
| 26 | 21.4 | 558 | C30H22O11 | Fukugetin
|
Flavonoid
-Antibacterial -Hypoglycaemic |
0.7 | Mass bank ID:
TY000181 |
| 27 | 21.5 | 270 | C15H10O5 | Apigenin
|
Flavonoid
-Inhibits skin and thyroid carcinogenesis |
3.7 | Mass bank ID:
PR020018 |
| 28 | 21.7 | 181 | C9H11NO3 | L-tyrosine
|
Amino-acid
-Non-essential amino acid -The precursor to neurotransmitters and hormones |
0.8 | Mass bank ID:
KNA00264 |
| 29 | 22.1 | 464 | C21H20O12 | Quercetin-3-beta-O-galactoside
|
Flavonoid
-Antinflammatory -Antiproliferative |
1.2 | Mass bank ID:
PR020075 |
| 30 | 22.2 | 314 | C17H14O6 | Velutin
|
Flavonoid
-Antinflammatory -Antifungal |
1.5 | Mass bank ID:
FIO00265 |
| 31 | 22.4 | 284 | C16H12O5 | Biochanin A
|
Flavone
-Phytoestrogen Inhibits prostate cancer |
2.3 | Mass bank ID:
PR0000261 |
| 32 | 22.8 | 448 | C21H20O11 | Maritimein
|
Phenol
-Antioxidant |
0.4 | Mass bank ID:
PR020067 |
| 33 | 25.3 | 290 | C15H14O6 | Epicatechin
|
Flavonol
-Antioxidant |
0.5 | Mass bank
ID: CE000092 |
| 34 | 26.4 | 418 | C20H18O10 | Juglanin
|
Flavonoid
-Inhibits hepatic diseases and hepatitis viruses |
0.1 | Mass bank
ID: Ty000222 |
| 35 | 26.5 | 286 | C15H10O6 | Kaempferol
|
Flavonol
-Antioxidant -Antinflammatory |
2.6 | Mass bank
ID: CE000175 |
| 36 | 26.6 | 578 | C27H30O14 | Kaempferitrin
|
Flavonol
-Insulinomimetic |
1.0 | Mass bank
ID: TY000224 |
| 37 | 27.0 | 328 | C18H16O6 | Salvigenin
|
Flavonoid
-Immunomodulatory -Antitumor |
1.7 | Mass bank
ID : Ty000249 |
| 38 | 27.3 | 286 | C15H10O6 | Fisetin
|
Flavonol
-Antioxidant -Enhances memory |
0.4 | Mass bank ID :
FIO00086 |
| 39 | 27.5 | 374 | C16H22O10 | Swertiamarin
|
Glycoside “Secoiridoid”
-Hepatoprotective -Antioxidant |
0.3 | Mass bank ID: Ty000077 |
| 40 | 27.5 | 300 | C16H12O6 | Diosmetin
|
Flavonoid
-Antioxidant -Anticancer -Cytoprotective |
8.5 | Mass bank
ID: Ty000129 |
| 41 | 27.5 | 272 | C15H12O5 | Naringenin
|
Flavonoid
-Antioxidant -Neuroprotective -Regulate HepG2 |
2.8 | Mass bank
ID: PR020017 |
| 42 | 28.0 | 300 | C16H12O6 | Hispidulin
|
Flavone
-Fights pancreatic cancer |
0.1 | Mass bank
ID: Ty000233 |
| 43 | 28.2 | 592 | C28H32O14 | Fortunellin
|
Flavonoid
-Antioxidant
|
0.1 | Mass bank
ID: PR020026 |
| 44 | 28.9 | 346 | C17H14O8 | Syringetin | Flavonol
- Inhibition of the procoagulant activity -Antiviral |
0.3 | Mass bank
ID: PR020084 |
| 45 | 29.2 | 330 | C17H14O7 | Cirsiliol
|
Flavone
-Sedative and hypnotic |
2.1 | Mass bank
ID: TY000127 |
| 46 | 29.2 | 462 | C22H22O11 | Scoparin
|
Flavonoid
-Anticancer |
5.0 | Mass bank
ID: TY000252 |
| 47 | 29.2 | 606 | C29H34O14 | Embinin
|
Flavone
-Antiviral -An influenza |
2.7 | Mass bank
ID: TY000131 |
| 48 | 29.9 | 224 | C11H12O5 | Sinapic acid
|
Phenolic acid
-Antinflammatory -Antioxidant |
4.6 | Mass bank
ID: PR020014 |
| 49 | 29.3 | 610 | C27H30O16 | Rutin
|
Flavonol
-Antioxidant -Antinflammatory -Hepatoprotective |
0.6 | Mass bank
ID: CE000140 |
| 50 | 29.4 | 538 | C30H18O10 | Amentoflavone
|
Bi-flavonoid
-Antinflammatory |
0.5 | Mass bank
ID: PR030006 |
RT= Retention time
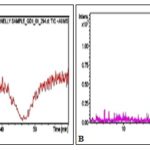
FIG. 1: LC/MS CHROMATOGRAMS OF THE METHANOLIC EXTRACT OF THE AERIAL PARTS OF ANTIGONON LEPTOPUS. A-Total ion chromatogram of the positive ion mode, B-Total ion masses of the identified compounds of the positive ion mode
The first experiment is to select the preference of ion polarity. The selection was based on the abundance of ions determined by the intensity of the peaks. In the second experiment, the preferred ion polarity was used for identification of the compounds. The selection of ion was automatically performed by the system. Compounds were characterized by their retention times, mass spectra and comparison to online mass bank database. Total ion chromatogram of the positive mode and the total masses of the identified compounds are shown in Fig. 1. Results of the identified compounds are displayed in Table 1.
CONCLUSION: The LC-MS analysis report has shown that the methanolic extract of the aerial parts of Antigonon leptopus contains various bio-active compounds. The compounds were flavonoids, flavones, flavonols, flavanones, bi-flavonoids, phenolics, glycosides, anthocyanins and amino-acids. All these diversified phytoconstituents might be responsible for the wide pharmacological actions of the vine.
ACKNOWLEDGEMENT: The author is thankful to the Department of Pharmacognosy, Faculty of Pharmacy, Cairo University for all the facilities provided. Special thanks are owed to the Environmental Studies and Research Institute, El-Sadat City, Egypt for carrying out LC-MS analysis of the sample.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Witcht M: Herbal Drugs and Phytopharmaceuticals; A Handbook for Practice on a Scientific Basis, Pages of the General Part, Edition 3rd, 2003.
- Burkill IH: A dictionary of the economic products of the Malay Peninsula. Revised reprint, 2 vols. Ministry of Agriculture and Co-operatives, Kuala Lumpur., Vol. 1 (A-H), 1-1240, Vol. 2, (I-Z), 1966; 1241-2444.
- Mitchell SA and Ahmad MH: A review of medicinal plant research at the University of the West Indies, Jamaica. West Indian Medical Journal 2006; 55(2): 243-269.
- Lans CA: Ethnomedicine used in Trinidad and Tobago for urinary problems and diabetes mellitus. Journal of Ethnobiology and Ethnomedicine 2006; 2: 45-55.
- Idu M and Onyibe HI: Medicinal plants of Edo State, Nigeria. Research J of Medicinal Plant 2007; 1(2): 32-41.
- Salazar SFM, Zepeda RE and Johnson DE: Plant folk medicine for gastrointestinal disorders among the main tribes of Sonora, Mexico Elsevier, Fitoterapia 2008; 79: 132-141.
- Angothu S, Lakshmi SM, Kumar AS and Reddy KY: Hepatoprotective activity of Antigonon leptopus Hook & Arn against carbon tetrachloride (CCl4) induced hepatotoxicity in Wistar albino rats. International Journal of Biological and Pharmaceutical Research 2010; 1(1): 27-32.
- Vanisree M, Alexander LRL, DeWitt DL and Nair MG: Health-beneficial phenolic aldehyde in Antigonon leptopus tea. Evidence-Based Complementary and Alternative Medicine 2011; 1-6.
- Marimuthu J, Aparna JS, Jeeva S, Sukumaran S and Anantham B: Preliminary phytochemical studies on the methanolic flower extracts of some selected medicinal plants from India. Asian Pacific Journal of Tropical Biomedicine 2012; 79-82.
- Evans WC and Trease GE: Textbook of Pharmacognosy, Bailliere Tindall, London, Edition 16th, 2009.
- Harborne JB: Phytochemical methods- A guide to modern techniques of plant analysis, Chapman and Hall, London, 1973: 49-188.
How to cite this article:
Ramadan NA: LC/MS identification of phytoconstituents of the methanolic extract of the aerial parts of Antigonon leptopus (Hook & Arn). Int J Pharmacognosy 2018; 5(2): 91-96. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(2).91-96.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.