ISOLATION OF SULFATED POLYSACCHARIDES FROM GREEN ALGAE ENTEROMORPHA INTESTINALIS AND ITS EFFECT ON THE GROWTH OF HEPATOCARCINOMA CELL LINE
HTML Full TextISOLATION OF SULFATED POLYSACCHARIDES FROM GREEN ALGAE ENTEROMORPHA INTESTINALIS AND ITS EFFECT ON THE GROWTH OF HEPATOCARCINOMA CELL LINE
Akhil Gopalakrishnan Chandrika, Reshma Sivan, Sunitha Mary Chacko, Prakash Kumar Bhaskara and Nevin Kottayath Govindan *
School of Biosciences, Mahatma Gandhi University, PD Hills, Kottayam - 686560, Kerala, India.
ABSTRACT: Identification of natural marine bioactive molecules and their therapeutic potential is valuable considering that there are reduced chances of undesirable side effects. This study was designed to isolate and purify the polysaccharides from E. intestinalis and evaluate its anti-cancer effect on human hepatocarcinoma cell lines (Hep3B). Polysaccharides from Enteromorpha intestinalis were isolated and purified. Preliminary characterization was done using UV-Vis spectrum and FTIR analysis. Total sulfate content of the polysaccharides was determined. MTT assay, AO / EB and DAPI staining were done to determine the cell growth pattern and nuclear changes effected by polysaccharides on Hep3B cells. Effect of polysaccharides on mitochondrial membrane potential and ROS production on Hep3B cells were evaluated using Rhodamine 123 and DCHF-DA staining. Results showed that polysaccharides from E. intestinalis were rich in sulfate groups (sPS). Cell viability, assessed in-vitro by MTT assay showed that the number of viable cells was less than 50% when exposed to concentrations exceeding 1 mg/ml of sPS. Cell viability and nuclear integrity assays depicted cells with compromised membrane integrity, cell damage, and nuclear perforation compared to control cells. Cells treated with sPS while stained with rhodamine 123 displayed a diffused pattern of staining with loss of intensity at higher concentrations indicating that loss of mitochondrial potential is a notable characteristic in sPS induced cytotoxicity. Hep-3B cells treated with sPS and stained with a ROS-specific fluorescent dye-DCFH-DA displayed cells with higher levels of ROS in comparison to control cells. These results may provide a basis on which further research in the development of marine drugs for the effective treatment of hepatocellular carcinoma.
| Keywords: |
Enteromorpha intestinalis, Hep3B, MTT, Polysaccharides, Mitochondrial membrane potential
INTRODUCTION:
Purpose and Rationale: Chemotherapy and all forms of treatment involving synthetic drugs are generally associated with side-effects, whose symptoms manifest itself in different degrees ranging from mild to severe. It is in this regard that research about alternative medicine derived from naturally occurring sources becomes significant.
Identification of bioactive molecules and their therapeutic potential is a lucrative proposition, considering that there are reduced chances of undesirable side effects 1, 2. As of now an estimated 71% of the Earth's surface is composed of water bodies.
Therefore, exploring marine sources such as marine algae for therapeutic bioactive molecules are sustainable and logical. Phenols, polysaccharides, peptides and other secondary metabolites unique to these algae have been identified for it's antitumor, anticoagulant, anti-inflammatory, antiviral and antiallergic properties 3 - 7. These algae are also a part of regular consumption in far-eastern regions and thereby their proposed use as medicinal food is of added advantage 8. Enteromorpha intestinalis is green algae found growing in fresh water and seawater and on a diverse number of substrates.
E. intestinalis has been used in Asia as a pharmaceutical and health care food since it is low in calories and rich in amino acids, fatty acids, vitamins and minerals 9. These algae are a rich source of sulfated polysaccharides (sPS) having various biological activities which are renowned for their multiple therapeutic effects over a range of ailments 10, 11. sPS is absent in higher plants and all marine algae produce at least one type of sPS 12. Moreover, their structure also varies from species to species which provides one with several alternatives for use in development 13.
This study was designed to isolate and purify the polysaccharides from E. intestinalis and evaluate its anti-cancer effect using hepatocarcinoma cell lines (Hep3B).
MATERIALS AND METHODS:
Reagents: Eagle’s minimum essential medium (EMEM), 3- (4, 5- dimethylthiazol- 2- yl)- 2, 5-diphenyltetrazolium bromide (MTT), Acridine orange, Ethidium bromide, Formaldehyde, Triton-X, Dinitrosalicylic acid (DNS) and formaldehyde were purchased from Himedia Laboratories, Mumbai, India. Dichlorofluorescein, Rhodamine -123 and DAPI were purchased from Sigma Aldrich chemical Pvt. Ltd., Bangalore, India. Bovine serum albumin was purchased from Sisco Research Laboratories Pvt. Ltd. Mumbai, India. All other reagents used were of the highest analytical grade possible.
Samples and Preparation of Polysaccharides: The algae Enteromorpha intestinalis were collected from the south coast of Thiruvananthapuram, India. Fresh algae (5 g) were mixed with 100 ml distilled water and heated in a water bath at 100 °C for 2 hours. The mixture was filtered using Whatman filter paper no. 54. Polysaccharides were then precipitated using absolute ethanol. The precipitated polysaccharide mixture was separated by centrifugation at 10,000 rpm. The precipitate containing polysaccharides were washed several times and finally dried under vacuum at 30 °C in a rotary evaporator. Proteins present in the mixture were determined using Bradford’s method 14.
Deproteinization of Polysaccharide Mixture: Polysaccharide mixture obtained from E. intestinalis was deproteinized by Sevag’s method described earlier 15. Crude polysaccharide extract was dissolved in distilled water and added a one-fifth volume of a mixture of the reagent [n-butanol and chloroform (1:4)]. The reaction mixture was shaken vigorously for 20 min. After 20 min the mixture was centrifuged at 4000 rpm for 20 min.
The proteins were precipitated as a gel in the Sevag reagent. The protocol was repeated a few more times to effectively deproteinize the polysaccharide.
Estimation of Total Carbohydrate and Reducing Sugars: Phenol-Sulfuric acid method was used to estimate the total carbohydrate content 16. 5 ml of 96% H2SO4 and 1 ml of 4% phenol were added to the samples in ethanol and shaken vigorously for few minutes. The samples were then incubated in a water bath for 20 min at 30 °C and cooled at room temperature. The orange-yellow color formed was measured at 490 nm using UV-visible spectrophotometer (Shimadzu, Japan). Dinitro-salicylic acid (DNSA) method was used for determining reducing sugars in the polysaccharide mixture 17. 1 ml of DNSA reagent was added to 1 ml of the mixture and incubated in a water bath at 100 °C for 10 min. After cooling at room temperature, 10 ml of distilled water was added to it, and the optical density was recorded at 540 nm using UV-visible spectrophotometer (Shimadzu, Japan). A standard curve was prepared using a standard glucose solution.
Estimation of Sulfate Content: Total sulfate content of the polysaccharide mixture was estimated by using Barium chloride method. Samples were pipetted into a test-tube, and 2 ml of barium chloride (1 mg/ml) was added, mixed well and the optical density was read at 620 nm using a UV-visible spectrophotometer (Shimadzu, Japan). Standard curves were prepared using sodium sulfate 18.
UV-Vis Spectrum and FT-IR Spectroscopy: UV-Vis spectrum of the polysaccharides in water were recorded using a UV visible spectrophotometer (UV 1800, Shimadzu). For FTIR spectrum, the dried polysaccharide mixture was grounded with KBr powder and pressed into a 1mm pellets. The spectrum was recorded at the transmittance from 400 - 4,000 cm-1 in an FTIR spectrophotometer (FTIR 8400, Shimadzu) 19.
Cell Lines: Hep3B (Human hepatic carcinoma cell line) were obtained from National Centre for Cell Sciences, Pune, India. Modified Eagles medium (MEM), supplemented with 10% FBS, penicillin (100 U/ml), streptomycin (100 mg/ml) and 2 mg/ml NaHCO3 at 37 °C in a humidified CO2 incubator.
Cytotoxicity Analysis:
MTT Assay: Cytotoxic activity of the polysaccharide mixture towards Hep 3B cells were determined by assaying the reduction of 3- (4, 5-dimethylthiazol-2-yl)-2, 5diphenyltetrazolium bromide (MTT) to formazan. Hep3B Cells were seeded in 48 well microtitre plates (2 × 104 per well) and left overnight to adhere before being exposed to different concentrations of polysaccharide. Different concentrations of polysaccharide (125 µg - 2 mg/ml) were added to the cells and incubated at 37 °C with 95% O2 and 5% CO2 for 24 hrs. After exposure to polysaccharide mixture, incubation of the wells was carried out in the dark at 37 °C for 4 h after the addition of 50 µl of 5 mM MTT solution. Subsequently, formazan crystals formed were dissolved in 200 µl of DMSO after the removal of MTT, and the absorbance was measured at 570 nm using a microplate reader (Thermo varioskan multimode reader) 20.
Detection of Cell and Nuclear Morphology: Acridine Orange / Ethidium Bromide Staining: Hep 3B cells were seeded in a 96 well plate. When the cell became 60 - 70% confluent, different concentrations of polysaccharide mixture (125 µg -2 mg/ml) were added and incubated at 37 °C with 95% O2 and 5% CO2 for 24 hrs. After the incubation of 24 hours, the media was removed, and cells were washed 2 - 3 times with Phosphate buffered saline (PBS). To the cells, added 10 µl of 1 mg/ml AO and EtBr mixture was added to each well and incubated at 37 °C for 15 - 30 min. The staining solution was removed, and the cells were again washed in PBS for a few times. After washing, the cells were observed under a fluorescent microscope (Olympus Co., Japan) with 20x magnification 21.
Nuclear Changes Using DAPI Staining: Hep 3B cells were seeded in a 96 well plate. When the cell became 60-70% confluent, different concentrations of polysaccharide mixture (125 µg - 2 mg/ml) were added and incubated at 37 °C with 95% O2 and 5% CO2 for 24 hrs. After 24 hr media was aspirated and the cells were washed with PBS several times. The cells were permeabilized by immersing in 0.2% Triton-X 100 for 5 minutes. Triton-X 100 was aspirated, and the cells were aging washed three times with PBS. After washing, cells were treated with 100 µl DAPI solution and incubated for 15 min at room temperature. Finally, the cells were washed with PBS and observed under a fluorescent microscope (Olympus Co., Japan) with 20x magnification 22.
Detection of Mitochondrial Membrane Potential (mmp): Rhodamine (R - 123) a lipophilic cationic indicator used to label mitochondria was used to estimate the electrical potential across the inner mitochondrial membrane. Hep3b cells were seeded in a 96 well plate and treated with different concentrations of polysaccharide mixture (125 µg -2 mg/ml) for 24 hr. After incubation, the cells were stained with 10 µg/ml Rhodamine - 123 in DMSO for 30 minutes at 37 °C. The cells were then washed with PBS, and the cellular images were taken at using fluorescent microscope (Olympus Co., Japan) at 20x magnification 23.
Detection of ROS Levels in Hep3b Cells: The ability of a compound to induce intracellular ROS formation was determined using DCHFA (Dichlorofluorescein). The Hep3B seeded plates after attaining 60 - 70% confluence were treated with different concentration of polysaccharides (125 µg - 2 mg/ml) for 24hr. After 24 hours of incubation, the media was removed, and the cells were washed with PBS for 3 times. After washing the cells were treated with 20 µl of DCHF-DA (10 µM) and incubated in a CO2 incubation for 15 - 30 min. The cells were again washed with PBS four times. The washed cells were observed under a fluorescence microscope at 20x magnification 24.
Statistical Analysis: All the data are expressed as the mean ± standard deviation of three independent determinations. Statistical comparison was performed using SPSS 19 software via a one-way analysis of variance (ANOVA).
RESULTS:
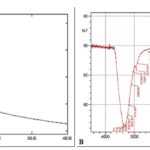
Deproteinized Algal Extract is Composed Primarily of Sulfated Polysaccharides: UV-Visible spectrum of deproteinized algal samples is enriched in polysaccharides as evident from the UV absorbance peak at 200 - 250 nm. FTIR spectrum of deproteinized samples shows a peak in the region of 3200 cm-1 which could correspond to the hydroxyl group stretches of the polysaccharide. Peaks at 2545 cm-1 correspond to weak C-H vibrations, and those at 1250 cm-1 indicates a sulfate -ester substitution. Absorption peaks at 846 cm-1 is a result of bending vibrations of C-O-S and peaks at 1253 cm-1 is due to stretching vibration of S-O which confirms the presence of sulfates in the deproteinized sample, thereby confirming the presence of sulfated polysaccharides (sPS).
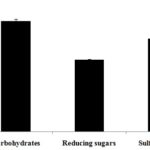
FIG. 1: TOTAL CARBOHYDRATE AND SULFATE CONTENT OF POLYSACCHARIDE ISOLATED FROM E. INTESTINALIS Values are expressed as mean ± SD of three separate experiments.
FIG. 2: UV-VIS AND FTIR SPECTRUM OF POLYSACCHARIDE ISOLATED FROM E. INTESTINALIS
Figures are a representation of three separate experiments. A. UV-Vis spectrum; B: FTIR spectrum.
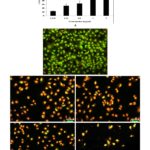
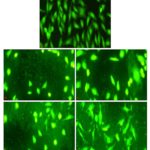
Sulfated Polysaccharides (sPS) Exhibit Significant Cytotoxicity in Cultured Hepatocarcinoma Cells: Cell viability was assessed in-vitro by MTT assay conducted following a 24 h treatment period in a dose-dependent manner. The number of viable cells was less than 50% in cells exposed to concentrations exceeding 1mg/ml of sPS. Cell health and viability were evaluated in terms of nuclear integrity through a dual fluorescent staining method which includes a cell-permeant - Acridine orange (AO) and a nucleic acid specific dye- Ethidium Bromide (EB). Fig. 3B depicts cells with compromised membrane integrity as observed through the bright yellow-orange stained nuclei.
FIG. 3: MTT ASSAY AND AO / EB STAINING OF Hep 3B CELLS TREATED WITH Sps
Cell morphology was examined and was photographed by a fluorescence microscope as described under Materials and Methods. Left panel (A) represents the MTT assay for cytotoxicity and right Panel (B) represents AO/EB staining, a- Control, b- 0.25mg/ml, c- 0.5mg/ml, d-1mg/ml, e- 2mg/ml. Magnification (20x). Figures are a representation of three separate experiments.
Dispersed EB stain indicates extensive cell damage and nuclear perforation when compared to control cells which display a homogenous green color throughout. The drastic reduction in the number of cells available for viability staining is in itself an indicator of the efficacy of the sPS extract in eliminating cancer cells in-vitro.
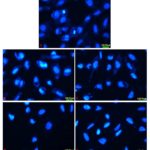
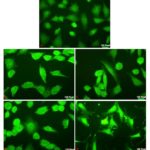
sPS Induced Cytotoxicity is Associated with Nuclear Damage and Loss of Mitochondrial Membrane Potential: The effect of treatment of sPS on mitochondrial membrane viability of Hep3B cells were assessed via Rhodamine - 123 staining and cells were subsequently stained with DAPI an AT-region specific binding dye observe various traits associated with nuclear damage. In Fig. 4, DAPI stained control nuclei display a visible number of rounded, evenly sized nuclei without aberrations of any kind in contrast to the treated modules which depict a misshaped, shrunken nuclear morphology and vacuole-like hollow spaces within the nuclei.
FIG. 4: NUCLEAR CHANGES INDUCED BY sPS TREATMENT IN Hep3B CELLS
Nuclear morphology and changes were examined and was photographed by a fluorescence microscope as described under Materials and Methods. a- Control, b- 0.25 mg/ml, c- 0.5 mg/ml, d-1 mg/ml, e- 2 mg/ml. Magnification (40x). Figures are a representation of three separate experiments.
FIG. 5: ROS PRODUCTION INDUCED BY sPS IN Hep3B CELLS
Hep3B at a density of 1 × 104 cells/well were treated with different concentration of polysaccharides and then harvested for staining with DCFH-DA to determine the production of ROS after 24hrs. Cell morphology was examined and was photographed by a fluorescence microscope as described under Materials and Methods. Figures are a representation of three independent experiments, magnification (20x).
In Fig. 6, cells treated with sPS when stained with the mitochondria-specific dye displayed a diffuse pattern of staining with loss of intensity at higher concentrations indicating that loss of mitochondrial potential is a notable characteristic in sPS induced cytotoxicity.
Elevated Levels of Cellular Oxidative Stress is Observed in sPS Treated Cells: Certain molecules create conditions of elevated oxidative stress in treated cells thereby tipping the redox balance of cells to an unfavorable status.
In Fig. 5, Hep-3B cells treated with sPS and stained with a ROS - specific fluorescent dye – DCFH - DA, displayed cells with higher levels of ROS in comparison to control cells as evaluated from brightly stained cells.
Rounded, dying cells displayed characteristically higher levels of ROS whereas the remaining cells showed a dispersed pattern of intense fluorescence throughout its structure.
FIG. 6: MITOCHONDRIAL MEMBRANE POTENTIAL (MMP) OF Hep3B CELL LINES TREATED WITH sPS
Hep3B at a density of 1 × 104 cells/well in were treated with different concentration of polysaccharides and then harvested for staining with Rhodamine 123to determine the MMP after 24 h. Cells were photographed by a fluorescence microscope as described under Materials and Methods. Figures are a representation of three independent experiments. a- Control, b- 0.25mg/ml, c- 0.5mg/ml, d-1mg/ml, e- 2mg/ml. Magnification (40x).
DISCUSSION: Rising numbers of afflicted persons and unpredictability makes cancer one of the most dreaded diseases in the present times. Cancers of the liver and gastrointestinal tract are on the rise partly due to an unhealthy lifestyle and partly due to genetic predisposition 25.
Conventional therapies reduce the risk of mortality but are often associated with side effects which affect the quality of life. Marine algae survive in non-growth conducive conditions producing several bioactive compounds of which sulfated polysaccharides have been particularly acclaimed for its anti-proliferative activity 26, 27. The possibility that such algae can be included in daily diet as a 'medicinal' food is a comfort from the painful and debilitating routine of chemotherapy.
This present study involved the isolation and characterization of a polysaccharide-rich algal extract. IR spectrum of the deproteinized algal sample showed peaks between 800 - 850 cm-1 which indicated the presence of sulfur groups associated with carbohydrates 28.
FTIR data and estimated values of total carbohydrate and sulfate further confirmed that the polysaccharides are significantly sulfated. Effects of the sPS on cell integrity were evaluated by fluorescent staining methods which indicated that a concentration as low as 0.25 mg/ml displayed considerable toxicity on cancer cells, whose numbers dwindled noticeably in subsequent higher concentrations indicating prominent cell death. Among marine macrophytes, marine green algae have been less studied compared to brown and red algae as sources of bioactive polysaccharides with cancer preventive properties. However, the anti-cancer properties reported was mainly on the polysaccharides from ulvans 29.
The rise in levels of oxidative stress and loss of mitochondrial membrane potential could also be observed which may be a direct or indirect consequence of the treatment. Cancer cells show persistent oxidative stress level compared to normal cells, rendering malignant cells more vulnerable to several drugs that boost increased ROS levels, such as marine drug, 10-acetylirciformonin B (10AB), a marine sponge furanoterpenoid derived from irciformonin 30. Mitochondrial dysfunctions leading to elevated ROS generation is another characteristics of cancer cells as a result the possibility of preferentially targeting cancer cell mitochondria and improve therapeutic selectivity is of great importance 31. Overall, it can be said that sPS exhibits desirable anticancer properties in vitro, whose action on normal cells at therapeutic concentrations needs to be evaluated to rule out non-specific toxicity.
CONCLUSION: Further studies with fractions purified and characterized based on their molecular weight are in progress so that the active component may be identified. Preliminary findings warrant a molecular level study of the role of sPS in caspase - 3 induced apoptosis and cell proliferation, differentiation and metastasis.
ACKNOWLEDGEMENT: The authors are grateful to School of Biosciences, Mahatma Gandhi University and Department of Biotechnology, Government of India for the excellent research facilities supported through the DBT-MSUB-IPLSARE (BUILDER) programme (No.BT/PR 4800/INF/22/152/2012).
CONFLICT OF INTEREST: Nil
REFERENCES:
- Clark AM: Natural products as a resource for new drugs. Pharmaceutical Res 1996; 13: 1133-41.
- Cragg GM and Newman DJ: Plants as a source of anti-cancer agents. J Ethnopharmacol 2005; 100: 72-79.
- Amaro HM, Barros R, Guedas, Sousa-Pinto C and Malcata FX: Microalgal compounds modulate carcinogenesis in the gastrointestinal tract. Trends Biotech 2013; 31: 92-98.
- Iwamoto K, Hiragun T, Takahagi S, Yanase Y, Morioke S, Mihara S, Kameyoshi Y and Hide M: Fucoidan suppresses IgE production in peripheral blood mononuclear cells from patients with atopic dermatitis. Arch Dermatol Res 2011; 303: 425-31.
- Sung-Myung K, Kil-Nam K, Seung-Hong L, Ahn G, Cha S, Kim A, Yang X, Kang M and Jeon Y: Anti-inflammatory activity of polysaccharide purified from AMG-assistant extract of Ecklonia cava in LPS-stimulated RAW 264.7 macrophages. Carbohydrate Pol 2011; 85: 80-85.
- Lee JB, Hayashi K, Maeda M and Hayashi T: Antiherpetic activities of sulfated polysaccharides from green algae. Planta Medica 2004; 70: 813-817.
- Lionel C, Foucault A, Chaubet F, Kervarec N, Sinquin C, Fisher A and Boisson-Vidal C: Further data on the structure of brown seaweed fucans: relationships with anticoagulant activity. Carbohydrate Res 1999; 3119: 154-65.
- Se-Kwon K, Thomas NV and Li X: Anticancer compounds from marine macroalgae and their application as medicinal foods. Adv Food Nutr Res 2011; 64: 213-224.
- Eun-Jeong L, Cho K, Kim J, Lee Y, Ho JN, Kim Y, and Cho H: The anticoagulant and anticancer activities of Enteromorpha intestinalis The Kor J Food Nutr 2008; 21: 7-14.
- Ladislava M, Škrovánková S, Samek D, Ambrožová J and Machu L: Health Benefits of Algal Polysaccharides in Human Nutrition. Adv Food Nutr Res 2012; 66: 75.
- You-Jin J, Wijesinghe WAJP and Kim S: Functional properties of brown algal sulfated polysaccharides, Fucoidans. Adv Food Nutr Res 2011; 64: 163-78.
- Myriam W and Clercq ED: Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen Pharmacol Vascular Sys 1997; 29: 497-11.
- Dietrich CP, Farias GGM, de Abreu LRD, Leite EL, da Silva LF and Nader HB: A new approach for the characterization of polysaccharides from algae: the presence of four main acidic polysaccharides in three species of the class Phaeophyceae. Plant Science 1995; 108: 143-53.
- Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976; 72: 248-54.
- Xia Q, Liu G, Ge CL and Xu DJ: Removal of proteins from crude astragalus polysaccharides by sevag method. Anhui Med Pharmaceutical J 2007; 11: 1069-70.
- Dubios M, Giles KA, Hamilton JK, Rebors PA and Smith F: Colorimetric method for determination of sugars and related substances. Anal Chem 1956; 28: 350-56.
- Miller GL: Use of dinitrosalisylic acid reagent for determination of reducing sugars. Anal Chemistry 1959; 31: 426-28.
- Ye H, Wang K, Zhou C, Liu J and Zheng X: Purification and antioxidant activities of in vitro of polysaccharides from the brown sea weed Sagassum palladium. Food Chem 2008; 111: 428-32.
- Zang Q, Li N, Liu X, Zhao Z, Li Z and Xu Z: The structure of a sulfated galactan from Porphyra lalenensis and its in-vivo anticancer activity. Carbohydrate Res 2004; 339: 105-11.
- Mosmann T: Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods 1983; 65: 55-63.
- Deborah R, Goldstein NB, Norris DA and Shellman YG: A simple technique for quantifying apoptosis in 96 well plates. BMC Biotech 2005; 5: 12.
- Chazotte B: Labelling nuclear DNA using DAPI. Cold Spring Harbour Protocols 2011; 1: 5556.
- Wang Y, Zhao X, Gao X, Nie X, Yang Y and Fan X: Development of fluorescence imaging-based assay for screening cardioprotective compounds from medicinal plants. Anal Chimica Acta 2011; 702: 87-94.
- Shen W, Hsieh C,Chen C, Yang K, Ma C, Choi P and C Lin: A modified fixed staining method for the simultaneous measurement of reactive oxygen species and oxidative responses. Biochem Biophy Res Commun 2013; 430: 442-47.
- Coyle YM: Lifestyle, genes and cancer. Cancer Epidemiol Mod Factors 2009; 25-56.
- Ngo DH and Kim SK: Sulfated polysaccharide as bioactive agents from marine algae. Int J Biol Macromol 2013; 62: 70-75.
- Noda H, Amano H, Arashima K and Nisizawa K: Antitumor activity of marine algae. Hydrobiologia 1990; 204: 577-84.
- Vishchuk OS, Ermakova SP and Zvyagintseva TN: Sulfated polysaccharides from brown seaweeds Saccharina japonica and Undaria pinnatifida: isolation, structural characteristics, and antitumor activity. Carbohydrate Res 2011; 346: 2769-76.
- Lahaye M and Robic A: Structure and functional properties of ulvan, a polysaccharide from green seaweeds. Biomacromolecules 2007; 8: 1765-74.
- Farooqi AA, Fayyaz S, Hou M, Li K, Tang J and Chang H: Reactive oxygen species and autophagy modulation in non-marine drugs and marine drugs. Marine Drugs 2014; 12: 5408-24.
- Tsai CF, Yeh WL, Huang SM, Tan TW and Lu DY: Wogonin induces reactive oxygen species production and cell apoptosis in human glioma cancer cells. Int J Mol Sci 2012; 13: 9877-92.
How to cite this article:
Chandrika AG, Sivan R, Chacko SM, Bhaskara PK and Govindan NK: Isolation of sulfated polysaccharides from green algae Enteromorpha intestinalis and its effect on the growth of hepatocarcinoma cell line. Int J Pharmacognosy 2018; 5(1): 51-60. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(1).51-60.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
51-60
727
1566
English
IJP
A. G. Chandrika, R. Sivan, S. M. Chacko, P. K. Bhaskara and N. K. Govindan *
School of Biosciences, Mahatma Gandhi University, Kottayam, Kerala, India.
nevinkg@gmail.com
21 July 2017
25 August 2017
17 September 2017
10.13040/IJPSR.0975-8232.IJP.5(1).51-60
01 January 2018