ISOLATION AND PURIFICATION OF ANTI-TUMOR ENZYME L-ASPARAGINASE FROM SOIL ISOLATES
HTML Full TextISOLATION AND PURIFICATION OF ANTI-TUMOR ENZYME L-ASPARAGINASE FROM SOIL ISOLATES
P. Kothari and D. V. Deshmukh *
MGM’s, Institute of Biosciences and Technology, Aurangabad - 431003, Maharashtra, India.
ABSTRACT: 16 different bacteria were isolated from soil collected from and near Aurangabad city. All of the isolates were screened for L-asparaginase activity using modified M-9 medium. Spectro-photometric determination of L-asparaginase was done, and it was partially purified using ammonium sulphate precipitation. Out of the bacterial species screened only two were found to be producers of L-asparaginase. The Biochemical identification revealed both the species to be of the genus Bacillus. The crude extract showed a low enzyme activity as compared to partially purified extract. Both the isolates gave a maximum enzyme activity at pH 8 and a temperature of 40 °C. The genus Bacillus has become a promising candidate for the production of L-asparaginase from the other reported bacteria.
| Keywords: |
L-asparaginase, M-9 modified medium, Enzyme activity
INTRODUCTION: The enzyme L-asparaginase is a pyrimidine derivative which has been considered as an important compound in acute lymphoblastic leukemia management. The pyrimidines are said to be an integral part of nucleic acid which also shows anti-viral, anti-bacterial, anti-cancer activities depending upon geometry and type of substituent attached to pyrimidine ring 1. The enzyme L-asparaginase consists of two forms: L-asparaginase I, an internal constitutive enzyme and L-asparaginase II, an external enzyme which is secreted in response to nitrogen starvation. The two enzymes are biochemically and genetically distinct. In many organisms, L-asparaginase is found in the soluble fraction of cytoplasm 2. The tetrameric enzyme L-asparaginase composed of four similar
monomers each monomer have 326 amino acid residue, and the whole efficient enzyme exists as a tetramer of identical subunits, with the molecular weight between 140 and 160 kDa mass 3, 4. Asparaginase occurs in many animal tissues, plants, vertebrates, bacteria, actinomycetes, and algae but not in human blood. L-asparaginase II is widely distributed in both eukaryotic and prokaryotic cells and has been intensively studied over the past five decades. L-asparaginase II is produced by a large number of organisms such as E. coli, Erwinia cartovora, Pseudomonas aeruginosa, Serratia marcescens 8. L-asparaginase was also reported from B. cadaveris and P. vulgaris which were isolated from soil 5. It has been observed that eukaryotic microorganisms like yeast 6 and fungi have a potential for asparaginase production 7.
Some methods have been suggested for the commercial production of the important enzymes. For liquid culture in huge bioreactors are preferred for the bulk production of therapeutic enzymes. Other processes which are used for production includes solid-state fermentation (SSF), immobilization and fermentation on an inert solid support. Submerged fermentation is the cultivation of microbial cells in liquid media under controlled conditions for the production of a desirable metabolite.
The physicochemical conditions for L-asparaginase production vary with the micro-organism. Some purification procedures have been developed for the purification of L-asparaginase which includes ethyl alcohol fractionation, DEAE Cellulose Chromatography, CM-Sephadex Chromatography and Polyacrylamide Electrophoresis 8. The important application of the L-asparaginase enzyme is in the treatment of acute lymphoblastic leukemia (mainly in children), acute myelocytic leukemia, acute myelomonocytic leukemia, chronic lymphocytic leukemia, lymphosarcoma treatment, and melanosarcoma. Though, many species producing L-asparaginase are available, only E. coli and E. cartovora asparaginases are currently in medical use as drugs in the treatment of lymphocytic leukemia, because of high substrate affinity.
Another, potentially important application to asparaginases is in the food industry. As a food processing aid, asparaginases can effectively reduce the level of acrylamide up to 90% in a range of starchy foods without changing the taste and appearance of the end product. On the other hand, reproductive toxicity, genotoxicity, and carcinogenicity are further potential human health risks, by animal studies. Therefore, the use of L-asparaginases to hydrolyze asparagine in food as well as in the development of an immunoassay (ELISA) for the analysis of asparagine content could be a useful approach to reduce the extent of acrylamide formation 9.
MATERIAL AND METHOD:
Sampling and Isolation: Soil samples were collected from nearby suburbs of Aurangabad City. Pre-sterilized plastic bags were used for the collection of soil samples. Bacterial cultures from the soil samples were isolated by using the serial dilution method, and the respected dilution was placed on nutrient agar medium containing (g/l) peptone, 5.0; beef extract, 3.0; sodium chloride, 3.0; agar, 15 and pH 7.0 ± 0.2. All the constituents were added in 50 ml distilled water and the volume was made up to 100ml and autoclaved for 15 min at 121 °C. All the inoculated plates were incubated at 37 °C for 24 h and observed for bacterial growth.
Screening of Asparaginase Producers: The well isolated colonies after the incubation period were plated on modified M9 medium 10 containing (g/l): KH2PO4, 2.0; L-asparagines, 6.0; MgSO4.7H2O, 1.0; CaCl2.2H2O, 1.0; glucose, 3.0; agar, 20.0; phenol red (few drops) as indicator. The inoculated agar plates were incubated at 37 °C overnight along with a control plate in which asparagine was absent. The pink colored colony was selected for further studies. The cultures were purified using subsequent transfer and were stored on slants at 4°C until further use.
Characterization of the Isolates: Gram staining was used for the morphological identification and characterization of the isolates. Biochemical characterization of the isolates was done based on a biochemical test such as IMViC, enzymes such as catalase, gelatinase, and amylase, sugar fermentation tests.
Production and Purification of L-Asparaginase: Submerged fermentation was used as a method for the production of L-asparaginase. The pressurized flasks containing (g/l): KH2PO4, 2.0; L-asparagine, 6.0; MgSO4.7H2O, 1.0; CaCl2.2H2O, 1.0 and glucose, 3.0 medium were inoculated with a loop-full of log phase bacterial cultures and was incubated on a rotary shaker at 37 °C for 24 h. After the incubation, the content of the fermentation broth was centrifuged at 10000 rpm for 10 min, and the crude enzyme was extracted. TLC of the extract was done to check the conversion of L-asparagine into L-Aspartic acid. The partial purification of the enzyme was done using fractional precipitation using ammonium sulphate at 35%, 50% and finally 80%. Protein estimation was done using the Biuret method. The partially purified enzyme was estimated spectrophotometrically by estimating the ammonia produced during L-asparaginase catalysis using Nessler’s reagent at 480 nm.
Effect of Temperature and pH on Asparaginase Activity: The assay mixture containing the partially purified enzyme and the substrate was tested for the effect of temperature and pH on the asparaginase activity. A series of the flask containing the enzyme mixture was exposed to a temperature regime of 20° C to 70° C for 10 min and a pH range of 5-8 respectively, the reaction was stopped by adding 0.1 ml of 1.5 M TCA. The mixture from all the tubes was centrifuged at 5000 rpm for 10 min, and the supernatant was collected, and the enzyme activity was measured as described above.
RESULT AND DISCUSSION: The enzyme L-asparaginase has been intensively investigated over the past two decades owing to its importance as an antineoplastic agent 11, although the enzyme has been found in a variety of bacteria, fungi, yeast, and mammals. Few of these purified preparations have proceeded antitumor activity.
TABLE 1: CHARACTERIZATION OF BACTERIAL ISOLATES BASED ON GRAM REACTION AND CELL MORPHOLOGY
| Bacterial isolates | Gram’s nature | Morphology |
| 1A | -ve | Rod-shaped |
| 2A | +ve | Short rods |
| 3A | +ve | Cocci in bunch |
| 4A | +ve | Rods in chain |
| 5B | +ve | Cocci |
| 6B | -ve | Rods |
| 7B | +ve | Rods |
| 8B | -ve | Rods |
| 9B | -ve | Chain of rods |
| 10C | +ve | Short rods |
| 11C | +ve | Rods |
| 12C | -ve | Rods |
| 13D | +ve | Rods |
| 14D | -ve | Chain of rods |
| 15D | +ve | Rods |
| 16D | -ve | Cocci |
Some investigators have been producing L-Asparaginase from different biological systems. Howard and James 12 has used a wild type E.coli k12 strain for the production of L-Asparaginase. Some bacterial species have been investigated by Peterson and Ciegler in 1969 13. They screened 44 species belonging to different genera for the enzyme production. A Bacillus species form soil has been investigated for the production of L-asparaginase. In the present study, 16 different bacteria were isolated from the soil samples collected in the nearby area of Aurangabad city. The bacterial isolates were designated as in Table 1. The morphological and gram nature of the isolate was studied.
All the 16 isolates were screened for their production of the enzyme asparaginase as according to the procedure describe above. Out of the total 16 isolates only two isolates (7B & 12 C) showed the presence of pink colored colony on Modified M-9 medium. These two isolates (7B & 12 C) were taken for futher studies. The biochemical characteriazation for the two isolates was done Table 2.
TABLE 2: BIOCHEMICAL CHARACTERIZATION OF 7B AND 12C ISOLATES
| Biochemical
Test |
Result | |
| 7B | 12C | |
| Indole Test | -ve | -ve |
| Methyl red | -ve | -ve |
| Vogus proscur | -ve | -ve |
| Citrate utilization | +ve | +ve |
| Gelatinase | +ve | +ve |
| Starch hydrolysis | +ve | +ve |
| Catalase | -ve | -ve |
| Carbohydrate fermentation | +ve | +ve |
-ve- Negative, +ve- Positive
The biochemical characterization revealed both the isolates to be Bacillus species they were designated as ASP-I and ASP-II. Similar, results were obtained by Moorthy et al., 2010, who has isolated a Bacillus species from soil and investigated for the production of asparaginase using modified M-9 medium.
The application of the enzyme L-asparaginase in the area of biomedical sciences demands a high degree of purity of this enzyme. The intracellular enzyme was purified from the E. caratovora using ammonium sulphate precipitation; dialysis and anion exchange chromatography 1, 14 has purified L-asparaginase from Erwinia carotovora by ammonium sulfate fractionation (60–70%), Sephadex G-100, CM-cellulose, and DEAE Sephadex chromatography. In the case of extracellular L-asparaginase from Bacillus the purification of the enzyme was done by ion exchange chromatography using DEAE column 5.
In this study both the isolated Bacillus species showed maximum growth at 24 h of submerged fermentation in the presence of glucose as a carbon source and nitrogen source as asparagines followed by production of the enzyme. The TLC results showed that an Rf value of sample ASP-I was 0.26 and Rf value of ASP-II was 0.25 is almost the same to that of standard L-aspartic acid, i.e. 0.27. The specific activity and the protein concentration of both crude and partially purified enzyme are reported in Table 3.
TABLE 3: PURIFICATION STEPS, TOTAL PROTEIN AND ENZYME ACTIVITY OF CRUDE AND PARTIALLY PURIFIED L-ASPARAGINASE FROM ASP-I AND ASPII
| Crude extract | Ammonium Sulphate precipitation | |||
| Isolates | Total Protein (mg) | Specific activity (U/mg) | Total Protein (mg) | Specific activity (U/mg) |
| ASP-I | 2.17 | 0.18 | 1.33 | 1.79 |
| ASP-II | 1.96 | 0.12 | 0.75 | 1.85 |
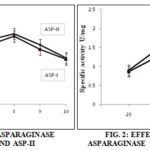
A rise in the specific activity of L-asparaginase from both the isolates and a decrease in the total protein concentration after partial purification of the enzyme have been observed. The isolate ASP-I showed 1.79 U/mg of specific activity with a total protein concentration of 1.33 mg, while ASP-II showed 1.85 U/mg of activity with a total protein of 0.75 mg. Similar, results were obtained by 5, where at a total protein concentration of 2.60 mg the activity of the crude extract was 0.10, while after ammonium sulphate precipitation and dialysis the increase in the specific activity of the enzyme was about 1.09 u/mg. 15 has used Bacillus cereus MNTG-7 for the production of L-asparaginase. 8, has achieved 0.950 U/mg of activity from E. coli. HAP strain. An increase in the specific activity from 0.123 U/mg to 1.41 U/mg was reported in the crude and ammonium sulphate precipitated extract respectively by 16. The activity of L-aparaginase was evaluated at different pH and temperature. The optimum activity of the enzyme was shown at pH 8, for both the isolate Fig. 1.
Has reported 17 a maximum activity of L- asparaginase from Streptomyces gulbargensis at pH 8.5, while 18. Reports an optimum pH as 7.5 produced by marine actinomycetes. L- asparaginase produced by bacterial species such as Serratia marcescens, Mycobacterium sp. and Pseudomonas sp. have shown the enzyme activity in the range of 8.0 to 8.5. These variations in pH optima for L-asparaginase may be due to the strain of the organism used, the chemical composition of the substrate, fermentation system and finally the conditions under which fermentation takes place 19. The activity of L-asparaginase was also studied for optimum temperature; it was found that both the cultures ASP-I and ASP-II were able to show maximum activity at a temperature of 40°C Fig. 2. A temperature of 37 °C has been observed by Moorthy et al., 2010, using a soil isolate of Bacillus species. An increase in the enzyme activity was observed in a temperature range of 25°C-35°C isolated from Aspergillus terreus through solid-state fermentation. Maximum activity of L-asparaginase was observed at 40 °C produced by Streptomyces albidoflavus 19.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kamble V, Sriniwasa Rao R, Borkar P, Khobragade C and Dawane B: purification of L-asparaginase from a bacteria Erwinia caratovora and effect of dihydropyrimidine derivatives on some of its parameters Indian Journal of Biochemistry and Biophysics 2006; 43: 391-394.
- Youssef M and Al-Omair M: Cloning, purification, characterization and immobilization of L-asparaginase II from coli W3110. Asian J Biochem 2008; 3: 337-350.
- Aung HP: Dynamics of the mobile loop at the active site of coli. Asparaginase Biochemistry Biophysics Acta 2000; 1481: 349-359
- Kozak: Crystallization and preliminary crystallographic studies of five crystal forms of Escherichia coli asparaginase II (Asp 90 Gi mutant) acta crystallography D: Biol. Crystallogr 2002; 58: 130-132.
- Moorthy V, Ramalingam A, Sumantha A and Shankar-narayan R: Production, purification and characterization of extracellular L-asparaginase from a soil isolate of Bacillus sp. African Journal of Microbiology Research. 2010; 4(18): 1862-1867. http://www.academicjournals. org/ajmr
- Dunlop P, Meyer G, Ban D and Roon R: Characterization of two forms of asparaginase in Saccharomyces cerevisiae The Journal of Biological Chemistry 1978; 253(4):1291-1394.
- de Moura Sarquis MI: Production of L-asparaginase by filamentous fungi. Mem Inst Oswaldo Cruz, Rio de Janeiro 2004; 99(5): 489-492
- Roberts J, Burson G and Hill J: New procedures for purification of L-asparaginase with high yield from Escherichia coli. Journal of Bacteriology 1968; 95(6): 2117-2123
- Sanches M, Krauchenco S and Polikarpov I: Structure, substrate complexation and reaction mechanism of bacterial asparaginases. Current Chemical Biology 2007; 1: 75-86.
- Kalingapplar S and Ligappa K: Screening & optimization of L-asparaginase-a tumor inhibitor from Aspergillus terreus through solid-state fermentation. 2010; 1(1): 55-60.
- Gulati R, Savena R and Gupta R: A rapid plate assay for screening L-asparaginase producing microorganisms. Ltrs Appl Microbiol 1997; 24: 23-26.
- Savitri, Asthana N and Azmi W: Microbial L-asparaginase: a potent antitumour enzyme. Indian Journal of Biotechnology 2003; 2: 184-194.
- Peterson R and Ciegler A: L-asparaginase production by various bacteria. Applied Microbiology 1969; 17: 929-930.
- Warangkar S and Khobragade C: Screening, enrichment and media optimization for the L-asparaginase producer. Journal of Cell and Tissue Research 2009; 9(3): 1963-68.
- Sunitha M, Ellaiah P and Devi R: Screening and optimization of nutrients for L-asparaginase production by Bacillus cereus MNTG-7 in SmF by plackett-burmann design. African J of Microbiology Res 2010; 4 (4): 297-03.
- Paul J: Isolation and characterization of a Chlamydomonas L-asparaginase. Biochemistry Journal 1982; 203: 109-115.
- Amena S, Vishalakshi N, Prabhakar M, Dayanand A and Lingappa K: Production, purification and characterization of L-asparaginase from Streptomyces gulbargensis. Brazilian Journal of Microbiology 2010; 41: 173-178.
- Basha S, Rekha R, Komala M and Ruby S: Production of extracellular anti-leukemic enzyme L-asparaginase from marine Actinomycetes by solidstate and submerged fermentation: Purification and Characterisation, Tropical Journal of Pharmaceutical Research 2009; 8(4): 353-360
- Poorani E, Saseetharan M and Dhevagi P: Isolation and characterization of L-asparaginase from marine actinomycetes Indian Journal of Biotechnology 2005; 5: 514-520.
How to cite this article:
Kothari P and Deshmukh DV: Isolation and purification of anti-tumor enzyme L-asparaginase from soil isolates. Int J Pharmacognosy 2014; 1(6): 384-88. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.1(6).384-88.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.