INNOVATIONS IN LIPOSOMAL DRUG DELIVERY: A COMPREHENSIVE REVIEW OF STRUCTURE, PREPARATION, APPLICATIONS, AND THERAPEUTIC POTENTIAL
HTML Full TextINNOVATIONS IN LIPOSOMAL DRUG DELIVERY: A COMPREHENSIVE REVIEW OF STRUCTURE, PREPARATION, APPLICATIONS, AND THERAPEUTIC POTENTIAL
Junaid Ahmad Dar, Satpal Kushwaha, Ranjan Kumar Singh * and Tanya Sharma
Faculty of Pharmaceutical Sciences, Mewar University, Gangrar, Chittorgarh, Rajasthan, India.
ABSTRACT: Liposomes are spherical vesicles with one or more concentric peel of phospholipid bilayers enclosing an aqueous core. Liposomes, which can be nontoxic or biodegradable, are a strong delivery system of tranches of drugs. Liposomes (or lipid vesicles) are a type of platform with different applications, for example as carriers that take drugs or other macromolecules into the bodies of humans and animals. The review covers traditional and advanced methods of liposomal preparation and stresses how these methods influence the marketability of liposomes. The review emphasises the need for research on properties of lecithin and modulators, which may improve the stability of liposomes. At present, liposomal vesicles are utilised to enhance the therapeutic efficacy of active compounds and to facilitate the controlled release of pharmaceuticals, thereby extending the biological half-life or diminishing the toxicity of the active ingredients. This paper looks at the latest changes in the way liposomal systems work, especially those that have happened in the last ten years. The procedures for system preparation, characterisation, and application are delineated. The text also talks about ways to change the surface of liposomes and how active substances are delivered.
Keywords: Bio-nanoparticle, Liposomes, Nanotechnology, Nanocarriers
INTRODUCTION: A vesicular system known as a liposome is created when phospholipids are distributed in water and spontaneously form a closed structure with an internal aqueous environment surrounded by phosphor lipid bi-layer membranes. Cholesterol, non-toxic surfactants, sphingolipids, glycolipids, long-chain fatty acids, and even membrane proteins can all be converted into liposomes, which are tiny spherical vesicles 1. Alec D. Bangham discovered liposomes in 1965. Since then, the lipid vessel has become a common way to encapsulate and deliver molecules to treat many different diseases.
Lipids and fatty acids are the main parts of liposomes. They are naturally found in cell membranes, so they are thought to be biocompatible and biodegradable. Liposomes are made up of amphipathic molecules that come together to form a bilayer sphere. The hydrophilic head groups face the outside water environment, and the hydrocarbon chains come together in the hydrophobic interior. Liposomes are great drug carriers because they have both polar and nonpolar parts. The knee joint is a complicated place, and OA affects its different parts in different ways.
The articular capsule surrounds the joint and holds the bones together. The inner layer, called synovium (also called synovial membrane), is a connective tissue layer with a lot of blood vessels 2. The intima layer of synovium has two types of cells: Type a macrophages that eat things and Type B fibroblast-like cells that make synovial fluid (SF) components. Blood and lymphatic vessels in the subintima layer move nutrients around and get rid of dead cells, drugs, and other things. Nonetheless, the application of PEG in pharmaceuticals is being examined due to the fact that patients receiving PEGylated medications can generate antibodies that attach to PEG, and anti-PEG antibodies are already present in numerous healthy individuals. Anti-PEG antibodies have been associated with diminished clinical efficacy and heightened adverse effects, rendering this a significant domain of investigation. As we have recently found out, anti-PEG antibodies are able to initiate complement activity and the release of drugs from PEGylated liposomal doxorubicin (PLD). This has been demonstrated with PLD as well as with the release of mRNA from lipid nanoparticles. The formulation of PLD consists of the nanorod crystalline form of the drug DOX-sulphate within an aqueous compartment of the liposomal delivery system covered with densely packed PEG. New formulations for liposomes, including transferosomes and ectosomes, were designed in order to increase the flexibility of the vesicles in a better permeability manner. Soluble polymers, insoluble or biodegradable microparticles, synthetic and natural polymers, microcapsules, cells, cell ghosts, lipoproteins, liposomes, and micelles are all considered drug carriers. It is feasible to create carriers that are location-specific, responsive to stimuli like pH or temperature changes, and slow to decay.
Targeting is the technique of directing the drug-loaded system to a certain location 3. Transfer some have been formulated using phospholipids and the presence of a surfactant as an edge activator whereas ectosomes include higher concentrations of ethanol as the main component in the composition. The two mentioned systems are more flexible than liposomes, and based on penetration enhancing components, the vesicles are able to enter the deeper parts of the skin. Bacterial conjunctivitis is an inflammatory disease of the tissue called conjunctiva due to a bacterial infection 1–3. Bacterial keratitis is a similar condition but affects the cornea. Symptoms include a reddened eye and a discharge containing. Fluoroquinolones are the antibiotic drugs primarily employed in treatment due to their wide-spectrum of action against gram-positive bacteria, gram-negative bacteria, and anaerobes and proven efficacy 4.
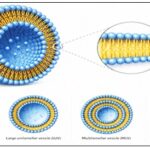
Structure of Liposomes: Liposomes are artificially formed vesicles that are made up of phospholipid bilayers. They possess the capability of entrapping functional agents inside the aqueous medium contained within the liposomes while being suspended in the external aqueous medium. Phospholipids are considered the basic building blocks of liposomes, whereas sterols, non-ionic, and ionic amphiphiles are utilized to increase the stability of liposomes (Large, Abdelmessih, Fink, & August, 2021). Liposome preparation is carried out by using mechanical force to form 5.
FIG. 1: VARIOUS TYPES OF LIPOSOMES
The structural arrangement within the liposome entails that there is an aqueous core surrounded by one or more layers of phospholipid bilayers. The phospholipid heads, being hydrophilic, interact with the aqueous medium both inside and outside the bilayer, whereas their tails interact with each other. Such an arrangement makes the membrane structure quite stable. The hydrophilic medicines remain enclosed in the aqueous medium inside, whereas hydrophobic medicines exist within the lipid bilayer. The liposomes can have either one bilayer or more than one layer 6. Liposomes could be unilamellar, having a single bilayer structure, or multilamellar, having a number of concentric layers similar to an onion, each having a bilayer structure. Cholesterol can be added to the liposomal membrane structure to increase rigidity, stability, and regulation of permeability of the membrane. Liposome size varies based on different methods of production from a few nanometers to even micrometers. Bilayer membrane structure is similar to biological membranes of living cells; therefore, it has better biocompatibility 7.
Types of Liposomes:
Small Unilamellar Vesicles (SUVs): Small unilamellar vesicles (SUVs) are liposomes that consist of one phospholipid bilayer, and their shape is spherical and small, typically between 20 to 100 nm in diameter. These vesicles are prepared using methods such as sonication and extrusion of multilamellar vesicles. Due to their large surface area, SUVs can easily load drugs 8.
Large Unilamellar Vesicles (LUVs): These can be used for incorporating both hydrophilic drugs into the internal aqueous compartment and lipophilic drugs into the lipid bilayer. Large unilamellar vesicles find wide application in drug delivery vehicles, gene transfection, and other target-oriented therapies because of their increased loading capacity and greater stability than small unilamellar vesicles 9.
Gaint Unilamellar Vesicles (GUVs): Giant unilamellar vesicles are liposomes that have only one phospholipid bilayer and their diameter is larger than 1 micron. They resemble biological cell membranes very much and therefore find application in the field of studying cell membrane structure, for drug delivery studies, microscopy, and transport studies 10.
Multilamellar Vesicles (MLVs): Multilamellar Vesicles (MLVs) are liposomes that consist of concentric phospholipid bilayers stacked upon each other like an onion. These liposomes contain aqueous layers sandwiched between them. MLVs are usually large (0.1–10 µm) and can enclose hydrophilic and hydrophobic drugs.
Oligolamellar Vesicles (OLVs): The oligolamellar vesicles consist of liposomes that comprise a number of concentric lamellae surrounding an aqueous core. The liposomes exhibit a medium size along with good drug loading capability and thus are successfully used in controlled delivery applications 11.
Multivesicular Liposomes (MVLs): Multivesicular liposomes are vesicles made of lipids that contain multiple aqueous compartments within the same outer layer. They offer sustained drug release, efficient drug loading, and have extensive applications in pharmaceutical delivery systems.
Prepration Methods:
Thin Flim Hydration Method: Film Hydration Technique: First, phospholipids and cholesterol are dissolved using organic solvent. This organic solvent is then evaporated off under vacuum pressure, resulting in the formation of a lipid film. Hydration of this lipid film leads to the formation of liposomes 12.
Sonication Method: The sonication technique involves breaking up multilamellar vesicles into smaller, homogeneous liposomes through the use of ultrasonic vibrations. The high-frequency sound waves decrease the size of the vesicles. It is easy and quick, making it the most common technique for lab-based production of liposomes 13.
Reverse Phase Evaporation Method: The reverse phase evaporation technique is employed for the production of liposomes with high efficiency of aqueous entrapment. The phospholipid layer is dissolved in an organic solvent and combined with aqueous phase; subsequently, emulsification takes place followed by dehydration of the mixture to form LUVs 14.
Ethanol Injection Method: The method of ethanol injection involves the use of ethanol to dissolve phospholipids and then injecting this solution into the aqueous medium while stirring. This causes the dispersion of ethanol, which results in the spontaneous formation of liposomes 15.
Detergent Removal Method: Liposome preparation through the method of detergent elimination uses detergents to dissolve phospholipids and produce mixed micelles. Lipid self-assembly occurs due to progressive extraction of the detergent by dialysis, gel filtration, or adsorption. This method can be used to study incorporation of proteins 16.
Microfluidization Method: Liposome formation via microfluidization involves the use of pressure to pass the lipid and aqueous solutions through thin channels. The process results in the formation of uniform and smaller vesicles due to collision and shear forces 17.
Advantages: Liposomes are extensively used in formulation sciences due to their ability to be biocompatible and biodegradable, thus being safe to use for medicinal purposes. They have the capacity to trap hydrophilic drugs in their aqueous core and lipophilic drugs in their lipid bilayer. The main purpose served by liposomes is that they help in decreasing the toxicity of drugs as well as preventing any adverse reactions to occur as a result of improper distribution of the drug inside the body. Liposomes are also very efficient in stabilizing highly sensitive drugs 18.
Disadvantages: Even with numerous advantages, there are specific disadvantages associated with liposomes. The preparation cost of liposomes is high due to high costs of phospholipids and advanced methods of preparation. The physical and chemical instability of liposomes could lead to aggregation, fusion, oxidation, and hydrolysis when stored for a considerable period. Leakage of the loaded drugs could lead to loss of efficacy. Liposomes have a limited lifespan and require particular storage conditions. It is challenging to manufacture liposomes on a large scale and sterilize them. In some instances, drug loading efficiency is poor 19.
Applications: Liposomes are used as the carriers for the active agents that may be hydrophilic or hydrophobic in nature. Liposomes carry the active agents (antioxidants, anti-inflammatory agents, antimicrobial or anti-cancer agents) to the selected target cells without degradation. Thus, liposomes help deliver higher concentrations of the drugs to the target sites.
Liposomes are extensively employed in the field of medicines and pharmaceuticals as vectors for delivering medicines, decreasing their toxicity levels, and increasing their efficiency. They are utilized in various fields like cancer treatment, vaccine development, gene delivery, and antimicrobial treatments. In cosmetology, they serve to increase the penetration of active components through the skin barrier 20. The stability, bioavailability, and targeted administration of phytochemicals are being positively impacted by recent advancements in nanocarrier systems, such as polymeric nanoparticles and antibody-conjugated formulations. Additionally, new pharmaceutical approaches for PCOS control have been made possible by our growing understanding of the manipulation of gut microbiota, functional genomics, and proteomic interactions 21.
CONCLUSION: Liposomes have been extensively considered as a vehicle for many types of drugs. As the immediate application of liposomes in the medical practice inspires the researchers to develop new liposomes to be used in diagnosis and treatment, in a broad spectrum of illnesses, the ability to control the behavior of the drug within the body, and its toxicity are the two key factors that should be taken into account when designing liposomal. Since the first use of liposome technology as drug carriers by Gregoriadis et al., phospholipid-based carriers have found applications as carriers of both hydrophilic and hydrophobic components in the pharmaceutical, cosmetic and food industries. The characteristics of liposomes will vary widely depending on the nature of their phospholipids, surface charge, size and mode of production. Liposomes can either be made up of natural or synthetic phospholipids but are still biocompatible and non-toxic.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Sakshi and Singh RK: “Liposomes - An Updated overview.” Int J Pharma Prof Res 2024; 15(1): 119–25.
- M. Allen T, Hansen C, Martin FCR and AYY: Liposomes containing synthetic lipid derivatives of poly (ethylene glycol) show prolonged circulation half-lives in-vivo. Biochim Biophys Acta 1991; 1066: 29–36.
- Kumar JP, Singh RK, Mm C, Rao P and Tanniru R: Tropical Journal of polymeric nano-carriers for drug delivery systems in cancer therapy - an exhaustive review. Trop J Pharm Life Sci 2025; 12(1): 33–44.
- Mehta AR, Lopez-Berestein GB, Hopfer RC, Km C and Rlj AA: liposomal amphotericin b is toxic to fungal cells but not to mammalian cells. Biochim Biophys Acta 1984; 770: 230–4.
- Luisi SB and Barbachan JJD: Behavior of Human Dental Pulp Cells Exposed to Transforming Growth Factor-Beta1 and Acidic Fibroblast Growth Factor in Culture. Basic Res Behav 2007; 33(7): 833–5.
- Bangham AD: Liposomes: the Babraham connection. Chem Phys Lipids 1993; 64: 275–85.
- Amarnath Sharma: USS Liposomes drug Deliv Prog limitations. International Journal of Pharmaceutics. 1997; 154.
- Jokerst JV, Lobovkina T, Richard N Zare & Sanjiv S Gambhir: Nanoparticle PEGylation for imaging and therapy R eview. Repr orders, please contact Repr 2011; 6: 715–28.
- Multiple Injections of Pegylated Liposomal Doxorubicin: Pharmacokinetics and Therapeutic Activity. Greg J R Charrois Theresa M Allen 2003; 306(3): 1058–67.
- Sara Nicolia, Giulio Ferrarib, Marinella Quartaa and Claudio Macalusob PS: In-vitro transscleral iontophoresis of high molecular weight neutral compounds. Eur J Pharm Sci 2008; 6: 486–92.
- Zook JM and Vreeland WN: Effects of temperature, acyl chain length, and flow-rate ratio on liposome formation and size in a microfluidic hydrodynamic focusing device. www.rsc.org/softmatter 2010; 1352–60.
- Magee WE, Goff CW, Schoknecht J, Smith MD and Cherian K: The interaction of cationic liposomes containing entrapped horseradish peroxidase with cells in culture. J Cell Biol 1974; 63(3): 492–504.
- Spden TRJA, Ason JUDTM, Ones NISJ, Owe JAL, Kaugrud ØYS and X LIIL: Chitosan as a Nasal Delivery System : The effect of chitosan solutions on in-vitro and in-vivo mucociliary transport rates in human turbinates and volunteers. Conf Rep 1997; 86(4): 509–13.
- Vance ME and Marr LC: Exposure to airborne engineered nanoparticles in the indoor environment. Atmos Environ [Internet]. 2014; 1–7. Available from: http://dx.doi.org/10.1016/j.atmosenv.2014.12.056
- Allen TM, Menezes DEL De and Moase EH: Immunoliposome-mediated targeting of anti-cancer drugs in-vivo. Ther Monoclonals 2000; (1993): 6444–8.
- Yao Y: Accelerated blood clearance phenomenon upon cross-administration of PEGylated nanocarriers in beagle dogs. Int J Nanomedicine 2015; 3533–45.
- John H. Venable and Richard Coggeshall: A simplified Lead citrate stain for use in electron microscopy. Dep Anat 1965;
- Liu Y, Hu Y and Huang L: Biomaterials In fl uence of polyethylene glycol density and surface lipid on pharmacokinetics and biodistribution of lipid-calcium-phosphate nanoparticles. Biomaterials [Internet]. 2014; 35(9): 3027–34. Available from: http://dx.doi.org/10.1016/j.biomaterials.2013.12.022
- Henriksen I, Vhgen SR, Sande SA, Smistad G and Karlsen J: Interactions between liposomes and chitosan II: effect of selected parameters on aggregation and leakage. Int J Pharm 1997; 146.
- Bianca P, Souza S, Marquez L, Luiz J, Anjos V and Antonio S: Journal of Drug Delivery Science and Technology Enhanced asiaticoside skin permeation by Centella asiatica-loaded lipid nanoparticles: Effects of extract type and study of stratum corneum lipid dynamics. J Drug Deliv Sci Technol 2019; 50(8): 305–12. Available from: https://doi.org/10.1016/j.jddst.2019.01.016
- Singh RK, Sharma K, Gupta MP, Ravikant, Rajpurohit MS and Saxena V: Exploring nanotechnology with traditional herbal pharmacology: nano carrier-based phytochemical delivery in PCOS treatment. Nanotechnol Next Gen from Treat to Transform Polycystic Ovary Syndr 2025; 49–78.
How to cite this article:
Dar JA, Kushwaha S, Singh RK and Sharma T: Innovations in liposomal drug delivery: a comprehensive review of structure, preparation, applications, and therapeutic potential. Int J Pharmacognosy 2026; 13(5): 431-35. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.13(5).431-35.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
6
431-435
627 KB
3
English
IJP
Junaid Ahmad Dar, Satpal Kushwaha, Ranjan Kumar Singh * and Tanya Sharma
Faculty of Pharmaceutical Sciences, Mewar University, Gangrar, Chittorgarh, Rajasthan, India.
rxsingh41800557@gmail.com
18 April 2026
06 May 2026
12 May 2026
10.13040/IJPSR.0975-8232.IJP.13(5).431-35
15 May 2026