INHIBITION OF PROTEIN (ENZYME) DHNA BY USING MOLECULAR DOCKING
HTML Full TextINHIBITION OF PROTEIN (ENZYME) DHNA BY USING MOLECULAR DOCKING
Pawan Kant * 1, Saugata Hazra 1, Rohit Kumar Bijauliya 2 and Dilip Kumar Chanchal 2
Department of Biotechnology 1, Indian Institute of Technology Roorkee, Roorkee - 247667, Uttarakhand,
India.
Department of Pharmacognosy 2, Institute of Pharmacy, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: Aim of this study was to generate a model of DHNA using protein sequence and homology modeling and then dock the modeled protein with an inhibitor. Molecular docking is a frequently used method in structure-based rational drug design. It is used for evaluating the complex formation of small ligands with large biomolecules, predicting the strength of the bonding forces and finding the best geometrical arrangements. For inhibition of final model built from the swiss model by using target sequence of DHNA from Shigella flexneri and template sequence from E. coli choose 4 types of ligand molecule in which 2 molecules (2-amino pyrimidine and neopterin) selected for docking with the help of Autodock Vina (software). And the final result is shown in docking result 1 and 2 respectively. Docking results shows mean binding energy -3.42 by neopterin and -2.77 by 2-amino, pyrimidine. Neopterin shows high mean binding energy in both of ligands so we can use neopterin as a strong inhibitor of DHNA.
| Keywords: |
DHNA enzyme, Microorganism, Molecular docking
INTRODUCTION: Folate Biosynthesis Pathway: Folate cofactors are important for living systems. Most of the microorganisms synthesize folates de novo, but in mammals, folate synthesis does not occur. Hence, the folate biosynthetic pathway is a perfect target for antimicrobial agents. It required for the transfer of one-carbon units in several metabolic steps, including the key methylation of dUMP to give dTMP, an essential nucleotide for DNA synthesis. Most microorganisms can synthesize the required folates from the simple precursor GTP, p-aminobenzoate (pABA) and glutamate. But folate biosynthetic pathway absent in mammals because of lack of all three enzymes which works in the middle of folates synthesis. So mammals take folates through diet 1.
Folate Biosynthetic Pathway:
FIG. 1: IT SHOWS THE SIMPLE FOLATE BIOSYNTHETIC PATHWAY IN WHICH GUANINE TRIPHOSPHATE (GTP) THOUGH USING DIFFERENT ENZYMES IN EACH STEPS LIKE DIHYDRONEOPTERINALDOLASE (DHNA); PYRO-PHOSPHOKINASE (PPPK); PARA-AMINOBENZOIC ACID (PABA); DIHYDROPTEROATE SYNTHASE (DHPS); DIHYDROFOLATE SYNTHASE (DHPS) 1
Enzyme: Dihydroneopterinaldolase (DHNA) is the first enzyme in the pathway and has a major role of the three enzymes that are absent in mammals and therefore an attractive target for developing antimicrobial agents 2.
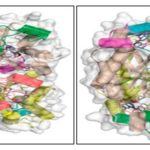
Structure, Function, Reaction, Inhibitors: DHNA has a hollow cylinder structure, 70Å in height, an outer diameter of 65 Å and an inner diameter of 13Å. Two tetrameric rings are placed head to head forming an octamer of cylindrical shape. The N and C termini are located on the top and bottom of the structure.
The figure represents the DHNA crystal structure with different subunits represented as distinguished with colors. The HP molecules are shown as stick models in atomic color scheme (Carbon in black, nitrogen in blue, and oxygen in red).
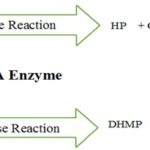
DHNA is a unique enzyme which works as aldolase as well as epimerase. It works as a unique type of aldolase it requires neither the construction of a Schiff’s base between the substrate and enzyme nor metal ions for catalysis. When it works as aldolase then conversion of DHNP to 6-hydroxymethyl-7, 8-dihydropterin (HP) with the generation of glycoaldehyde (GA) and the epimerization of 7, 8-dihydroneopterin (DHNP) to 7, 8-dihydromonapterin (DHMP).
FIG. 2: TWO VIEWS (SIDE, ON THE LEFT; TOP, ON THE RIGHT) OF THE SaDHNA-HP OCTAMER (PDB ENTRY 2DHN)
The reaction of DHNA it works as aldolase and epimerase both.
FIG. 3: IT SHOWS ONLY ONE ENZYME DHNA (DIHYDRO-NEOPTERINALDOLASE) PROCEEDS TWO TYPE OF MECHANISM FIRST ALDOLASE REACTION WHICH FORMS HP(6-HYDROXYMETHYL-7, 8-DIHYDROPTERIN) WITH GA (GLYCOLATE ALDEHYDE) BY DHNP (7,8-DIHYDRO-D-NEOPTERIN) AND WITH SIMILAR INTER-MEDIATE EPIMERASE RECTION GET PROCEEDS DHMP (7, 8-DIHYDRO-L-MONAPTERIN) MOLECULE GET FORMS 2
Inhibitors: Neopterin (oxidized form of DHNP) and monopterin (oxidized form of DHMP) are known inhibitors of DHNA. After the oxidation of DHNP and DHMP forms a double bond between C7 and N8, which may make the protonation of N5 much harder so that NP and MP may not undergo chemical reaction catalyzed by DHNA. Thus, these two blocks DHNA catalysis. 2-amino pyrimidine, a substrate analog, forms the same hydrogen bonding with the enzyme as the substrate and is also a good inhibitor.
Organism from Where the Enzyme is taken: Scientific name of organism Shigella flexneri (Uniprot id-P0AC18).
Classification of Organism:
Kingdom: Bateria
Phylum: Proteobacteria
Class: Gammaproteobacteria
Order: Enterobacteriales
Genus: Shigella
Species: flexneri 3
Some Reported Work on DHNA in Other Organisms:
TABLE 1: SOME REPORTED WORK ON DHNAS AND ITS ORGANISM 8, 9, 10, 11, 12
| S. no. | Enzymes | Organism | Work | Reference |
| 1 | EcDHNA | E. coli | First identified of DHNA | Mathis and Brown 1970 |
| 2 | SaDHNA | S. aureus | Crystal structure of DHNA complex with the product HP | Hennig
and coworkers 1998 |
| 3 | SaDHNA | S. aureus | aldolase and epimerase activities and determined the steady-state kinetic parameters for both reactions | Haussmann and coworkers 1998 |
| 4 | SaDHNA | S. aureus | the total sequential resonance assignment of the 110-kDa homo-octomeric SaDHNA | Wu thrich 2000 |
| 5 | SaDHNA | S. aureus | pKaof N5 of SaDHNA-bound 7,8-dihydrobiopterin
by Raman spectroscopy |
Deng and coworkers 2000 |
| 6 | SaDHNA | S. aureus | protonation of the reaction
intermediate prefers the pro-S position |
Illarionova
and coworkers 2002 |
The objective of Study: Objective of the study was to generate a model of DHNA using protein sequence and homology modeling and then dock the modeled protein with an inhibitor.
METHDOLOGY: Using uniprot id P0AC18 of Dihydroneopterin-aldolase from Shigella flexneri run FASTA for align sequence of that enzyme. Then using blasta do pBlast of fasta sequence in pdb formate. I have got chain A, atomic resolution crystal structure of E. coli dihydroneopterin-aldolase in complex with neopterin. It gives a maximum, total score of alignment is 246 with 100% quality cover, identity and E value 2e-84 and accession no. is 2O90_A. Using 2O90_A accession no. or PDB id in RCSB download template .pdb file. Then in swiss modeler, put the target template in FASTA format and upload a .pdb file of the template and build the model. The model obtained was docked with an appropriate ligand using Autodock Vina 4, 5.
RESULTS AND DISCUSSION:
FIG. 4: FIRST RED COLOUR LINE REPRESENT 100% IDENTITY IN E. COLI AND 85% IDENTITY IN YERSINIA PESTRIS
FIG. 5: TOTAL 18 RESULT SPECIES HAVE FOUND WHICH SHOWS THE PRESENCE OF DHNA ENZYME
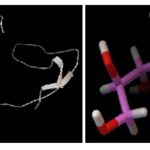
FIG. 6: FINAL MODEL BUILT FROM THE SWISS-MODEL WITH THE HELP OF TARGET SEQUENCE OF DHNA FROM SHIGELLA FLEXNERI AND TEMPLATE SEQUENCE FROM E. COLI. AND IT'S FRONT OF LIGAND (2-AMINO, PYRIMIDINE) STRUCTURE 7
Docking Result 1 of Fig. 6 Protein and Ligand (2-amino, pyrimidine):
FIG. 7: FINAL MODEL BUILT FROM THE SWISS-MODEL WITH THE HELP OF TARGET SEQUENCE OF DHNA FROM SHIGELLA FLEXNERI AND TEMPLATE SEQUENCE FROM E. COLI. AND IT’S FRONT OF LIGAND (NEOPTERIN) STRUCTURE
Docking Result 2 of Fig. 7 Protein and Ligand (Neopterin):
CONCLUSION: For inhibition of final model built from the swiss model by using target sequence of DHNA from Shigella flexneri and template sequence from E. coli choose 4 types of ligand molecule in which 2 molecules (2-aminopyrimidine and neopterin) selected for docking with the help of Autodock Vina (software).
And the final result is shown in docking results 1 and 2 respectively. Docking result shows mean binding energy -3.42 by neopterin and -2.77 by 2-amino, pyrimidine. Neopterin shows high mean binding energy in both of ligands so we can use neopterin as a strong inhibitor of DHNA.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Dittrich S, Mitchell SL, Blagborough AM,Wang Q, Wang P, Sims PF and Hyde JE: An atypical orthologue of 6-pyruvoyltetrahydropterin synthase can provide the missing link in the folate biosynthesis pathway of malaria parasites. Mol Microbiol 2008; 67: 609-18.
- Wang Y, Li Y, Wu Y and Yan H: Mechanism of dihydro-neopterinaldolase NMR, equilibrium and transient kinetic studies of the Staphylococcus aureus and Escherichia coli The febs J 2007; 274: 2240-52.
- Castellani, Chalmers, Flemmingscheutz and Nancy: a strockbine, genus Escherichia family enterobacteriaceae. 1919; 24: 607-24.
- Magnani G, Lomazzi M and Peracchi A: Completing the folate biosynthesis pathway in Plasmodium falciparum: p-aminobenzoate is produced by a highly divergent promiscuous amino-deoxychorismatelyase. Biochem J 2013; 2: 149-55.
- Klaus SMJ, Wegkamp A, Sybesma W, Hugenholtz J, Gregory JF and Hanson AD: A nudix enzyme removes pyrophosphate from dihydroneopterin triphosphate in the folate synthesis pathway of bacteria and plants. The Journal of Biological Chemistry 2005; 280: 5274-80.
- Blaszczyk J, Li Y, Gan J, Yan H and Ji X: structural basis for the aldolase and epimerase activities of Staphylococcus aureus dihydro-neopterin aldolase. J Mol Biol 2007; 1: 161-69.
- Sanders WJ, Nienaber VL, Lerner CG, McCall JO, Merrick SM, Swanson SJ, Harlan JE, Stoll VS, Stamper GF, Betz SF, Condroski KR, Meadows RP, Severin JM, Walter KA, Magdalinos P, Jakob CG, Wagner R and Beutel BA: Discovery of potent inhibitors of dihydro-neopterinaldolase using Crystal LEAD high-throughput X-ray Crystallographic Screening and Structure-Directed. J Med Chem 2004; 47:1709-18.
- Mathis JB anf Brown GM: The biosynthesis of folic acid. XI. Purification and properties of dihydroneopterin-aldolase. J Biol Chem 1970; 245: 3015-25.
- Hennig MD, Arcy A, Hampele IC, Page MGP, Oefner C and Dale GE: Crystal structure and reaction mechanism of 7,8-dihydroneopterin aldolase from Staphylococcus aureus. Nat Struct Biol 1998; 5: 357-62.
- Salzmann M, Pervushin K, Wider G, Senn H and Wu thrich K: NMR assignment and secondary structure determination of an octameric 110 kDa protein using TROSY in triple resonance experiments. J Am Chem Soc 2000; 122: 7543-48.
- Deng H, Callender R and Dale GE: A vibrational structure of 7, 8-dihydrobiopterin bound to dihydroneopterin aldolase. J Biol Chem 2000; 275: 30139-43.
- Illarionova V, Eisenreich W, Fischer M, Haussmann C, Romisch W, Richter G and Bacher A: Biosynthesis of tetrahydrofolate: stereochemistry of dihydroneopterin aldolase. J BiolChem 2002; 277: 28841-47.
How to cite this article:
Kant P, Hazra S, Bijauliya RK and Chanchal DK: Inhibition of protein (enzyme) DHNA by using molecular docking. Int J Pharmacognosy 2018; 5(10): 682-87. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(10).682-87.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
7
682-687
843
1453
English
IJP
P. Kant *, S. Hazra, R. K. Bijauliya and D. K. Chanchal
Department of Biotechnology, Indian Institute of Technology Roorkee, Roorkee, Uttarakhand, India.
pawan.iitrbiotech1315@gmail.com
05 August 2018
24 September 2018
30 September 2018
10.13040/IJPSR.0975-8232.IJP.5(10).682-87
01 October 2018