INDOLE-3-CARBINOL: A REVIEW ON CURRENTTRENDS AND FUTURE PROSPECTS
HTML Full TextINDOLE-3-CARBINOL: A REVIEW ON CURRENTTRENDS AND FUTURE PROSPECTS
Tisha Pandav and Ashwin Mali *
Department of Pharmaceutics, Bharati Vidyapeeth Deemed University, Poona College of Pharmacy, Erandawane, Pune, Maharashtra, India.
ABSTRACT: Indole-3-carbinol, a phytoactive constituent obtained from cruciferous vegetables like Brussels, sprouts, cabbage and cauliflower. It has chemopreventive properties supported by numerous studies carried out to prevent breast, lung, prostate, leukemia and ovarian cancer. Indole-3-carbinol possesses therapeutic benefits like antimicrobial, neuroprotective and antitumor properties. However, it has got poor oral bioavailability due to limitations like metabolic instability in acidic media, thermal instability, etc. The review focuses on the chemistry, anticancer mechanism, limitations, bioavailability, pharmacokinetics, and toxicities studies related to I3C. It also focuses on analytical methods and current approaches for developing an improved delivery system for Indole 3 carbinol by overcoming bioavailability and toxicity limitations. I3C has reported showing limited efficacy in clinical trials due to its poor oral bioavailability. Hence administration of I3C from alternative routes is explored by formulating it as nanoparticles, nanocapsules, suspensions, microemulsions, liposome.
Keywords: Indole-3-carbinol, Brassica; Cruciferae, Anticarcinogen, DIM
INTRODUCTION: Numerous epidemiological studies suggest that plant bio-actives obtained from cruciferous vegetables belonging to the brassica species family are a rich source of bioactive components like glycosinolates, flavonoids and vitamins 1. The various sources of cruciferous vegetables are Brussel sprouts, cabbage, cauliflower, collards, kale, mustard, rape, etc. These plant bio-actives were recognized first in the early 1990s for their chemopreventive properties which were later supported by various studies carried out for the prevention of breast 2, lung 3, prostate, leukemia 4 and ovarian cancer 5. I3C also causes gene modulation by various mechanisms like apoptosis, xenobiotic metabolism, anti-oxidant, transcription factor, cell cycle, inflammation and angiogenesis 6.
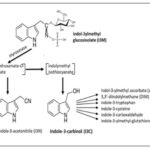
A study was carried out on 3 types of cruciferous vegetables like cabbage, brussel sprouts and cauliflower. Follow-up studies have attributed this activity to the metabolic products of glucosinolates, a class of secondary sulfur-containing metabolites produced by crucifers 7. Glucosinates and their derived forms; isothiocyanates possess properties like antimicrobial, neuroprotective and antitumor properties. Indo methyl glucosinolate (glucobrassicin) in presence of enzyme myrosinase which is activated when a vegetable is chewed or macerated and leads to hydrolysis which results in the formation of products like indole-3-carbinol(I3C),3,3'diiodolyl-methane (DIM) and indole acetonitrile 8. The strongest inducing activity was shown by Indole-3-carbinol.
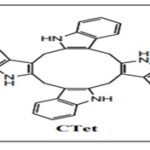
Although I3C possess several advantages, the limitations of I3C involved its highly unstable nature in the presence of acidic conditions of the stomach and its rapid oligomerization to multiple products like DIM [bis(3’-indolyl) methane, a dimer], ICZ (indole[3,2b]- carbazole, a dimer), LTr1 (a liner trimer), CTr (a cyclic trimer) and CTet (a cyclic tetramer).
The intrinsic instability in acidic medium was said to arise from the presence of vinyl hemiaminal moiety' of indole ring, which rapidly leads to the acid-catalyzed degradation and polymerization of I3C into various dimers, trimers and tetramers 9. ICZ,LTr1, and CTr are mainly associated with ERα and AhR; CTet suppressed breast cancer growth 9.
TABLE 1: NUTRITIONAL COMPOSITION OF RAW CRUCIFEROUS VEGETABLES PER 100 G OF FRESH PRODUCE 7
| Broccoli | Kale | cauliflower | Brussel sprouts | Mustard spinach | |
| Proximates | |||||
| Water | 89.3 | 84.04 | 92.07 | 86 | 92.2 |
| Energy | 34 | 49 | 0.25 | 43 | 22 |
| Protein | 2.82 | 4.28 | 1.92 | 3.38 | 2.2 |
| Total lipid | 0.37 | 0.93 | 0.28 | 0.3 | 0.3 |
| Carbohydrate | 6.62 | 8.75 | 4.97 | 8.95 | 3.9 |
| Dietary fiber | 2.6 | 0.6 | 2 | 3.8 | 2.8 |
| Sugar | 1.7 | 2.26 | 1.91 | 2.2 | N/A |
| Minerals | |||||
| Calcium. Ca | 47 | 150 | 22 | 42 | 210 |
| Iron | 0.73 | 1.47 | 0.42 | 1.4 | 1.5 |
| Magnesium | 21 | 47 | 15 | 23 | 11 |
| Phosphorous | 66 | 92 | 44 | 69 | 28 |
| Potassium | 316 | 491 | 299 | 389 | 449 |
| Sodium | 33 | 38 | 30 | 25 | 21 |
| Zinc | 0.41 | 0.56 | 0.27 | 0.42 | 0.17 |
| Vitamins | |||||
Vitamin C |
89.2 | 120 | 48.2 | 85 | 130 |
Thiamin |
0.071 | 0.11 | 0.05 | 0.139 | 0.068 |
Riboflavin |
0.117 | 0.13 | 0.06 | 0.09 | 0.093 |
Niacin |
0.639 | 1 | 0.507 | 0.745 | 0.678 |
Vitamin B6 |
0.175 | 0.271 | 0.184 | 0.219 | 0.153 |
Folate DFE |
0.063 | 0.141 | 0.057 | 0.061 | 0.159 |
Vitamin B12 |
0 | 0 | 0 | 0 | 0 |
Vitamin A |
0.031 | 0.5 | 0 | 0.038 | 0.495 |
Vitamin E |
0.78 | 1.54 | 0.08 | 0.88 | N/A |
Vitamin D |
0 | 0 | 0 | 0 | 0 |
Vitamin K |
0.101 | 0.704 | 0.015 | 0.177 | N/A |
| Lipids | |||||
Saturated fatty acids |
0.039 | 0.091 | 0.13 | 0.062 | 0.015 |
Monounsaturated fatty acids |
0.011 | 0.052 | 0.034 | 0.023 | 0.138 |
Polyunsaturated fatty acids |
0.038 | 0.338 | 0.031 | 0.153 | 0.057 |
Trans fatty acids |
0 | 0 | 0 | 0 | 0 |
Cholesterol |
0 | 0 | 0 | 0 | 0 |
Indole 3 carbinol is a phytoactive constituent obtained from cruciferous vegetables like Brussels, sprouts, cabbage and cauliflower.
Type of Inducers of Activity: Indo methyl glycocinate upon hydrolysis in presence of myrosinase enzyme formed I3C, DIM and indole-3-acetonitrile. I3C showed strongest inducing activity 10. The anticancer activity shown by cruciferous Brassica vegetables like Brussel sprouts, cabbage, cauliflower are most likely mediated through “bioactive compounds” that induce a variety of physiological processes including direct or indirect antioxidant action, detoxifying enzymes, inducing apoptosis and cellcycle regulation 11. Preventive effect of I3C in animal models of chemically induced carcinogenesis of various tissues including colon, liver, lung mammary gland, cervix endometrium and skin 10 and respiratory papillomatosis 12.
Indole 3 Carbinol: The chemical name of Indole-3-carbinol is 1H-Indole-3-methanol (9CI); indole-3-methanol (8CI) with a molecular weight of 147.18 g /mol. It structurally belongs to the heterocyclic class and is commercially available as an off-whitesolid 13, having a melting point and boiling point around 96-99 °C (Sigma, 1998) (Aldrich Chemical Co) and 278.28 °C respectively. It was considered to be stable between 2-80 °C. The physical and chemical properties were reported by Pubchem and Drug bank.
It's water solubility was reported to be around 3.75mg/ml, the estimated pKa value was found to be 15.1 (strongest acidic) and -2.7(strongest basic). It is found to be unstable in an acidic medium, where it rapidly undergoes acid-catalyzed degradation and forms multiple condensation products.
Solubility was reported to be around 7µg/ml. National Center for Biotechnology Information.
Pubchem database. Indole-3-carbinol CID = 3712
Anticancer Mechanism of I3C: The anticancer mechanism of I3C included, Modulation of epigenetic alteration as mentioned below –
- DNA methylation
- Histone modification
- Aberrant expression of miRs deregulation of DNA methylation
DNA is hypermethylated via DNA methyl- transferase (DNMT) mediated addition of a methyl group at the 5' position of cytosine within CpG dinucleotides, resulting in transcriptional silencing of tumor suppressor genes. On the other hand, DNA hypomethylation, resulting from DNMT inactivation or feeding of methyl-deficient diets, may lead to over expression of proto-oncogenes involved in carcinogenesis 14, 15.
Histone modification is produced by acetylation, methylation, phosphorylation and ubiquitination which occur systemicallyand regulate processes like gene transcription, DNA replication and DNA repair 16.
Alterations occur as a function of histone-modifying complexes by removal of acetyl group in the presence of histone deacetylases (HDACs) or by methylation of a lysine residue of histone-by-histone methyltransferase, which leads to transcriptionally re-expressed chromatin and decreases gene transcription.Onthe contrary, methylation (H3K4, H3K27, H4K20) is associated with transcriptionally active chromatin. DIM is a potent inhibitor of aberrant histone modification. DIMs exposure to androgen-dependent LnCAPn prostate cancer cell line leads to an increase in H3K4Me3 in the promoter region of TGFBR1 and CYR61 genes 17. Another study carried out on Androgen independent P3C and androgen-dependent LnCAP prostate cell line led to drug decrease HDAC activity and suppressed expression of HDAC2 without affecting HDAC1, HDAC3, HDAC6, or HDAC8 levels.
In the same study, they reported that I3C inhibits the activity of HDAC in LNCa Pcells but not in PC-3 cells. In human colon cell line studies, DIM selectively led to the induction of class 1 histone deacetylases in-vivo and in-vitro. They also enhanced the expression ofcyclin-dependent kinase inhibitors p21WAF1 and p27KIP2 and triggered cell cycle arrest in G (2) phase 18.
Aberrant expression of miRs Deregulation of DNA Methylation: Micro RNAs represent a class of small 21-23 nucleotides non-coding RNAs and are part of post-transcriptional control of gene expression and are also important regulators of gene expression 19. Specific targeting of miRNAs by natural agents could open newer possibilities for the prevention and treatment of cancer 20. The first report on dietary feeding of I3C led to significant suppression of miR21, miR31. MiR130, miR146b, and miR377 were overexpressed due to vinyl carbamate-induced lung cancer. Further studies on miRs revealed miR21’s potential targets are the tumor suppressor genes PTEN, pdcd4 and RECK 21. The further results in the case of pancreatic cancer model and hepatocellular carcinoma resulted in the downregulation of miR21 by I3C and upregulation of pdcd4 and PTEN, respectively. Hence, enhanced sensitivity of the pancreatic cell to gemcitabine and repression of the ART pathway was observed in the case of pancreatic cancer models and hepatocellular carcinoma.
More research is being carried out on I3C because of its antioxidant properties. In the case of mouse lung carcinogenesis, chemically induced deregulation of miRNA expression occurs by upregulation of miR21, miR31, mi130a, miR146 and miR377, which can be prevented by I3C 21. Downregulation of microRNA expression in the lungs of rats exposed to the cigarette smoke carcinogen. Cigarette smoke-induced alteration in lung tissues microRNA expression pattern and consequences of these changes are partially reversed by I3C 22.
Also, further studies of DIM in the pancreatic cancer cell line resulted in the induction of re-expression of miR200, thus reducing the expression of 2EB1, slug, and vimentin genes that are involved inthe drug resistance 23. The re-expression of miR21 and induction of its target genes which are involved in cell survival and regulation, maybe responsible for the chemopreventive activity of I3C.
Indole-3-carbinol Structure-Activity Relationship: I3C is seen as a potential antitumor agent because it possesses anti-tumor properties.
The structure of I3C consist of vinyl hemiaminal moiety of indole group, which is highly unstable in acidic medium and hence leads to acid-catalyzed degradation and polymerization to form various oligomeric products like DIM,ICZ, LTr1, CTr and CTet9.
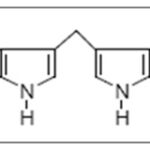
FIG. 1: DIM
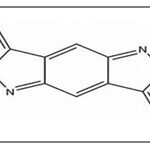
FIG. 2: ICZ
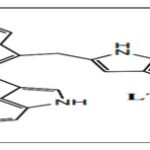
FIG. 3: LTr
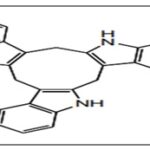
FIG. 4: CTr
FIG. 5: CTet
Each of these oligomeric products of I3C possessits chemopreventive effect; hence the collective chemopreventive effect of I3C is attributed to these metabolic products.
DIM is found to be the major condensation product. It exhibits anti-proliferative and apoptotic effects and cell cycle arrest by targeting Akt-NF kappa B signaling, caspase activation, cyclin-dependent kinase activities, estrogen metabolism, estrogen receptor signaling, endoplasmic reticulum stress, and BRCA geneexpression 10, 24. The other oligomers like ICZ, LTr1, and CTr are associated with ERα and AhR (aryl hydrocarbon receptor) 25, 26. CTr is a strong ligand for estrogen receptors and increases estrogen-dependent MCF-7cell proliferationbut not independent MDA-MB 231 cells, which suggests that it possesses estrogenicactivity 27.
CTet is responsible for suppressing breast cancer growth by inhibiting the expression of (CDK)6 Cyclin-dependent kinase and other cell regulatory proteins with 5-fold higher potency than I3C.
I3C Chemistry: The compound that sets cruciferous foods apart from other vegetables is glucosinolates. Glucosinolates are beta thio-glucoside-N-hydroxy sulfates with a side chain and a sulfur linked beta pyronase moiety glucosinolate scan be divided into 3 groups based upon the structure of amino acid precursors i)aliphatic- methionine, isoleucine, leucine and valine ii) aromatic- derived from phenylalanine and tyrosine iii) indole - derived from tryptophan. Myronase is physically separated from glucosinolates in the intact plant cells, catalysis the hydrolysis ofglycosinolates 7.
The concentration of glucosinolates in vegetables varies with factors like i) cultivator selector ii) growing condition iii) preparation or processing, plant cells on damage insert glycosinolates which in the presence of myrosinase results into the active derivative.
The degree to which this occurs depends upon i) preparation method ii) chewing iii) heat which inactivates myronise iv) gastric pH and gut floraalso affectsthe formation of heterodimer formation of I3C.
Glucosinolates and myrosinase are present in separate plant compartments and meet upon cell rupture. Indole 3 methyl glucosinolate in the presence of myronisae leads to the formation of thiohyroxamate-O-sulfonate and iodolylmethyl isothiocyanate which further forms indole 3 acetonitrile and indole 3carbinol, respectively 28.
Limitations of I3C: Indole 3 carbinol is highly unstable in acidic Condition and forms condensation products like DIM, and 4 others 9.
A study carried out in women which consisted of single-dose and multiple-dose administration of I3C, where doses of 400mg,600mg upto 1200mg were administered indicated that the only detectable product of I3C in plasma was found to be DIM. No other condensation product of I3C was detected. The mean Cmax of DIM increased from 61ng/mL at 400mg dose to 607ng/mL at 1000mg dose. The AUC also increased from 392 h ng/mL at 400mg dose to 3376 h ng/mL at 1000 mg dose. The Tmax was ~2h. The limit of quantitation of DIM was below 15 ng/ ml by 24 h.The limit of detection was 1.0 ng/ml for major metabolite DIM, which were analyzed by the HPLC MS method. The calculations were done bywinnonlin v.4.0 using a non-compartmental model. The plasma DIM levels were set to zero and the AUC was calculated using linear trapezoidal interpolation. It also reported that oral administration of I3C lead to the formation of primary product DIM and the production and uptake of this compound was at 1000mg dose.
Also,a comparable increase in AUC and Cmax was found at dose 400mg and 600mg, but further non-linear increases in DIM Cmax and AUC were observed above 600mg. Thus the biological activity of I3C oral administration is attributed by its oligomerization product DIM and its other derivatives and perhaps not by I3C in humans 29. Also, DIM had not been reported from in-vivo studies. In another study carried out in mice which was dosed with either I3C or DIM, no DIM metabolite was detected in tissue and plasma upon oral administration of I3C, infact, I3C was abundantly found in plasma. But DIM was found to be extremely stable in acidic medium 6. The thermal stability of I3C is much poorer than that of DIM. It was observed that I3C content decreased within boiling time of 30 minutes at 37 °C. It was reported that I3C oligomerized into 3 products during the thermal treatment, with DIM as the major product 30.
The lack ofan objective biomarker of phytonutrient exposure and uptake is a critical issue hindering the validation of cruciferous vegetable consumption as a chemopreventive strategy. For example, glucosinolate concentration in vegetables varies from factors such as cultivar selection, growing conditions, and cooking or processing 31. Plant cells, upon damage from inert glucosinolates in the presence of myrosinase, get converted to active derivative DIM which may be affected by preparation method, chewing and essentially with anything that damages the plant cell
Also, gastric pH and gut flora can affect the relative proportion of heterodimer formation from I3C, whereby more acidic pH favors higher-order heterodimers
Bioavailability of I3C and DIM: A Phase I trials study was carried out in women as I3C was proposed to be a potential chemopreventive agent for breast cancer. The study was initially carried out with a single 400mg I3C dose daily. Further multiple doses were carried out where women were injected dose of 200 mg twice daily and later 400mg twice daily for 4 weeks. The study reported that no other condensation product of I3C other than DIM was found in humans after a single dose of I3C. Indole {3,2b} carbazole and higher oligomeric products were detected in the 'acid-catalyzed model study' when analyzed by HPLC-MS but were not detected in plasma 3.
Single-dose pharmacokinetics studies of DIM showed inter-individual variability. Also, a non-linear increase in DIM's Cmax and AUC was observed at dose >600mg.
A pharmacokinetic study on oral administration of I3C to mice was carried out in which the highest concentration of I3C was found in the liver and also peak kidney concentration was significantly higher than plasma, lung heart and brain, which had nearly the sameconcentration. According to the study, the concentration of I3C was considerably higher compared to other condensation products involving DIM,LTr1,HIM, and ICZ. Rapid absorption and elimination were seen 9. The variability between the different results obtained between I3C ingestion in animal and human models maybe be explained by I3C doses, I3C chemistry and different species in gastric physiology. The dose administered in mice was 10-40 times higher than that administered in humans, which may change the gastric concentration of I3C.The gastric pH of rodentsis higher than humans, which leads to slower consumption of I3C in rodents, resulting in a different distribution of products depending upon the pH conditions 32. Also, all subjects for the I3C pharmacokinetic study had been fasting, ultimately resulting in a highly gastric environment and thus rapid consumption of I3C. The mouse requires less gastric emptying time than humans, which causes less exposure of I3C in the reactive condition, ultimately resulting in more I3C availability for absorption. Thus, the factors like gastric pH fasted state or fed state and gastric emptying time can be instrumental in the availability of I3C in humans and mice.
Absorption, Distribution, Metabolism and excretion of I3C: An article about pharmacokinetic studies provided the report of a study carried on women who received oral doses of I3C with the initial dose of 400 mg, which further ranged upto 1200 mg, their plasma was analyzed by the HPLC-MS method for detection of I3C and DIM. The results confirmed that I3C remained undetected but its bioactive metabolite DIM could be detected33. Initially, a high plasma value was seen, which later decreased after 12 hrs. sampling time 34. PBPK model was useful for plasma and tissue concentration detection. It provided the concentration data of DIM and helped to compare the pharmacokinetic properties and the biodistribution of pure crystalline DIM and a novel formulation (Bio Response DIM) of DIM after it was orally administeredto the mice. I3C was found to rapidly metabolize into its condensation product DIM, Ltr and HI-IM, CTr, ICZ, NI3C, CTeT; hence it is considered hard to formulate.But the major I3C condensation product DIM is found to be stable in acidic medium and upon exposure to high temperature and humidity as well. Oral doses of I3C to female CD-1 mice were reported 35; they studied the disposition of I3C and its acid condensation products DIM, ICZ, Ltr (1) and HI- IM in various tissues (blood, liver, kidney, lung, heart, and brain). It was concluded that I3C was rapidly absorbed, distributed, and eliminated from plasma and tissues, falling below the limit of detection by 1 h 35. It was also reported that BR-DIM did not undergo degradation and had proven to be stable in an acidic medium. I3C & DIM disappear within 45 min after oral administration.
Metabolism and Excretion: Under the acidic conditions of the gastric tract, I3C undergoes condensation to form several oligomeric products, particularly DIM. The tissue distribution of I3C has been determined in mice by radio-labeled I3C 36. Both I3C and DIM have been detected in kidneys, lungs, heart, liver, plasma, and brain samples ofmice treated with 250mg/kg of I3C as early as 15 min after administration36. These results suggest that I3C is rapidly absorbed and spread to several well-perfused tissues, where it is transformed to DIM to perform its anticancer actions 36.
In the presence of acidic conditions of the stomach, I3C oligomerizes into its Acid condensation product DIM. Radiolabeled I3C was used to determine the tissue distribution of the acid condensation product; from this it can be concluded that I3C gets rapidly absorbed and it undergoes distribution in the highly perfused tissues where the effect of DIM isseen 36.
I3C Toxicity:Many short-term studies had reported the liver as the target organ of I3C. In male guinea pigs, induced steatosis in the liver was observed. Male CD-1 mice, a dose of 500 to 750mg/kg per day are fed for 5 days, increased weight and microsomal protein content, and altered cholesterol homeostasis.At the 500-750 mg/kg dose, hepatic acyl COA: cholesterol acyltransferase activity. The toxicity studies performed in various animals had the liver as their target organ and also the nervous system.In the case of male guinea pigs, induced steatosis in the liver was observed. In male CD-1 mice, I3C altered cholesterol homeostasis, and also increase in liver weight and microsomal protein content was observed. In the case of male and female CD rats, increased liver weight and hepatic CYP450 activity were noted. Sprague Dawley rats with a 50mg/kg dose showed a significant increase in hepatic CYP450 levels (CYP1A1 and CYP1A2) in both sexes.Also, increased expression of hepatic CYP3A2 and CYP1A1 in the colon of both sexes of rats was observed. The nervous system as a target organ for I3C was administered to Sprague Dawley rats with a subcutaneous dose of 225,300 or 500mg/ml showed induced sedation ataxia, loss of righting reflex, and sleep. Three out of four rats died upon exposure to a dose of 500mg/kg and they were found to be in comatose before death.
Male CD mice upon receiving a dose between 100-500mg/kg resulted in a dose-dependent increase in neurological impairment. Male guinea pigs who received a dose of 0.3mg/kg wt. in Cremophor castor oil showed moderate depression, trembling, tachypnea, polypnea, irregular breathing, and increased vesicular lung soundin lung, I3C induced interstitial pneumonia with septal hyperemia.
Analytical Methods used for I3C: Analytical methods like High-performance liquid chromatography (HPLC), High-performance liquid chromatography with mass detector and UV detector (HPLC-MS37, HPLC-UV) 38, 39, Liquid chromatography Tandem mass spectrometry (LC-MS/MS) 40. Various analytical methods have been developed for the analysis of I3C and its metabolites, like DIM in human subjects likereverse-phase high-performance liquid chromatography-mass spectrometry HPLC-MS 29 and Liquid chromatographic assay method 40 in human and mouse plasma, respectively, very sensitive liquid chromatography-tandem mass spectrometry (LS-MS/MS) method 41 for quantification of a low amount of glucobrassic in metabolite in plasma and urine samples. Also, HPLC-MS 37 was used to access pulmonary bioavailability in A/J mice. Few analytical chemists have validated an HPLC -UV method for the simultaneous quantification of indole-3-carbinol and 3,3'-diindolylmethane in mouse plasma, liver, and kidney tissues 42.
Formulations of Indole-3-carbinol: A comprehensive study about the fate of I3C in-vivo in mice reported the concentration of I3C and its condensation products in plasma as well as the well-perfused organs like the liver, lung, heart, kidney and brain. I3C was not detected in a lot of studies due to its rapid absorption followed by rapid distribution in the tissues. The maximum concentration was found in the liver. Also, the peak kidney concentration was significantly higher than the other tissues, which had nearly similar concentrations. The oligomerization process was found to be pH-dependent, and thus the extent of oligomerization varied with fasted state pH and fed state pH.
- Nanoparticulate Systems: Nanoparticles are solid,colloidal particles consisting of macromolecular substances that vary in size from 10 nm to1000 nm in which the active principle is dissolved, entrapped, or encapsulated and/or to whichthe active principle is adsorbed or attached, which can be administered in fluid form with a liquidcarrier. They are mostly used in cancer therapy anddiagnosis 43, 44. The different nanotechnological drug delivery systems include Solid lipid nanoparticles (SLNs), nanoparticles, nanoemulsions, polymeric nanoparticles, nanocapsules, nanospheres and many more.
Here, zein coated by carboxymethylchitosan (Z/CMCS) nanoparticles were formulated by encapsulating I3C and DIMby using liquid-liquid phase separation and ionic gelation method45. The encapsulation technology was used for formulation I3C and DIM in Z/CMCS nanoparticles with an aim to protect hydrophobic nutrients46like I3C and DIM from harsh conditions, mask off the odor of relevant ingredients and provide control release property 47, their thermal and photo stabilities were studied test which would help monitor the effect of encapsulation on dimerization of I3C and DIM based upon their physicochemical properties by comparing the two delivery systems. The denaturation of zein protein was prevented due to the presence of CMCS. The results showed that the particle size of Z/I and Z/D was about 250nm. After being coated with ZC/I and ZC/D the size reduced to 113nm and 89 nm, respectively. The size reduction may have been caused by the high repulsive forces caused by the high surface charge of CMCS coating. The decrease in zeta potential indicated the adsorption of negatively charged CMCS to the surface of zein.The PDI was found to be within 0.2, indicating the small distribution of particle size. Encapsulation efficiency for zein was between 60-70% for I3C and DIM. For CMCS it increased almost 80%. The particles were spherical in shape and had a smooth surface. CMCS coating reduced the particle size of both nanoparticles and more uniform nanoparticles were formed. Controlled release profiles indicated that both I3C and DIM showed similar effects and showed burst release after 0.5 h and sustained release for 6hrs. The effect of CMCS on kinetic release profiles was not prominent. During thermal stability measurement, precipitation of zein nanoparticle was observed after incubation for 24 h at 37 °C; thus, denaturation of zein protein may result in degradation of I3C, but a coating of CS may reduce the denaturation of zein and thus may prevent the degradation of I3C and an only small amount of DIM and HIIM was formed after 24 h.
- Nanocapsules: Nanocapsules are a characteristic class of nanoparticles, made up of one or more active material (core) and a protective matrix (shell) in which the therapeutic substance maybe confined. I3C is said to have the ability to neutralize free radicals and also to inhibit the lipid peroxidation, reducing both hepatotoxicity and carcinogenesis. However, upon exposure to highly acidic conditions, it is found to be unstable. Hence in order to protect the substances against photo and chemical degradation 48, a study was reported the formulation of polymeric nanocapsules. Polymeric nanocapsules are composed of a polymeric wall surrounding an oil core; these systems were reported to enhancethe substance's stability and its antioxidantactivity 48, 38, improved therapeutic efficacy and reduction in toxicity. The group prepared I3C loaded nanoparticles consisting of Poly (E caprolactone) PLC, a biodegradable polyester, by using interfacial deposition of preformed polymertechnique 39, 49. Here, MCT (medium-chain triglycerides)/RHO (Rose hip oil) were used as oily core.
Two formulations incorporating I3C were formulated as a nano capsule suspension by interfacial deposition technique NC-MCT-I3C and I3C-RHO-I3C, respectively. TheNC-MCT-I3C and NC-MCT-I3C nanoparticles showed high-encapsulation efficiency 41% and 42%, with particle size and zeta potential of (236±5 nm, 240±6nm) and -8,08 ± 0.40 mV, -10.03 ± 0.25 mV, respectively.
The photostability studies were performed, and HPLC analyzed the drug content after 300mins exposure to UVC; MS-I3C showed only 20% I3C left, whereas NC-RHO-I3C and NC-MCT-I3C had 77% and 87%, respectively. Hence, the improved photostability suggested that of nanocarriers have the ability to scatter UV light due to their colloidal size.
Both NC-RHO-I3C and NC-MCT-I3Csignificantly improved the I3C radical scavenging activity to 81 and 74%, respectively, at 75 μg/mL with no significant difference between these formulations (p N >0.05). However, only NC-RHO-I3C showedDPPH radical scavenging activity comparable (p N 0.05) to the positive control (ascorbic acid). NC-RHO-I3C was found to be more effective than NC-MCT-I3C. This result is supported by evidence from another study that showed improved radical scavenging activity by nanocapsules.
Hemocompatibility evaluation was performed as it is important for systemic administration, free I3C shows no hemolytic activity but nanocapsules tend to show concentration-dependent hemolysis. According to the statistical analysis, there was no significant difference between NC-MCT-I3C and NC-RHO-I3C, and as 5% hemolysis is considered as an adequate limit50, only the concentration of 10 μg/mL of NCs could be used intravenously without causing damage to the red blood cells. Both the formulations showed biphasic release profiles, which included the rapid onset of drug and prolonged release to maintain drug effect for long periods. The burst effect was observed due to partial encapsulation. RHO NCs were able to increase the antitumor effect of I3C, reducing MCF-7 and C6 cell viability, without significant reduction in the viability of primary astrocyte cells 39.
- Liposomes: Liposomes were prepared by lipid film hydration technique. 7.5mg DPPC, 2.5mg Cholesterol and 5mg I3C is taken in an organic solvent, chloroform: methanol 6:1. Further, the solvent was evaporated on a rotary evaporator at a speed of 100 rpm for hr. at room temperature. Then after the formation of dry firm occurs, it was subjected to hydration by addition of HEPES buffer, which leads to the formation of liposomal dispersion. The resultant dispersion was sonicated using a probe sonicator on an ice bath for 2 min at an output of 15 W.
The particle size and zeta potential were measured by Dynamic light scattering. To calculate the drug loading, the liposomal suspension was centrifuged at 14000 rpm for 15mins. The supernatant was discarded, and the pellet was extracted with 500µL of acetonitrile.
The results indicated that the liposomes were submicron in size (664.4 ±212.3), the PDI value was found to be 0.277±0.078, zeta potential (mW) was found to be negative (-6.7±1.08). Its encapsulation efficiency was found to be (7.9±4.0). These liposomes were formulated for the purpose of intranasal administration; thus, a 100-fold lower dose of I3C led to an equivalent concentration of the drug in the lungs. Also, the decline in lung concentration of I3C and DIM was much slower compared to an oral formulation. The enhanced lung bioavailability of I3C resulted in a decrease in the formation of O6-methyl- G in lung tissues of NNK treated mice. A 10-fold increase in detoxification enzyme CYP1A1 was seen.
- Microemulsion: An emulsion is a non-homogenous dispersion of two or more immiscible liquids stabilized by surfactant, sub-surfactant and emulsifiers. Drug packaging in the inner core of emulsion facilitates the sustained release of drugs over a long period of time. The size of emulsion droplets plays a significant role in the targeted distribution of the drug. Emulsion helps to concentrate lipophilic drug moiety into the liver, spleen, and kidney while hydrophilic drug into the lymphatic system via intramuscular or subcutaneous injection. I3C was dissolved in an oil phase consisting of Cremophor EL, carbitol and Captex 355(6:3:1).
The oil phase was diluted with saline before dosing. The pulmonary bioavailability of the oral microemulsion (250mg/kg) was given to A/J mice. It was sedated with isoflurane. The AUC of I3C and DIM in lungs was determined by trapezoidal rule and it was found to be 5088.3 for I3C and 54537.5 for DIM. In case of microemulsion 0.0008% i.e., 2 folds higher concentration than of suspension administered dose of I3C was found in lung but within 4hr and 24 h the conc. of microemulsion declined.
- Suspension: Suspensions are heterogeneous systems consisting of two phases. The continuous or external phase is usually liquid or semisolid and therefore the dispersed or internal phase is formed from particulate that's essentially insoluble in but dispersed throughout the continuous phase.
A suspension of I3C was formulated by dispersing I3C into cottonseed oil immediately before dosing. The pulmonary bioavailability of I3C was studied in 5–6 weeks-old female A/J mice which were treated with an oral suspension (250 mg/kg). Only 0.0004% of the administered dose was found in the lungs at 30 min post-dose upon administration of suspension formulation. And further, after 4h and 24h the level of I3C rapidly declined when administered as a suspension formulation.
Theconcentration of I3C was 2-fold higher when it had been administered duringa microemulsion formulation. The area under the curve for I3C and DIM was determined upon administration as an oral suspension. The levels of I3C and DIM were found in 5088.3 ng h/g and 54537.5 ng h/g. This maybe due to the poor solubility of I3C, which may interfere with its absorption and thereby reduce its bioavailability 37.
CONCLUSION: In conclusion, the developed formulations have shown that Indole-3-carbinol can have therapeutic uses in preventingdiseases like breast, lung, prostate, leukemia and overian cancer. This review article provides a wide overview suggesting that the metabolite DIM could have potential benefit for chemoprevention. In addition, DIM could be useful as an adjunct to conventional therapeutics for the prevention of tumor progression or treatment of human malignancies in the future.
The experimental studies also suggest that I3C and DIM, the components of cruciferous vegetables, have therapeutic potential for the prevention and treatment of cancer. In most clinical trials, indole 3 carbinol has major problem of its poor oral bioavailability due to the instability of indole 3 carbinol in an acidic medium. Future research could concentrate on the benefits of I3C and DIM in various clinical studies to combat the cancer epidemic in society through the development of formulations by various technologies. Future research can focus on the advantages of I3C and DIM in various clinical studies to overcome the cancer epidemic among the population by the application of diverse technologies.
ACKNOWLEDGEMENT: The authors would like to acknowledge the support from Bharati Vidyapeeth DeemedUniversity, Poona College of Pharmacy, Pune.
CONFLICTS OF INTEREST: The authors declare that they have no conflicts of interest concerning this article.
REFERENCES:
- Moreno DA, Carvajal M, López-BerenguerCand García-VigueraC: Chemical and biological characterisation of nutraceutical compounds of broccoli. J Pharm Biomed Anal 2006; 41: 1508-22.
- Tin AS, Park AH, Sundar SNand Firestone GL: Essential role of the cancer stem/progenitor cell marker nucleostemin for indole-3-carbinol anti-proliferative responsiveness in human breast cancer cells. BMC Biol 2014; 12: 1-22.
- Qian X, Melkamu T, Upadhyaya Pand Kassie F: Indole-3-carbinol inhibited tobacco smoke carcinogen-induced lung adenocarcinoma in A/J mice when administered during the post-initiation or progression phase of lung tumorigenesis. Cancer Lett 2011; 311: 57-65.
- SafaM: Indole-3-carbinol suppresses NF-κB activity and stimulates the p53 pathway in pre-B acute lymphoblastic leukemia cells. Tumor Biol 2015; 36: 3919-30.
- Taylor-HardingB: Indole-3-carbinol synergistically sensitises ovarian cancer cells to bortezomib treatment. Br. J. Cancer 2012; 106: 333-43.
- Maruthanila VL, Poornima J and Mirunalini S: Attenuation of carcinogenesis and the mechanism underlying by the influence of indole-3-carbinol and its metabolite 3,3'-diindolylmethane: a therapeutic marvel. Adv Pharmacol Sci 2014; 2014: 832161.
- Lafarga T, Bobo G, Viñas I, Collazo Cand Aguiló-Aguayo I: Effects of thermal and non-thermal processing of cruciferous vegetables on glucosinolates and its derived forms. J Food Sci Technol 2018; 55l 1973-81.
- SchnekenburgerM: Research on cruciferous vegetables, indole-3-carbinol, and cancer prevention: A tribute to Lee W. Wattenberg. MolNutrFood Res 2018; 60: 1228-38.
- Weng JR, Omar HA, Kulp SK and Chen CS: Pharmacological exploitation of indole-3-carbinol to develop potent antitumor agents. Mini Rev Med Chem 2010; 10(5): 398-404.
- Fujioka N, Fritz V, Upadhyaya P, Kassie Fand Hecht SS: Research on cruciferous vegetables, indole-3-carbinol, and cancer prevention: A tribute to Lee W. Wattenberg. Mol. Nutr. Food Res 2016; 60: 1228-38.
- BanerjeeS: Attenuation of multi-targeted proliferation-linked signaling by 3,3’-diindolylmethane (DIM): From bench to clinic. MutatRes RevMutatRes 2011; 728: 47-66.
- Rosen CA, Woodson GE, Thompson JW, Hengesteg APand Bradlow HL: Preliminary results of the use of indole-3-carbinol for recurrent respiratorypapillomatosis. Otolaryngol. - Head Neck Surg 1998; 118: 810-15.
- LicznerskaBand Baer-DubowskaW: Indole-3-carbinol and its role in chronic diseases in Advances in Experimental Medicine and Biology 2016; 928: 131-54.
- Sharma S, Kelly TKand Jones PA: Epigenetics in cancer. Carcinogenesis 2009; 31: 27-36.
- ShapiroOand BratslavskyG: Genetic Diseases. Brenner’s Encycl. Genet. Second Ed 2013; 429: 246-47.
- SawanCand HercegZ: Chapter 3 - Histone Modifications and Cancer. Epigenetics and Cancer, Part A vol. 70 (Elsevier Inc., 2010).
- Wong CP: Effects of sulforaphane and 3,3′-diindolylmethane on genome-wide promoter methylation in normal prostate epithelial cells and prostate cancer cells. PLoS One 2014; 9.
- Li Y, Li Xand Guo B:Chemopreventive agent 3,3′-diindolylmethane selectively induces proteasomal degradation of class I histone deacetylases. Cancer Res 2010; 70: 646-54.
- Karius T, Schnekenburger M, Dicato Mand DiederichM: MicroRNAs in cancer management and their modulation by dietary agents. Biochem. Pharmacol 2012; 83: 1591-1601.
- Li Y, Kong D, Wang Zand Sarkar FH: Regulation of microRNAs by Natural Agents : An Emerging Field in Chemoprevention and Chemotherapy Research 2010; 1027–1041. doi:10.1007/s11095-010-0105-y.
- Melkamu T, Zhang X, Tan J, Zeng Yand Kassie F: Alteration of microRNA expression in vinyl carbamate-induced mouse lung tumors and modulation by the chemopreventive agent indole-3-carbinol. Carcinogenesis 2010; 31: 252-58.
- IzzottiA: Downregulation of microRNA expression in the lungs of rats exposed to cigarette smoke. FASEB J 2009; 23: 806-12.
- LiY: Up-regulation of miR-200 and let-7 by natural agents leads to the reversal of epithelial-to-mesenchymal transition in gemcitabine-resistant pancreatic cancer cells. Cancer Res 2009; 69: 6704-12.
- KassieF: Inhibition of vinyl carbamate-induced pulmonary adenocarcinoma by indole-3-carbinol and myo-inositol in A/J mice. Carcinogenesis 2010; 31: 239-45.
- Liu H, Wormke M, Safe SHand Bjeldanes LF:Indolo[3,2-b]carbazole: a Dietary-Derived Factor That Exhibits Both Antiestrogenic and Estrogenic Activity 1994; 86: 1758-65.
- Herrmann S, Seidelin M, Bisgaard HCand Vang O: Indolo [3, 2- b] carbazole inhibits gap junctional intercellular communication in rat primary hepatocytes and acts as a potential tumor promoter of I3C. The aim of the present study was to investigate the (GJIC) in primary cultured rat hepatocytesc 2002; 23: 1861-68.
- Riby JE: The Major Cyclic Trimeric Product of Indole-3-carbinol is a Strong Agonist of the Estrogen Receptor Signaling Pathway 2000; 910-18.
- Chamovitz DA, Katz Eand Nisani S: Indole-3-carbinol: A plant hormone combatting cancer [version 1; referees: 2 approved]. F1000Research 2018; 7: 1-9.
- Reed G: Single-Dose and Multiple-Dose Administration of Indole-3-Carbinol to Women : Pharmacokinetics Based on 3 , 3 ¶ -Diindolylmethane 2006; 15: 2477-2482.
- LuoY: Encapsulation of indole-3-carbinol and 3,3′-diindolylmethane in zein/carboxymethyl chitosan nanoparticles with controlled release property and improved stability. Food Chem 2013; 139: 224-30.
- Lü H, Pope CNand Xu KW: Black p-branes and their vertical dimensional reduction. Nucl. PhysB 1997; 489: 264-78.
- Martinez V: High basal gastric acid secretion in somatostatin receptor subtypes 2 knockout mice. Gastroenterology 1998; 114: 1125-32.
- Hayes JD, Kelleher MOand Eggleston IM: The cancer chemopreventive actions of phytochemicals derived from glucosinolates. Eur JNutr 2008; 47: 73-88.
- Gong Y, Firestone GLand Bjeldanes LF: 3,3′-Diindolylmethane is a novel topoisomerase IIα catalytic inhibitor that induces S-phase retardation and mitotic delay in human hepatoma HepG2 cells. Mol. Pharmacol 2006; 69: 1320-27.
- Anderton MJ, Manson MM, Verschoyle RD, Gescher A, Lamb JH, Farmer PB, Steward WP and Williams ML: Pharmacokinetics and tissue disposition of indole-3-carbinol and its acid condensation products after oral administration to mice. Clin Cancer Res 2004; 10(15): 5233-41.
- KassieF: Indole-3-carbinol inhibits 4-(methyl-nitrosamino)-1-(3-pyridyl)-1-butanone plus benzo(a)pyrene-induced lung tumorigenesis in A/J mice and modulates carcinogen-induced alterations in protein levels. Cancer Res 2007; 67: 6502-11.
- Song JM: Intranasal delivery of liposomal indole-3-carbinol improves its pulmonary bioavailability. Int JPharm 2014; 477: 96-101.
- GehrckeM:Nanocapsules improve indole-3-carbinol photostability and prolong its antinociceptive action in acute pain animal models. Eur. J. Pharm. Sci 2018; 111: 133-41.
- GehrckeM: Enhanced photostability, radical scavenging and antitumor activity of indole-3-carbinol-loaded rose hip oil nanocapsules. Mater. Sci. Eng. C 2017; 74: 279-86.
- Anderton MJ: Liquid chromatographic assay for the simultaneous determination of indole-3-carbinol and its acid condensation products in plasma 2003; 787: 281-91.
- Hauder J, Winkler S, Bub A, Rüfer CE, Pignitter M and Somoza V: LC-MS/MS quantification of sulforaphane and indole-3-carbinol metabolites in human plasma and urine after dietary intake of selenium-fortified broccoli. J Agric Food Chem 2011; 59(15): 8047-57.
- Moussata J, Wang Zand Wang J: Development and validation of an HPLC method for the simultaneous quantification of indole-3-carbinol acetate, indole-3-carbinol, and 3,3’-diindolylmethane in mouse plasma, liver, and kidney tissues. J. Chromatogr. B Anal. Technol. Biomed. Life Sci 2014; 958: 1-9.
- KothamasuP:Nanocapsules: The weapons for novel drug delivery systems. BioImpacts 2012; 2: 71-81.
- Pawar A, Rajalakshmi S, Mehta P, Shaikh Kand Bothiraja C: Strategies for formulation development of andrographolide. RSC Advan 2016; 6: 69282-300.
- Luo Y: Encapsulation of indole-3-carbinol and 3,3′-diindolylmethane in zein/carboxymethyl chitosan nanoparticles with controlled release property and improved stability. Food Chem 2013; 139: 224-30.
- Luo Y, Teng Z and Wang Q: Development of zein nanoparticles coated with carboxymethyl chitosan for encapsulation and controlled release of vitamin D3. J Agric Food Chem 2012; 60(3): 836-43.
- Arvanitoyannis IS: Encapsulation and Controlled Release Technologies in Food Systems. Int J Food Sci Technol 2009; 44: 1462-63.
- Ros Dand Bulho LOS: Nanostructured Systems Containing Rutin : In Vitro Antioxidant Activity and Photostability Studies 2010; 1603-10.
- Fessi H, Puisieux F, Devissaguet JPH, Ammoury N and Benita S: Nanocapsules formation by interfacial polymer deposition following solvent displacement. International Journal of Pharmaceutics 1989; 55(1): 1-4.
- Das D, Chandra B, Phukon Pand Kumar S: Colloids and Surfaces B :Biointerfaces Synthesis of ZnO nanoparticles and evaluation of antioxidant and cytotoxic activity. Colloids Surfaces B Biointerfaces 2013; 111: 556-60.
How to cite this article:
Pandav T and Mali A: Indole-3-carbinol: a review on currenttrends and future prospects. Int J Pharmacognosy 2021; 8(9): 373-84.doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(9).373-84.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
373-384
413 KB
1045
English
IJP
Tisha Pandav and Ashwin Mali *
Department of Pharmaceutics, Bharati Vidyapeeth Deemed University, Poona College of Pharmacy, Erandawane, Pune, Maharashtra, India.
ashwinjmali@rediffmail.com
28 June 2021
16 September 2021
19 September 2021
10.13040/IJPSR.0975-8232.IJP.8(9).373-84
30 September 2021