IN-VITRO FREE RADICAL SCAVENGING AND HYPOGLYCEMIC EVALUATION OF FRUIT EXTRACT AND SOLVENT FRACTIONS OF ZIZIPHUS OENOPLIA MILL (RHAMNACEAE)
HTML Full TextIN-VITRO FREE RADICAL SCAVENGING AND HYPOGLYCEMIC EVALUATION OF FRUIT EXTRACT AND SOLVENT FRACTIONS OF ZIZIPHUS OENOPLIA MILL (RHAMNACEAE)
Praveen Kr Goyal * 1, 2, G. Jeyabalan 1 and Yogendra Singh 3
Alwar Pharmacy College 1, Alwar - 301030, Rajasthan, India.
Rajasthan University of Health Sciences 2, Jaipur - 302033, Rajasthan, India.
Maharaja Agrasen University 3, Solan - 174103, Himachal Pradesh, India.
ABSTRACT: Objective: Stem Bark, Root, and fruit of Ziziphus oenoplia (Rhamnaceae) has been used in traditional health systems to treat diabetes and many more disorders in southern Asia as India, Sri Lanka, and Burma. However, the anti-oxidant and hypoglycemic potential of the fruit of this valuable plant is not scientifically validated till date. The aim of the present study is to evaluate in vitro free radical scavenging and hypoglycemic effect of ethanol extracts and solvent fractions of Ziziphus oenoplia fruit. Methods: The ethanol extracts and three solvent fractions of Ziziphus oenoplia fruit were evaluated at different concentration (20- 640 µg/ml) for free radical scavenging activity by using in-vitro DPPH method and different concentration (25-800 µg/ml) for hypo-glycemic potentials by using α – glucosidase and α-amylase enzymes (intestinal enzymes which slow the digestion of carbohydrates and delay glucose absorption) inhibitory activity. Results: The outcome of present study indicates that maximum anti-oxidant activity was produced by ethanol crude extract (87.66±1.54% Inhibition at 640 μg/mL) with IC50 value 2.72±0.62 followed by chloroform fraction (66.76±0.92% Inhibition) compare to ascorbic acid standard (90.72±0.76 Inhibition) with IC50 value 1.33±0.45. In similar manner maximum α – glucosidase inhibitory and α-amylase effect shown by ethanol crude extract 85.2 ± 1.7 % Inhibition and 88.43 ± 0.58 % Inhibition at 800 μg/mL respectively compare to acarbose as reference drug 97.2 ± 0.48 and 99.12± 0.72% Inhibition. Conclusion: The result showed the potential effects of Z. oenoplia fruit extract and fractions as anti-oxidant by scavenging diphenyl-2-picrylhydrazyl (DPPH) free radicals and as postprandial hypoglycemic by inhibiting α–amylase, α–glucosidase enzyme in dose dependant manner compare to ascorbic acid and acarbose as standard drug. The claimed traditional use as anti-oxidant and hypoglycemic has a scientific background.
| Keywords: |
Antioxidant, Antidiabetic, DPPH, α-amylase, α–glucosidase, Ziziphus oenoplia
INTRODUCTION: Diabetes mellitus is one of the foremost reasons of mortality and morbidity in human being 1. According to an estimate by the International Diabetes Federation, near about 375
million persons are suffering from this disease, and this is continuously increasing day by day; in India, about 40-45 million people are suffering from this disorder, which is expected to increase to about 60-65 million in the future 2-3.
Diabetes mellitus is caused by an absolute or relative deficiency of insulin and/or reduced insulin activity or inherited and/or acquired deficiency in the production of insulin. It represents serious, chronic heterogeneous group of metabolic disorder which finally produce hyperglycemia and irregularity in sugar, lipid and protein metabolism 4-6. Chronic hyperglycemia is associated with dysfunction of heart, eyes, blood vessels, kidneys, nerves etc. and it is characterized by damage of pancreatic β cells, oxidative stress and cardio-vascular complications 2, 7, 8. Globally type 2 diabetes is common form which considered as one of the most recurrent lifestyle diseases. Prevalence of Type 2 is also more than type 1. Type 2 diabetes mellitus (T2DM) is a non-communicable disorder that is major causes of death worldwide because it is associated with long-term side effects like retinopathy, nephropathy, neuropathy, skin complications, etc. For the treatment of DM there are various classes of oral hypoglycemic agents existing along with a variety of insulin, but due to so many side effects and long duration treatment, there is an increasing demand by the patients to use natural products for control hyperglycemia 9-12.
Despite considerable progress within the management of Type 2 DM by synthetic drugs, the design for natural anti-diabetic plant products for controlling diabetes goes on. There are many hypoglycemic plants known through the folklore but their introduction into the modern therapy system awaits the invention of an animal test system that closely parallel to the pathological course of diabetes in human beings 13-16.
Some medicinal herbs with proven anti-diabetic and related beneficial effects utilized in the treatment of diabetes are Tinospora cordifolia, Gymnema sylvestre, Casearia esculenta, Syzygium cumini, Commiphora wightii, Gmelina arborea, Asparagus racemosus, Boerhavia diffusa, Sphaeranthus indicus, Pterocarpus marsupium, Tribulus terrestris, Phyllanthus amarus, Swertia chirata, Glycyrrhiza glabra, Gossypium herbaceum, Berberis aristata, Piper nigrum 10, 17, 18.
A traditional medicinal plant, broadly distributed through Australia, tropical and subtropical Asia including India, known as jackal jujube, in Hindi known as Makora, Botanically identified as Ziziphus oenoplia Mill. belongs to the family Rhamnaceae 19-20. Ziziphus oenoplia is a straggling shrub, which has green flowers in subsessile axillary cymes. The fruits are containing a single seed having globose drupe, black and shiny when ripe 19, 21.
There are large numbers of traditional benefits of Ziziphus plants, since ancient times- leaves, fruits, seeds, and barks of these plants have been used medicinally. Z. oenoplia plant is widely used in Ayurveda for the treatment of various diseases, such as ulcer, Stomach ache, obesity, asthma, and it has astringent, digestive, antiseptic, hepato-protective, wound healing and diuretic property. Ziziphus plants are traditionally used as medicine for the treatment of various diseases such as digestive disorders, urinary troubles, diabetes, skin infections, diarrhea, fever, bronchitis, liver complaints, and anaemia. Aerial parts exhibit diuretic, hypoglycemic, and hypotensive activity 19, 21-24. The phytochemical analysis of the Z. oenoplia showed the presence of alkaloids, steroids, flavonoids, carbohydrate, and pectin compounds 25.
The fruit of Z. oenoplia (Rhamnaceae) has been used in the treatment of Diabetes mellitus in tropical and subtropical Asia, including India, as folk-medicine without any scientific base for safety and efficacy 20, 23. One interesting approach is to decrease postprandial hyperglycemia by reducing glucose uptake through the inhibition of carbohydrate-hydrolyzing enzymes, such as α- glucosidase and α-amylase 26-27. Thus, the objective of the present study is to evaluate the in vitro anti-oxidant and anti-diabetic activity of fruit crude extract and solvent fractions.
MATERIALS AND METHODS:
Reagents and Drugs: 2,2- diphenyl-1-picrylhydrazyl (DPPH), 2-Nitrophenyl- α -D-galactopyranoside, α-amylase, α -Glucosidase from Saccharomyces cerevisiae, Acarbose, Folin-Ciocalteu reagent, and ascorbic acid were purchased from Sigma-Aldrich (France). All other reagents of analytical grade were obtained from Merck Chemicals.
Plant Material:
Plant Collection, Identification and Authentication: The fruit of Z. oenoplia was collected from Sariska forest Alwar district, Rajasthan, India, in September 2019. The collected plant material was botanically identified and authenticated by Dr. L. K. Sharma, Department of Botany, Raj Rishi Govt. College, Alwar (India). The voucher specimens (001-APC/2019) are deposited in the herbarium.
The crude drug was cleaned, dried in the shade for 48-72 h, and coarsely powdered. The powdered drug is used for further studies.
Extraction of Plant Material: The extraction of fruit with ethanol was carried out by using the hot continuous percolation method in the soxhlet apparatus. 95% ethanol was used as a solvent. After completion of extraction, the extract was collected directly from the round-bottomed flask, and the solvent was evaporated using a vacuum evaporator.
Fractionation of Crude Extract: Fractionation was performed by dissolving ethanol extract in 200 ml of distilled water and partitioned between pet. ether (200 ml × 3 times) and water to obtain pet. ether fraction (PEF) and remaining aqueous portion further partitioned with chloroform (200 ml × 3 times) to obtain chloroform fraction (CF) and residual aqueous fraction (AF). The fractions were subjected individually to evaporation using a rotary evaporator.
Preliminary Phytochemical Screening of Extract and Solvent Fractions: Standard Preliminary phytochemical qualitative test of the extract and fractions was carried out for detection of phyto-constituents. The crude extract was screened for the presence or absence of secondary metabolites such as Reducing sugars, Alkaloids, Steroidal compounds, Phenolic compounds, Cardiac glycosides, Flavonoids, Saponins, Tannins, and Anthraquinones using standard procedures 28-29.
In-vitro Antioxidant Activity Model:
DPPH Free Radical Scavenging Activity Assay: Radical scavenging potential of the plant extracts, solvent fractions, and standard drug were measured by the stable free radical DPPH using standard procedure reported previously 30-31. The variable concentration of ethanol fruit extract and solvent fractions (20-640 𝜇g/ml) were mixed in DMSO and dissolved individually into 0.1 ml methanol solution of DPPH (0.3 mM). The mixture was left in the dark for 30 min, and then absorbance at 517 nm was determined using a spectrophotometer, and the percentage inhibition effect was calculated. Ascorbic acid was used as anti-oxidant standard. IC50 values were determined by using linear regression analysis, which indicates the amount of the sample needs to scavenge 50% DPPH-free radicals. The percentage (%) of inhibition of the DPPH-free radical was determined by the formula:
(A0-A1)/A0 × 100
A0 is the absorbance of the control, and A1 is the absorbance of the extract/fractions/standard.
In-vitro Models of Anti-diabetic Activity of Crude Extract and Solvent Fractions:
𝛼 -Glucosidase Inhibitory activity: The 𝛼 -Glucosidase inhibitory activity was determined by the use of substrate p-Nitrophenyl 𝛼-d-gluco-pyranoside (pNPG) method reported previously 32-33. pNPG is hydrolyzed by enzyme 𝛼 -Glucosidase to produce p-Nitrophenyl (a colored compound that can be observed at 405 nm). The variable concentration of ethanol fruit extract and solvent fractions (25-800 𝜇g/ml) were used for the study. Acarbose was used as standard 𝛼 –Glucosidase inhibitor. The results are presented as percentage inhibition, and IC50 value (concentrations of inhibitor required to inhibit 50% of enzyme activity) was determined by using linear regression analysis.
𝛼 - Amylase Inhibitory Activity: 𝛼-Amylase inhibition effect of different samples was determined according to standard procedure 31, 34 with little modification. At first, 0.5% Starch solution (5 mg/ml) was prepared by heating 40 mg of starch in 8 ml of 10 mM sodium phosphate buffer (pH 6.9) for 10 min. The porcine pancreatic enzyme solution (0.5mg/ml) was prepared by mixing 1 mg of α-amylase in 2 ml of the above phosphate buffer.
Fruit extracts and solvent fractions in different concentration (25-800 𝜇g/ml) was mixed individually with 550 μl of starch solution and 40 μl of the enzyme solution in a tube and incubated at 37 °C for 15 min. Now, 1 mL of di-nitro salicylic acid (DNSA) was added and the reaction ended by boiling in a water bath for 10 min. After cooling, the absorbance was measured at 540 nm. The Control group contains all reagents and the enzyme except the test sample, while the α-amylase inhibitory potential was indicated as percentage inhibition. Acarbose was utilized as the standard or positive control. α-Amylase inhibitory activity was calculated as follows:
Inhibition (%) = (1 − B/A) × 100, where A = absorbance of control and B = absorbance of test samples.
Statistics: The IC50 values were determined by curves between log of inhibitor concentration versus percentage inhibition. All values are expressed as Mean ±SEM. The differences were compared using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison tests. P values <0.05 were considered as significant.
RESULTS:
Percentage Yield of Extract and Solvent Fractions: After extraction, the fruit crude extract was 16%, and the yield of different fractions was 18.2%, 28.6, and 36.5 for petroleum ether, aqueous, and chloroform, respectively
Preliminary Phytochemical Analysis of Extract and Solvent Fractions: Preliminary phytochemical analysis was done for the fruit crude extract, and solvent fractions of Z. oenoplia resulted in the presence of Alkaloids, Saponins, Tannins, Phenols, Flavonoids, and Steroids. Terpenoids and Glycosides were not present in the phytochemical screening Table 1.
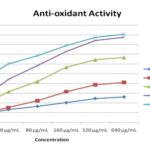
In-vitro Antioxidant Activity by DPPH Free Radical Scavenging Assay: Ethanol extract of Ziziphus oenoplia (EEZO), solvent fraction and reference material ascorbic acid with different concentrations (20–640 𝜇g/ml) was used for study. EEZO and different fraction had demonstrated the enormous potential to scavenge DPPH radicals with an IC50 value of 11.92 ± 0.53, 6.98 ± 0.32, 5.74 ± 1.12, 2.72 ± 0.62, 1.33 ± 0.45 for Pet. Ether fraction, Aqueous fraction, Chloroform Fraction, Ethanol crude extract, and standard drug, respectively. Data indicates a dose-dependent enhancement in the percentage of free Radical Scavenging activity for all concentrations and all groups tested.
The highest antioxidant activity was observed in the Ethanol crude extract with 87.66 ± 1.54% inhibition at a concentration of 640 μg/mL, which is comparable to that of standard Ascorbic acid 90.72 ± 0.76% inhibition Table 2 and Fig. 1.
TABLE 1: PHYTOCHEMICAL ANALYSIS OF FRUIT CRUDE EXTRACT AND SOLVENT FRACTIONS
| S. no. | Phyto-Constituents | Ethanol
Extract |
Pet. ether
fraction |
chloroform
fraction |
Aqueous
fraction |
| 1 | Alkaloids | + | - | + | - |
| 2 | Carbohydrates | + | - | - | + |
| 3 | Proteins | - | - | - | - |
| 4 | Tannins | + | - | + | - |
| 5 | Terpenoids | - | - | - | - |
| 6 | Phenols | + | - | + | + |
| 7 | Flavonoids | + | - | + | + |
| 8 | Glycosides | - | - | - | - |
| 9 | Steroids | + | + | - | + |
TABLE 2: IN-VITRO ANTIOXIDANT POTENTIAL OF THE FRUIT EXTRACT AND SOLVENT FRACTIONS BY DPPH ASSAY:
|
Concentration |
Percentage (%) Inhibition of DPPH | ||||
| Pet. Ether Fraction | Aqueous fraction | Chloroform Fraction | Ethanol Extract | Ascorbic Acid | |
| 20 μg/mL | 8.14±1.12 | 8.54±0.68 | 16.35±1.12 | 22.62 ± 1.92 | 42.4 ± 1.42 |
| 40 μg/mL | 13.32±0.76 | 15.23±0.45 | 31.54±0.94 | 44.3 ± 1.34 | 60.24 ± 1.2 |
| 80 μg/mL | 16.62±0.82 | 22.24±0.72 | 42.12±0.62 | 58.52 ± 2.82 | 68.62 ± 0.94 |
| 160 μg/mL | 20.33±0.65 | 31.53±0.63 | 56.74±1.43 | 72.91 ± 2.32 | 78.97 ±1.12 |
| 320 μg/mL | 24.65±0.94 | 38.62±0.75 | 64.46±0.86 | 84.32 ± 3.12 | 87.4 ± 1.4 |
| 640 μg/mL | 26.24±0.62 | 41.12±0.43 | 66.76±0.92 | 87.66 ± 1.54 | 90.72 ± 0.76 |
| IC50 μg/mL | 11.92±0.53 | 6.98±0.32 | 5.74±1.12 | 2.72 ± 0.62 | 1.33 ± 0.45 |
Notes: Each value of Percentage inhibition of DPPH free radical is presented as means ± S.E.M., n = 3 Abbreviations: DPPH, 2,2-diphenyl-1-picrylhydrazinel IC50, half maximal inhibitory concentration, SEM- standard error of mean
In-vitro α-glucosidase and α-amylase Inhibitory Activity of EEZO and its Fractions: In the present study, the EEZO extract and solvent fractions were evaluated for their potential to inhibit both of these enzymes; on the basis of the effect of crude extract and fractions result indicates that EEZO and CF have potent inhibition of pancreatic α-glucosidase and α-amylase in a concentration-dependent manner.
FIG. 1: IN-VITRO DPPH RADICAL SCAVENGING POTENTIAL OF EEZO AND ITS FRACTIONS IN COMPARISON WITH ASCORBIC ACID AS AN ANTIOXIDANT STANDARD
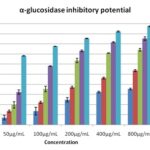
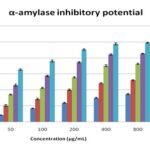
EEZO and solvent fraction with different concentrations (25–800 𝜇g/ml) were used for the study, and Acarbose was used as a reference under a specific set of assay conditions. α-glucosidase and α-amylase inhibitory analysis show that maximum inhibition produced by EEZO crude extract 85.2 ± 1.7% and 88.43 ± 0.58% respectively and followed by chloroform fraction 74.16 ± 1.44% and 72.58 ± 0.82 respectively, which was comparable with the standard drug acarbose. IC50 value of PEF, AF, CF, EEZO was 32.54 ± 0.82, 46.27 ± 1.48, 70.92 ± 0.24, 81.54± 0.58 μg/ml, respectively, for α-glucosidase effect and for α-amylase IC50 value was 70.83± 0.15, 49.74 ± 0.86, 37.42 ± 0.43, 19.76 ± 0.26 μg/ml respectively. The results were shown in Table 3 and 4 and Fig. 2 and 3.
TABLE 3: α - GLUCOSIDASE INHIBITORY POTENTIAL OF THE FRUIT EXTRACT AND SOLVENT FRACTIONS OF ZIZIPHUS OENOPLIA
| Conc. (μg/mL) | Pet. Ether Fraction | Aqueous fraction | Chloroform fraction | EEZO crude extract | Std Acarbose |
| 25 | 3.45 ± 1.72 | 8.56 ± 1.54 | 10.12 ± 0.76 | 24.74 ± 2.14 | 47.47 ± 1.67 |
| 50 | 7.16± 2.23 | 13.65 ± 2.7 | 19.76 ± 3.15 | 32.4 ± 2.24 | 68.12 ± 0.32 |
| 100 | 13.65 ± 3.78 | 22.34 ± 2.26 | 35.45 ± 2.67 | 58.08 ± 0.36 | 77.58 ± 0.12 |
| 200 | 24.82 ± 2.18 | 37.12 ± 3.86 | 63.42 ± 2.15 | 73.16 ± 0.8 | 85.32 ± 0.27 |
| 400 | 32.54 ± 0.82 | 46.27 ± 1.48 | 70.92 ± 0.24 | 81.54± 0.58 | 92.04 ± 0.54 |
| 800 | 35.5 ± 0.79 | 53.58 ± 2.36 | 74.16 ± 1.44 | 85.2 ± 1.7 | 97.2 ± 0.48 |
| IC50 | 70.83± 0.15 | 52.57± 1.42 | 34.89± 0.8 | 22.87± 0.45 | 5.2± 0.2 |
Abbreviation: IC50- half maximal inhibitory concentration. Each value of percentage inhibition of α - glucosidase is presented as means + standard error of the mean (SEM), n =3
TABLE 4: α -AMYLASE INHIBITORY POTENTIAL OF THE FRUIT EXTRACT AND SOLVENT FRACTIONS OF ZIZIPHUS OENOPLIA
| Conc.(μg/mL) | Pet. Ether Fraction | Aqueous fraction | Chloroform fraction | EEZO crude extract | Std Acarbose |
| 25 | 3.54 ± 0.65 | 13.32 ± 0.83 | 23.53±1.14 | 34.46 ± 0.76 | 48.24 ± 1.12 |
| 50 | 8.54 ± 0.16 | 20.54 ± 1.23 | 34.25 ± 0.54 | 46.12 ± 1.42 | 65.44 ± 0.92 |
| 100 | 16.88 ± 0.46 | 25.66 ± 1.08 | 42.52 ± 0.76 | 57.6 ± 1.08 | 76.1 ±1.35 |
| 200 | 23.76 ± 0.72 | 37.32 ± 0.84 | 55.82 ± 0.83 | 74.48 ± 0.84 | 90.67 ± 0.86 |
| 400 | 30.12 ± 0.62 | 43.31 ± 0.74 | 70.23 ± 1.14 | 84.24 ± 0.86 | 97.54 ± 1.21 |
| 800 | 34.92 ± 0.8 | 46.16 ± 0.76 | 72.58 ± 0.82 | 88.43 ± 0.58 | 99.12 ± 0.72 |
| IC50 | 72.32 + 0.54 | 49.74 + 0.86 | 37.42 + 0.43 | 19.76 + 0.26 | 3.54 + 0.32 |
Abbreviation: IC50- half maximal inhibitory concentration. Each value of percentage inhibition of α -amylase is presented as means + standard error of the mean (SEM), n = 3
FIG. 2: INHIBITORY EFFECT OF THE FRUIT EXTRACT/ SOLVENT FRACTIONS OF ZIZIPHUS OENOPLIA AND ACARBOSE ON Α-GLUCOSIDASE The test was performed in triplicate; values (% enzyme activity), each value is presented as means ± S.E.M
FIG. 3: INHIBITORY EFFECT OF THE FRUIT EXTRACT/ SOLVENT FRACTIONS OF ZIZIPHUS OENOPLIA AND ACARBOSE ON Α-AMYLASE The test was performed in triplicate; values (% enzyme activity), each value is presented as means ± S.E.M
DISCUSSION: This work is accomplished to detect natural remedy which can be utilized in the prevention or treatment of diabetes. Phyto-constituents from plants have a traditional history of treating diabetes 35-36. Numerous compounds extracted from plants exhibit promising antioxidant and hypoglycemic potential, and sometimes they are more potent than oral hypoglycemic agents 37-38. It is proven that the effect of plant extract as α-amylase and α-glucosidase inhibitor is linked with the presence of some secondary metabolites as flavonoids, phenols, and tannins phytochemicals with antioxidant potential 39-40. So the present work analyzed the in vitro anti-oxidant and anti-diabetic potential of the ethanol crude extract and solvent fractions of Z. oenoplia fruit due to the presence of the phenolic and flavonoids compounds.
The preliminary phytochemical analysis of the ethanol crude extract and solvent fractions of Z. oenoplia indicates the presence of Alkaloids, Flavonoids, Saponins, Phenols, Tannins, and carbohydrates Table 1, which are responsible for anti-oxidant and hypoglycemic potential. This result was also reported in leaf by Soman S. et al, 2016 41.
In our study, the first report states the free radical scavenging potential of fruit crude extracts and frations of Z. oenoplia by using the DPPH assay method. During research EEZO show maximum antioxidant effect (87.66 ± 1.54% Inhibition at 640 μg/mL) followed by CF (66.76±0.92% Inhibition at 640 μg/mL) in dose dependant manner when compare to standard drug Ascorbic acid (90.72 ± 0.76% Inhibition) Table 2.
This could be attributed due to the presence of vitamin C, flavonoids and phenolic compounds in fruit of ZO. Vit. C and polyphenols have the capability to decrease oxidative stress; they can also prevent enzymes responsible for hydrolysis of carbohydrate as α-amylase and α-glucosidase 42-43.
According to the literature, flavonoids and polyphenols are able to prevent intestinal α-glucosidases and pancreatic α-amylase enzymes. So they can be used to treat postprandial hyper-glycemia 44-46. Fruit extract and solvent fractions of ZO in different concentrations inhibit both the enzymes in a dose-dependent manner, as presented in Table 3 and Table 4. The highest inhibition of α-glucosidases shown by EEZO (88.54 ± 1.7%
Inhibition at 800 μg/mL) followed by CF (84.16 ± 1.44% Inhibition) compare to reference drug acarbose (97.2 ± 0.48% Inhibition) while the PEF (70.34 ± 0.79%) produce the least Inhibition. In similar manner the highest inhibition of α- amylase shown by EEZO (85.43 ± 0.58% Inhibition at 800 μg/mL) followed by CF (72.58 ± 0.82% Inhibition) compare to reference drug acarbose (99.12 ± 0.72% Inhibition). Flavonoids, Tannins, and other poly-phenolic compounds are a major group of a secondary metabolite that has been reported to possess α-glucosidases and α-amylase enzyme inhibitory activity.
CONCLUSION: The result of this research revealed that fruit crude extract and solvent fractions of Z. oenoplia contain poly-phenols as flavonoids and tannin, which may be responsible for the DPPH free radical scavenging effect of crude extract and solvent fractions. The outcome of study also shows the α-amylase and α -glucosidase enzyme inhibitory potential of extracts and solvent fractions which may contribute to the hypo-glycemic and hypolipidemic effects. The outcome produces scientific support for the use of the plant as a folk remedy for the management of diabetes and its related complications.
ACKNOWLEDGEMENT: Nil
CONFLICTS OF INTEREST STATEMENT: We declare that we have no conflict of interest.
REFERENCES:
- Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM, Patel HR, Ahima RS and Lazar MA: The hormone resistin links obesity to diabetes. Nature 2001; 409(6818): 307-12
- International Diabetes Federation, DIABETES ATLAS, Ninth edition 2019
- Kaveeshwar SA and Cornwall J: The current state of diabetes mellitus in India. Australas Med J 2014; 7(1): 45-48
- Roglic G: WHO Global report on diabetes: A summary. Int J Non-Commun Dis 2016; 1(1).
- Okur ME, Karantas ID and Siafaka PI: Diabetes mellitus: a review on pathophysiology, current status of oral pathophysiology, current status of oral medications and future perspectives. Acta Pharm Sci 2017; 55(1): 61.
- Kifle ZD, Yesuf JS and Atnafie SA: Evaluation of in-vitro and in-vivo anti-diabetic, anti-hyperlipidemic and anti-oxidant activity of flower crude extract and solvent fractions of Hagenia Abyssinica (Rosaceae). Journal of Exp Pharmacol. 2020;12: 151-67.
- Shewasinad A, Bhoumik D, ZERO HISHE H and Masresha B: Antidiabetic activity of methanol extract and fractions of thymus schimperi ronniger leaves in normal and streptozotocin induce dia- betic mice. Iran J Pharmacol Ther 2018; 16(1): 1–8.
- Vinuthan M, Kumar V and Ravindra J: Effect of extracts of Murraya koenigii leaves on levels of blood glucose and plasma insulin in alloxan-induced diabetic rats. Indian Journal of Physiology and Pharmacology 2004; 48 (3): 348-52.
- Association AD: Diagnosis and classification of diabetes mellitus. Diabetes Care 2014; 37(Supplement 1): S81–S90.
- Mitra A, Dewanjee D and Dey B: Mechanistic studies of lifestyle interventions in Type 2 diabetes. World J Diabetes 2012; 3: 201-7.
- Hara Y and Honda M: The Inhibition of α-Amylase by Tea Polyphenols. Agri and Biolog Chem 1990; 54(8): 1939-45.
- Tsang MW: The management of type 2 diabetic patients with hypoglycaemic agents. ISRN Endocrinol 2012; 478120: 11-19
- Wadkar K, Magdum C, Patil S, Naikwade N. Anti-diabetic potential and Indian medicinal plants. J Herb Med Toxicol 2008; 2(1):45–50.
- Rizvi SI, Matteucci E and Atukeren P: Traditional medicine in management of type 2 diabetes mellitus. J Diabetes Res 2013; 1-2
- Elujoba AA, Odeleye O and Ogunyemi C: Traditional medicine devel- opment for medical and dental primary health care delivery system in Africa. Afr J Tradit Complement Altern Med. 2005; 2(1): 46–61.
- Mamun-or-Rashid A, Hossain MS, Hassan N, Dash BK, Sapon MA and Sen MK: A review on medicinal plants with antidiabetic activity. Int J Pharmacogn Phytochem 2014; 3(4): 149-59.
- Shukia R, Sharma S, Puri D, Prabhu K and Murthy P: Medicinal plants for treatment of diabetes mellitus. Indian J Clin Biochem 2000; 15(1): 169-77.
- Modak M, Dixit P, Londhe J, Ghaskadbi S and Devasagayam TP: Indian herbs and herbal drugs used for the treatment of diabetes. J Clin Biochem Nutr 2007; 40(3): 163-73.
- Mourya P, Shukla A, Rai G and Lodhi S: Hypoglycemic and hypolipidemic effects of ethanolic and aqueous extracts from Ziziphus oenoplia (L) Mill on alloxan-induced diabetic rats. Beni-Suef University Journal of Basic and Applied Sciences 2016; 6(1): 1-9
- Eswari ML, Bharathi RV and Jayshree N: Pharmacognostical and Physico-Chemical Standardization on the Leaves of Ziziphus oenoplia. International Journal of Green Pharmacy 2014; 267-70.
- Shukla A, Garg A, Mourya P and Jain CP: Zizyphus oenoplia Mill: A review on Pharmacological aspects. Advance Pharmaceutical Journal 2016; 1(1): 8-12
- Prema G and Chitra M: Anatomical Studies of the Fruit of Ziziphus rugosa. Asian Journal of Pharm Clin Res 2019; 12(8): 284-87.
- Thirugnanasampandan R, Ramya G and Bhuvaneswari G: Preliminary phyto-chemical analysis and evaluation of antioxidant, cytotoxic and inhibition of lipo-polysaccaride-induced NOS (iNOS) expression in BALB/c mice liver by Ziziphus oenoplia fruit. J Journal of Complementary and Integrative Medicine 2017; 231-37.
- Tambekar DH, Tiwari BK, Shirsat SD and Jaitalkar DS: Antimicrobial potential and phytochemical analysis of medicinal plants from Lonar Lake. International Journal of Life Sciences Biotechnology and Pharma Research 2013; 2(3): 1-4
- Singhal U, Goyal A, Solanki NS, Jain VK and Goyal PK: Pharmacognostical study on fruit of Ziziphus xylopyrus (retz.) Willd. International Journal of Drug Development & Research 2012; 4(3): 263-67.
- Bhandari MR, Jong-Anurakkun N, Hong G and Kawabata J: α -Glucosidase and α -amylase inhibitory activities of Nepalese medicinal herb Pakhanbhed (Bergenia ciliata, Haw.). Journal of Food Chemistry and Nutrition 2008; 106(1): 247-52
- Puls W, Keup U, Krause HP, Tomas G and Hofmeister F: Glucosidase inhibition. A new approach to the treatment of diabetes, obesity, and hyper lipo-proteinaemia. The Science of Nature 1977; 64(10): 536-37.
- Evans WC. Trease and Evans Pharmacognosy, 15th ed. New Delhi: Sounders-an imprint of Elsevier; 2002.
- Harborne JB: Phytochemical methods: A Guide to Modern Techniques of Plant Analysis, 3rd, New Delhi: Springer (India) 1998.
- Moein S, Farzami B, Khaghani S, Moein MR and Larijani B: Antioxidant properties and prevention of cell cytotoxicity of Phlomis persica Daru-Journal of Faculty of Pharmacy 2007: 15(2): 83-88.
- Moein S, Pimoradloo E, Moein M and Vessal M: Evaluation of antioxidant potentials and 𝛼-amylase inhibition of different fractions of labiatae plants extracts: as a model of antidiabetic compounds properties. Bio Med Research International 2017; 1-9.
- Omari N, Sayah K, Fettach S and El Blidi O: Evaluation of in-vitro antioxidant and antidiabetic activities of Aristolochia longa Evidence-based Complementary and Alternative Medicine 2019; 1-10.
- Marmouzi I, Karym EM and Saidi N: In-vitro and in-vivo antioxidant and anti-hyperglycemic activities of moroccan oat cultivars. Antioxidants 2017; 6(4): 102-07.
- Ademiluyi AO, Oboh G, Boligon AA and Athayde ML: Effect of fermented soybean condiment supplemented diet on 𝛼-amylase and 𝛼-glucosidase activities in Streptozotocin induced diabetic rats. Journal of Functional Foods 2014; 9(1): 1–9
- Moradi AF, Asghari B and Saeidnia S: In-vitro α-glucosidase inhibitory activity of phenolic constituents from aerial parts of Polygonum hyrcanicum. DARU, Journal of Pharmaceutical Sciences 2012; 20(1): 37.
- Kunyanga CN, Imungi JK, Okoth MW, Biesalski HK and Vadivel V: Total phenolic content, antioxidant and antidiabetic properties of methanolic extract of raw and traditionally processed Kenyan indigenous food ingredients. LWT Food Sci and Tech 2012; 45(2): 269-76.
- Rai PK, Srivastava AK, Sharma B, Dhar P, Mishra AK and Watal G: Use of laser-induced breakdown spectroscopy for the detection of glyce- mic elements in Indian medicinal plants. Evid Based Complement Alternat Med 2013: 1–9.
- Watal G, Dhar P, Srivastava SK, Sharma B. Herbal medicine as an alternative medicine for treating diabetes: the global burden. Evid Based Complement Alternat Med. 2014; 2014: 1–2.
- Hoon Jo S, Ka E and Lee H: Comparison of antioxidant potential and rat intestinal a-glucosidases inhibitory activities of quercetin, rutin, and isoquercetin. Int Journal of App Research in Natural Products 2009; 2(4): 52–60.
- Asadi S, Khodagholi F and Esmaeili MA: Chemical composition analysis, antioxidant, antiglycating activities and neuroprotective effects of choloroleuca, S. mirzayanii and S. santolinifolia from Iran. American Journal of Chinese Medicine 2011; 39(3): 615-38.
- Soman S and Ray JG: Silver nanoparticles synthesized using aqueous leaf extract of Ziziphus oenoplia (L.) Mill: Characterization and assessment of antibacterial activity. J of Photochemistry and Photobiology. 2016; 163: 1-10
- Ramkumar KM, Thayumanavan B, Palvannan T and Rajaguru P: Inhibitory effect of Gymnema montanum leaves on 𝛼-glucosidase activity and 𝛼-amylase activity and their relationship withpolyphenolic content. Medicinal Chemistry Research. 2010; 19(8): 948-61
- Padilla-Camberos E, Lazcano-Dıaz E and Flores-Fernandez JM: Evaluation of the inhibition of carbohydrate hydrolyzing enzymes, the antioxidant activity and the polyphenolic content of citrus limetta peel extract. The Scientific World Journal 2014; 4: 1-7.
- Mai TT, Thu NN, Tien PG and Chuyen N: Alpha-glucosidase inhibitory and antioxidant activities of Vietnamese edible plants and their relationships with polyphenol contents. JNSV 2007; 53(3): 267-76.
- Ramkumar KM, Thayumanavan B, Palvannan T and Rajaguru P: Inhibitory effect of Gymnema montanum leaves on α-glucosidase activity and α-amylase activity and their relationship withpolyphenolic content. Medicinal Chemistry Research 2010; 19(8): 948-61.
- Bothon FT, Debiton E and Avlessi F: In-vitro biological effects of two anti-diabetic medicinal plants used in Benin as folk medicine. BMC Complementary and Alternative Medicine 2013; 13(51): 326-31.
How to cite this article:
Goyal PK, Jeyabalan G and Singh Y: In-vitro free radical scavenging and hypoglycemic evaluation of fruit extract and solvent fractions of Ziziphus oenoplia mill (rhamnaceae). Int J Pharmacognosy 2021; 8(5): 216-23. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP. 8(5).216-23.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.