IN-VITRO CYTOTOXICITY OF ASSAY OF LEAVES OF PORTULACA QUADRIFIDA USING BRINE SHRIMP ASSAY
HTML Full TextIN-VITRO CYTOTOXICITY OF ASSAY OF LEAVES OF PORTULACA QUADRIFIDA USING BRINE SHRIMP ASSAY
R. Thirumalai Kumaran * and Nivetha Naidu
Department of Pharmacognosy, Faculty of Pharmacy, Sri Ramachandra Medical College and Research Institute, Porur, Chennai - 600116, Tamil Nadu, India.
ABSTRACT: Portulaca quadrifida Linn. (Portulacaceae) is commonly known as a chicken weed in English. In the traditional system, it is used to treat rheumatism and gynecological diseases, urinary tract infections, worm diseases, dysentery and dermatitis. To assess the cytotoxic potential of hydroalcoholic extract of P. quadrifida leaves using brine shrimp assay. Potassium dichromate was considered a standard reference drug. The concentrations of 10, 100, 1000 μg/ml were used in the experiment. The mature shrimps were taken, and it was treated with the extract and the standard in the respective concentrations. LC50 value was calculated for both standard and extract, and the probit analysis was made by using SPSS software. From the probit analysis, the cytotoxicity of the extract was determined by comparing it with the standard. The graph was plotted for both the standard and the extract; the extract was found to be non- toxic.
| Keywords: |
Brine shrimp assay, Cytotoxicity, SPSS Software, Portulaca quadrifida
INTRODUCTION: About the Plant: Portulaca quadrifida is mostly found in bare patches of ground and among rocks, on sandy or stony soils, from sea-level up to 2000 m altitude. Mostly it is growing on sand or sandy loams. P. quadrifida may occur on alkaline soils but is not so common on saline soils 1.
Cytotoxicity Studies: The term cytotoxicity is the quality of being toxic to the cells. When a particular cell is being treated with a cytotoxic compound, it can result in a variety of cell fates such as necrosis in which the cell membrane will lose its integrity, and cell lysis will take place. The membrane integrity can be assessed by monitoring the passage of substances that are normally sequestered inside cells to the outside.
Brine Shrimp assay is to measure the cytotoxicity of the extract of Portulaca quadrifida Linn, as it’s a new method and it hasn’t been carried out in many plants. In this method brine shrimp (Artemia salina) was used to determine the cytotoxicity when the extract was used as a feed substance for it and kept for few h. This procedure determines the LD50 values in microgram/ml of extract in the brine medium. The cytotoxicity studies are done to prove whether the plant is harmful or not since plants are used worldwide mainly as medicines and cosmetics. The cytotoxicity analysis was carried out on Portulaca quadrifida as it was found to be an anticancer drug and thus by doing the studies we wanted to prove whether it was toxic or not.
MATERIALS AND METHODS:
Preparation of Plant Extract: The leaves were initially separated and washed thoroughly with water and distilled water. The leaves were allowed to air dry for about 10 - 20 days. It is then finely powdered and placed in a container 3. The powdered crude drug was subjected to extraction with hydro-alcohol (1:1 ratio of ethanol and distilled water). The crude drug was subjected to a cold maceration for 72 h, and the extract was filtered out. The filtered extract was subjected to distillation to remove the solvent and further evaporated to concentrate the extract 4.
Brine Shrimp Lethality Assay:
Hatching of Artemia salina Cysts: 33.3g of red sea salt was dissolved in 500ml of sterilized distilled water. About 100g of cysts were allowed to soak in sterilized distilled water to bring back to room temperature. Then it is transferred to a separating funnel consisting of sterilized seawater. Suitable hatching environment should be maintained 5.
Dilution of Extracts: A concentration of 1000 µg/ml, 100 µg/ml, 10 µg/ml were prepared. A stock of 10 mg in 10 ml of sterilized distilled water was prepared. From stock, 1 ml was pipetted out to give 1000 µg/ml of concentration. 0.1 ml of stock was pipetted out, and the volume made up to 1 ml with sterilized distilled water to produce a concentration of 100 µg/ml. Similarly, 0.01 ml of stock was pipetted out, and the volume made up to 1ml with sterilized distilled water to produce a concentration of 10 µg/ml 6.
Plating of Nauplii to the Extracts: Potassium dichromate was used as an agent which induces cytotoxicity to cells. A solution of potassium dichromate was prepared with the same sterilized distilled water with a concentration of 1% potassium dichromate. In two different 24 well plates, 1% potassium dichromate and the dilutions prepared with extract were added in triplicates.
Potassium dichromate was also prepared for the same concentration as that of the extract. With 1ml of potassium dichromate and extract in the plates, the volume was made up with sterilized sea water to 3/4th of the capacity of the wells. Sterilized distilled water and seawater was used as control 6.
RESULTS AND DISCUSSION:
TABLE 1: NAUPLII OBSERVED IN HYDROALCOHOLIC PLANT EXTRACT OF PORTULACA QUADRIFIDA
| Concentration | No. of Nauplii Alive (24 h) | No. of Nauplii Alive (48) | ||||||
| T1 | T2 | T3 | Average | T1 | T2 | T3 | Average | |
| 10 µg/ml | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| 100 µg/ml | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| 1000 µg/ml | 8 | 7 | 8 | 8 | 7 | 8 | 7 | 7 |
| Control | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
TABLE 2: NAUPLII OBSERVED IN POTASSIUM DICHROMATE
| Concentration | No. of Nauplii Alive (24 h) | No. of Nauplii Alive (48 h) | ||||||
| T1 | T2 | T3 | Average | T1 | T2 | T3 | Average | |
| 10 µg/ml | 8 | 8 | 7 | 8 | 7 | 6 | 7 | 7 |
| 100 µg/ml | 5 | 4 | 5 | 5 | 3 | 4 | 4 | 4 |
| 1000 µg/ml | 3 | 3 | 2 | 3 | 2 | 2 | 3 | 2 |
| Control | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
Where, T1- Trial 1, T2 -Trial 2, T3 -Trial 3
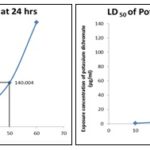
TABLE 3: LETHALITY DOSE FOR POTASSIUM DICHROMATE AT 24 h
| Log
Concentration |
Exposure Concentration
(µg/ml) |
Confidence Limits for Concentration | |
| Lower | Upper | ||
| 10 | 1.812 | 0 | 17.648 |
| 20 | 8.060 | 0 | 47.334 |
| 30 | 23.639 | 0 | 118.369 |
| 40 | 59.283 | 0.099 | 461.894 |
| 50 | 140.004 | 9.487 | 12160.922 |
| 60 | 330.639 | 61.07 | 4742346.880 |
TABLE 4: LETHALITY DOSE FOR POTASSIUM DICHROMATE AT 48 h
| Log
Concentration |
Exposure Concentration
(µg/ml) |
Confidence Limits For Concentration | |
| Lower | Upper | ||
| 10 | 1.812 | 0 | 9.092 |
| 20 | 8.060 | 0 | 22.698 |
| 30 | 23.639 | 0 | 48.0346 |
| 40 | 59.283 | 0.098 | 112.218 |
| 50 | 140.004 | 0.182 | 448.755 |
| 60 | 330.639 | 10.408 | 13871.776 |
FIG. 1: LETHALITY DOSE FOR POTASSIUM DICHROMATE AT 24 h and 48 h
From the number of shrimps that were alive after the treatment of extract and the standard, the LC50 value was calculated for both standard, and the extract and the Probit analysis were made by using SPSS software. Using Probit analysis, the cytotoxicity of the extract was determined by comparing it with the standard. The graph was plotted for both the standard and the extract; the extract was found to be non- toxic.
CONCLUSION: The Brine shrimp results in this study are interpreted as follows:
LC50 <1.0 µg/ml - Highly toxic
LC50 1.0 – 10.0 µg/ml - Toxic
LC50 10.0 – 30.0 µg/ml - Moderately Toxic
LC50 <100 µg/ml- Mildly Toxic
LC50 >100 µg/ml - Non-Toxic
The brine shrimp test for the plant Portulaca quadrifida results indicates that the leaf extract had LC50 values > 100 µg/ml which suggests that they are practically non-toxic when compared to that of Potassium dichromate. So, In conclusion, the leaf extracts of Portulaca quadrifida exhibited against the brine shrimp. This may be due to non-toxic active principles present in the extract. This confirms that the leaf can be used as a nutraceutical.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Naidu LM and Babu NK: Evaluation of the in-vitro antimicrobial activity of quadrifida Linn. International Journal of Plant Sciences 2009; 4(2): 494-497.
- Murty BS, Shankar P, Baldev R, Rath BB and Murday J: Textbook of Nanoscience and Nanotechnology. Springer Science and Business Media 2013; 216.
- Odeja O: phytochemical screening, antioxidant and anti-microbial activities of Senna occidentalis (Linn.) leaves extract - clinical phytoscience. International Journal of Phytomedicine and Phytotherapy 2015; 1: 6.
- Tiwari P, Kumar B, Kaur M, Kaur G and Kaur H: Phytochemical screening and extraction: A review. Internationale Pharmaceutica Sciencia 2011; 1(1): 98-106.
- Abosede WO, Sunday A and Jide AA: Toxicological investigations of Aloe ferox Mill. Extracts using Brine shrimp (Artemia salina) assay - Pakistan Journal of Pharmaceutical Sciences 2015; 28(2): 635-640.
- Suraj SK and Chaterjee P: Brine Shrimp cytotoxic activity of 50% aqueous ethanolic leaf extract of Calotropis procera Br. Asian Journal of Pharmaceutical and Clinical Research 2015; 8( 6): 215-217.
How to cite this article:
Kumaran RT and Naidu N: In-vitro cytotoxicity of assay of leaves of Portulaca quadrifida using Brine Shrimp assay. Int J Pharmacognosy 2018; 5(7): 437-39. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(7).437-39.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
10
437-439
531
1404
English
IJP
R. T. Kumaran * and N. Naidu
Department of Pharmacognosy, Faculty of Pharmacy, Sri Ramachandra Medical College and Research Institute, Porur, Chennai, Tamil Nadu, India.
kumarancognosist@gmail.com
27 March 2018
12 June 2018
17 June 2018
10.13040/IJPSR.0975-8232.IJP.5(7).437-439
01 July 2018