IN-VITRO AND IN-VIVOANTI-OXIDANT AND ANTI-DIABETIC EVALUATION OF CLADODES CRUDE EXTRACT AND SOLVENT FRACTIONS OF OPUNTIA ELATIOR MILL (CACTACEAE)
HTML Full TextIN-VITRO AND IN-VIVOANTI-OXIDANT AND ANTI-DIABETIC EVALUATION OF CLADODES CRUDE EXTRACT AND SOLVENT FRACTIONS OF OPUNTIA ELATIOR MILL (CACTACEAE)
Praveen Kumar Goyal * and G. Jeyabalan
Alwar Pharmacy College, MIA, Alwar, Rajasthan, India.
ABSTRACT: Objective: Ariel part of Opuntia elatior (Cactaceae) has been used in folklore health systems to treat diabetes in America, Mexico, and India. However, the anti-diabetic potential of cladodes of this medicinal plant is not scientifically validated and authenticated. The present study aimed to evaluate in-vitro and in-vivo anti-oxidant and anti-diabetic potential of methanol extracts and solvent fractions of O. elatior cladodes. Methods: The methanol extracts and solvent fractions of Opuntia elatior cladodes were evaluated at different concentrations (12.5-400 µg/ml) for anti-oxidant activity by using DPPH method and different doses (200 and 400 mg/kg body weight) for anti-diabetic potentials in staptozotocin induced diabetic albino rats. The extracts were administered for three weeks in different groups. Results: The acute toxicity study of Opuntia elatior cladodes extract, and fractions did not show mortality in the animals at the limit dose of 2000mg/kg during the observation period. The outcome of the present study indicates that extract and different fraction shows potential anti-oxidant activity. Cladodes also extract significantly decreases elevated level of blood glucose in dose dependant manner and also caused to reverse of the cholesterol, triglyceride, HDL, and LDL values when compared to untreated diabetic rats. Conclusions: The result indicates the beneficial effects of Opuntia elatior cladodes extract by inhibiting α–amylase, α–glucosidase, scavenging diphenyl-2-picryl-hydrazyl (DPPH) free radicals and improving serum lipid profile levels. The cladodes crude extract of Opuntia elatior are effective in lowering blood glucose and improving insulin levels in diabetic rats. The claimed traditional use as anti-diabetic has scientific ground.
Keywords: Antioxidant, Antidiabetic, α-amylase, α–glucosidase, Streptozotocin, Opuntia elatior
INTRODUCTION: One of the foremost causes of mortality and morbidity in humans is none other than diabetes mellitus 1. According to an estimation of the International Diabetes Federation, approximately 375 million people are suffering from diabetes, and this may double by 2030, in India 42 million people are suffering from diabetes, which is expected to increase to about 65 million people by the year 2025 2.
Diabetes mellitus represents a serious, chronic heterogeneous group of a metabolic disorder caused by an absolute or relative lack of insulin and/or reduced insulin activity or inherited and/or acquired deficiency in production of insulin which ends up in hyperglycemia and abnormalities in carbohydrate, fat and protein metabolism 3-4. Chronic hyperglycemia is associated with dysfunction of the heart, eyes, blood vessels, kidneys, nerves etc and it is characterized by symptoms like polyuria and polydipsia 2, 5.
Type 2 diabetes mellitus is considered one of the most recurrent lifestyle diseases. Type 2 is more prevalent than type 1, with about more than 90% of the total diabetic patients suffering from it. Type 2 diabetes (T2DM) is a disease caused by an imbalance between blood glucose absorption and insulin secretion.
Type 2 DM is a non-communicable disease which is leading causes of mortality worldwide due to associated long term side effects as ketoacidosis, hyperosmolarcoma accompanied with chronic disorders, retinopathy, nephropathy, neuropathy, skin complications, as well as increasing cardiovascular risks. Management of DM with none toxic effects remains a challenge for medical system. This results in an increasing search for improved anti-diabetic drugs 6-7.
Few of plant extract utilized in traditional medicine for diabetes received scientific scrutiny and WHO has recommended that this area warrants attention 6, 8. Despite considerable progress within the management of Type 2 DM by synthetic drugs, the design for natural anti-diabetic plant products for controlling diabetes is goes on. There are many hypoglycemic plants known through the folklore but their introduction into the modern therapy system awaits the invention of animal test system that closely parallel to the pathological course of diabetes in human beings 9-11.
Some medicinal herbs with proven anti-diabetic and related beneficial effects utilized in treatment of diabetes are Tinospora cordifolia, Gymnema sylvestre, Casearia esculenta, Syzygium cumini, Commiphora wightii, Gmelina arborea, Asparagus racemosus, Boerhavia diffusa, Sphaeranthus indicus, Pterocarpus marsupium, Tribulus terrestris, Phyllanthus amarus, Swertia chirata, Glycyrrhiza glabra, Gossypium herbaceum, Berberis aristata, Piper nigrum 9, 12, 13.
A traditional medicinal plant, widely distributed in the Rajasthan, known as Cactus, Prickly pears or Hathlo thore, Botanically identified as Opuntia elatior Mill. of family Cactaceae. Opuntia elatior is a large, succulent shrub, trunk-forming, segmented cactus that may grow upto 5–7 m with a crown of possibly 10 ft in diameter and a trunk diameter of 1 m 9 Cladodes are green to blue-green, bearing few spines up to 2.5 cm (0.98 in) or maybe spineless 14-17.
- elatior, whole or their parts like phylloclade, fruit, stem, flowers, leaves, and thorns were reported for their traditional-medicinal uses. O. elatior is traditionally used in different disease conditions like burning sensation in the stomach, abscess & wound, diphtheria 18-20, anaemia, hyperglycemia, antihyperlipidemic 21-23 analgesic, anti-inflammatory, anticancer and hyper cholesterolemic, anti-oxidant, antiulcer, antiviral, diuretics 24, asthma, cough, refrigerant, gonorrhea, Ophthalmia 25 Antileukemic 26 immunomodulatory, antiasthmatic, improve platelet function, neuro-protective, wound healing, monoamino-oxidase inhibitor and nutritional important etc. by tribal of Rajasthan 27-30. The phytochemical analysis of the Opuntia cladodes showed the presence of flavonoid, carbohydrate, tannin, protein, and pectin compounds. The cladodes of Opuntia elatior (Cactaceae) have been used in the treatment of Diabetes mellitus in American and Indian folk-medicine without any scientific verification for safety and efficacy 31–34. Thus, the objective of the present study is to evaluate the in-vitro and in-vivo anti-oxidant and anti-diabetic activity of cladodes crude extract and solvent fractions.
MATERIAL AND METHODS:
Plant Collection, Identification and Authentication: The cladodes of Opuntia elatior was collected from Sariska forest Alwar district, Rajasthan, India, in November 2019. The collected plants material were botanically identified and authenticated by Dr. L. K. Sharma, Department of Botany, Raj Rishi Govt. College, Alwar (India). The voucher specimens (002-APC/2019) are deposited in herbarium. The crude drug were cleaned, dried in shade and coarsely powdered. The powdered samples were kept in airtight container for further studies.
Chemicals and Drugs: Streptozotocin, Glibenclemide, DPPH and Methanol were purchased from Sigma-Aldrich Chemicals, St. Louis. Chemical kits for estimation of blood glucose, cholesterol, triglyceride, LDL and HDL were purchased from Erba Diagnostics, Mannheim.
Animals: The male albino rats (Wistar strain weighing 150-180 g) and albino mice ( weighing 20-30 g, Age 6-8 weeks) were procured from animal house of the Department of Pharmacology, Alwar Pharmacy Pharmacy, Alwar (Raj) India. They were kept at 27 ± 3 ºC (Relative humidity: 65% ± 10% and light / dark cycle for 12 h. All the animals were fed with rodent pellet diet and water was allowed ad-libitum under strict hygienic conditions. Institutional Animal Ethics Committee (IAEC) approved all the protocol of study (Reg. No. 963/PO/Re/S/06/CPCSEA).
Preparation of Plant Crude Extract: Cleaned, dried cladodes were weighed by digital weighing balance and a total of 80 g of coarsely powdered cladodes were used for extraction. An extract is prepared using the hot continous method. 80 g coarse cladodes powdered were defatted with 250 ml petroleum ether (50-60 ºC) using soxhlet apparatus. The dried residue (mark) obtained after defattation was packed in soxhlet apparatus and extracted with 250 ml methanol. Extraction was continued until a drop of outcome from the siphon tube, when taken on TLC plate and sprayed with conc. H2So4 does not give a spot. Light greenish brown extract obtained and solvent was evaporated first at water bath than under reduced pressure.
Fractionation of Crude Extract: Fractionation was performed by dissolving crude methanol extract in distilled water and partitioned with chloroform (1.5l × 3 times), ethyl acetate (1.5 l × 3 times) respectively. The fractions were subjected individually to evaporation using rotary evaporator.
Preliminary Phytochemical Screening of Cladodes Crude Extracts: Standard Preliminary phytochemical qualitative analysis of the extract was carried out for various plant constituents. The crude extract was screened for the presence or absence of secondary metabolites such as Reducing sugars, Alkaloids, Steroidal compounds, Phenolic com- pounds, Cardiac glycosides, Flavonoids, Saponins, Tannins and Anthraquinones using standard procedures 35-36.
Acute Oral Toxicity Study: The acute oral toxicity studies of crude extracts were carried out as per the guidelines of the Organization for Economic Co-operation Development (OECD), draft guidelines 423 adopted on 17th December 2001 37. An acute toxicity of MEOE (Methanol Extract of Opuntia elatior) was carried out in 5 female albino mice (25-30 g, 6–8 weeks). The mice were divided in to three groups (n=6). All mice were fasted (food but not water) for 3 h before and 1 h after administration of the extract. A limit dose of 2000 mg/kg was given for the first mice.
On the basis of result, four additional mice were dosed sequentially. The animals were housed separately and observed for the manifestation of gross behavioural and physical toxicities like changes in skin, urination, lacrimation, reduction in feeding activity, excitation, paw licking, increased respiratory rate, decreased motor activity, diarrhea, weight loss and paralysis continuously for the first 30 min and intermittently for 4 h, over a period of 24 h and later followed for 14 days for an interval of 24 hrs for any lethality.
Experimental Group and Dose: In the experiment, a total of 30 rats were divided into 5 groups (n=6) for the oral/i.p. administration of extracts/drugs or vehicle Table 1.
TABLE 1: GROUPING AND DOSE OF DRUG FOR ANTI-DIABETIC ACTIVITY
| Group | Drug and Doses |
| Streptozotocin -induced diabetes | |
| Group I | Normal Control
(Group NC, Received 0.5% CMC) |
| Group II | Diabetic Control Streptozotocin (60mg/kg i.p) |
| Group III | O. elatior 200mg (oral) and inducing material(i.p.) |
| Group IV | O. elatior 400mg (oral) and inducing material(i.p.) |
| Group V | GLC 5mg/kg (oral) and inducing material(i.p.) |
Induction of Diabetes in Rats: Streptozotocin (STZ) dissolved in 0.1 M citrate buffer (pH=4.5) was injected in rats intraperitoneally at dose of 60 mg/ kg body weight for induction of diabetes. After 72 h of STZ injection, the rats were screened for DM. Rats that showed FBG level >200 mg/dl were used for the study.
Biochemical Estimation: Serum samples from all the experimental rats were collected for estimation of biochemical parameters, serum glucose (GOD-POD method), cholesterol (CHOD-PAP method), triglycerides (GPO-Triender method), HDL and LDL 29.
Statistics: All values are expressed as Mean ±SEM. The differences were compared using one way analysis of variance (ANOVA) followed by Tukey’s multiple comparison tests. P values < 0.05 were considered as significant.
Results:
Preliminary Phytochemical Screening: Preliminary phytochemical screening was done for the cladodes crude extract of O. elatior resulted in the presence of Alkaloids, Saponins, Tannins, Phenols, Flavonoids, and Steroids.
Terpenoids and Glycosides were not present in the phytochemical screening Table 2.
TABLE 2: PHYTOCHEMICAL SCREENING OF CLADODES CRUDE EXTRACT
| S. no. | Secondary Metabolites | Test Results |
| 1 | Alkaloids | + |
| 2 | Saponins | + |
| 3 | Tannins | + |
| 4 | Terpenoids | + |
| 5 | Phenols | + |
| 6 | Flavonoids | + |
| 7 | Glycosides | + |
| 8 | Steroids | + |
| 9 | Anthraquinones | + |
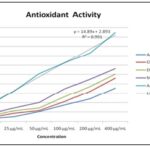
In-vitro Antioxidant Potential: DPPH Free Radical Scavenging Activity: The experiment was performed to assess the anti-oxidant potential of MEOE by scavenging the DPPH free radicals. The better anti-oxidant activity was reflected by, the lower IC50 (Concentrations that led to 50% of inhibition) values. The concentration of ascorbic acid, cladodes crude extracts, and solvent fractions varied from 12.5 to 400 µg/mL. MEOE and solvent fractions exhibited significant anti-oxidant activity as the extract contains Vitamin C. Among extracts, the highest inhibitory activities were shown by the crude cladodes methanol extract (MEOE) with an IC50 value 5.53 ± 0.54 µg/mL (56.21% inhibition at 400 µg/mL) using Ascorbic Acid as reference standard displaying IC50 value 3.56 ± 0.94 µg/mL (94.64% inhibition at 400 µg/mL). There was a dose-dependent increase in the percentage of free radical scavenging potency for all concentrations tested. The IC50 values of aqueous fraction, chloroform fraction and ethyl acetate fraction were not significant. The potential of the MEOE was found to be less than that of the reference anti-oxidant value; however, the MEOE has demonstrated significant DPPH radical scavenging potential Table 3 and Fig. 1.
% scavenging effect = Control absorbance - sample absorbance × 100
Control Absorbance: Each experiment was carried out in triplicate and results are expressed as means % scavenging effect ± SD.
TABLE 3: ANTI-OXIDANT ACTIVITIES OF THE CRUDE EXTRACT AND SOLVENT FRACTIONS
|
Concentration |
Percentage (%) Inhibition of DPPH | ||||
| Aqueous Extract | Chloroform Fraction | Ethyl Acetate Fraction | Methanol Crude Extract | Ascorbic Acid | |
| 12.5 μg/mL | 3.14±0.16 | 4.02±0.34 | 5.32±0.43 | 8.12±0.34 | 17.24±0.35 |
| 25 μg/mL | 5.14±0.43 | 8.65±0.76 | 11.32±0.44 | 14.32±0.54 | 32.12±0.54 |
| 50 μg/mL | 10.43±0.21 | 11.65±0.94 | 14.32±0.65 | 20.12±0.54 | 50.82±0.12 |
| 100 μg/mL | 17.34±0.45 | 21.12±0.14 | 26.12±0.43 | 34.72±0.82 | 62.24±0.76 |
| 200 μg/mL | 24.23±0.12 | 32.12±0.43 | 37.32±0.82 | 45.14±0.32 | 73.08±0.92 |
| 400 μg/mL | 35.52±0.62 | 46.32±0.76 | 50.43±0.12 | 56.21±0.15 | 94.64±0.72 |
| IC50 μg/mL | 08.84±0.13 | 7.36±0.64 | 6.37±0.54 | 5.53±0.54 | 3.56±0.94 |
Notes: Each value of Percentage inhibition of DPPH free radical is presented as means ± S.E.M., n = 3
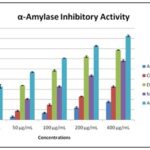
In-vitro α-amylase Inhibitory Activity of MEOE Cladodes and Solvent Fractions: α -amylase is the key enzyme associated with the hydrolysis of polysaccharides into the disaccharides and absorbable monomers.
In this study, the potential of MEOE cladodes as anti-hyperglycemic was evaluated along with its fractions to inhibit this enzyme; acarbose was used as a positive control Table 4 and Fig. 2.
FIG. 1: IN-VITRO ANTI-OXIDANT ACTIVITY OF MEOE CRUDE EXTRACT AND FRACTIONS BY DPPH INHIBITION ASSAY
TABLE 4: Α-AMYLASE INHIBITORY EFFECT OF THE CRUDE EXTRACT AND FRACTIONS
| Concentration | Percentage (%) Inhibition | ||||
| Aqueous fraction | Chloroform Fraction | Ethyl Acetate Fraction | Methanol Crude Extract | Acarbose | |
| 25 μg/mL | 1.12±0.92 | 3.21±0.60 | 15.32±0.75 | 8.76±0.91 | 32.76±1.87 |
| 50 μg/mL | 2.95±0.78 | 8.54±0.48 | 33.65±0.12 | 20.48±0.48 | 46.92±0.39 |
| 100 μg/mL | 6.78±0.45 | 14.65±0.94 | 48.44±0.67 | 32.72±0.62 | 60.58±0.14 |
| 200 μg/mL | 11.8±0.87 | 22.87±0.37 | 62.05±0.82 | 43.42±0.67 | 72.71±0.49 |
| 400 μg/mL | 17.43±0.72 | 32.54±0.91 | 68.63±0.28 | 58.07±0.94 | 82.62±0.87 |
| IC50 μg/mL | 13.14±0.63 | 07.61±0.36 | 3.32±0.92 | 4.42±0.86 | 2.27±0.48 |
FIG. 2: Α-AMYLASE INHIBITORY POTENTIAL OF THE CRUDE EXTRACT AND FRACTIONS
The result showed that aqueous fraction (AF) was inactive while the ethyl-acetate fraction (EAF) was the most potent inhibitor of α- Amylase (IC50 = 3.32±0.92) followed by methanol crude extract (IC50 = 4.42 ± 0.86) with competing IC50 values.
The Chloroform fraction (CF) was found to be very less potent for inhibition of α- amylase. These results suggest that the EAF and MEOE displayed the potential to inhibit this enzyme with greater potency compared to that of standard inhibitor Acarbose (IC50 = 2.27±0.48) Table 4.
Acute Toxicity Test: The acute oral toxicity study indicated that the crude extract caused no mortality in limit dose of 2000 mg/kg within the first 24 h as well as for the following 14 follow-up days. Physical and behavioral observations of the experimental mice and rats also revealed no visible signs of overt toxicity. This indicates that the extract's median lethal dose (LD50) is greater than 2000 mg/kg.
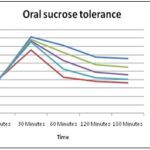
Oral Sucrose Tolerance in Rats: The methanol extract was evaluated in-vivo at three different concentrations under specified conditions for sucrose tolerance test in rats fed with sucrose. Among these concentrations, the 400 mg/kg displayed potent anti-hyperglycemic activity and significantly prevented a sudden rise in blood glucose levels.
The observations were in line with the results obtained in in-vitro enzyme inhibition assays and lead to conclude that the methanol extract inhibited the enzymes responsible for the hydrolysis of sucrose into absorbable sugar in the mice model. The results are shown in Table 5 and Fig. 3.
TABLE 5: ORAL SUCROSE TOLERANCE TEST IN RATS
|
Group |
Blood Glucose Level (mg/dl) | ||||
| 0 Min | 30 Min | 60 Min | 120 Min | 180 Min | |
| DW 10 | 80.32±2.54 | 182.45±2.44 | 162.34±6.72 | 134.67±3.72 | 130.65±4.68 |
| DC | 80.32±2.54 | 182.45±2.44 | 178.12±1.65 | 173.43±0.44 | 172.65±1.82 |
| GLC5 | 81.87±1.98 | 150.82±3.32 | 86.12±6.10 | 76.67±5.12 | 72.67±3.76 |
| MEOE100 | 82.12±2.12 | 176.33±4.65 b | 145.50±9.62 a | 116.20±5.92 | 110.43±5.42 a |
| MEOE200 | 82.42±2.45 | 172.67±3.82 b | 126.33±8.08 | 98.6±3.65 a | 92.6±3.92 a |
| MEOE400 | 80.92±3.45 | 170.26 ±4.65 b | 104.60 ±4.42 a | 84.42±3.65 | 80.8±2.82 |
Notes: Values are expressed as mean ± SEM (n=6) a P<0.05 b P<0.01 compared to diabetic control.and analyzed by one-way ANOVA followed by Tukey’s post test.; Abbreviations: MEOE-Methanol Extract of Opuntia elatior; DW, distilled water; GLC, glibenclamide.
FIG. 3: EFFECT OF O. ELATIOR CLADODES CRUDE EXTRACT ON THE BLOOD GLUCOSE LEVEL OF ORAL SUCROSE LOADED MICE
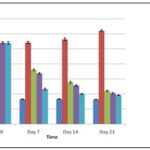
Anti-diabetic activity in Streptozotocin-induced Diabetes Rat model: MEOE on oral administration had significantly lowered the blood glucose level on the administration of MEOE over the experimental period of 21 days.
The experimental animals treated with MEOE with a dose of 200 mg/kg and 400 mg/kg exerted a notable reduction (P < 0.05) in blood glucose levels on the 7th, 14th and 21st days for 400 mg/kg dose and 14th and 21st days in case of 200 mg/kg dose respectively in comparison with STZ treated rats. The results were displayed in Table 6 and Fig. 4.
Administration of MEOE 200 and 400 mg/kg for seven-days STC, STG and LDL levels were significantly (P<0.001) increased whereas HDL was decreased in diabetic rats compared to normal rats Table 7.
TABLE 6: BLOOD GLUCOSE-LOWERING EFFECT OF MEOE
| Groups | Blood Glucose Level (mg/dl) | |||
| Day 0 | Day 7 | Day 14 | Day 21 | |
| Normal Control 0.5% CMC (1 ml/kg; p.o) | 84.25 ± 2.65 | 82.18 ± 2.24 | 82.89 ± 1.54 | 81.85 ± 2.76 |
| Diabetic Control Streptozotocin (60 mg/kg; b.wt; i.p) | 275.78 ±4.20 | 271.45±3.87 | 282.19 ± 4.12 | 310.48 ±2.67 |
| Streptozotocin +MEOE (200 mg/kg, b.wt; p.o) | 270.49 ± 3.75 | 180.25 ± 2.76 | 138.82 ± 3.22 | 110.22 ± 2.43 |
| Streptozotocin +MEOE (400 mg/kg, b.wt; p.o) | 270.49 ± 3.65 | 168.25 ± 2.34 | 127.82 ± 3.12 | 102.22 ± 1.78 |
| Streptozotocin+Glibinclamide (5 mg/kg, b.wt; p.o) | 269.78 ± 4.45 | 115.85 ± 3.65 | 100.01 ± 1.87 | 95.92 ± 1.24 |
FIG. 4: EFFECT OF MEOE ON STREPTOZOTOCIN-INDUCED DIABETIC RATS
TABLE 7: BIOCHEMICAL PARAMETERS OF NORMAL AND EXPERIMENTAL ANIMALS ON 7TH DAY POST TREATMENT
|
Groups |
Treatment |
Serum Lipid Parameters ((mg/dl)) | |||
| Total Cholesterol | Triglycerides | HDL | LDL | ||
| I | Normal control | 66.34±2.65 | 84.82±2.72 | 42.60± 3.72 | 46.53±4.36 |
| II | Diabetic Control | 128.28±4.01 | 157.24±3.12 | 23.14 ±5.42 | 128.23±5.12 |
| III | Diabetic + MEOE 200 mg/kg | 96.76±3.32 * | 115.60±2.86*** | 30.14±4.82** | 112.34±3.52*** |
| IV | Diabetic + MEOE 400 mg/kg | 90.12±1.45 ** | 98.58±3.18*** | 34.30±3.72*** | 82.24±2.65** |
| V | Diabetic + Glibenclamide 5 mg/kg | 78.56±2.32*** | 92.42±3.34** | 38.62±3.34* | 64.62±4.37** |
Values are expressed as Mean ±SEM; (n = 5), * P < 0.05, ** p < 0.01, *** P<0.001 compared with untreated diabetic rats
Assessment of Serum Insulin Level on MEOE Treatment: This experimental investigation displays the anti-diabetic potential of MEOE where streptozotocin-induced diabetic rats significantly (p<0.05) reduced the insulin level, and MEOE could bring the reduced insulin level to near normal limit (p<0.001)
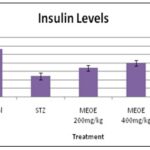
FIG. 5: EFFECT OF MEOE ON SERUM INSULIN LEVEL IN DIABETIC RATS The data are presented as mean ± S.E.M. n = 5 and analyzed using One-way ANOVA followed by post hoc Tukey’s test. * P < 0.05 was considered significant as compared to the control group.
TABLE 8: EFFECT OF MEOE ON SERUM INSULIN LEVEL IN DIABETIC RATS
| Groups | Insulin(μlU/ml) |
| Control 0.5% ,CMC (1 ml/kg; p.o) | 12.24 ± 0.65 |
| Streptozotocin, (60 mg/kg; b.wt; i.p) | 4.82 ± 0.82 |
| Streptozotocin + MEOE,
(200 mg/kg, b.wt; p.o) |
6.72± 0.66* |
| Streptozotocin + MEOE
(400 mg/kg, b.wt; p.o) |
7.94± 0.66* |
| Streptozotocin + Glibinclamide
(5 mg/kg, b.wt; p.o) |
9.65 ± 0.76 |
DISCUSSION: In the present study methanol crude extract and fractions of Opuntia elatior cladodes were evaluated for their in-vitro as well as in-vivo anti-oxidant and anti-diabetic activities via DPPH inhibition assay, α-Amylase Inhibitory potential, glucose tolerance test as well as improvement in blood glucose and insulin profile in STZ induced wistar albino rats as compared to the standard glibenclamide, a hypoglycaemic drug in order to validate their effect. Diabetes being a metabolic disorder, bears a lot of complications if it is not properly managed. Unavailability of suitable anti-diabetic agents and side effects associated with synthetic drug regimes prompt us to search out newer anti-diabetic agents from natural resources. This study sketches out the anti-diabetic potential of medicinal plants from Sariska forest of the Alwar district of India. Preliminary phytochemical analysis showed the presence of secondary metabolites as saponins, phenolic acid, flavonoids, phytosterols, and alkaloids in cladodes of this plant. The anti-diabetic activity in plants may be due to the presence of phenolic and flavonoid compounds. In this study, the crude extract and ethyl acetate fraction of selected plants were found more active than other fractions, indicating that compounds having anti-diabetic activity are polar in nature which established the significant correlations of polyphenolic and flavonoids to lower down the diabetic effect.
Free radical scavenging effect is very important to prevent the deleterious role of radicals in various disorders, including diabetes. DPPH free radical scavenging is an accepted mechanism by which anti-oxidants act to inhibit lipid peroxidation. The DPPH radical scavenging activity of crude extract and EA fraction increased with an increase in concentration Fig. 1. In DPPH assay, the extract and EA fraction showed a notable radical scavenging activity in a dose-dependent manner and was significantly different (p < 0.05). α- amylase inhibitory activity of the crude extract and solvent fractions of O. elatior was in a dose-dependent manner. Most potent inhibition was shown by the ethyl acetate solvent fraction while the aqueous fraction showed the least effect. OSTT is a measure of the body’s ability to utilize sugars. At 30 min of sucrose administration, the peak of blood glucose level became higher and then consequently lowered. The postprandial glucose-lowering ability of the extract may be accredited to the embarrassment of glucose absorption, stimulation of peripheral glucose utilization, reduction in glycogenolysis, and gluconeogenesis. This finding gives confident proof that the claimed medicinal plant has anti-diabetic activity. An acute oral toxicity study revealed the non-toxic nature of the methanol extract of cladodes.
STZ causes alkylation of pancreatic deoxy-ribonucleic acid by entering to the 𝛽-cell via glucose transporter 2 and induces activation of poly (ADP-ribosylation) that causes depletion of cellular nicotinamide adenine dinucleotide (NAD+) and adenosine triphosphate. As a result, the generation of free radicals causes pancreatic 𝛽-cells necrosis 38. In this study, induction of type 2 diabetes showed significantly increased blood glucose level and decreased insulin level compared to control rats which confirm the induction of diabetes, and it may be due to partial necrosis of pancreatic 𝛽cell by STZ. Oral administration of MEOE (200 and 400 mg/kg dose) and glibenclamide to the diabetic rats showed a significant reduction of blood glucose and increase in insulin level than diabetic control rats. Hence, MEOE mediated the above effect possibly due to its preventive effect on STZ-mediated 𝛽-cell damage in diabetic rats and thereby increases insulin release.
CONCLUSION: The present study indicated that the cladodes of the plant O. elatior possessed the highest phenolic and flavonoid content and produces strong anti-diabetic and anti-oxidant activities, which were comparable to the commercial anti-dibetic drug Glibenclemide and anti-oxidant ascorbic acid. The results were also verified by inhibition of intestinal α-amylase and free radical scavenging activity by the extracts, contributing to the anti-hyperglycemic and anti-oxidant activity. This seems that the Opuntia elatior extract can be used as a natural anti-diabetic and anti-oxidant agent.
ACKNOWLEDGEMENT: The authors are gratefully acknowledged the Management of Alwar Pharmacy College, IET group of Institution Alwar, (Raj) for facilities provided during the study.
CONFLICTS OF INTEREST STATEMENT: We declare that we have no conflict of interest.
REFERENCES:
- Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM, Patel HR, Ahima RS and Lazar MA: The hormone resistin links obesity to diabetes. Nature 2001; 409(6818): 307-12
- International Diabetes Federation, DIABETES ATLAS, Ninth edition 2019.
- Roglic G: WHO Global report on diabetes: A summary. Int J Non-Commun Dis. 2016; 1(1): 1-12
- Okur ME, Karantas ID and Siafaka PI: Diabetes mellitus: a review on pathophysiology, current status of oral pathophysiology, current status of oral medications and future perspectives. Acta Pharm Sci 2017; 55(1): 61.
- Shewasinad A, Bhoumik D, Zero Hishe H and Masresha B: Anti-diabetic activity of methanol extract and fractions of thymus Schimperi ronniger leaves in normal and streptozotocin induce dia- betic mice. Iran J Pharmacol Ther. 2018; 16(1): 1-8.
- Mitra A, Dewanjee D and Dey B: Mechanistic studies of lifestyle interventions in Type 2 diabetes. World J Diabetes 2012; 3: 201-7.
- Association AD: Diagnosis and classification of diabetes mellitus. Diabetes Care 2014; 37(Supplement 1): S81–S90.
- Wadkar K, Magdum C, Patil S and Naikwade N: Anti-diabetic potential and Indian medicinal plants. J Herb Med Toxicol. 2008; 2(1): 45–50.
- Rizvi SI, Matteucci E and Atukeren P: Traditional medicine in manage- ment of type 2 diabetes mellitus. J Diabetes Res 2013; 2013.
- Elujoba AA, Odeleye O and Ogunyemi C: Traditional medicine devel- opment for medical and dental primary health care delivery system in Africa. Afr J Tradit Complement Altern Med 2005; 2(1): 46–61.
- Mamun-or-Rashid A, Hossain MS, Hassan N, Dash BK, Sapon MA and Sen MK: A review on medicinal plants with anti-diabetic activity. Int J Pharmacogn Phytochem 2014; 3(4):149-59.
- Shukia R, Sharma S, Puri D, Prabhu K and Murthy P: Medicinal plants for treatment of diabetes mellitus. Indian J Clin Biochem. 2000; 15(1):169-77.
- Modak M, Dixit P, Londhe J, Ghaskadbi S, Devasagayam TP: Indian herbs and herbal drugs used for the treatment of diabetes. J Clin Biochem Nutr 2007; 40(3): 163-73.
- Prajapati S, Harisha CR and Acharya RN: Pharmaco-gnostic evaluation of stem of elatior Mill. (Nagaphani). Euro J of Biomedi and Pharma Sci 2015; 2(2): 351-57.
- Kaur M, Kaur A and Sharma R: Pharmacological actions of Opuntia ficus indica: A Review, Journal of Applied Pharmaceutical Science 2012; 2(7): 15-18.
- Kuti JO: Anti-oxidant compounds from four Opuntia cactus pear fruits varieties, Food Chemis 2004; 85: 527-33.
- New Delhi, India: National Institute of Science Communication, CSIR; IV; 2001. Anonymous, A Dictionary of Indian Raw Material and Industrial products, raw material; The Wealth of India; 223-224.
- Kirtikar KR and Basu BD: Indian Medicinal Plants. 2nd edition. Pub by Int Bo Dist Dehradun India 1999; 783-84.
- Khare CP: Indian Medicinal Plants. Published by Springer-Verlag Heidelberg; 2004; 451-52.
- Prajapati S and Acharya R: Opuntia elatior (Nagaphani): A Review on its Ethnobotany, Phyto-chemical and Pharmacological Properties, Annals of Ayurvedic Medicine2015; 4(3): 107-15
- Patel H, Prajapati S and Acharya R: A preliminary phyto-pharmacognostical evaluation of Opuntia elatior (Nagaphani or Hathalo-thore) fruit, International Journal of Ayurvedic Medicine 2015; 6(4): 310-16.
- Wadie N: An Overview on Nutraceuticals and Herbal Supplements for Diabetes and Metabolic Syndrome, Nutritional and Therapeutic Interventions for Diabetes and Metabolic Syndrome 2012; 355-65.
- Arumugam G, Manjula P and Paari N: A review: Anti diabetic medicinal plants used for diabetes mellitus, Journal of Acute Disease, ELSEVIER 2013; 196-00.
- Caroline AG, Ekavi NG, Duane DM, Andrew M and Naumovski N: Effects of the Consumption of Prickly Pear Cacti (Opuntia spp.) and its Products on Blood Glucose Levels and Insulin: A Sys Rev Medic 2019; 55(138): 1-18.
- Chauhan SP: Phytochemical and pharmacological screening of fruit of Opuntia elatior , PhD thesis, Saurashtra University, Rajkot 2010.
- Trivedi PC: Medicinal Plant Utilization and Conservation. 2nd Edition. Published by Aavishkar publishers and Distributer; Jaipur; 2016: 165-66.
- Itankar P, Acharya S and Arora SK: Opuntia elatior: exploring antileukemic potential, published by: LAP LAMBERT Academic Publishing 2013; 23-26.
- Chauhan SP, Sheth NR and Rathod IS: Phytochemical Screening of Fruits of Opuntia elatior , American Journal of Pharmtech Research 2013; 3(2): 1-16.
- Dashahre AK, Navaneethan B, Bhutt P and Mahato S: Medicinal Plants of Sariska Tiger Reserve (Rajasthan) India. J of Medi Plants and Studies 2014; 2(2): 137-46.
- Itankar P, Acharya S, Arora SK and Thakre P: Phytochemical study and evaluation of antileukemic activity of ripe fruit of Opuntia elatior , Ancient Science of Life 2012; 32(1): 47.
- Galal, TM, Hassan LM, Youssef AK and Heba I: Micro-morphology and Phytochemical Screening of Opuntia littoralis englem. Cladodes. Egyptian Journal of Desert Research 2017; 67(1): 153-68.
- Chauhan SP, Sheth NR, Rathod IS, Suhagia BN and Maradia RB: Phytochemical Screening of Fruits of Opuntia elatior American Journal of Pharmtech Research 2013; 3(2): 1-16.
- Santos Diaz MS and Barbarosa AP: Opuntia spp.: Characterization and Benefits in Chronic Diseases, Oxidative Medicine and Cellular Longevity 2017; 1-25.
- Sanyal S, Rabinarayan A, Kumar M and Gamit R: Toxicological study of Opuntia elatior , Fruit (ripen) juice: A folklore medicinal plant. International Journal of Green Pharmacy 2016; 9.
- Evans WC: Trease and Evans Pharmacognosy, 15th ed. New Delhi: Sounders-an imprint of Elsevier; 2002.
- Harborne JB: Phytochemical methods: A Guide to Modern Techniques of Plant Analysis, 3rd, New Delhi: Springer (India) 1998.
- Organization for Economic Co-operation and Development (OECD). OECD guidelines for the testing of chemicals. Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure 2008.
- Szkudelski T: “The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas”, Physiological Research 2001; 50(6): 537-46.
How to cite this article:
Goyal PK and Jeyabalan G: In-vitro and in-vivo anti-oxidant and anti-diabetic evaluation of cladodes crude extract and solvent fractions of Opuntia elatior mill (cactaceae). Int J Pharmacognosy 2021; 8(6): 257-65. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.8(6).257-65.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.