IN VITRO AND IN VIVO TOXICOLOGICAL EVALUATION OF AQUEOUS EXTRACTS FROM BENINESE PLANTS: SENNA SIAMEA LEAVES AND SARCOPCEPHALUS LATIFOLUS STEM ROOTS
HTML Full TextIN-VITRO AND IN-VIVO TOXICOLOGICAL EVALUATION OF AQUEOUS EXTRACTS FROM BENINESE PLANTS: SENNA SIAMEA LEAVES AND SARCOPCEPHALUS LATIFOLUS STEM ROOTS
Kpèdétin Wilfrid Dieu-Donné Agbodjogbe 1, 4, Alban Gouton Houngbeme * 3, Mahamane Haïdara 4, Rokia Sanogo 4, 5, Anatole Laleye 2 and Houndjovi Pierre Dansou 1
Laboratoire de Physiologie de l’Effort 1, Institut National de la Jeunesse, d’Education Physique et du Sport (INJEPS), Université d’Abomey-Calavi, BP 169 Porto-Novo, Bénin.
Laboratoire de Cytogénétique and Biologie Cellulaire 2, Faculté des Sciences de la Santé, Université d’Abomey-Calavi 01BP 188 Cotonou, Bénin.
Laboratoire National de Pharmacognosie 3, Institut de Recherche et d’Expérimentation en Médecine et Pharmacopée Traditionnelles (IREMPT), Centre Béninois de Recherche Scientifique et Innovation (CBRSI), Benin
Faculté de Pharmacy 4, USTTB, Benin
Département de la Médecine Traditionnelle du Mali 5, Benin.
ABSTRACT: The toxicity of aqueous extracts of S. siamea leaves, S. latifolius roots collected in Benin and their mixture (1/1, w/w), was evaluated on female non-pregnant rats, Albino wistar genus. They were treated for two weeks, with various extracts administered by the oral way at 2000 mg/kg body weight. The biochemical parameters were determinate, histological analysis of the rats was done, and the urinary concentration of neutral gelatinase-associated lipocalin (NGAL) was measured. The treatment of the rats with different extracts not caused undesirable effects and deaths during the fourteen days of observation. Significant changes are relative of a histological aspect of hepatocyte and renal cortex as well as significant increases in the activity of enzyme serum alanine aminotransferases (ALT) and aspartate aminotransferases (AST). The aqueous extracts at 2000 mg/kg were disrupted some biochemical parameters. These changes were confirmed by the rate raised from NGAL noted in the urine of the treated rats.
| Keywords: |
S. siamea, S. latifolius, Aqueous extract, Toxicity, Benin
INTRODUCTION: The plants are harvested and fluently used for foods and medicines purposes 1, 2. Alcoholic extracts of leaves of Senna siamea have been shown anti-cancerous and antioxidant activities 3.
In West African countries, its peel and its eraser are used to treat cancers and the tumors of the ear, eyes and genital organs. In Benin, the decoction of the root is used against fever, constipation, hypertension and insomnia 4. In the north of Nigeria, the peel of Sarcocephalus latifolius is used in brewing as a diuretic and anti-helminthic 5. Particularly, its root is used to fight against various infections such as malaria and stomach pains 6. Other studies used the extracts of peel stems to treat pain, inflammation and malaria effects 7, 8. Unfortunately, very little studies have done on the acute toxicity of those plants which are recognized to have enormous pharmacological wealth. The objective of the present work is to evaluate the toxicity of aqueous extracts of S. siamea leaves, of S. latifolius roots and their mixture (w/w) on the rats.
MATERIAL AND METHODS:
Material: The leaves of S. siamea and the roots of S. latifolius were collected in March 2013, respectively in Adjarra and Porto-Novo at Benin. The plants were authenticated at the National herbarium from Abomey-Calavi University. The voucher specimens were kept under the numbers: AA6414/HNB (S. siamea) and AA6415/HNB (S. latifolius). They were dried in the laboratory room at constant temperature (air conditioning) and carefully powdered with an electric grinder (of Flour MILLS NIGERIA, El MOTOR No 1827). Eggs of Shrimp larvae (Artemia salina Leach) commercialized by German company JBL GmbH & Co. KG was used for assessing the cytotoxicity. Male and female albino Wistar rats weighing 150-200 g at the age of months were used for this study. Animals were housed in polypropylene cages and maintained under standard conditions with an alternated cycle of 12 h light and 12 h dark with free access to food and water. The room temperature was maintained at 25 °C with a relative humidity of 35-60%.
Methods:
Extraction and Cytotoxicity Assay: 50 g of each powdered plant were macerated with 500 ml of distilled water for 72 h and filtered three times successively. Then the filtrate was evaporated to dryness at 40 °C using a rotary evaporator (Heidolph efficient Laborota 4000) coupled to a water chiller (Julabo FL 300) to give the crude aqueous extracts 9.
The cytotoxicity test was performed on Artemia salina Leach by the method used by Houngbèmè et al. 9 The eggs of Artenia salina were incubated in seawater until hatching of young larvae (48 h). Then, a series of solutions of each tested crud extract at varying and progressive concentrations were prepared. A defined number (16) of larvae were introduced into each solution. All solutions and control solution containing no active substance was left under stirring for 24 h. Counting under a microscope the number of dead larvae in each solution was used to evaluate the toxicity of the solution. In the case, where there was death in the control medium, the data was corrected by Abbott's formula 10:
% death = 100 × [(test - control) / control)]
Data (dose-response) are transformed by logarithm, and the LC50 was determined by linear regression 11. Tests were carried out in duplicate.
Acute Toxicity Assay: This test was carried out for aqueous extract of S. siamea leaves, S. latifolius roots and their mixture on Wistar rats according to Organization for Economic Cooperation and Development (OECD) guidelines 12. Wistar rats (3 males + 3 females) weighing 150-200 g were used for the test. Animals were kept out on fasting overnight before aqueous extract of Momordica charantia administration.
After the period of fasting, they were weighed, and plant extract was administered by gavage at the dose of 2000 mg/kg. Following administration of extract, food was held for further 3-4 h followed by observation after 30 min, 1 h, 2 h, 3 h, 4 h, 24 h and once a day till the fourteenth day. Rats were examined for various observations like weight change, tremors, convulsion, salivation, diarrhea, lethargy, sleep, coma, and death. Along with that, the cage side observations like changes in the skin, fur, eyes and behavioral pattern were studied.
Sub-acute Toxicity Assay: The rats were unfortunately distributed in a lot of five animals. 3 lots received 2000 mg/kg respectively of the aqueous extract of leaves of C. siamea, of S. latifolius roots and their mixture. The animals were treated for 14 days, and the observation continued after two weeks following the treatment. The dose of the daily one hold managed extract was 2000 mg/kg. During the 4 weeks of the survey, the clinic's signs and the toxicity were observed in all animals daily. The blood withdrawal was done since the first day (J0). It was continued to the 3rd, 7th, 14th and 28th days after administration of the extract. At 7th day, a rat was sacrificed in every group and was dissected; the withdrawal of the kidneys and liver were done for the histological survey.
Determination of Biochemical Parameters: The blood and serum samples were collected by cervical decapitation from anesthetized rats into heparinized bottles for hematological studies. Blood samples collected into non-heparinised bottles were allowed to clot. The serum was separated from the clot and centrifuged into clean bottles for biochemical analysis.
Hematological Measurements Blood samples were collected from retro-orbital of the experimental rats in capillary tubes coated with ethylene diamine tetra-acetic acid (EDTA). The tubes were immediately capped, kept at -4 °C and were immediately analyzed for blood parameters using automated Coagulating Sysmex apparatus Type 8999. The parameters included Aspartate amino-transferase (AST) and alanine aminotransferase (ALT) were determined using a photoelectric colorimeter (Gallenkamp® and Sons Ltd.; England) 13, 14. Serum urea and creatinine levels were determined using the same photoelectric colorimeter 12-14.
Histological Analysis of Rats Treated at 2000 mg/kg with Extracts: The kidneys and the liver were conserved in formalin (10%) during one week, and then they were cutting in small pieces. These samples are dehydrated in three successive ethanol baths for 30 min (70-75°; 90-95° and 100°) before solved in toluene baths, during 20 min. Then, they were put in two baths of paraffin for every 2 h. The operation was automated with an automaton (TISSUE-TEK® II°) device. The definitive inclusion was then achieved in metallic molds (Shandon [OH] Histocentre). The gotten blocks of kinds of paraffin were cut by a microtome (LEICA RM 2145). The cuts of 5µm of thickness were spread on blades and were dried for 1 h at 37 °C, dehydrated, colored to the hematoxylin-eosin (SHANDON [OH]).
Urinary Concentration Determination of Neutral Gelatinized Associated Lipocalin (NGAL) of Treated Rats: It is about to cram the animals at 2000 mg/kg. Urine was collected at 0, 2, 4, 6, 8, 10, 12, 24 h after stuffing. The NGAL test urinary ELISAT is an immunological micro particular determination by reading at a wavelength of 450 nm for its quantitative determination in urine. It was used to detect sharp renal lesions.
Statistical Analysis: All the data obtained were submitted to statistical analysis using Minitab software Version 1.0. The results were expressed as mean. Once the variance analysis (ANOVA) was performed, the differences were pointed out using the Kruskal-Wallis test. Results were considered statistically significant for p<0.05. Correlations between different values were expressed as a graph using Graph Pad PRISM software version 5.
RESULTS:
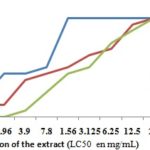
Cytotoxicity Activity: The Fig. 1 show the sensibility of extracts of S. siamea leaves, S. latifolius roots, and their mixture. So, the LC50 values are respectively 0.78 mg/ml, 1.56 mg/ml and of 3.16 mg/ml. The statistical analysis of these results, done by the Student test in non matched sets, permitted us to observe a meaningful difference (p<0.05) between the variables of every extract.
FIG. 1: COMPARATIVE GRAPHS OF THE TOXICITY OF THE EXTRACTS ON THE SHRIMP LARVAE. Sesia: S. siamea, Salat: S. latifolius; Sesia-Salat: the mixture of S. siamea leaves and S. latifolius roots (1/1)
The aqueous extract of the mixture decorated less poisonous on the larvae that one of S. latifolius roots and S. siamea leaves. According to the correspondence table of Mousseux 15, LC50 values proved that the extracts were not toxic. There is a good correlation between toxicity against shrimp larvae and cytotoxicity on 9KB and 9PS (nasopharygien human carcinoma) cells, on the one hand, and cytotoxicity on A-549 carcinoma lung cells and HT -29 colon carcinoma cells, on another hand 9. So the aqueous extracts and their mixture are not cytotoxic.
Acute Toxicity: No overt undesirable effect and no deaths were recorded during the 14 days of observation for regular clinic of sequential doses, 300 mg/kg and 2000 mg/kg of body weight of the animal treated with aqueous extracts of S. siamea leaves, S. latifolius roots and their mixture (v/v). At the time of the experimentation progress, the absence of undesirable effect to the sequential dose of 2000 mg/kg of the body weight of the animal entailed the end of the test.
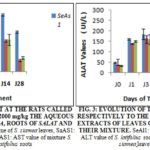
Evolution of Transaminases on the Treated Rats: The Fig. 2 reveals an increase of the AST; 22.27 times 24 hr after within the rats treated to the aqueous extract of Salat to the dose 2000 mg/kg. This increase remains obstinate and reaches 460.6 ± 11.2 UI/L on the 7th day is 30.71 times the value of J0. One week after (J14), this value regressed and reached 300 ± 10.2 UI/L. Two weeks after the stop of the treatment (J28), the regression persisted and reached a value of 110.2 ± 10.2 UI/L is 7.35 times the value in J0. To the level of the rats treated to the aqueous extract of leaves of S. siamea to the same dose, the report was the same with an increase of 26.53 times 24 h after. This increase persisted and reached on the 14th day value of 550 ± 12.4 U.I/L is 36.67 times J0. Two weeks after the stop of the treatment and there is a regression that reached a value of 385 ± 11.5 UI/L (25.67 times J0). For the rats treated in the mixture of leaves of S. siamea and roots of S. latifolius (1/1), there was an increase of 9.82 times J0 24 h after the treatment, this increase appreciably and reached a value of 310.2 ± 52.2 UI/L (10.34 times J0). Two weeks after the treatment, this value regressed and reached 135 ±11.5 UI/L (4.5 times J0).
About the ALAT, Fig. 3 revealed, an increase of 3.5 and 3.3 times J0, 24 h after the treatment respectively at the rats treated to the aqueous extracts of roots of S. latifolius and leaves of S. siamea. This increase persisted and reached his/her/its pick on the day J14 with values 262.0 ±16.0 UI/L and 235 ± 15.2 UI/L respectively at the rats treated with aqueous extract of roots of Salat and leaves of Sesia. Two weeks after the end of the treatment (J28), there was a regression that reached respective values of 140.8±2.1 UI/L (3.5 J0) and 150.8 ± 2.7 UI/L (3.1 J0). To the dose 2000 mg/kg, we observed that the extracts had all provoked a dizzy increase of the transaminases. This increase was least at the rats treated at the extracts of the S. siamea - S. latifolius mixture and two weeks after the end of treatment, the rate of the AST fell considerably.
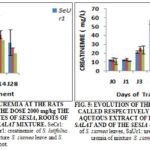
Evolution of Uremia and Creatinemia on Treated Rats: To the dose 2000 mg/kg of aqueous extracts of roots of S. latifolius, of the leaves of S. siamea and their mixture, we observed, at the treated rats, a meaningful increase of the uremia of 1, 83; 2, 6 and 2.42) since the 3rd day of the treatment equivalent to respective values of 1.1 ± 0.0; 1.3 ± 00 and 0.97 ± 01g/L. This increase remained obstinate until the 14th day of treatment with respective values of 3.0 ± 0.3; 3.5 ± 0.3 and 2.61 ± 0.1 g/L. It is necessary to notice that already on the 7th day the rate of urea was 4.67; 5.8 and 4.5 times the rate of J0 at the rats treated respectively to the roots of S. latifolius, the leaves of S. siamea and their mixture. Two weeks after the stop of the treatment (J28), we observed a reduction that brings back the values respectively to 2 ± S 00; 2,4 ± 00 and 1.58 ± 0.4 g/L for the rats treated to the aqueous extract of roots of S. latifolius, the leaves of S. siamea and their mixture. The creatinine increased very quickly and reached on the 14th day, 56.0 ± 2.0; 60.0 ± 1.1et 26.0 ± 2.0 mg/L is 4.48; 5 and 2,38 times J0 at the rats treated respectively to the aqueous extracts of roots of S. latifolius, the leaves of S. siamea and their mixture. Two weeks after the stop of the treatment (J28), we observed, a meaningful reduction, the creatinemie reached the values of 20.7 ±1.3 mg/L, 30 ± 2.1 and 12.0 mg/L at the rats treated respectively to the aqueous extracts of roots of S. latifolius, the leaves of S. siamea and their mixture.
These results were confirmed by histological analysis which proved that the renal cortex of the rats treated with the aqueous extract of leaves of S. siamea to the dose 2000 mg/kg presented a disorganization of the structure of the renal cortex that become hardly recognizable with a hyalinisation of the regions glomerular giving a picture of " kidney in bread to seal " on the 7th day of treatment.
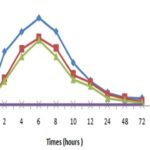
Evolution of the Concentration of Urinary NGAL at the Treated Rats: The Fig. 6 below showed the curves translating the urinary NGAL expression in the time of the rats treated respectively to the aqueous extract of roots of S. latifolius and of leaves of S. siamea presented the same pace. These curves have an ascending pace that reaches 2h after the treatment, the values of 3281 ng/ml; 1660ng/ml and 1500ng/ml respectively at the rats treated to the aqueous extract of leaves of S. siamea of roots of S. latifolius and their mixture. These values increased and reached their picks at the 6th hrs with 532ng/ml; 4120 ng/ml and 4000 ng/ml respectively at the rats treated to the aqueous extract of leaves of S. siamea, of roots of S. latifolius and their mixture.
FIG. 6: EVOLUTION OF THE URINARY CONCENTRATION OF NGAL AT THE WITNESS AND THE RATS TREATED TO THE DIFFERENT EXTRACTED OF PLANTS. (n = 5)
We observed a significant regression of these values of the 8th hrs at the 12th hrs (p = 0.0002 on average). This regression was no meaningful of the 24th the 72nd h with respective values of 456 ng/ml; 260 ± 0 ng/ml and 200 ng/ml at the rats treated to the extract of leaves of S. siamea, of roots of S. latifolius and the mixture. The concentrations of NGAL to the level of the rats witnesses evolved around a middle value of 68.7ng/ml.
The histological cut of some organs appropriated at the rats milked respectively to the dose of 2000 mg/kg of the aqueous extract of leaves of S. siamea, of roots of S. latifolius and the mixture. To- normal histological different studied organ aspect.
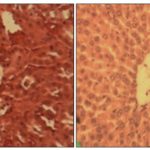
FIG. 7: NORMAL HISTOLOGY OF THE LIVER AND THE RENAL CORTEX OF WISTAR RAT. A) NORMAL RENAL CORTEX OF WISTAR RAT SHOWING A RENAL GLOMERULAR (G X 100). B) NORMAL HEPATIC LOBULE OF WISTAR RAT (G X 100)
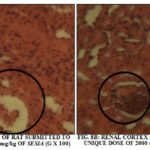
The histological aspects of the renal cortex of rat wistar after sharp administration of the dose 2000 mg/kg respectively of the aqueous extract of leaves of S. siamea and roots of S. latifolius are presented in the Fig. 8A and 8B. The Fig. 9A and 9B show the histological aspects of the liver of Wistar rat respectively after sharp administration of dose 2000 mg/kg of the aqueous extract of leaves of S. siamea or roots of S. latifolius. The administration of the aqueous extract of leaves of S. siamea to the dose 2000 mg/kg creates some signs of necroses at the rats that the level of the renal cortex that hepatocyte what was not observed at those treated to the aqueous extract of roots of S. latifolius to the same dose.
Sub-Acute Toxicity: Histological impacts of the treatment of the rat’s Wistar to the dose of 2000 mg/kg of body weight of the aqueous extract of leaves of S. siamea and roots of S. latifolius during 7 days.
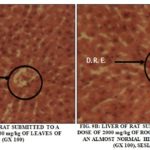
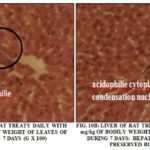
The Liver Aspects: The Fig. 10A and 10B show the histological aspects of the liver of rat wistar after 7 days of treatment to the lethal dose of 2000 mg/kg of body weight of the aqueous extract of leaves of S. siame and roots of S. latifolius.
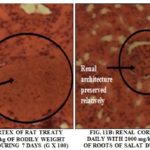
Renal aspects: The histological aspects of the renal cortex of wistar rat after 7 days of treatment at the daily dose of 2000 mg/Kg of body weight for aqueous extract of S. siamea leaves and S. latifolius roots were presented in Fig 11A and 11B. The aqueous extract of leaves of S. siamea to the dose 2000 mg/kg destroyed the renal cortex and created of necrosis to the level hepatocyte with pycnose of the core.
DISCUSSION: To guarantee the innocuity of the products destined to the man in the conditions of jobs foreseen, some toxicological studies must be achieved on laboratory animals. The different tests of toxicity permitted to determine the Lethal Dose 50 % (LD50) of leaves of S. siamea, of roots of S. latifolius, and the one of their mixture. The LD50 of these different plants and mixture were superior to 2000 mg/kg, our results corroborated those of Mohammed and coll. 16 that indicated that the Lethal Dose50 of the aqueous extract of S. siamea was superior to 5000 mg/kg. The aqueous extract of leaves of S. siamea was slightly more poisonous than the one of roots of S. latifolius and of the mixture of S. siamea leaves - S. latifolius roots with as lethal concentrations (LC50) respective of 0,78; 1,56, and 3,16 mg/ml.
According to the scale of toxicity of Hodge and Sterner 17 and of the WHO 18, our extracts were weakly poisonous and classified in the III class of the chemical substances.
According to José and coll. 19, an interrelationship existed between the toxicity of the shrimp larvae (Artemia salina) and pulmonary cell toxicity. The larvas of shrimp pickle were sensitive to the poisonous effects of the aqueous extracts of leaves of S. siamea, of roots, and the one of the mixtures of S. siamea leaves - S. latifolius roots. The lethal concentrations gotten at the shrimp larvae pickle were too weak to predict toxicity at the man.
Several poisonous plants compound accumulated in the liver where they are detoxified 20. The liver was the first target of the toxicity and the first organ exposed to everything that is absorbed in the spindly intestine; he/it metabolizes the substances foreign to compounds that can be hepatotoxic 21. The liver worked in association with the kidneys to suppress the poisonous substances of blood 22.
A survey of the renal and hepatic function can prove to be therefore useful to value the poisonous medicinal plant effects. These tests consisted mainly of the determination of the AST, the ALT, the creatinemy, the urea and other 23 and all hepatic cell necrosis derived to a meaningful rise of the AST enzymes, ALT in the blood serum 24.
The ALT and the AST were the cytotoxic enzymes whose rates rise quickly when the liver was damaged for various reasons including the hepatic cellular necroses, hepatitis, the cirrhosis as well as the hepatotoxicity of some drugs 25.
The seric studies done on the rats treated by the aqueous extract of leaves of S. siamea, of roots of S. latifolius and their mixture to the dose 2000 mg/kg showed a meaningful increase of the parameters: ASAT, ALAT, the urea and the creatinemy in relation to the witnesses These increases would translate a reach hepatocytary 26. This reach has been confirmed, by the reduction of the spaces vascular and some signs of hepatocytary necrosis in the centrolobular region FIG. 9A. This poisonous action could be assigned to the different secondary metabolites as the saponosides, the tannins, the flavonoïdes, present in the aqueous extract respectively of the leaves of S. siamea and roots of S. latifolius as Agbaje and coll, 27 suggested it, at the time of the survey of the toxicity subaigüe of Syzigium aromaticum on the rats.
To the level of the rats treated to the dose 2000 mg/kg of aqueous extract of leaves of S. siamea, there was a disorganization of the structure of the renal cortex that become hardly recognizable with a hyalinisation of the glomerular regions giving a picture of " kidney in bread to seal " on the 7th day of treatment Fig. 11A.
Of this fact, he/it was probable that the aqueous extracts of the leaves of S. siamea, of the roots of S. latifolius and of their mixture, misled a vascular proliferation to the level of the centrolobular zone and the space carried from the successive metabolisms bound to the daily stuffings to single dose. The successive increases of the rates of the transaminases found all their explanations in the numerous metabolism complexities that took place after the stuffings of the animals to the extracts of plants in the case of experimentation.
In all aggressions by the xenobiotical, the hepatic metabolism of the proteins was modified toward the production of the defense systems and the neoglucogenesis. The deterioration of the compounds proteic hepatic could explain the increase of the urea and seric creatinin at the treated rats, where the proteins can be damaged then in amino acids in urea and creatinin. So these formed amino acids could turn under the action of the seric transaminases into carboxylic compounds as the pyruvic acidic 28. It implies the strong enzymatic activity of the transaminases.
The seric rates of creatinine and urea that was of excellent scorers of the renal function are generally constant in the normal conditions 29. The increase or the reduction of these scorers reflected a renal dysfunction 30. The interest of the measure of the rate of the urea, consisted in valuing the intensity of the protidic destruction, herself reflection of the lesions associated to all metabolic process. The changes can reach the numerous structures of the kidneys in a reversible or chronic way.
The results of this survey permitted to appreciate the exhibition of the population to the medicinal plant use. It puts the problematic of the security and the health of the Beninese population. These different arrangements were not sufficient to confirm the toxicity of the extracts of the leaves of S. siamea, of the roots of S. latifolius and the mixture.
In the animal model, the NGAL was one of the proteins, induced the most precociously after a renal reach of ischemic type or poisonous 31. He is used in different situations clinics as in postoperative of cardiac surgery, at the time of a renal transplantation 32, after an administration of products of contrasts iodized or in intensive care. A concentration of NGAL in urine above 350 ng/ml or in plasma 400 ng/ml was above strongly likely (> 90 %, positive value predictive) to be due to the renal damages that can lead to the renal failure. A sudden elevation of the urinary concentration of NGAL above the patient's previous values can indicate the renal damages even though the value limits is not passed, and the narrow surveillance of the renal function is recommended.
The dosage of NGAL in this survey has for objective to detect precocious manner the effect of the administration by oral way of the aqueous excerpt of leaves of S. siamea, of roots of S. latifolius and their mixture to the dose 2000 mg/kg. The gotten results showed a dizzy rise of the urinary NGAL rate Fig. 6 enters 0 and 6 h whatever is the extract, there is a meaningful difference respectively between the rate of NGAL of the three extracts of plants at the time 2 h, 4 h and 6 h.
The rate raised from NGAL could signal damages of the kidney 33. This result confirms those especially gotten on the biochemical and histological side for the aqueous extract of Sesia.
Of this fact, we will say that the dose 2000 mg/kg of these different extracts apparently without effect on motility, the lucidity of the animals, create some damages to the levels of the organs as the liver and the kidneys.
CONCLUSION: The toxicity of the aqueous extract of leaves of Senna siamea, of roots of Sarcocephalus latifolius and their mixture (1/1) to the dose 2000 mg/kg has been performed. Information pulled from the set of the results presented in this work on the applied data of the acute toxicity at the rats Albino wistars suggested classifying leaves of Senna siamea, of roots of Sarcocephalus latifolius and their mixture in the weakly toxicity plant's category by the oral way.
However, the sub-acute toxicity showed that the aqueous extract of these plants and their mixture (1/1) at 2000 mg/kg disrupts some biochemical parameters bound to the hepatic, renal and muscular function. The histological observation was especially characterized by structural changes of the liver and the kidney on the 7th day. These changes were confirmed by the rate raised from NGAL in the urine, noted since the first hours of the rats treated. The further works will be focused on the pharmacological activities and toxicological on human cells studies for moderation of security in the use of these plants.
ACKNOWLEDGEMENT: The authors thank Prof. Anatole Laleye, M. Brumo Agboton and Prof. Pierre H. Dansou for their technical control and financial support respectively.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Assogbadjo AE, Glele Kakaı R, Chadare FJ, Thomson L, Kyndt T, Sinsin B and Van Damme P: Folk classification, perception and preferences of baobab products in West Africa: consequences for species conservation and improvement. Econ Bot 2008; 62: 74-84.
- Vodouhe FG, Coulibaly O, Greene C and Sinsin B: Estimating the local value of non-timber forest products to Pendjari Biosphere Reserve dwellers in Benin. Econ Bot 2009; 63: 397- 412.
- kalaivani T, Rajasekaran C, Suthindhiran K and Mathew L: Free radical scavenging, cytotoxic and hemolytic activities from leaves of Acasia nilotica (L.) Wild. ex. Delite subsp. indica (Benth.) Brenan. Evidence-Based Complementary and Alternative Medicine (Advance Access) 2010; 1-8.
- Allabi AC, Busia K, Ekanmian V and Bakiono F: The use of medicinal plants in self-care in the Agonlin region of Benin. Journal of Ethnopharmacology 2011; 133: 234-243.
- Ademola IO, Fagbemi BO and Idowu SO: Nauclea latifolia extract against gastrointestinal nematodes of sheep: in-vitro and in-vivo. African Journal of Traditional Complementary and Alternative Medicines 2007; 4(2): 148-156.
- Yesufu HB, Mohammed GT, Muazu J, Yakubu S, Sadiq UG and Wazis CH: Effect of sub chronic administration of ethanolic root bark extract of Sarcocephalus latifolius (Smith) Bruce (Rubiaceae) on hematological parameters of rats. Res J Pharm Biol Chem Sci 2011; 2(3): 499.
- Nsonde Ntandou GF: Etude des propriétés chimiques pharmacologiques et anti vectorielles de Cassia siamea (Fabacae) sur le paludisme. Thèse de Doctorat d’Etat. Physiologie animale-pharmacologie-Biochimie. Université Marien NGOUABI 2010; 165.
- Odugbemi T: Outlines and Pictures of Medicinal Plants from Nigeria. University of Lagos Press. Revised Edition. 2008: 54.
- Houngbeme AG, Gandonou C, Yehouenou B, Kpoviessi SDS, Sohounhloue D, Moudachirou M and Gbaguidi FA: Phytochemical analysis, toxicity and antibacterial activity of Benin medicinal plants extracts used in the treatment of sexually transmitted infections associated with hiv/aids. Int J Pharm Sci Res 2014; 5(5): 1739-1745.
- Abbott WS: A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925; 18: 265.
- Solis PN, Wright CW, Aderson MM, Gupta MP and Phillipson JD: A microwell cytotoxicity assay using Artemia salina. Plant Med 1999; 59: 250-2.
- Organisation de Coopération et de Développement Economique (OCDE). Essai n° 423: Toxicité orale aigue. Méthode par classe de toxicité. Bookshop 2002.
- Duncan JR, Prasse KW and Mahaffey EA: Veterinary Laboratory Medicine (Clinical Pathology), Iowa State University Press, Ames 1994: 94-96.
- Coles EH: Veterinary. Clinical Pathology. W.B. Saunders’ Co., Philadelphia 1986; 5-87.
- Mousseux M: Test de toxicité sur larves d'Artemia salina: entretien d'un élevage de balanes; Nouméa, Centre Universitaire de Nouvelle Calédonie 1995; 20.
- Mohammed A, Liman ML and Atiku MK: Chemical composition of the methanolic leaf and stem bark extracts of Senna siamea Lam». Journal of Pharmacognosy and Phytotherapy 2013; 5: 98-100.
- Hodge HG and Sterner JH: Determination of substance acute toxicity by LD50. American Industrial Hygien Asssociation 1943; 1093-101.
- OMS: Médecine traditionnelle: Rapport du secrétariat. Conseil exécutif. 111ième session. Point 5.7 de l’ordre du jour provisoire. EB 111/9 du 12 Décembre 2002; 28.
- José Luis C, Zaira L, Hernandez I, Pilar P, Maria D and Garcia G: A comparison between two brine shrimp assays to detect in-vitro cytotoxicity in marine natural products. BMC Biotechnology 2002; 2: 17-35.
- Clarke EGC and Clarke ML: Veterinary Toxicology. Ed Cassel and Collier Macmillan, London, 1977: 268-277.
- Rhiouani H, El-Hilaly J, Israili ZH and Lyoussi B: Acute and sub-chronic toxicity of an aqueous extract of the leaves of Herniaria glabra in rodents. Journal of Ethnopharmacology 2008; 118: 378-386.
- Tulsawani R: Ninety day repeated gavage administration of Hipphophae rhamnoides extract in rats. Food and Chemical Toxicology 2010; 48: 2483-2489.
- Tilkian SM: Clinical Implications of Laboratory Tests. Missouri: The C.V. Mosby Company, 1979: 11-17.
- Adeneye AA, Ajagbonna OP, Adeleke TI and Bello SO: Preliminary toxicity and phytochemical studies of the stem bark aqueous extract of Musanga cecropioides in rats. Journal of Ethnopharmacology 2006; 105: 374-379.
- Palanivel MG, Rajkapoor B, Kumar RS, Einstein JW and Prem E: Effet hépatoprotecteur et antioxydant de Pisonia aculeata L. contre la CCl4 lésions hépatiques induites chez le rat. Sci. Pharm 2008; 76: 203-15.
- Adedapo AA, Abatan MO and Olorunsogo OO: Toxic effects of some plants in the genus of Euphorbia on the hematological and biochemical parameters of rats. Vet Arhiv 2004; 1(77): 29-38.
- Agbaje EO and Daramola AO: Biochemical and toxicological studies of aqueous extract of Syzigium aromaticum (L.) Merr and Perry (myrtaceae) in rodents. Afr J Trad 2009; 3(6): 241-254.
- Bergmeyer H: Dietary nitrate in man: friend or foe? Clin Chem Acta 1980; 105: 147-152.
- Whitby IG, Smith AF and Beckett GJ: Lecture note of clinical chemistry. Blackwell Scientific Publications, Oxford, Edition 4th, 1988: 153- 4.
- Sirwal IA, Banday KA, Reshi AR, Bhat MA and Wani MM: Estimation of glomerular filteration rate (GFR). JK Science 2004; 6: 121-3.
- Mishra J, Ma Q, Prada A, Mitsnefes M, Zahedi K and Yang J: Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury. J Am Soc Nephrol 2003; 14: 2534-43.
- Malyszko J, Malyszko JS, Bachorzewska-Gajewska H, Poniatowski B, Dobrzycki S and Mysliwiec M: Neutrophil gelatinase-associated lipocalin is a new and sensitive marker of kidney function in chronic kidney disease patients and renal allograft recipients. Transplant Proc 2009; 41: 158-61.
- Devarajan P: NGAL in acute Kidney Injury: from serendipity to utility. Am J Kid Dis 2008; 52(3): 395-99.
How to cite this article:
Agbodjogbe KWDD, Houngbeme AG, Haïdara M, Sanogo R, Laleye A and Dansou HP: In-vitro and in-vivo toxicological evaluation of aqueous extracts from Beninese plants: Senna siamea leaves and Sarcopcephalus latifolus stem roots. Int J Pharmacognosy 2017; 4(2): 60-70. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(2).60-70.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
60-70
731
1562
English
IJP
K. W. Dieu-Donné Agbodjogbe, A. G. Houngbeme *, M. Haïdara, R. Sanogo, A. Laleye and H. P. Dansou
Laboratoire National de Pharmacognosie 3, Institut de Recherche et d’Expérimentation en Médecine et Pharmacopée Traditionnelles (IREMPT), Centre Béninois de Recherche Scientifique et Innovation (CBRSI), Benin
albanusphd@yahoo.fr
25 November 2016
22 December 2016
26 December 2016
10.13040/IJPSR.0975-8232.IJP.4(2).60-70
01 February 2017